Sinol™d
ee82eb62-7e52-4a38-8f38-656e24f5d214
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts:
Purpose
Relief of nasal congestion and post nasal drip
Medication Information
Purpose
Relief of nasal congestion and post nasal drip
Description
Drug Facts:
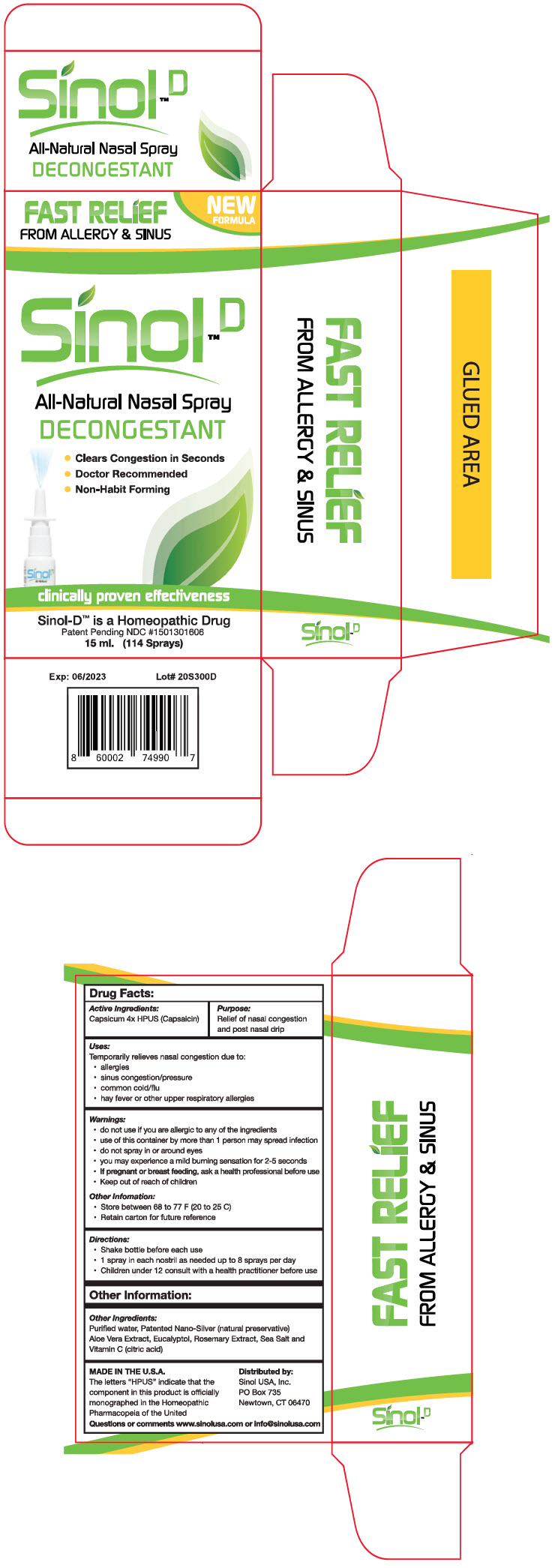
Uses
Temporarily relieves nasal congestion due to:
- allergies
- sinus congestion/pressure
- common cold/flu
- hay fever or other upper respiratory allergies
Section 42229-5
Drug Facts:
Section 50565-1
- Keep out of reach of children
Section 50567-7
- use of this container by more than 1 person may spread infection
- do not spray in or around eyes
- you may experience a mild burning sensation for 2-5 seconds
Section 50570-1
- do not use if you are allergic to any of the ingredients
Section 53414-9
- If pregnant or breast feeding, ask a health professional before use
Directions
- Shake bottle before each use
- 1 spray in each nostril as needed up to 8 sprays per day
- Children under 12 consult with a health practitioner before use
Other Information
- Store between 68 to 77 F (20 to 25 C)
- Retain carton for future reference
Other Ingredients
Purified water, Patented Nano-Silver (natural preservative) Aloe Vera Extract, Eucalyptol, Rosemary Extract, Sea Salt and Vitamin C (citric acid)
Active Ingredients
Capsicum 4× HPUS (Capsaicin)
Questions Or Comments
www.sinolusa.com or [email protected]
Principal Display Panel 15 Ml Bottle Carton
FAST RELIEF
FROM ALLERGY & SINUS
NEW
FORMULA
Sinol™ D
All-Natural Nasal Spray
DECONGESTANT
- Clears Congestion in Seconds
- Doctor Recommended
- Non-Habit Forming
clinically proven effectiveness
Sinol-D™ is a Homeopathic Drug
Patent Pending NDC #1501301606
15 ml. (114 Sprays)
Structured Label Content
Uses
Temporarily relieves nasal congestion due to:
- allergies
- sinus congestion/pressure
- common cold/flu
- hay fever or other upper respiratory allergies
Section 42229-5 (42229-5)
Drug Facts:
Section 50565-1 (50565-1)
- Keep out of reach of children
Section 50567-7 (50567-7)
- use of this container by more than 1 person may spread infection
- do not spray in or around eyes
- you may experience a mild burning sensation for 2-5 seconds
Section 50570-1 (50570-1)
- do not use if you are allergic to any of the ingredients
Section 53414-9 (53414-9)
- If pregnant or breast feeding, ask a health professional before use
Purpose
Relief of nasal congestion and post nasal drip
Directions
- Shake bottle before each use
- 1 spray in each nostril as needed up to 8 sprays per day
- Children under 12 consult with a health practitioner before use
Other Information
- Store between 68 to 77 F (20 to 25 C)
- Retain carton for future reference
Other Ingredients
Purified water, Patented Nano-Silver (natural preservative) Aloe Vera Extract, Eucalyptol, Rosemary Extract, Sea Salt and Vitamin C (citric acid)
Active Ingredients
Capsicum 4× HPUS (Capsaicin)
Questions Or Comments (Questions or comments)
www.sinolusa.com or [email protected]
Principal Display Panel 15 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 15 ml Bottle Carton)
FAST RELIEF
FROM ALLERGY & SINUS
NEW
FORMULA
Sinol™ D
All-Natural Nasal Spray
DECONGESTANT
- Clears Congestion in Seconds
- Doctor Recommended
- Non-Habit Forming
clinically proven effectiveness
Sinol-D™ is a Homeopathic Drug
Patent Pending NDC #1501301606
15 ml. (114 Sprays)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:56.948574 · Updated: 2026-03-14T23:17:15.281880