These Highlights Do Not Include All The Information Needed To Use Abilify Maintena Safely And Effectively. See Full Prescribing Information For Abilify Maintena.
ee49f3b1-1650-47ff-9fb1-ea53fe0b92b6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1) ].
Indications and Usage
ABILIFY MAINTENA (aripiprazole) is indicated: for the treatment of schizophrenia in adults for maintenance monotherapy treatment of bipolar I disorder in adults
Dosage and Administration
Only to be administered by intramuscular injection in the deltoid or gluteal muscle by a healthcare professional ( 2.1 ) For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating ABILIFY MAINTENA ( 2.1 ) Recommended dosage is 400 mg administered monthly as a single injection. Dose can be reduced to 300 mg in patients with adverse reactions ( 2.2 ) There are two ways to initiate treatment with ABILIFY MAINTENA 1-day initiation: Administer two separate intramuscular injections of ABILIFY MAINTENA 400 mg and a single oral dose of aripiprazole 20 mg ( 2.2 ) 14-day initiation: In conjunction with first ABILIFY MAINTENA 400 mg dose, take 14 consecutive days of concurrent oral aripiprazole (10 mg to 20 mg) or current oral antipsychotic ( 2.2 ) Dosage adjustments are required for missed doses ( 2.3 ) Known CYP2D6 poor metabolizers: Recommended starting and maintenance dose is 300 mg administered monthly as a single injection ( 2.4 ) See full prescribing information for ABILIFY MAINTENA dosage modifications due to drug interactions ( 2.4 ). ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe ( 2.6 ), and 2) Vials ( 2.7 ).
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities) ( 5.2 ) Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring ( 5.3 ) Tardive Dyskinesia: Discontinue if clinically appropriate ( 5.4 ) Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain ( 5.5 ) – Hyperglycemia and Diabetes Mellitus: Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with and at risk for diabetes ( 5.5 ) – Dyslipidemia: Undesirable alterations have been observed in patients treated with atypical antipsychotics ( 5.5 ) – Weight Gain: Gain in body weight has been observed; clinical monitoring of weight is recommended ( 5.5 ) Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation ( 5.6 ) Orthostatic Hypotension: Use with caution in patients with known cardiovascular or cerebrovascular disease ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell count (WBC)/absolute neutrophil count (ANC). Consider discontinuation if clinically significant decline in WBC/ANC in the absence of other causative factors ( 5.9 ) Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold ( 5.10 ) Potential for Cognitive and Motor Impairment: Use caution when operating machinery ( 5.11 )
Contraindications
ABILIFY MAINTENA is contraindicated in patients with a known hypersensitivity to aripiprazole. Hypersensitivity reactions ranging from pruritus/urticaria to anaphylaxis have been reported in patients receiving aripiprazole [see Adverse Reactions (6.1 and 6.2) ] .
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Increased Mortality in Elderly Patients with Dementia - Related Psychosis Use [see Boxed Warning and Warnings and Precautions (5.1) ] Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning and Warnings and Precautions (5.2) ] Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.3) ] Tardive Dyskinesia [see Warnings and Precautions (5.4) ] Metabolic Changes [see Warnings and Precautions (5.5) ] Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions (5.6) ] Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.7) ] Falls [see Warnings and Precautions (5.8) ] Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.9) ] Seizures [see Warnings and Precautions (5.10) ] Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.11) ] Body Temperature Regulation [see Warnings and Precautions (5.12) ] Dysphagia [see Warnings and Precautions (5.13) ]
Drug Interactions
CYP2D6 inhibitors and CYP3A4 Inhibitors : See full prescribing information for ABILIFY MAINTENA dosage modifications when used concomitantly with CYP2D6 inhibitors and/or CYP3A4 inhibitors for greater than 14 days ( 7.1 ) CYP3A4 Inducers : Avoid concomitant use for greater than 14 days ( 7.1 ) See full prescribing information for additional clinically significant drug interactions ( 7.1 )
Medication Information
Warnings and Precautions
Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities) ( 5.2 ) Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring ( 5.3 ) Tardive Dyskinesia: Discontinue if clinically appropriate ( 5.4 ) Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain ( 5.5 ) – Hyperglycemia and Diabetes Mellitus: Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with and at risk for diabetes ( 5.5 ) – Dyslipidemia: Undesirable alterations have been observed in patients treated with atypical antipsychotics ( 5.5 ) – Weight Gain: Gain in body weight has been observed; clinical monitoring of weight is recommended ( 5.5 ) Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation ( 5.6 ) Orthostatic Hypotension: Use with caution in patients with known cardiovascular or cerebrovascular disease ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell count (WBC)/absolute neutrophil count (ANC). Consider discontinuation if clinically significant decline in WBC/ANC in the absence of other causative factors ( 5.9 ) Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold ( 5.10 ) Potential for Cognitive and Motor Impairment: Use caution when operating machinery ( 5.11 )
Indications and Usage
ABILIFY MAINTENA (aripiprazole) is indicated: for the treatment of schizophrenia in adults for maintenance monotherapy treatment of bipolar I disorder in adults
Dosage and Administration
Only to be administered by intramuscular injection in the deltoid or gluteal muscle by a healthcare professional ( 2.1 ) For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating ABILIFY MAINTENA ( 2.1 ) Recommended dosage is 400 mg administered monthly as a single injection. Dose can be reduced to 300 mg in patients with adverse reactions ( 2.2 ) There are two ways to initiate treatment with ABILIFY MAINTENA 1-day initiation: Administer two separate intramuscular injections of ABILIFY MAINTENA 400 mg and a single oral dose of aripiprazole 20 mg ( 2.2 ) 14-day initiation: In conjunction with first ABILIFY MAINTENA 400 mg dose, take 14 consecutive days of concurrent oral aripiprazole (10 mg to 20 mg) or current oral antipsychotic ( 2.2 ) Dosage adjustments are required for missed doses ( 2.3 ) Known CYP2D6 poor metabolizers: Recommended starting and maintenance dose is 300 mg administered monthly as a single injection ( 2.4 ) See full prescribing information for ABILIFY MAINTENA dosage modifications due to drug interactions ( 2.4 ). ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe ( 2.6 ), and 2) Vials ( 2.7 ).
Contraindications
ABILIFY MAINTENA is contraindicated in patients with a known hypersensitivity to aripiprazole. Hypersensitivity reactions ranging from pruritus/urticaria to anaphylaxis have been reported in patients receiving aripiprazole [see Adverse Reactions (6.1 and 6.2) ] .
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Increased Mortality in Elderly Patients with Dementia - Related Psychosis Use [see Boxed Warning and Warnings and Precautions (5.1) ] Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning and Warnings and Precautions (5.2) ] Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.3) ] Tardive Dyskinesia [see Warnings and Precautions (5.4) ] Metabolic Changes [see Warnings and Precautions (5.5) ] Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions (5.6) ] Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.7) ] Falls [see Warnings and Precautions (5.8) ] Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.9) ] Seizures [see Warnings and Precautions (5.10) ] Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.11) ] Body Temperature Regulation [see Warnings and Precautions (5.12) ] Dysphagia [see Warnings and Precautions (5.13) ]
Drug Interactions
CYP2D6 inhibitors and CYP3A4 Inhibitors : See full prescribing information for ABILIFY MAINTENA dosage modifications when used concomitantly with CYP2D6 inhibitors and/or CYP3A4 inhibitors for greater than 14 days ( 7.1 ) CYP3A4 Inducers : Avoid concomitant use for greater than 14 days ( 7.1 ) See full prescribing information for additional clinically significant drug interactions ( 7.1 )
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1) ].
Section 42229-5
Preparation Prior to Reconstitution
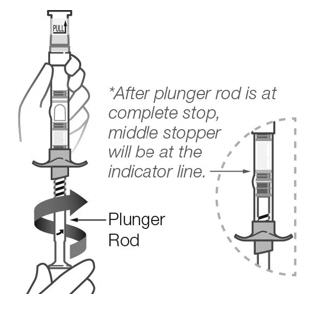
For deep intramuscular deltoid or gluteal injection by healthcare professionals only. Do not administer by any other route. Inject full syringe contents immediately following reconstitution. Administer once monthly.
Lay out and confirm that components listed below are provided in the kit:
- One ABILIFY MAINTENA (aripiprazole) pre-filled dual chamber syringe (400 mg or 300 mg as appropriate) for extended-release injectable suspension containing lyophilized powder and Sterile Water for Injection
- One 23-gauge, 1-inch (25 mm) hypodermic safety needle with needle protection device for deltoid administration in non-obese patients
- One 22-gauge, 1.5-inch (38 mm) hypodermic safety needle with needle protection device for gluteal administration in non-obese patients or deltoid administration in obese patients
- One 21-gauge, 2-inch (51 mm) hypodermic safety needle with needle protection device for gluteal administration in obese patients
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 03/2025 | ||
|
MEDICATION GUIDE
ABILIFY MAINTENA® (a-BIL-i-fy main-TEN-a) (aripiprazole) for extended-release injectable suspension, for intramuscular use |
|||
|
What is the most important information I should know about ABILIFY MAINTENA?
ABILIFY MAINTENA may cause serious side effects, including:
|
|||
|
What is ABILIFY MAINTENA?
ABILIFY MAINTENA is a prescription medicine given by injection by a healthcare provider:
|
|||
|
Who should not receive ABILIFY MAINTENA? Do not receive ABILIFY MAINTENA if you are allergic to aripiprazole or any of the ingredients in ABILIFY MAINTENA. See the end of this Medication Guide for a complete list of ingredients in ABILIFY MAINTENA. |
|||
Before receiving ABILIFY MAINTENA, tell your healthcare provider about all of your medical conditions, including if you:
ABILIFY MAINTENA and other medicines may affect each other causing possible serious side effects. ABILIFY MAINTENA may affect the way other medicines work, and other medicines may affect how ABILIFY MAINTENA works. Your healthcare provider can tell you if it is safe to receive ABILIFY MAINTENA with your other medicines. Do not start or stop any medicines during treatment with ABILIFY MAINTENA without talking to your healthcare provider first. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I receive ABILIFY MAINTENA?
|
|||
What should I avoid while receiving ABILIFY MAINTENA?
|
|||
|
What are the possible side effects of ABILIFY MAINTENA? ABILIFY MAINTENA may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
These are not all the possible side effects of ABILIFY MAINTENA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of ABILIFY MAINTENA.
If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about ABILIFY MAINTENA that is written for healthcare professionals. |
|||
|
What are the ingredients in ABILIFY MAINTENA? Active ingredient: aripiprazole monohydrate Inactive ingredients: carboxymethylcellulose sodium, mannitol, sodium phosphate monobasic monohydrate and sodium hydroxide ABILIFY MAINTENA is a trademark of Otsuka Pharmaceutical Co., Ltd. ©2025, Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan For more information, go to www.ABILIFYMAINTENA.com or call 1-800-441-6763. |
Section 43683-2
Section 44425-7
Storage
5.8 Falls
Antipsychotics, including ABILIFY MAINTENA, may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
5.10 Seizures
As with other antipsychotic drugs, use ABILIFY MAINTENA cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in a population of 65 years or older.
11 Description
Aripiprazole is an atypical antipsychotic which is present in ABILIFY MAINTENA as its monohydrate polymorphic form. Aripiprazole monohydrate is 7-[4-[4-(2,3-dichlorophenyl)-1-piperazinyl] butoxy]-3,4 dihydrocarbostyril monohydrate. The empirical formula is C23H27Cl2N3O2∙H2O and its molecular weight is 466.40. The chemical structure is:
ABILIFY MAINTENA (aripiprazole) is an extended-release injectable suspension available in 400 mg or 300 mg strength pre-filled dual chamber syringes and 400 mg or 300 mg strength vials. The labeled strengths are calculated based on the anhydrous form (aripiprazole). Inactive ingredients (per administered dose) for 400 mg and 300 mg strength products, respectively, include carboxymethyl cellulose sodium (16.64 mg and 12.48 mg), mannitol (83.2 mg and 62.4 mg), sodium phosphate monobasic monohydrate (1.48 mg and 1.11 mg) and sodium hydroxide (pH adjuster).
5.13 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use, including ABILIFY MAINTENA. ABILIFY MAINTENA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia [see Warnings and Precautions (5.1)].
2.3 Missed Doses
If the second or third doses are missed:
- If more than 4 weeks and less than 5 weeks have elapsed since the last injection, administer the injection as soon as possible.
- If more than 5 weeks have elapsed since the last injection, restart treatment with either 1-day initiation or 14-day initiation with ABILIFY MAINTENA [see Dosage and Administration (2.2)].
If the fourth or subsequent doses are missed:
- If more than 4 weeks and less than 6 weeks have elapsed since the last injection, administer the injection as soon as possible.
- If more than 6 weeks have elapsed since the last injection, restart treatment with either 1-day initiation or 14-day initiation with ABILIFY MAINTENA [see Dosage and Administration (2.2)].
8.4 Pediatric Use
ABILIFY MAINTENA has not been studied in children 18 years of age or younger. However, juvenile animal studies have been conducted in rats and dogs.
8.5 Geriatric Use
Clinical studies of oral aripiprazole did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. Other reported clinical experience and pharmacokinetic data have not identified differences in responses between the elderly and younger patients [see Clinical Pharmacology (12.3)]. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
In single-dose and multiple-dose pharmacokinetic studies, there was no detectable age effect in the population pharmacokinetic analysis of oral aripiprazole in schizophrenia patients [see Clinical Pharmacology (12.3)]. No dosage adjustments are recommended based on age alone. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis [see also Boxed Warning and Warnings and Precautions (5.1)].
14.1 Schizophrenia
The efficacy of ABILIFY MAINTENA for treatment of schizophrenia was established in:
- One short-term (12-week), randomized, double-blind, placebo-controlled trial in acutely relapsed adults, Protocol 31-12-291 (Study 1)
- One longer-term, double-blind, placebo-controlled, randomized-withdrawal (maintenance) trial in adults, Protocol 31-07-246 (Study 2).
4 Contraindications
ABILIFY MAINTENA is contraindicated in patients with a known hypersensitivity to aripiprazole. Hypersensitivity reactions ranging from pruritus/urticaria to anaphylaxis have been reported in patients receiving aripiprazole [see Adverse Reactions (6.1 and 6.2)].
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia - Related Psychosis Use [see Boxed Warning and Warnings and Precautions (5.1)]
- Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning and Warnings and Precautions (5.2)]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.3)]
- Tardive Dyskinesia [see Warnings and Precautions (5.4)]
- Metabolic Changes [see Warnings and Precautions (5.5)]
- Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions (5.6)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.7)]
- Falls [see Warnings and Precautions (5.8)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.9)]
- Seizures [see Warnings and Precautions (5.10)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.11)]
- Body Temperature Regulation [see Warnings and Precautions (5.12)]
- Dysphagia [see Warnings and Precautions (5.13)]
7 Drug Interactions
- CYP2D6 inhibitors and CYP3A4 Inhibitors: See full prescribing information for ABILIFY MAINTENA dosage modifications when used concomitantly with CYP2D6 inhibitors and/or CYP3A4 inhibitors for greater than 14 days (7.1)
- CYP3A4 Inducers: Avoid concomitant use for greater than 14 days (7.1)
- See full prescribing information for additional clinically significant drug interactions (7.1)
12.2 Pharmacodynamics
Aripiprazole exhibits high affinity for dopamine D2 and D3 (Kis 0.34 and 0.8 nM, respectively), serotonin 5-HT1A and 5-HT2A receptors (Kis 1.7 and 3.4 nM, respectively), moderate affinity for dopamine D4, serotonin 5-HT2C and 5-HT7, alpha1-adrenergic and histamine H1 receptors (Kis of 44 nM, 15 nM, 39 nM, 57 nM, and 61 nM, respectively), and moderate affinity for the serotonin reuptake site (Ki=98 nM). Aripiprazole has no appreciable affinity for cholinergic muscarinic receptors (IC50>1000 nM). Actions at receptors other than D2, 5-HT1A, and 5-HT2A could explain some of the other adverse reactions of aripiprazole (e.g., the orthostatic hypotension observed with aripiprazole may be explained by its antagonist activity at adrenergic alpha1 receptors).
12.3 Pharmacokinetics
Steady-state concentrations for the typical subject were attained by the fourth dose for both sites of administration. Approximate dose-proportional increases in aripiprazole and dehydro-aripiprazole exposure were observed after every four-week ABILIFY MAINTENA injections of 300 mg and 400 mg.
5.5 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that include hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. While all drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.4 Tardive Dyskinesia
A syndrome of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment, itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and, thereby, may possibly mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, ABILIFY MAINTENA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that 1) is known to respond to antipsychotic drugs and 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient treated with ABILIFY MAINTENA drug discontinuation should be considered. However, some patients may require treatment with ABILIFY MAINTENA despite the presence of the syndrome.
1 Indications and Usage
ABILIFY MAINTENA (aripiprazole) is indicated:
- for the treatment of schizophrenia in adults
- for maintenance monotherapy treatment of bipolar I disorder in adults
12.1 Mechanism of Action
The mechanism of action of aripiprazole in the treatment of schizophrenia and bipolar I disorder is unknown.
The efficacy of aripiprazole could be mediated through a combination of partial agonist activity at dopamine D2 and serotonin 5-HT1A receptors and antagonist activity at 5-HT2A receptors.
5 Warnings and Precautions
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities) (5.2)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring (5.3)
- Tardive Dyskinesia: Discontinue if clinically appropriate (5.4)
-
Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain (5.5)
-
–Hyperglycemia and Diabetes Mellitus: Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with and at risk for diabetes (5.5)
-
–Dyslipidemia: Undesirable alterations have been observed in patients treated with atypical antipsychotics (5.5)
-
–Weight Gain: Gain in body weight has been observed; clinical monitoring of weight is recommended (5.5)
-
- Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation (5.6)
- Orthostatic Hypotension: Use with caution in patients with known cardiovascular or cerebrovascular disease (5.7)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell count (WBC)/absolute neutrophil count (ANC). Consider discontinuation if clinically significant decline in WBC/ANC in the absence of other causative factors (5.9)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold (5.10)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery (5.11)
2 Dosage and Administration
- Only to be administered by intramuscular injection in the deltoid or gluteal muscle by a healthcare professional (2.1)
- For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating ABILIFY MAINTENA (2.1)
- Recommended dosage is 400 mg administered monthly as a single injection. Dose can be reduced to 300 mg in patients with adverse reactions (2.2)
- There are two ways to initiate treatment with ABILIFY MAINTENA
- 1-day initiation: Administer two separate intramuscular injections of ABILIFY MAINTENA 400 mg and a single oral dose of aripiprazole 20 mg (2.2)
- 14-day initiation: In conjunction with first ABILIFY MAINTENA 400 mg dose, take 14 consecutive days of concurrent oral aripiprazole (10 mg to 20 mg) or current oral antipsychotic (2.2)
- Dosage adjustments are required for missed doses (2.3)
- Known CYP2D6 poor metabolizers: Recommended starting and maintenance dose is 300 mg administered monthly as a single injection (2.4)
- See full prescribing information for ABILIFY MAINTENA dosage modifications due to drug interactions (2.4).
- ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe (2.6), and 2) Vials (2.7).
3 Dosage Forms and Strengths
For extended-release injectable suspension: 300 mg and 400 mg of lyophilized powder for reconstitution in:
- single-dose, pre-filled, dual chamber syringe
- single-dose vial
The reconstituted extended-release injectable suspension is a uniform, homogeneous suspension that is opaque and milky-white in color.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of oral aripiprazole or ABILIFY MAINTENA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: occurrences of allergic reaction (anaphylactic reaction, angioedema, laryngospasm, pruritus/urticaria, or oropharyngeal spasm), pathological gambling, hiccups, blood glucose fluctuation, drug reaction with eosinophilia and systemic symptoms (DRESS), and fecal incontinence.
8.6 Cyp2d6 Poor Metabolizers
Dosage adjustment is recommended in known CYP2D6 poor metabolizers due to high aripiprazole concentrations. Approximately 8% of Caucasians and 3% to 8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers (PM) [see Dosage and Administration (2.4)].
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.12 Body Temperature Regulation
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing ABILIFY MAINTENA for patients who will be experiencing conditions which may contribute to an elevation in core body temperature, (e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide)
5.3 Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) may occur with administration of antipsychotic drugs, including ABILIFY MAINTENA. Rare cases of NMS occurred during aripiprazole treatment in the worldwide clinical database.
Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to exclude cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system pathology.
The management of NMS should include: 1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy; 2) intensive symptomatic treatment and medical monitoring; and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
2.5 Aripiprazole Formulations and Kits
ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe available in 300 mg or 400 mg strength syringes [see Dosage and Administration (2.6)], and 2) Single-dose vials available in 300 mg or 400 mg strength vials [see Dosage and Administration (2.7)].
The 200 mg and 160 mg dosage adjustments are obtained only by using the 300 mg or 400 mg strength vials.
5.7 Orthostatic Hypotension and Syncope
ABILIFY MAINTENA may cause orthostatic hypotension, perhaps due to its α1-adrenergic receptor antagonism. In the short-term, placebo-controlled trial in adults with schizophrenia, the adverse event of presyncope was reported in 1/167 (0.6%) of patients treated with ABILIFY MAINTENA, while syncope and orthostatic hypotension were each reported in 1/172 (0.6%) of patients treated with placebo. During the stabilization phase of the randomized-withdrawal (maintenance) study in adult patients with schizophrenia, orthostasis-related adverse events were reported in 4/576 (0.7%) of patients treated with ABILIFY MAINTENA, including abnormal orthostatic blood pressure (1/576, 0.2%), postural dizziness (1/576, 0.2%), presyncope (1/576, 0.2%) and orthostatic hypotension (1/576, 0.2%).
In the short-term placebo-controlled trial in adults with schizophrenia, there were no patients in either treatment group with a significant orthostatic change in blood pressure (defined as a decrease in systolic blood pressure ≥20 mmHg accompanied by an increase in heart rate ≥25 bpm when comparing standing to supine values). During the stabilization phase of the randomized-withdrawal (maintenance) study in adult patients with schizophrenia, the incidence of significant orthostatic change in blood pressure was 0.2% (1/575).
2.1 Important Administration Information
For patients who have never taken aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ABILIFY MAINTENA. Due to the half-life of oral aripiprazole (i.e., 75 hours and 94 hours for aripiprazole and dehydro-aripiprazole, respectively), it may take up to 2 weeks to fully assess tolerability.
ABILIFY MAINTENA must be administered by intramuscular injection by a healthcare professional. Do not administer by any other route.
For detailed preparation and administration instructions, [see Dosage and Administration (2.6, 2.7)].
Principal Display Panel 5 Ml Vial Label
NDC 59148-100-01
5mL
Sterile Water
for Injection, USP
For single use only.
Discard unused portion.
Store at 25 °C (77 °F), excursions
permitted between 15 °C and 30 °C
(59 °F to 86 °F) [See USP].
09US12L-1008
Rx only
2.2 Recommended Dosage for Abilify Maintena
The recommended dose of ABILIFY MAINTENA is 400 mg monthly (no sooner than 26 days after the previous injection).
There are two ways to initiate treatment with ABILIFY MAINTENA in patients receiving oral antipsychotics:
1-day initiation:
- Administer two intramuscular injections of ABILIFY MAINTENA 400 mg in two different injection sites (in either the deltoid or gluteal muscle), and one dose of oral aripiprazole 20 mg on the first day of treatment with ABILIFY MAINTENA.
- Do not administer both injections into the same muscle.
14-day initiation:
- When ABILIFY MAINTENA injection is initiated in patients receiving oral aripiprazole administer one intramuscular injection of ABILIFY MAINTENA 400 mg in either the deltoid or gluteal muscle and continue treatment with oral aripiprazole (10 mg to 20 mg) for 14 consecutive days to achieve therapeutic aripiprazole concentrations during initiation of therapy.
- For patients already stable on another oral antipsychotic (and known to tolerate aripiprazole), administer one intramuscular injection of ABILIFY MAINTENA 400 mg in either the deltoid or gluteal muscle and continue treatment with the oral antipsychotic for 14 consecutive days to maintain therapeutic antipsychotic concentrations during initiation of therapy.
If there are adverse reactions with the 400 mg dosage, the dosage may be reduced to 300 mg once monthly.
Principal Display Panel 300 Mg Kit Carton
NDC 59148-018-71
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
300 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena™
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.5 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
Principal Display Panel 300 Mg Vial Label
NDC 59148-018-70
Rx only
300 mg per vial
Abilify Maintena™ (aripiprazole)
for extended release injectable suspension
Single use only. Sterile. Discard any unused portion.
For deltoid or gluteal intramuscular injection only.
Usual Dosage: See package insert.
Manufactured by: Otsuka Pharmaceutical Co., Ltd.
Distributed and Marketed by: Otsuka America Pharmaceutical, Inc.
Marketed by: Lundbeck
Principal Display Panel 400 Mg Kit Carton
NDC 59148-019-71
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
400 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena™
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.9 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
Principal Display Panel 400 Mg Vial Label
NDC 59148-019-70
Rx only
400 mg per vial
Abilify Maintena™ (aripiprazole)
for extended release injectable suspension
Single use only. Sterile. Discard any unused portion.
For deltoid or gluteal intramuscular injection only.
Usual Dosage: See package insert.
Manufactured by: Otsuka Pharmaceutical Co., Ltd.
Distributed and Marketed by: Otsuka America Pharmaceutical, Inc.
Marketed by: Lundbeck
Principal Display Panel 300 Mg Syringe Label
NDC 59148-045-80
300 mg
Abilify Maintena®
(aripiprazole)
for extended release
injectable suspension
Single use only.
Sterile. Discard any
unused portion.
For deltoid or gluteal
intramuscular
injection only.
Usual Dosage:
See package insert.
516628AA
Principal Display Panel 400 Mg Syringe Label
NDC 59148-072-80
400 mg
Abilify Maintena®
(aripiprazole)
for extended release
injectable suspension
Single use only.
Sterile. Discard any
unused portion.
For deltoid or gluteal
intramuscular
injection only.
Usual Dosage:
See package insert.
516629AA
Principal Display Panel 300 Mg Syringe Carton
For deltoid or
gluteal intramuscular
injection only
NDC 59148-045-80
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
300 mg PER PRE-FILLED
DUAL CHAMBER SYRINGE
Single use only.
Keep out of reach
of children.
Each injection must be administered by a healthcare professional only.
Attention: Dispense an enclosed Medication Guide to each patient.
Rx Only
Principal Display Panel 400 Mg Syringe Carton
For deltoid or
gluteal intramuscular
injection only
NDC 59148-072-80
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
400 mg PER PRE-FILLED
DUAL CHAMBER SYRINGE
Single use only.
Keep out of reach
of children.
Each injection must be administered by a healthcare professional only.
Attention: Dispense an enclosed Medication Guide to each patient.
Rx Only
5.9 Leukopenia, Neutropenia, and Agranulocytosis
In clinical trials and post-marketing experience, leukopenia and neutropenia have been reported temporally related to antipsychotic agents, including ABILIFY MAINTENA. Agranulocytosis has also been reported [see Adverse Reactions (6.1)].
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC)/absolute neutrophil count (ANC) and a history of drug-induced leukopenia/neutropenia. In patients with a history of a clinically significant low WBC/ANC or drug-induced leukopenia/neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of ABILIFY MAINTENA at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue ABILIFY MAINTENA in patients with severe neutropenia (absolute neutrophil count <1000/mm3) and follow their WBC counts until recovery.
Principal Display Panel 300 Mg Vial Kit Carton
NDC 59148-232-12
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
300 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.5 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
Principal Display Panel 400 Mg Vial Kit Carton
NDC 59148-245-12
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
400 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.9 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
5.11 Potential for Cognitive and Motor Impairment
ABILIFY MAINTENA, like other antipsychotics, may impair judgment, thinking, or motor skills. Patients should be cautioned about performing activities that require mental alertness such as operating hazardous machinery, or operating a motor vehicle, until they are reasonably certain that therapy with ABILIFY MAINTENA does not affect them adversely.
14.2 Bipolar I Disorder – Maintenance Monotherapy
The efficacy of ABILIFY MAINTENA for the maintenance treatment of bipolar I disorder was established in a 52-week, double-blind, placebo-controlled, randomized withdrawal trial in adult patients who were experiencing a manic episode at trial entry, met DSM-IV-TR criteria for bipolar I disorder, and had a history of at least one previous manic or mixed episode with manic symptoms of sufficient severity to require one of the following interventions: hospitalization and/or treatment with a mood stabilizer, and/or treatment with an antipsychotic agent.
Clinical ratings during this trial included:
- Young Mania Rating Scale (YMRS)-an 11-item, clinician-rated scale used to assess the degree of manic symptomatology, in a range with 0 representing no symptoms, and 60 representing worst symptoms.
- Montgomery-Asberg Depression Rating Scale (MADRS) – a 10-item clinician-related scale used to assess the degree of depressive symptomatology, with 1 representing no symptoms, and 60 representing worst symptoms.
- Clinical Global Impression Bipolar Version Severity of Illness (CGI-BP-S) a scale of 1 (normal, not at all ill) to 7 (very severely ill patient) based on the patient's severity of illness mania, depression, and overall bipolar illness.
This trial included:
- A 4 to 6 week, open-label, oral conversion phase for patients on treatments for bipolar I disorder other than aripiprazole. A total of 466 patients entered this phase.
- A 2 to 8 week, open-label, oral aripiprazole stabilization phase (target dose of 15 mg to 30 mg once daily). A total of 632 patients entered this phase. Patients were 18 to 65 years old (mean 40.7 years) and 60% were female. The mean (range) baseline scores were: YMRS total, 16.9 MADRS total, 5.7, and CGI-BP-S overall, 3.4 (mildly to moderately ill). Prior to the next phase, stabilization was required. Stabilization was defined as having all of the following at one bi-weekly visit: Outpatient status, YMRS total score ≤12, MADRS total score ≤12, no active suicidality; with active suicidality defined as a score of 4 or more on the MADRS item 10 OR an answer of "yes" on question 4 or 5 on the Columbia Suicide Severity Rating Scale (C-SSRS).
- A minimum 12-week, uncontrolled, single-blind ABILIFY MAINTENA stabilization phase (treatment with 400 mg of ABILIFY MAINTENA given every 4 weeks in conjunction with oral aripiprazole [10 mg to 20 mg/day] for the first 2 weeks). The dose of ABILIFY MAINTENA was allowed to be decreased to 300 mg due to adverse reactions. A total of 425 patients entered this phase. The mean (range) baseline scores were: YMRS total, 5.8, MADRS total 3.7, and CGI-BP-S overall, 2.1 (minimally ill). Prior to the next phase, stabilization was required (see above for the definition of stabilization) for 8 consecutive weeks starting at week 6.
- A double-blind, placebo-controlled, randomized-withdrawal phase to observe for recurrence to a mood episode (defined below) for up to 52 weeks. A total of 266 patients were randomized 1:1 to the same dose of ABILIFY MAINTENA they were receiving at the end of the stabilization phase, (400 mg or 300 mg administered once every 4 weeks) or placebo. The mean (range) baseline scores were: YMRS total, 2.8 (0 to 12), MADRS total, 2.7 (0 to 12), and CGI-S overall, 1.7 (minimally ill). The dose could be decreased to 300 mg for tolerability and returned once to 400 mg.
The primary efficacy endpoint was time from randomization to recurrence of any mood episode. Recurrence was defined as the first occurrence of one or more of the following criteria:
- Hospitalization for any mood episode OR
- Any of the following:
- YMRS total score ≥15 OR
- MADRS total score ≥15 OR
- Clinical Global Impression - Bipolar Version-Severity (CGI-BP-S) score >4 (overall score) OR
- Serious adverse event (SAE) of worsening disease (bipolar I disorder) OR
- Discontinuation due to lack of efficacy or discontinuation due to an adverse event (AE) of worsening disease OR
- Clinical worsening with the need for addition of a mood stabilizer, antidepressant treatment, antipsychotic medication, and/or increase greater than the allowed benzodiazepine doses for treatment of symptoms of an underlying mood disorder OR
- Active suicidality, which is defined as a score of 4 or more on the MADRS item 10 OR an answer of "yes" on question 4 or 5 on the C-SSRS
Analysis demonstrated a statistically significantly longer time to recurrence of any mood episode in subjects randomized to the ABILIFY MAINTENA group than compared to placebo-treated subjects. The Kaplan-Meier curves of the time of recurrence to any mood episode during the double-blind treatment phase for ABILIFY MAINTENA and placebo groups are shown in Figure 26.
|
Figure 26: Kaplan-Meier Estimation of Cumulative Recurrence Rate for Any Mood Episode This figure is based on a total of 103 recurrence events.
|
Analysis by type of mood recurrence demonstrated a statistically significantly longer time to recurrence for both manic and mixed mood episodes in subjects treated with ABILIFY MAINTENA compared to those treated with placebo. There was no substantial difference between treatment groups in delaying time to recurrence of depressive mood episodes.
An examination of subgroups did not reveal any clear evidence of differential responsiveness on the basis of age, sex, or race.
5.6 Pathological Gambling and Other Compulsive Behaviors
Post-marketing case reports suggest that patients can experience intense urges, particularly for gambling, and the inability to control these urges while taking aripiprazole. Other compulsive urges, reported less frequently, include: sexual urges, shopping, eating or binge eating, and other impulsive or compulsive behaviors. Because patients may not recognize these behaviors as abnormal, it is important for prescribers to ask patients or their caregivers specifically about the development of new or intense gambling urges, compulsive sexual urges, compulsive shopping, binge or compulsive eating, or other urges while being treated with aripiprazole. It should be noted that impulse-control symptoms can be associated with the underlying disorder. In some cases, although not all, urges were reported to have stopped when the dose was reduced, or the medication was discontinued. Compulsive behaviors may result in harm to the patient and others if not recognized. Consider dose reduction or stopping the medication if a patient develops such urges.
2.4 Dosage Modifications for Cytochrome P450 Considerations
Refer to Table 1 and Table 2 for dosage modifications for patients who are CYP2D6 poor metabolizers and/or in patients taking concomitant CYP3A4 inhibitors or CYP2D6 inhibitors for greater than 14 days.
If the CYP3A4 inhibitor or CYP2D6 inhibitor is withdrawn, the ABILIFY MAINTENA dosage may need to be increased to the previous dose [see Dosage and Administration (2.2)].
Avoid the concomitant use of CYP3A4 inducers with ABILIFY MAINTENA for greater than 14 days because the blood levels of aripiprazole are decreased and may be below the effective levels.
Dosage modifications are not recommended for patients with concomitant use of CYP3A4 inhibitors, CYP2D6 inhibitors or CYP3A4 inducers for less than 14 days.
| Factors | Adjusted Dose No change for oral dosage required during initiation.
|
|---|---|
| CYP2D6 Poor Metabolizers | |
| Known CYP2D6 Poor Metabolizers | 300 mg |
| Known CYP2D6 Poor Metabolizers taking concomitant CYP3A4 inhibitors | Avoid use |
| Concomitant use with CYP Inhibitors and/or Inducers | |
| Strong CYP2D6 or CYP3A4 inhibitors | 300 mg |
| CYP2D6 and CYP3A4 inhibitors | Avoid use |
| CYP3A4 inducers | Avoid use |
| Factors | Adjusted Dose No change for oral dosage required during initiation.
|
|---|---|
| CYP2D6 Poor Metabolizers | |
| Known CYP2D6 Poor Metabolizers | 300 mg |
| Known CYP2D6 Poor Metabolizers taking concomitant CYP3A4 inhibitors | 200 mg 200 mg and 160 mg dosage adjustment is obtained only by using the 300 mg or 400 mg strength vials.
|
| Patients Taking 400 mg of ABILIFY MAINTENA | |
| Strong CYP2D6 or CYP3A4 inhibitors | 300 mg |
| CYP2D6 and CYP3A4 inhibitors | 200 mg |
| CYP3A4 inducers | Avoid use |
| Patients Taking 300 mg of ABILIFY MAINTENA | |
| Strong CYP2D6 or CYP3A4 inhibitors | 200 mg |
| CYP2D6 and CYP3A4 inhibitors | 160 mg |
| CYP3A4 inducers | Avoid use |
ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe [see Dosage and Administration (2.6)], and 2) Vials [see Dosage and Administration (2.7)].
7.1 Drugs Having Clinically Important Interactions With Abilify Maintena
| Concomitant Drug Name or Drug Class | Clinical Rationale | Clinical Recommendation |
|---|---|---|
| Strong CYP3A4 Inhibitors (e.g., ketoconazole) or strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine) | The concomitant use of oral aripiprazole with strong CYP3A4 or CYP2D6 inhibitors increased the exposure of aripiprazole [see Clinical Pharmacology (12.3)]. | With concomitant use of ABILIFY MAINTENA with a strong CYP3A4 inhibitor or CYP2D6 inhibitor for more than 14 days, reduce the ABILIFY MAINTENA dosage [see Dosage and Administration (2.4)]. |
| Strong CYP3A4 Inducers (e.g., carbamazepine) | The concomitant use of oral aripiprazole and carbamazepine decreased the exposure of aripiprazole [see Clinical Pharmacology (12.3)]. | Avoid use of ABILIFY MAINTENA in combination with carbamazepine and other inducers of CYP3A4 for greater than 14 days [see Dosage and Administration (2.4)]. |
| Antihypertensive Drugs | Due to its alpha-adrenergic antagonism, aripiprazole has the potential to enhance the effect of certain antihypertensive agents. | Monitor blood pressure and adjust dose accordingly [see Warnings and Precautions (5.7)]. |
| Benzodiazepines (e.g., lorazepam) | The intensity of sedation was greater with the combination of oral aripiprazole and lorazepam as compared to that observed with aripiprazole alone. The orthostatic hypotension observed was greater with the combination as compared to that observed with lorazepam alone [see Warnings and Precautions (5.7)]. | Monitor sedation and blood pressure. Adjust dose accordingly. |
7.2 Drugs Having No Clinically Important Interactions With Abilify Maintena
Based on pharmacokinetic studies with oral aripiprazole, no dosage adjustment of ABILIFY MAINTENA is required when administered concomitantly with famotidine, valproate, lithium, lorazepam [see Clinical Pharmacology (12.3)].
In addition, no dosage adjustment is necessary for substrates of CYP2D6 (e.g., dextromethorphan, fluoxetine, paroxetine, or venlafaxine), CYP2C9 (e.g., warfarin), CYP2C19 (e.g., omeprazole, warfarin), or CYP3A4 (e.g., dextromethorphan) when coadministered with ABILIFY MAINTENA. Additionally, no dosage adjustment is necessary for valproate, lithium, lamotrigine, lorazepam, or sertraline when coadministered with ABILIFY MAINTENA. [See Clinical Pharmacology (12.3)].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group.
Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis.
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
5.2 Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients With Dementia Related Psychosis
In placebo-controlled clinical studies (two flexible-dose and one fixed-dose study) of dementia-related psychosis, there was an increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack), including fatalities, in oral aripiprazole-treated patients (mean age: 84 years; range: 78 to 88 years). In the fixed-dose study, there was a statistically significant dose response relationship for cerebrovascular adverse reactions in patients treated with oral aripiprazole. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis.
Structured Label Content
Section 42229-5 (42229-5)
Preparation Prior to Reconstitution
For deep intramuscular deltoid or gluteal injection by healthcare professionals only. Do not administer by any other route. Inject full syringe contents immediately following reconstitution. Administer once monthly.
Lay out and confirm that components listed below are provided in the kit:
- One ABILIFY MAINTENA (aripiprazole) pre-filled dual chamber syringe (400 mg or 300 mg as appropriate) for extended-release injectable suspension containing lyophilized powder and Sterile Water for Injection
- One 23-gauge, 1-inch (25 mm) hypodermic safety needle with needle protection device for deltoid administration in non-obese patients
- One 22-gauge, 1.5-inch (38 mm) hypodermic safety needle with needle protection device for gluteal administration in non-obese patients or deltoid administration in obese patients
- One 21-gauge, 2-inch (51 mm) hypodermic safety needle with needle protection device for gluteal administration in obese patients
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 03/2025 | ||
|
MEDICATION GUIDE
ABILIFY MAINTENA® (a-BIL-i-fy main-TEN-a) (aripiprazole) for extended-release injectable suspension, for intramuscular use |
|||
|
What is the most important information I should know about ABILIFY MAINTENA?
ABILIFY MAINTENA may cause serious side effects, including:
|
|||
|
What is ABILIFY MAINTENA?
ABILIFY MAINTENA is a prescription medicine given by injection by a healthcare provider:
|
|||
|
Who should not receive ABILIFY MAINTENA? Do not receive ABILIFY MAINTENA if you are allergic to aripiprazole or any of the ingredients in ABILIFY MAINTENA. See the end of this Medication Guide for a complete list of ingredients in ABILIFY MAINTENA. |
|||
Before receiving ABILIFY MAINTENA, tell your healthcare provider about all of your medical conditions, including if you:
ABILIFY MAINTENA and other medicines may affect each other causing possible serious side effects. ABILIFY MAINTENA may affect the way other medicines work, and other medicines may affect how ABILIFY MAINTENA works. Your healthcare provider can tell you if it is safe to receive ABILIFY MAINTENA with your other medicines. Do not start or stop any medicines during treatment with ABILIFY MAINTENA without talking to your healthcare provider first. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I receive ABILIFY MAINTENA?
|
|||
What should I avoid while receiving ABILIFY MAINTENA?
|
|||
|
What are the possible side effects of ABILIFY MAINTENA? ABILIFY MAINTENA may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
|
|||
These are not all the possible side effects of ABILIFY MAINTENA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of ABILIFY MAINTENA.
If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about ABILIFY MAINTENA that is written for healthcare professionals. |
|||
|
What are the ingredients in ABILIFY MAINTENA? Active ingredient: aripiprazole monohydrate Inactive ingredients: carboxymethylcellulose sodium, mannitol, sodium phosphate monobasic monohydrate and sodium hydroxide ABILIFY MAINTENA is a trademark of Otsuka Pharmaceutical Co., Ltd. ©2025, Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan For more information, go to www.ABILIFYMAINTENA.com or call 1-800-441-6763. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
5.8 Falls
Antipsychotics, including ABILIFY MAINTENA, may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
5.10 Seizures
As with other antipsychotic drugs, use ABILIFY MAINTENA cautiously in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in a population of 65 years or older.
11 Description (11 DESCRIPTION)
Aripiprazole is an atypical antipsychotic which is present in ABILIFY MAINTENA as its monohydrate polymorphic form. Aripiprazole monohydrate is 7-[4-[4-(2,3-dichlorophenyl)-1-piperazinyl] butoxy]-3,4 dihydrocarbostyril monohydrate. The empirical formula is C23H27Cl2N3O2∙H2O and its molecular weight is 466.40. The chemical structure is:
ABILIFY MAINTENA (aripiprazole) is an extended-release injectable suspension available in 400 mg or 300 mg strength pre-filled dual chamber syringes and 400 mg or 300 mg strength vials. The labeled strengths are calculated based on the anhydrous form (aripiprazole). Inactive ingredients (per administered dose) for 400 mg and 300 mg strength products, respectively, include carboxymethyl cellulose sodium (16.64 mg and 12.48 mg), mannitol (83.2 mg and 62.4 mg), sodium phosphate monobasic monohydrate (1.48 mg and 1.11 mg) and sodium hydroxide (pH adjuster).
5.13 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use, including ABILIFY MAINTENA. ABILIFY MAINTENA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia [see Warnings and Precautions (5.1)].
2.3 Missed Doses
If the second or third doses are missed:
- If more than 4 weeks and less than 5 weeks have elapsed since the last injection, administer the injection as soon as possible.
- If more than 5 weeks have elapsed since the last injection, restart treatment with either 1-day initiation or 14-day initiation with ABILIFY MAINTENA [see Dosage and Administration (2.2)].
If the fourth or subsequent doses are missed:
- If more than 4 weeks and less than 6 weeks have elapsed since the last injection, administer the injection as soon as possible.
- If more than 6 weeks have elapsed since the last injection, restart treatment with either 1-day initiation or 14-day initiation with ABILIFY MAINTENA [see Dosage and Administration (2.2)].
8.4 Pediatric Use
ABILIFY MAINTENA has not been studied in children 18 years of age or younger. However, juvenile animal studies have been conducted in rats and dogs.
8.5 Geriatric Use
Clinical studies of oral aripiprazole did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. Other reported clinical experience and pharmacokinetic data have not identified differences in responses between the elderly and younger patients [see Clinical Pharmacology (12.3)]. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
In single-dose and multiple-dose pharmacokinetic studies, there was no detectable age effect in the population pharmacokinetic analysis of oral aripiprazole in schizophrenia patients [see Clinical Pharmacology (12.3)]. No dosage adjustments are recommended based on age alone. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis [see also Boxed Warning and Warnings and Precautions (5.1)].
14.1 Schizophrenia
The efficacy of ABILIFY MAINTENA for treatment of schizophrenia was established in:
- One short-term (12-week), randomized, double-blind, placebo-controlled trial in acutely relapsed adults, Protocol 31-12-291 (Study 1)
- One longer-term, double-blind, placebo-controlled, randomized-withdrawal (maintenance) trial in adults, Protocol 31-07-246 (Study 2).
4 Contraindications (4 CONTRAINDICATIONS)
ABILIFY MAINTENA is contraindicated in patients with a known hypersensitivity to aripiprazole. Hypersensitivity reactions ranging from pruritus/urticaria to anaphylaxis have been reported in patients receiving aripiprazole [see Adverse Reactions (6.1 and 6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia - Related Psychosis Use [see Boxed Warning and Warnings and Precautions (5.1)]
- Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning and Warnings and Precautions (5.2)]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.3)]
- Tardive Dyskinesia [see Warnings and Precautions (5.4)]
- Metabolic Changes [see Warnings and Precautions (5.5)]
- Pathological Gambling and Other Compulsive Behaviors [see Warnings and Precautions (5.6)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.7)]
- Falls [see Warnings and Precautions (5.8)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.9)]
- Seizures [see Warnings and Precautions (5.10)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.11)]
- Body Temperature Regulation [see Warnings and Precautions (5.12)]
- Dysphagia [see Warnings and Precautions (5.13)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- CYP2D6 inhibitors and CYP3A4 Inhibitors: See full prescribing information for ABILIFY MAINTENA dosage modifications when used concomitantly with CYP2D6 inhibitors and/or CYP3A4 inhibitors for greater than 14 days (7.1)
- CYP3A4 Inducers: Avoid concomitant use for greater than 14 days (7.1)
- See full prescribing information for additional clinically significant drug interactions (7.1)
12.2 Pharmacodynamics
Aripiprazole exhibits high affinity for dopamine D2 and D3 (Kis 0.34 and 0.8 nM, respectively), serotonin 5-HT1A and 5-HT2A receptors (Kis 1.7 and 3.4 nM, respectively), moderate affinity for dopamine D4, serotonin 5-HT2C and 5-HT7, alpha1-adrenergic and histamine H1 receptors (Kis of 44 nM, 15 nM, 39 nM, 57 nM, and 61 nM, respectively), and moderate affinity for the serotonin reuptake site (Ki=98 nM). Aripiprazole has no appreciable affinity for cholinergic muscarinic receptors (IC50>1000 nM). Actions at receptors other than D2, 5-HT1A, and 5-HT2A could explain some of the other adverse reactions of aripiprazole (e.g., the orthostatic hypotension observed with aripiprazole may be explained by its antagonist activity at adrenergic alpha1 receptors).
12.3 Pharmacokinetics
Steady-state concentrations for the typical subject were attained by the fourth dose for both sites of administration. Approximate dose-proportional increases in aripiprazole and dehydro-aripiprazole exposure were observed after every four-week ABILIFY MAINTENA injections of 300 mg and 400 mg.
5.5 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that include hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. While all drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.4 Tardive Dyskinesia
A syndrome of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment, itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and, thereby, may possibly mask the underlying process. The effect of symptomatic suppression on the long-term course of the syndrome is unknown.
Given these considerations, ABILIFY MAINTENA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that 1) is known to respond to antipsychotic drugs and 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient treated with ABILIFY MAINTENA drug discontinuation should be considered. However, some patients may require treatment with ABILIFY MAINTENA despite the presence of the syndrome.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ABILIFY MAINTENA (aripiprazole) is indicated:
- for the treatment of schizophrenia in adults
- for maintenance monotherapy treatment of bipolar I disorder in adults
12.1 Mechanism of Action
The mechanism of action of aripiprazole in the treatment of schizophrenia and bipolar I disorder is unknown.
The efficacy of aripiprazole could be mediated through a combination of partial agonist activity at dopamine D2 and serotonin 5-HT1A receptors and antagonist activity at 5-HT2A receptors.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack, including fatalities) (5.2)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring (5.3)
- Tardive Dyskinesia: Discontinue if clinically appropriate (5.4)
-
Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain (5.5)
-
–Hyperglycemia and Diabetes Mellitus: Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with and at risk for diabetes (5.5)
-
–Dyslipidemia: Undesirable alterations have been observed in patients treated with atypical antipsychotics (5.5)
-
–Weight Gain: Gain in body weight has been observed; clinical monitoring of weight is recommended (5.5)
-
- Pathological Gambling and Other Compulsive Behaviors: Consider dose reduction or discontinuation (5.6)
- Orthostatic Hypotension: Use with caution in patients with known cardiovascular or cerebrovascular disease (5.7)
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts in patients with a history of a clinically significant low white blood cell count (WBC)/absolute neutrophil count (ANC). Consider discontinuation if clinically significant decline in WBC/ANC in the absence of other causative factors (5.9)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that lower the seizure threshold (5.10)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery (5.11)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Only to be administered by intramuscular injection in the deltoid or gluteal muscle by a healthcare professional (2.1)
- For patients naïve to aripiprazole, establish tolerability with oral aripiprazole prior to initiating ABILIFY MAINTENA (2.1)
- Recommended dosage is 400 mg administered monthly as a single injection. Dose can be reduced to 300 mg in patients with adverse reactions (2.2)
- There are two ways to initiate treatment with ABILIFY MAINTENA
- 1-day initiation: Administer two separate intramuscular injections of ABILIFY MAINTENA 400 mg and a single oral dose of aripiprazole 20 mg (2.2)
- 14-day initiation: In conjunction with first ABILIFY MAINTENA 400 mg dose, take 14 consecutive days of concurrent oral aripiprazole (10 mg to 20 mg) or current oral antipsychotic (2.2)
- Dosage adjustments are required for missed doses (2.3)
- Known CYP2D6 poor metabolizers: Recommended starting and maintenance dose is 300 mg administered monthly as a single injection (2.4)
- See full prescribing information for ABILIFY MAINTENA dosage modifications due to drug interactions (2.4).
- ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe (2.6), and 2) Vials (2.7).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For extended-release injectable suspension: 300 mg and 400 mg of lyophilized powder for reconstitution in:
- single-dose, pre-filled, dual chamber syringe
- single-dose vial
The reconstituted extended-release injectable suspension is a uniform, homogeneous suspension that is opaque and milky-white in color.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of oral aripiprazole or ABILIFY MAINTENA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: occurrences of allergic reaction (anaphylactic reaction, angioedema, laryngospasm, pruritus/urticaria, or oropharyngeal spasm), pathological gambling, hiccups, blood glucose fluctuation, drug reaction with eosinophilia and systemic symptoms (DRESS), and fecal incontinence.
8.6 Cyp2d6 Poor Metabolizers (8.6 CYP2D6 Poor Metabolizers)
Dosage adjustment is recommended in known CYP2D6 poor metabolizers due to high aripiprazole concentrations. Approximately 8% of Caucasians and 3% to 8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers (PM) [see Dosage and Administration (2.4)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.12 Body Temperature Regulation
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing ABILIFY MAINTENA for patients who will be experiencing conditions which may contribute to an elevation in core body temperature, (e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide)
5.3 Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) may occur with administration of antipsychotic drugs, including ABILIFY MAINTENA. Rare cases of NMS occurred during aripiprazole treatment in the worldwide clinical database.
Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to exclude cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system pathology.
The management of NMS should include: 1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy; 2) intensive symptomatic treatment and medical monitoring; and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
2.5 Aripiprazole Formulations and Kits
ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe available in 300 mg or 400 mg strength syringes [see Dosage and Administration (2.6)], and 2) Single-dose vials available in 300 mg or 400 mg strength vials [see Dosage and Administration (2.7)].
The 200 mg and 160 mg dosage adjustments are obtained only by using the 300 mg or 400 mg strength vials.
5.7 Orthostatic Hypotension and Syncope
ABILIFY MAINTENA may cause orthostatic hypotension, perhaps due to its α1-adrenergic receptor antagonism. In the short-term, placebo-controlled trial in adults with schizophrenia, the adverse event of presyncope was reported in 1/167 (0.6%) of patients treated with ABILIFY MAINTENA, while syncope and orthostatic hypotension were each reported in 1/172 (0.6%) of patients treated with placebo. During the stabilization phase of the randomized-withdrawal (maintenance) study in adult patients with schizophrenia, orthostasis-related adverse events were reported in 4/576 (0.7%) of patients treated with ABILIFY MAINTENA, including abnormal orthostatic blood pressure (1/576, 0.2%), postural dizziness (1/576, 0.2%), presyncope (1/576, 0.2%) and orthostatic hypotension (1/576, 0.2%).
In the short-term placebo-controlled trial in adults with schizophrenia, there were no patients in either treatment group with a significant orthostatic change in blood pressure (defined as a decrease in systolic blood pressure ≥20 mmHg accompanied by an increase in heart rate ≥25 bpm when comparing standing to supine values). During the stabilization phase of the randomized-withdrawal (maintenance) study in adult patients with schizophrenia, the incidence of significant orthostatic change in blood pressure was 0.2% (1/575).
2.1 Important Administration Information
For patients who have never taken aripiprazole, establish tolerability with oral aripiprazole prior to initiating treatment with ABILIFY MAINTENA. Due to the half-life of oral aripiprazole (i.e., 75 hours and 94 hours for aripiprazole and dehydro-aripiprazole, respectively), it may take up to 2 weeks to fully assess tolerability.
ABILIFY MAINTENA must be administered by intramuscular injection by a healthcare professional. Do not administer by any other route.
For detailed preparation and administration instructions, [see Dosage and Administration (2.6, 2.7)].
Principal Display Panel 5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 5 mL Vial Label)
NDC 59148-100-01
5mL
Sterile Water
for Injection, USP
For single use only.
Discard unused portion.
Store at 25 °C (77 °F), excursions
permitted between 15 °C and 30 °C
(59 °F to 86 °F) [See USP].
09US12L-1008
Rx only
2.2 Recommended Dosage for Abilify Maintena (2.2 Recommended Dosage for ABILIFY MAINTENA)
The recommended dose of ABILIFY MAINTENA is 400 mg monthly (no sooner than 26 days after the previous injection).
There are two ways to initiate treatment with ABILIFY MAINTENA in patients receiving oral antipsychotics:
1-day initiation:
- Administer two intramuscular injections of ABILIFY MAINTENA 400 mg in two different injection sites (in either the deltoid or gluteal muscle), and one dose of oral aripiprazole 20 mg on the first day of treatment with ABILIFY MAINTENA.
- Do not administer both injections into the same muscle.
14-day initiation:
- When ABILIFY MAINTENA injection is initiated in patients receiving oral aripiprazole administer one intramuscular injection of ABILIFY MAINTENA 400 mg in either the deltoid or gluteal muscle and continue treatment with oral aripiprazole (10 mg to 20 mg) for 14 consecutive days to achieve therapeutic aripiprazole concentrations during initiation of therapy.
- For patients already stable on another oral antipsychotic (and known to tolerate aripiprazole), administer one intramuscular injection of ABILIFY MAINTENA 400 mg in either the deltoid or gluteal muscle and continue treatment with the oral antipsychotic for 14 consecutive days to maintain therapeutic antipsychotic concentrations during initiation of therapy.
If there are adverse reactions with the 400 mg dosage, the dosage may be reduced to 300 mg once monthly.
Principal Display Panel 300 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 300 mg Kit Carton)
NDC 59148-018-71
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
300 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena™
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.5 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
Principal Display Panel 300 Mg Vial Label (PRINCIPAL DISPLAY PANEL - 300 mg Vial Label)
NDC 59148-018-70
Rx only
300 mg per vial
Abilify Maintena™ (aripiprazole)
for extended release injectable suspension
Single use only. Sterile. Discard any unused portion.
For deltoid or gluteal intramuscular injection only.
Usual Dosage: See package insert.
Manufactured by: Otsuka Pharmaceutical Co., Ltd.
Distributed and Marketed by: Otsuka America Pharmaceutical, Inc.
Marketed by: Lundbeck
Principal Display Panel 400 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 400 mg Kit Carton)
NDC 59148-019-71
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
400 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena™
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.9 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
Principal Display Panel 400 Mg Vial Label (PRINCIPAL DISPLAY PANEL - 400 mg Vial Label)
NDC 59148-019-70
Rx only
400 mg per vial
Abilify Maintena™ (aripiprazole)
for extended release injectable suspension
Single use only. Sterile. Discard any unused portion.
For deltoid or gluteal intramuscular injection only.
Usual Dosage: See package insert.
Manufactured by: Otsuka Pharmaceutical Co., Ltd.
Distributed and Marketed by: Otsuka America Pharmaceutical, Inc.
Marketed by: Lundbeck
Principal Display Panel 300 Mg Syringe Label (PRINCIPAL DISPLAY PANEL - 300 mg Syringe Label)
NDC 59148-045-80
300 mg
Abilify Maintena®
(aripiprazole)
for extended release
injectable suspension
Single use only.
Sterile. Discard any
unused portion.
For deltoid or gluteal
intramuscular
injection only.
Usual Dosage:
See package insert.
516628AA
Principal Display Panel 400 Mg Syringe Label (PRINCIPAL DISPLAY PANEL - 400 mg Syringe Label)
NDC 59148-072-80
400 mg
Abilify Maintena®
(aripiprazole)
for extended release
injectable suspension
Single use only.
Sterile. Discard any
unused portion.
For deltoid or gluteal
intramuscular
injection only.
Usual Dosage:
See package insert.
516629AA
Principal Display Panel 300 Mg Syringe Carton (PRINCIPAL DISPLAY PANEL - 300 mg Syringe Carton)
For deltoid or
gluteal intramuscular
injection only
NDC 59148-045-80
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
300 mg PER PRE-FILLED
DUAL CHAMBER SYRINGE
Single use only.
Keep out of reach
of children.
Each injection must be administered by a healthcare professional only.
Attention: Dispense an enclosed Medication Guide to each patient.
Rx Only
Principal Display Panel 400 Mg Syringe Carton (PRINCIPAL DISPLAY PANEL - 400 mg Syringe Carton)
For deltoid or
gluteal intramuscular
injection only
NDC 59148-072-80
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
400 mg PER PRE-FILLED
DUAL CHAMBER SYRINGE
Single use only.
Keep out of reach
of children.
Each injection must be administered by a healthcare professional only.
Attention: Dispense an enclosed Medication Guide to each patient.
Rx Only
5.9 Leukopenia, Neutropenia, and Agranulocytosis
In clinical trials and post-marketing experience, leukopenia and neutropenia have been reported temporally related to antipsychotic agents, including ABILIFY MAINTENA. Agranulocytosis has also been reported [see Adverse Reactions (6.1)].
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC)/absolute neutrophil count (ANC) and a history of drug-induced leukopenia/neutropenia. In patients with a history of a clinically significant low WBC/ANC or drug-induced leukopenia/neutropenia, perform a complete blood count (CBC) frequently during the first few months of therapy. In such patients, consider discontinuation of ABILIFY MAINTENA at the first sign of a clinically significant decline in WBC in the absence of other causative factors.
Monitor patients with clinically significant neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur. Discontinue ABILIFY MAINTENA in patients with severe neutropenia (absolute neutrophil count <1000/mm3) and follow their WBC counts until recovery.
Principal Display Panel 300 Mg Vial Kit Carton (PRINCIPAL DISPLAY PANEL - 300 mg Vial Kit Carton)
NDC 59148-232-12
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
300 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.5 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
Principal Display Panel 400 Mg Vial Kit Carton (PRINCIPAL DISPLAY PANEL - 400 mg Vial Kit Carton)
NDC 59148-245-12
Rx only
Attention: Dispense an enclosed Medication Guide to each patient.
400 mg
per vial
Single use only. Discard Unused Portion.
For deltoid or gluteal
intramuscular injection only.
Abilify Maintena®
(aripiprazole) for extended release injectable suspension
Keep out of reach of children. Each injection must be administered by a healthcare professional only.
To reconstitute: Add 1.9 mL of Sterile Water for Injection, USP (supplied in kit) to make injectable suspension containing 200 mg/mL.
Excess water will be left in the vial; discard vial with the unused portion.
5.11 Potential for Cognitive and Motor Impairment
ABILIFY MAINTENA, like other antipsychotics, may impair judgment, thinking, or motor skills. Patients should be cautioned about performing activities that require mental alertness such as operating hazardous machinery, or operating a motor vehicle, until they are reasonably certain that therapy with ABILIFY MAINTENA does not affect them adversely.
14.2 Bipolar I Disorder – Maintenance Monotherapy
The efficacy of ABILIFY MAINTENA for the maintenance treatment of bipolar I disorder was established in a 52-week, double-blind, placebo-controlled, randomized withdrawal trial in adult patients who were experiencing a manic episode at trial entry, met DSM-IV-TR criteria for bipolar I disorder, and had a history of at least one previous manic or mixed episode with manic symptoms of sufficient severity to require one of the following interventions: hospitalization and/or treatment with a mood stabilizer, and/or treatment with an antipsychotic agent.
Clinical ratings during this trial included:
- Young Mania Rating Scale (YMRS)-an 11-item, clinician-rated scale used to assess the degree of manic symptomatology, in a range with 0 representing no symptoms, and 60 representing worst symptoms.
- Montgomery-Asberg Depression Rating Scale (MADRS) – a 10-item clinician-related scale used to assess the degree of depressive symptomatology, with 1 representing no symptoms, and 60 representing worst symptoms.
- Clinical Global Impression Bipolar Version Severity of Illness (CGI-BP-S) a scale of 1 (normal, not at all ill) to 7 (very severely ill patient) based on the patient's severity of illness mania, depression, and overall bipolar illness.
This trial included:
- A 4 to 6 week, open-label, oral conversion phase for patients on treatments for bipolar I disorder other than aripiprazole. A total of 466 patients entered this phase.
- A 2 to 8 week, open-label, oral aripiprazole stabilization phase (target dose of 15 mg to 30 mg once daily). A total of 632 patients entered this phase. Patients were 18 to 65 years old (mean 40.7 years) and 60% were female. The mean (range) baseline scores were: YMRS total, 16.9 MADRS total, 5.7, and CGI-BP-S overall, 3.4 (mildly to moderately ill). Prior to the next phase, stabilization was required. Stabilization was defined as having all of the following at one bi-weekly visit: Outpatient status, YMRS total score ≤12, MADRS total score ≤12, no active suicidality; with active suicidality defined as a score of 4 or more on the MADRS item 10 OR an answer of "yes" on question 4 or 5 on the Columbia Suicide Severity Rating Scale (C-SSRS).
- A minimum 12-week, uncontrolled, single-blind ABILIFY MAINTENA stabilization phase (treatment with 400 mg of ABILIFY MAINTENA given every 4 weeks in conjunction with oral aripiprazole [10 mg to 20 mg/day] for the first 2 weeks). The dose of ABILIFY MAINTENA was allowed to be decreased to 300 mg due to adverse reactions. A total of 425 patients entered this phase. The mean (range) baseline scores were: YMRS total, 5.8, MADRS total 3.7, and CGI-BP-S overall, 2.1 (minimally ill). Prior to the next phase, stabilization was required (see above for the definition of stabilization) for 8 consecutive weeks starting at week 6.
- A double-blind, placebo-controlled, randomized-withdrawal phase to observe for recurrence to a mood episode (defined below) for up to 52 weeks. A total of 266 patients were randomized 1:1 to the same dose of ABILIFY MAINTENA they were receiving at the end of the stabilization phase, (400 mg or 300 mg administered once every 4 weeks) or placebo. The mean (range) baseline scores were: YMRS total, 2.8 (0 to 12), MADRS total, 2.7 (0 to 12), and CGI-S overall, 1.7 (minimally ill). The dose could be decreased to 300 mg for tolerability and returned once to 400 mg.
The primary efficacy endpoint was time from randomization to recurrence of any mood episode. Recurrence was defined as the first occurrence of one or more of the following criteria:
- Hospitalization for any mood episode OR
- Any of the following:
- YMRS total score ≥15 OR
- MADRS total score ≥15 OR
- Clinical Global Impression - Bipolar Version-Severity (CGI-BP-S) score >4 (overall score) OR
- Serious adverse event (SAE) of worsening disease (bipolar I disorder) OR
- Discontinuation due to lack of efficacy or discontinuation due to an adverse event (AE) of worsening disease OR
- Clinical worsening with the need for addition of a mood stabilizer, antidepressant treatment, antipsychotic medication, and/or increase greater than the allowed benzodiazepine doses for treatment of symptoms of an underlying mood disorder OR
- Active suicidality, which is defined as a score of 4 or more on the MADRS item 10 OR an answer of "yes" on question 4 or 5 on the C-SSRS
Analysis demonstrated a statistically significantly longer time to recurrence of any mood episode in subjects randomized to the ABILIFY MAINTENA group than compared to placebo-treated subjects. The Kaplan-Meier curves of the time of recurrence to any mood episode during the double-blind treatment phase for ABILIFY MAINTENA and placebo groups are shown in Figure 26.
|
Figure 26: Kaplan-Meier Estimation of Cumulative Recurrence Rate for Any Mood Episode This figure is based on a total of 103 recurrence events.
|
Analysis by type of mood recurrence demonstrated a statistically significantly longer time to recurrence for both manic and mixed mood episodes in subjects treated with ABILIFY MAINTENA compared to those treated with placebo. There was no substantial difference between treatment groups in delaying time to recurrence of depressive mood episodes.
An examination of subgroups did not reveal any clear evidence of differential responsiveness on the basis of age, sex, or race.
5.6 Pathological Gambling and Other Compulsive Behaviors
Post-marketing case reports suggest that patients can experience intense urges, particularly for gambling, and the inability to control these urges while taking aripiprazole. Other compulsive urges, reported less frequently, include: sexual urges, shopping, eating or binge eating, and other impulsive or compulsive behaviors. Because patients may not recognize these behaviors as abnormal, it is important for prescribers to ask patients or their caregivers specifically about the development of new or intense gambling urges, compulsive sexual urges, compulsive shopping, binge or compulsive eating, or other urges while being treated with aripiprazole. It should be noted that impulse-control symptoms can be associated with the underlying disorder. In some cases, although not all, urges were reported to have stopped when the dose was reduced, or the medication was discontinued. Compulsive behaviors may result in harm to the patient and others if not recognized. Consider dose reduction or stopping the medication if a patient develops such urges.
2.4 Dosage Modifications for Cytochrome P450 Considerations
Refer to Table 1 and Table 2 for dosage modifications for patients who are CYP2D6 poor metabolizers and/or in patients taking concomitant CYP3A4 inhibitors or CYP2D6 inhibitors for greater than 14 days.
If the CYP3A4 inhibitor or CYP2D6 inhibitor is withdrawn, the ABILIFY MAINTENA dosage may need to be increased to the previous dose [see Dosage and Administration (2.2)].
Avoid the concomitant use of CYP3A4 inducers with ABILIFY MAINTENA for greater than 14 days because the blood levels of aripiprazole are decreased and may be below the effective levels.
Dosage modifications are not recommended for patients with concomitant use of CYP3A4 inhibitors, CYP2D6 inhibitors or CYP3A4 inducers for less than 14 days.
| Factors | Adjusted Dose No change for oral dosage required during initiation.
|
|---|---|
| CYP2D6 Poor Metabolizers | |
| Known CYP2D6 Poor Metabolizers | 300 mg |
| Known CYP2D6 Poor Metabolizers taking concomitant CYP3A4 inhibitors | Avoid use |
| Concomitant use with CYP Inhibitors and/or Inducers | |
| Strong CYP2D6 or CYP3A4 inhibitors | 300 mg |
| CYP2D6 and CYP3A4 inhibitors | Avoid use |
| CYP3A4 inducers | Avoid use |
| Factors | Adjusted Dose No change for oral dosage required during initiation.
|
|---|---|
| CYP2D6 Poor Metabolizers | |
| Known CYP2D6 Poor Metabolizers | 300 mg |
| Known CYP2D6 Poor Metabolizers taking concomitant CYP3A4 inhibitors | 200 mg 200 mg and 160 mg dosage adjustment is obtained only by using the 300 mg or 400 mg strength vials.
|
| Patients Taking 400 mg of ABILIFY MAINTENA | |
| Strong CYP2D6 or CYP3A4 inhibitors | 300 mg |
| CYP2D6 and CYP3A4 inhibitors | 200 mg |
| CYP3A4 inducers | Avoid use |
| Patients Taking 300 mg of ABILIFY MAINTENA | |
| Strong CYP2D6 or CYP3A4 inhibitors | 200 mg |
| CYP2D6 and CYP3A4 inhibitors | 160 mg |
| CYP3A4 inducers | Avoid use |
ABILIFY MAINTENA comes in two types of kits. See instructions for reconstitution/injection/disposal procedures for 1) Pre-filled Dual Chamber Syringe [see Dosage and Administration (2.6)], and 2) Vials [see Dosage and Administration (2.7)].
7.1 Drugs Having Clinically Important Interactions With Abilify Maintena (7.1 Drugs Having Clinically Important Interactions with ABILIFY MAINTENA)
| Concomitant Drug Name or Drug Class | Clinical Rationale | Clinical Recommendation |
|---|---|---|
| Strong CYP3A4 Inhibitors (e.g., ketoconazole) or strong CYP2D6 inhibitors (e.g., paroxetine, fluoxetine) | The concomitant use of oral aripiprazole with strong CYP3A4 or CYP2D6 inhibitors increased the exposure of aripiprazole [see Clinical Pharmacology (12.3)]. | With concomitant use of ABILIFY MAINTENA with a strong CYP3A4 inhibitor or CYP2D6 inhibitor for more than 14 days, reduce the ABILIFY MAINTENA dosage [see Dosage and Administration (2.4)]. |
| Strong CYP3A4 Inducers (e.g., carbamazepine) | The concomitant use of oral aripiprazole and carbamazepine decreased the exposure of aripiprazole [see Clinical Pharmacology (12.3)]. | Avoid use of ABILIFY MAINTENA in combination with carbamazepine and other inducers of CYP3A4 for greater than 14 days [see Dosage and Administration (2.4)]. |
| Antihypertensive Drugs | Due to its alpha-adrenergic antagonism, aripiprazole has the potential to enhance the effect of certain antihypertensive agents. | Monitor blood pressure and adjust dose accordingly [see Warnings and Precautions (5.7)]. |
| Benzodiazepines (e.g., lorazepam) | The intensity of sedation was greater with the combination of oral aripiprazole and lorazepam as compared to that observed with aripiprazole alone. The orthostatic hypotension observed was greater with the combination as compared to that observed with lorazepam alone [see Warnings and Precautions (5.7)]. | Monitor sedation and blood pressure. Adjust dose accordingly. |
7.2 Drugs Having No Clinically Important Interactions With Abilify Maintena (7.2 Drugs Having No Clinically Important Interactions with ABILIFY MAINTENA)
Based on pharmacokinetic studies with oral aripiprazole, no dosage adjustment of ABILIFY MAINTENA is required when administered concomitantly with famotidine, valproate, lithium, lorazepam [see Clinical Pharmacology (12.3)].
In addition, no dosage adjustment is necessary for substrates of CYP2D6 (e.g., dextromethorphan, fluoxetine, paroxetine, or venlafaxine), CYP2C9 (e.g., warfarin), CYP2C19 (e.g., omeprazole, warfarin), or CYP3A4 (e.g., dextromethorphan) when coadministered with ABILIFY MAINTENA. Additionally, no dosage adjustment is necessary for valproate, lithium, lamotrigine, lorazepam, or sertraline when coadministered with ABILIFY MAINTENA. [See Clinical Pharmacology (12.3)].
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis (5.1 Increased Mortality in Elderly Patients with Dementia- Related Psychosis)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group.
Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis.
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis (WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
5.2 Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients With Dementia Related Psychosis (5.2 Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis)
In placebo-controlled clinical studies (two flexible-dose and one fixed-dose study) of dementia-related psychosis, there was an increased incidence of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack), including fatalities, in oral aripiprazole-treated patients (mean age: 84 years; range: 78 to 88 years). In the fixed-dose study, there was a statistically significant dose response relationship for cerebrovascular adverse reactions in patients treated with oral aripiprazole. ABILIFY MAINTENA is not approved for the treatment of patients with dementia-related psychosis.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:42.669136 · Updated: 2026-03-14T22:25:27.698826