These Highlights Do Not Include All The Information Needed To Use Levetiracetam In Sodium Chloride Injection Safely And Effectively. See Full Prescribing Information For Levetiracetam In Sodium Chloride Injection.
ee014183-a857-457b-9573-0cc168e560f3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.5 ) 3/2024
Indications and Usage
Levetiracetam in Sodium Chloride Injection is indicated for adjunct therapy in adults (≥16 years of age) with the following seizure types when oral administration is temporarily not feasible: Partial-onset seizures ( 1.1 ) Myoclonic seizures in patients with juvenile myoclonic epilepsy ( 1.2 ) Primary generalized tonic-clonic seizures ( 1.3 )
Dosage and Administration
For intravenous infusion only ( 2.1 ) Do not dilute prior to its use ( 2.1 ) Administer dose-specific bag intravenously over 15-minutes ( 2.1 ) Initial Exposure to Levetiracetam Partial-Onset Seizures: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to a maximum recommended dose of 1500 mg twice daily ( 2.2 ). Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to the recommended daily dose of 1500 mg twice daily ( 2.2 ) Primary Generalized Tonic-Clonic Seizures: Initial dose is 500 mg twice daily. Increase dose by 500 mg daily every 2 weeks to the recommended dose of 1500 mg twice daily. ( 2.2 ). Switching from or to oral Levetiracetam: The total daily dosage/frequency of levetiracetam injection should be equivalent to those of oral levetiracetam ( 2.3 , 2.4 ). Renal Impairment: Dose adjustment necessary based on creatinine clearance ( 2.5 ).
Warnings and Precautions
Psychiatric Reactions: Behavioral abnormalities including psychotic symptoms, suicidal ideation, irritability, and aggressive behavior have been observed. Monitor patients for psychiatric signs and symptoms ( 5.1 ) Somnolence and Fatigue: Monitor patients for these symptoms and advise patients not to drive or operate machinery until they have gained sufficient experience on levetiracetam ( 5.2 ) Serious Dermatological Reactions: Discontinue Levetiracetam at the first sign of rash unless clearly not drug related. ( 5.4 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: Discontinue if no alternative etiology ( 5.5 ) Coordination Difficulties: Monitor for ataxia, abnormal gait, and incoordination. ( 5.6 ) Withdrawal Seizures: Levetiracetam must be gradually withdrawn ( 5.7 )
Contraindications
Levetiracetam in Sodium Chloride Injection is contraindicated in patients with a hypersensitivity to levetiracetam. Reactions have included anaphylaxis and angioedema [ see Warnings and Precautions ( 5.3 ) ].
Adverse Reactions
The following serious adverse reactions are discussed in more details in other sections of labeling: Psychiatric Reactions [ see Warnings and Precautions ( 5.1 ) ] Somnolence and Fatigue [ see Warnings and Precautions ( 5.2 ) ] Anaphylaxis and Angioedema [ see Warnings and Precautions ( 5.3 ) ] Serious Dermatological Reactions [ see Warnings and Precautions ( 5.4 ) ] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [ see Warnings and Precautions ( 5.5 ) ] Coordination Difficulties [ see Warnings and Precautions ( 5.6 ) ] Withdrawal Seizures [ see Warnings and Precautions ( 5.7 ) ] Hematologic Abnormalities [ see Warnings and Precautions ( 5.8 ) ] Seizure Control During Pregnancy [ see Warnings and Precautions ( 5.9 ) ]
How Supplied
Levetiracetam in Sodium Chloride Injection is a clear, colorless, sterile solution that is available in a single-dose 100 mL dual port bag with an aluminum over wrap. The container closure is not made with natural rubber latex. It is available in the following presentations: Strength Package NDC Number 500 mg (5 mg/mL) 1 single-dose bag 14789-110-08 10 bags per carton 14789-110-05 1000 mg (10 mg/mL) 1 single-dose bag 14789-220-08 10 bags per carton 14789-220-10 1500 mg (15 mg/mL) 1 single-dose bag 14789-330-08 10 bags per carton 14789-330-15
Medication Information
Warnings and Precautions
Psychiatric Reactions: Behavioral abnormalities including psychotic symptoms, suicidal ideation, irritability, and aggressive behavior have been observed. Monitor patients for psychiatric signs and symptoms ( 5.1 ) Somnolence and Fatigue: Monitor patients for these symptoms and advise patients not to drive or operate machinery until they have gained sufficient experience on levetiracetam ( 5.2 ) Serious Dermatological Reactions: Discontinue Levetiracetam at the first sign of rash unless clearly not drug related. ( 5.4 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: Discontinue if no alternative etiology ( 5.5 ) Coordination Difficulties: Monitor for ataxia, abnormal gait, and incoordination. ( 5.6 ) Withdrawal Seizures: Levetiracetam must be gradually withdrawn ( 5.7 )
Indications and Usage
Levetiracetam in Sodium Chloride Injection is indicated for adjunct therapy in adults (≥16 years of age) with the following seizure types when oral administration is temporarily not feasible: Partial-onset seizures ( 1.1 ) Myoclonic seizures in patients with juvenile myoclonic epilepsy ( 1.2 ) Primary generalized tonic-clonic seizures ( 1.3 )
Dosage and Administration
For intravenous infusion only ( 2.1 ) Do not dilute prior to its use ( 2.1 ) Administer dose-specific bag intravenously over 15-minutes ( 2.1 ) Initial Exposure to Levetiracetam Partial-Onset Seizures: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to a maximum recommended dose of 1500 mg twice daily ( 2.2 ). Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to the recommended daily dose of 1500 mg twice daily ( 2.2 ) Primary Generalized Tonic-Clonic Seizures: Initial dose is 500 mg twice daily. Increase dose by 500 mg daily every 2 weeks to the recommended dose of 1500 mg twice daily. ( 2.2 ). Switching from or to oral Levetiracetam: The total daily dosage/frequency of levetiracetam injection should be equivalent to those of oral levetiracetam ( 2.3 , 2.4 ). Renal Impairment: Dose adjustment necessary based on creatinine clearance ( 2.5 ).
Contraindications
Levetiracetam in Sodium Chloride Injection is contraindicated in patients with a hypersensitivity to levetiracetam. Reactions have included anaphylaxis and angioedema [ see Warnings and Precautions ( 5.3 ) ].
Adverse Reactions
The following serious adverse reactions are discussed in more details in other sections of labeling: Psychiatric Reactions [ see Warnings and Precautions ( 5.1 ) ] Somnolence and Fatigue [ see Warnings and Precautions ( 5.2 ) ] Anaphylaxis and Angioedema [ see Warnings and Precautions ( 5.3 ) ] Serious Dermatological Reactions [ see Warnings and Precautions ( 5.4 ) ] Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [ see Warnings and Precautions ( 5.5 ) ] Coordination Difficulties [ see Warnings and Precautions ( 5.6 ) ] Withdrawal Seizures [ see Warnings and Precautions ( 5.7 ) ] Hematologic Abnormalities [ see Warnings and Precautions ( 5.8 ) ] Seizure Control During Pregnancy [ see Warnings and Precautions ( 5.9 ) ]
How Supplied
Levetiracetam in Sodium Chloride Injection is a clear, colorless, sterile solution that is available in a single-dose 100 mL dual port bag with an aluminum over wrap. The container closure is not made with natural rubber latex. It is available in the following presentations: Strength Package NDC Number 500 mg (5 mg/mL) 1 single-dose bag 14789-110-08 10 bags per carton 14789-110-05 1000 mg (10 mg/mL) 1 single-dose bag 14789-220-08 10 bags per carton 14789-220-10 1500 mg (15 mg/mL) 1 single-dose bag 14789-330-08 10 bags per carton 14789-330-15
Description
Warnings and Precautions ( 5.5 ) 3/2024
Section 42229-5
Partial-Onset Seizures
In clinical trials of oral levetiracetam, daily doses of 1000 mg, 2000 mg, and 3000 mg, given as twice-daily dosing, were shown to be effective. Although in some studies there was a tendency toward greater response with higher dose [see Clinical Studies (14.1)], a consistent increase in response with increased dose has not been shown.
Treatment should be initiated with a daily dose of 1000 mg/day, given as twice-daily dosing (500 mg twice daily). Additional dosing increments may be given (1000 mg/day additional every 2 weeks) to a maximum recommended daily dose of 3000 mg. Doses greater than 3000 mg/day have been used in open-label studies with levetiracetam tablets for periods of 6 months and longer. There is no evidence that doses greater than 3000 mg/day confer additional benefit.
Section 43683-2
| Warnings and Precautions (5.5) | 3/2024 |
Section 51945-4
Principal Display Panel – 5mg/mL Carton Label
NDC 14789-110-05
Levetiracetam
in 0.82% Sodium Chloride Injection
For Intravenous Infusion Only
Rx ONLY
10 Single Use Bags
500 mg/
100 mL
(5 mg/mL)
NEXUS
PHARMACEUTICALS
16.2 Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
11 Description
Levetiracetam in Sodium Chloride Injection is an antiepileptic drug available as a clear, colorless, sterile solution for intravenous administration.
The chemical name of levetiracetam, a single enantiomer, is (-)-(S)-α-ethyl-2-oxo-1-pyrrolidine acetamide, its molecular formula is C8H14N2O2 and its molecular weight is 170.21.
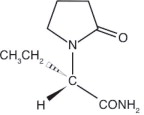
Levetiracetam is chemically unrelated to existing antiepileptic drugs (AEDs). It has the following structural formula:
Levetiracetam is a white to off-white crystalline powder with a faint odor and a bitter taste. It is very soluble in water (104.0 g/100 mL). It is freely soluble in chloroform (65.3 g/100 mL) and in methanol (53.6 g/100 mL), soluble in ethanol (16.5 g/100 mL), sparingly soluble in acetonitrile (5.7 g/100 mL) and practically insoluble in n-hexane. (Solubility limits are expressed as g/100 mL solvent.)
Levetiracetam in Sodium Chloride Injection is a clear, colorless, sterile solution that is available in a single-dose dual port bag with an aluminum over wrap. The container closure is not made with natural rubber latex.
500 mg/100 mL: One 100 mL bag contains 500 mg of levetiracetam (5 mg/mL), water for injection, 820 mg sodium chloride, 5.5 mg of glacial acetic acid and buffered at approximately pH 5.5 with glacial acetic acid and 164 mg sodium acetate trihydrate.
1000 mg/100 mL: One 100 mL bag contains 1000 mg of levetiracetam (10 mg/mL), water for injection, 750 mg sodium chloride, 6.5 mg of glacial acetic acid and buffered at approximately pH 5.5 with glacial acetic acid and 164 mg sodium acetate trihydrate.
1500 mg/100 mL: One 100 mL bag contains 1500 mg of levetiracetam (15 mg/mL), water for injection, 540 mg sodium chloride, 7.5 mg of glacial acetic acid and buffered at approximately pH 5.5 with glacial acetic acid and 164 mg sodium acetate trihydrate.
10.3 Hemodialysis
Standard hemodialysis procedures result in significant clearance of levetiracetam (approximately 50% in 4 hours) and should be considered in cases of overdose. Although hemodialysis has not been performed in the few known cases of overdose, it may be indicated by the patient's clinical state or in patients with significant renal impairment.
16.1 How Supplied
Levetiracetam in Sodium Chloride Injection is a clear, colorless, sterile solution that is available in a single-dose 100 mL dual port bag with an aluminum over wrap. The container closure is not made with natural rubber latex. It is available in the following presentations:
| Strength | Package | NDC Number |
| 500 mg (5 mg/mL) | 1 single-dose bag | 14789-110-08 |
| 10 bags per carton | 14789-110-05 | |
| 1000 mg (10 mg/mL) | 1 single-dose bag | 14789-220-08 |
| 10 bags per carton | 14789-220-10 | |
| 1500 mg (15 mg/mL) | 1 single-dose bag | 14789-330-08 |
| 10 bags per carton | 14789-330-15 |
8.4 Pediatric Use
Safety and effectiveness of levetiracetam injection in patients below the age of 16 years have not been established.
8.5 Geriatric Use
There were 347 subjects in clinical studies of levetiracetam that were 65 years old and over. No overall differences in safety were observed between these subjects and younger subjects. There were insufficient numbers of elderly subjects in controlled trials of epilepsy to adequately assess the effectiveness of levetiracetam in these patients.
Levetiracetam is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Clinical Pharmacology (12.3)].
14 Clinical Studies
All clinical studies supporting the efficacy of levetiracetam utilized oral formulations. The finding of efficacy of levetiracetam injection is based on the results of studies using an oral formulation of levetiracetam, and on the demonstration of comparable bioavailability of the oral and parenteral formulations [see Pharmacokinetics (12.3)].
4 Contraindications
Levetiracetam in Sodium Chloride Injection is contraindicated in patients with a hypersensitivity to levetiracetam. Reactions have included anaphylaxis and angioedema [see Warnings and Precautions (5.3)].
6 Adverse Reactions
The following serious adverse reactions are discussed in more details in other sections of labeling:
- Psychiatric Reactions [see Warnings and Precautions (5.1)]
- Somnolence and Fatigue [see Warnings and Precautions (5.2)]
- Anaphylaxis and Angioedema [see Warnings and Precautions (5.3)]
- Serious Dermatological Reactions [see Warnings and Precautions (5.4)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5)]
- Coordination Difficulties [see Warnings and Precautions (5.6)]
- Withdrawal Seizures [see Warnings and Precautions (5.7)]
- Hematologic Abnormalities [see Warnings and Precautions (5.8)]
- Seizure Control During Pregnancy [see Warnings and Precautions (5.9)]
8.6 Renal Impairment
Clearance of levetiracetam is decreased in patients with renal impairment and is correlated with creatinine clearance [see Clinical Pharmacology (12.3)]. Dosage adjustment is recommended for patients with impaired renal function and supplemental doses should be given to patients after dialysis [see Dosage and Administration (2.5)].
12.3 Pharmacokinetics
Equivalent doses of intravenous (IV) levetiracetam and oral levetiracetam result in equivalent Cmax, Cmin, and total systemic exposure to levetiracetam when the IV levetiracetam is administered as a 15 minute infusion.
1.4 Limitations of Use
Levetiracetam in Sodium Chloride Injection is an antiepileptic drug indicated for adult patients (16 years and older) when oral administration is temporarily not feasible.
1 Indications and Usage
Levetiracetam in Sodium Chloride Injection is indicated for adjunct therapy in adults (≥16 years of age) with the following seizure types when oral administration is temporarily not feasible:
5.7 Withdrawal Seizures
As with most antiepileptic drugs, levetiracetam should be withdrawn gradually because of the risk of increased seizure frequency and status epilepticus. But if withdrawal is needed because of a serious reaction, rapid discontinuation can be considered.
12.1 Mechanism of Action
The precise mechanism(s) by which levetiracetam exerts its antiepileptic effect is unknown.
A saturable and stereoselective neuronal binding site in rat brain tissue has been described for levetiracetam. Experimental data indicate that this binding site is the synaptic vesicle protein SV2A, thought to be involved in the regulation of vesicle exocytosis. Although the molecular significance of levetiracetam binding to synaptic vesicle protein SV2A is not understood, levetiracetam and related analogs showed a rank order of affinity for SV2A which correlated with the potency of their antiseizure activity in audiogenic seizure-prone mice. These findings suggest that the interaction of levetiracetam with the SV2A protein may contribute to the antiepileptic mechanism of action of the drug.
5.1 Psychiatric Reactions
In some patients levetiracetam causes behavioral abnormalities. The incidences of behavioral abnormalities in the myoclonic and primary generalized tonic-clonic seizure studies were comparable to those of the adult partial-onset seizure studies.
A total of 13.3% of adult levetiracetam-treated patients compared to 6.2% of placebo patients experienced non-psychotic behavioral symptoms (reported as aggression, agitation, anger, anxiety, apathy, depersonalization, depression, emotional lability, hostility, irritability, and nervousness).
A total of 1.7% of adult levetiracetam-treated patients discontinued treatment due to behavioral adverse events, compared to 0.2% of placebo patients. The treatment dose was reduced in 0.8% of adult levetiracetam-treated patients and in 0.5% of placebo patients.
One percent of adult levetiracetam-treated patients experienced psychotic symptoms compared to 0.2% of placebo patients.
Two (0.3%) adult levetiracetam-treated patients were hospitalized and their treatment was discontinued due to psychosis. Both events, reported as psychosis, developed within the first week of treatment and resolved within 1 to 2 weeks following treatment discontinuation.
The above psychiatric signs and symptoms should be monitored.
1.1 Partial Onset Seizures
Levetiracetam in Sodium Chloride Injection is indicated as adjunctive therapy in the treatment of partial-onset seizures in adults with epilepsy.
5 Warnings and Precautions
- Psychiatric Reactions: Behavioral abnormalities including psychotic symptoms, suicidal ideation, irritability, and aggressive behavior have been observed. Monitor patients for psychiatric signs and symptoms (5.1)
- Somnolence and Fatigue: Monitor patients for these symptoms and advise patients not to drive or operate machinery until they have gained sufficient experience on levetiracetam (5.2)
- Serious Dermatological Reactions: Discontinue Levetiracetam at the first sign of rash unless clearly not drug related. (5.4)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: Discontinue if no alternative etiology (5.5)
- Coordination Difficulties: Monitor for ataxia, abnormal gait, and incoordination. (5.6)
- Withdrawal Seizures: Levetiracetam must be gradually withdrawn (5.7)
5.2 Somnolence and Fatigue
In some patients, levetiracetam causes somnolence and fatigue. The incidences of somnolence and fatigue provided below are from controlled adult partial-onset seizure studies. In general, the incidences of somnolence and fatigue in the myoclonic and primary generalized tonicclonic studies were comparable to those of the adult partial-onset seizure studies.
In controlled trials of adult patients with epilepsy experiencing partial-onset seizures, 14.8% of levetiracetam-treated patients reported somnolence, compared to 8.4% of placebo patients. There was no clear dose response up to 3000 mg/day. In a study where there was no titration, about 45% of patients receiving 4000 mg/day reported somnolence. The somnolence was considered serious in 0.3% of the treated patients, compared to 0% in the placebo group. About 3% of levetiracetam-treated patients discontinued treatment due to somnolence, compared to 0.7% of placebo patients. In 1.4% of treated patients and in 0.9% of placebo patients the dose was reduced, while 0.3% of the treated patients were hospitalized due to somnolence.
In controlled trials of adult patients with epilepsy experiencing partial-onset seizures, 14.7% of levetiracetam-treated patients reported asthenia, compared to 9.1% of placebo patients.
Treatment was discontinued due to asthenia in 0.8% of treated patients as compared to 0.5% of placebo patients. In 0.5% of treated patients and in 0.2% of placebo patients the dose was reduced due to asthenia.
Somnolence and asthenia occurred most frequently within the first 4 weeks of treatment.
Patients should be monitored for these signs and symptoms and advised not to drive or operate machinery until they have gained sufficient experience on levetiracetam to gauge whether it adversely affects their ability to drive or operate machinery.
10.2 Management of Overdose
There is no specific antidote for overdose with levetiracetam. If indicated, elimination of unabsorbed drug should be attempted by emesis or gastric lavage; usual precautions should be observed to maintain airway. General supportive care of the patient is indicated including monitoring of vital signs and observation of the patient's clinical status. A Certified Poison Control Center should be contacted for up to date information on the management of overdose with levetiracetam.
14.1 Partial Onset Seizures
The effectiveness of levetiracetam as adjunctive therapy (added to other antiepileptic drugs) in adults was established in three multicenter, randomized, double-blind, placebo-controlled clinical studies in patients who had refractory partial-onset seizures with or without secondary generalization. The tablet formulation was used in all these studies. In these studies, 904 patients were randomized to placebo, 1000 mg, 2000 mg, or 3000 mg/day. Patients enrolled in Study 1 or Study 2 had refractory partial-onset seizures for at least two years and had taken two or more classical AEDs. Patients enrolled in Study 3 had refractory partial-onset seizures for at least 1 year and had taken one classical AED. At the time of the study, patients were taking a stable dose regimen of at least one and could take a maximum of two AEDs. During the baseline period, patients had to have experienced at least two partial-onset seizures during each 4-week period.
2 Dosage and Administration
- For intravenous infusion only (2.1)
- Do not dilute prior to its use (2.1)
- Administer dose-specific bag intravenously over 15-minutes (2.1)
Initial Exposure to Levetiracetam
- Partial-Onset Seizures: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to a maximum recommended dose of 1500 mg twice daily (2.2).
- Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to the recommended daily dose of 1500 mg twice daily (2.2)
- Primary Generalized Tonic-Clonic Seizures: Initial dose is 500 mg twice daily. Increase dose by 500 mg daily every 2 weeks to the recommended dose of 1500 mg twice daily. (2.2).
Switching from or to oral Levetiracetam: The total daily dosage/frequency of levetiracetam injection should be equivalent to those of oral levetiracetam (2.3, 2.4).
Renal Impairment: Dose adjustment necessary based on creatinine clearance (2.5).
2.4 Switching to Oral Dosing
At the end of the intravenous treatment period, the patient may be switched to levetiracetam oral administration at the equivalent daily dosage and frequency of the intravenous administration.
3 Dosage Forms and Strengths
Injection: Levetiracetam in Sodium Chloride Injection is a clear, colorless solution, packaged in a single-dose bag and available in three strengths:
- 500 mg Levetiracetam in 0.82 % sodium chloride injection (500 mg/100 mL)
- 1000 mg Levetiracetam in 0.75 % sodium chloride injection (1000 mg/100 mL)
- 1500 mg Levetiracetam in 0.54% sodium chloride injection (1500 mg/100 mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of levetiracetam. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In addition to the adverse reactions listed above [see Adverse Reactions (6.1)], the following adverse reactions have been reported in patients receiving marketed levetiracetam worldwide. The listing is alphabetized: abnormal liver function test, acute kidney injury, agranulocytosis, anaphylaxis, angioedema, choreoathetosis, drug reaction with eosinophilia and systemic symptoms (DRESS), dyskinesia, erythema multiforme, hepatic failure, hepatitis, hyponatremia, muscular weakness, pancreatitis, pancytopenia (with bone marrow suppression identified in some of these cases), panic attack, weight loss, and worsening of seizures Alopecia has been reported with levetiracetam use; recovery was observed in majority of cases where levetiracetam was discontinued.
5.6 Coordination Difficulties
Coordination difficulties were only observed in the adult partial-onset seizure studies. A total of 3.4% of adult levetiracetam-treated patients experienced coordination difficulties, (reported as either ataxia, abnormal gait, or incoordination) compared to 1.6% of placebo patients. A total of 0.4% of patients in controlled trials discontinued levetiracetam treatment due to ataxia, compared to 0% of placebo patients. In 0.7% of treated patients and in 0.2% of placebo patients the dose was reduced due to coordination difficulties, while one of the treated patients was hospitalized due to worsening of pre-existing ataxia. These events occurred most frequently within the first 4 weeks of treatment.
Patients should be monitored for these signs and symptoms and advised not to drive or operate machinery until they have gained sufficient experience on levetiracetam to gauge whether it adversely affects their ability to drive or operate machinery.
5.8 Hematologic Abnormalities
Levetiracetam can cause hematologic abnormalities. Hematologic abnormalities occurred in clinical trials and included decreases in white blood cell (WBC), neutrophil, and red blood cells counts (RBC); decreases in hemoglobin and hematocrit; and increases in eosinophil counts.
Cases of agranulocytosis, pancytopenia, and thrombocytopenia have been reported in the postmarketing setting. A complete blood count is recommended in patients experiencing significant weakness, pyrexia, recurrent infections, or coagulation disorders.
8 Use in Specific Populations
5.3 Anaphylaxis and Angioedema
Levetiracetam can cause anaphylaxis or angioedema after the first dose or at any time during treatment. Signs and symptoms in cases reported in the postmarketing setting with levetiracetam have included hypotension, hives, rash, respiratory distress, and swelling of the face, lip, mouth, eye, tongue, throat, and feet. In some reported cases, reactions were lifethreatening and required emergency treatment. If a patient develops signs or symptoms of anaphylaxis or angioedema, levetiracetam should be discontinued and the patient should seek immediate medical attention.
Levetiracetam should be discontinued permanently if a clear alternative etiology for the reaction cannot be established [see Contraindications ( 4 )].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The adverse reactions that result from levetiracetam injection use include all of those reported for levetiracetam tablets and oral solution. Equivalent doses of intravenous (IV) levetiracetam and oral levetiracetam result in equivalent Cmax, Cmin, and total systemic exposure to levetiracetam when the IV levetiracetam is administered as a 15-minute infusion.
2.3 Switching to Intravenous Dosing
When switching from oral levetiracetam, the initial total daily intravenous dosage of levetiracetam should be equivalent to the total daily dosage and frequency of oral levetiracetam.
2.7 Discontinuation of Levetiracetam
Avoid abrupt withdrawal from levetiracetam in order to reduce the risk of increased seizure frequency and status epilepticus [see Warnings and Precautions (5.7)].
5.4 Serious Dermatological Reactions
Serious dermatological reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported in patients treated with levetiracetam. The median time of onset is reported to be 14 to 17 days, but cases have been reported at least four months after initiation of treatment. Recurrence of the serious skin reactions following rechallenge with levetiracetam has also been reported. Levetiracetam should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed and alternative therapy should be considered.
5.9 Seizure Control During Pregnancy
Physiological changes may gradually decrease plasma levels of levetiracetam throughout pregnancy. This decrease is more pronounced during the third trimester. It is recommended that patients be monitored carefully during pregnancy. Close monitoring should continue through the postpartum period especially if the dose was changed during pregnancy.
2.2 Initial Exposure to Levetiracetam
Levetiracetam can be initiated with either intravenous or oral administration.
2.1 General Information – Administration
Levetiracetam in Sodium Chloride Injection is for intravenous infusion only. It is available in the following concentrations: three single-dose 100 mL bags, each containing a different total dosage of levetiracetam (500 mg [5 mg/mL], 1000 mg [10 mg/mL], or 1500 mg [15 mg/mL]).
A single-dose bag should be administered intravenously over a 15-minute IV infusion period.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Levetiracetam in Sodium Chloride Injection should not be further diluted prior to use. Any unused portion of the Levetiracetam in Sodium Chloride Injection contents should be discarded.
1.3 Primary Generalized Tonic Clonic Seizures
Levetiracetam in Sodium Chloride Injection is indicated as adjunctive therapy in the treatment of primary generalized tonic-clonic seizures in adults with idiopathic generalized epilepsy.
2.5 Adult Patients With Impaired Renal Function
Levetiracetam dosing must be individualized according to the patient's renal function status. Recommended doses and adjustment for dose for adults are shown in Table 1. To use this dosing table, an estimate of the patient's creatinine clearance (CLcr) in mL/min is needed.
|
1 Following dialysis, a 250 to 500 mg supplemental dose is recommended. |
|||
| Group | Creatinine Clearance (mL/min) | Dosage (mg) | Frequency |
| Normal | greater than 80 | 500 to 1,500 | Every 12 hours |
| Mild | 50 to 80 | 500 to 1,000 | Every 12 hours |
| Moderate | 30 to 50 | 250 to 750 | Every 12 hours |
| Severe | less than 30 | 250 to 500 | Every 12 hours |
| ESRD patients using dialysis | --- | 500 to 1,000 | 1Every 24 hours |
14.3 Primary Generalized Tonic Clonic Seizures
The effectiveness of levetiracetam as adjunctive therapy in patients with idiopathic generalized epilepsy experiencing primary generalized tonic-clonic (PGTC) seizures was established in one multicenter, randomized, double-blind, placebo-controlled study, conducted at 50 sites in 8 countries. Eligible patients on a stable dose of 1 or 2 antiepileptic drugs (AEDs) experiencing at least 3 PGTC seizures during the 8-week combined baseline period (at least one PGTC seizure during the 4 weeks prior to the prospective baseline period and at least one PGTC seizure during the 4-week prospective baseline period) were randomized to either levetiracetam or placebo. The 8-week combined baseline period is referred to as “baseline” in the remainder of this section. The population included 164 patients (levetiracetam N=80, placebo N=84) with idiopathic generalized epilepsy (predominately juvenile myoclonic epilepsy, juvenile absence epilepsy, childhood absence epilepsy, or epilepsy with Grand Mal seizures on awakening) experiencing primary generalized tonic-clonic seizures. Each of these syndromes of idiopathic generalized epilepsy was well represented in this patient population. Patients were titrated over 4 weeks to a target dose of 3000 mg/day for adults or a pediatric target dose of 60 mg/kg/day and treated at a stable dose of 3000 mg/day (or 60 mg/kg/day for children) over 20 weeks (evaluation period). Study drug was given in 2 equally divided doses per day.
The primary measure of effectiveness was the percent reduction from baseline in weekly PGTC seizure frequency for levetiracetam and placebo treatment groups over the treatment period (titration + evaluation periods). There was a statistically significant decrease from baseline in PGTC frequency in the levetiracetam-treated patients compared to the placebo-treated patients.
|
*Statistically significant versus placebo |
||
| Placebo (N=84) | Levetiracetam (N=78) | |
| Percentage reduction in PGTC seizure frequency | 44.6% | 77.6%* |
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in PGTC seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 4.
Figure 4: Responder Rate (≥50% Reduction from Baseline) in PGTC Seizure Frequency per Week
2.6 Compatibility With Other Antiepileptic Drugs
Levetiracetam in Sodium Chloride Injection is found to be physically compatible and chemically stable for at least 24 hours when mixed with lorazepam, diazepam, and valproate sodium and stored at controlled room temperature 15° to 30°C (59° to 86°F).
There are no data to support the physical compatibility of levetiracetam injection with antiepileptic drugs that are not listed above.
1.2 Myoclonic Seizures in Patients With Juvenile Myoclonic Epilepsy
Levetiracetam in Sodium Chloride Injection is indicated as adjunctive therapy in the treatment of myoclonic seizures in adults with juvenile myoclonic epilepsy.
14.2 Myoclonic Seizures in Patients With Juvenile Myoclonic Epilepsy
The effectiveness of levetiracetam as adjunctive therapy in patients with juvenile myoclonic epilepsy (JME) experiencing myoclonic seizures was established in one multicenter, randomized, double-blind, placebo-controlled study, conducted at 37 sites in 14 countries. Of the 120 patients enrolled, 113 had a diagnosis of confirmed or suspected JME. Eligible patients on a stable dose of 1 antiepileptic drug (AED) experiencing one or more myoclonic seizures per day for at least 8 days during the prospective 8-week baseline period were randomized to either levetiracetam or placebo (levetiracetam N=60, placebo N=60). Patients were titrated over 4 weeks to a target dose of 3000 mg/day and treated at a stable dose of 3000 mg/day over 12 weeks (evaluation period). Study drug was given in 2 divided doses. The primary measure of effectiveness was the proportion of patients with at least 50% reduction in the number of days per week with one or more myoclonic seizures during the treatment period (titration + evaluation periods) as compared to baseline. Table 10 displays the results for the 113 patients with JME in this study. Of 120 patients enrolled, 113 had a diagnosis of confirmed or suspected JME. The results are displayed in Table 10.
|
*Statistically significant versus placebo |
||
| Placebo (N=59) | Levetiracetam (N=54) | |
| Percentage of responders | 23.7% | 60.4%* |
10.1 Signs, Symptoms and Laboratory Findings of Acute Overdosage in Humans
The highest known dose of oral levetiracetam received in the clinical development program was 6000 mg/day. Other than drowsiness, there were no adverse reactions in the few known cases of overdose in clinical trials. Cases of somnolence, agitation, aggression, depressed level of consciousness, respiratory depression and coma were observed with levetiracetam overdoses in postmarketing use.
5.5 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as multiorgan hypersensitivity, has been reported in patients taking antiepileptic drugs, including levetiracetam. These events can be fatal or life-threatening, particularly if diagnosis and treatment do not occur as early as possible. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis, sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Levetiracetam should be discontinued if an alternative etiology for the signs or symptoms cannot be established [see Contraindications (4)].
Structured Label Content
Section 42229-5 (42229-5)
Partial-Onset Seizures
In clinical trials of oral levetiracetam, daily doses of 1000 mg, 2000 mg, and 3000 mg, given as twice-daily dosing, were shown to be effective. Although in some studies there was a tendency toward greater response with higher dose [see Clinical Studies (14.1)], a consistent increase in response with increased dose has not been shown.
Treatment should be initiated with a daily dose of 1000 mg/day, given as twice-daily dosing (500 mg twice daily). Additional dosing increments may be given (1000 mg/day additional every 2 weeks) to a maximum recommended daily dose of 3000 mg. Doses greater than 3000 mg/day have been used in open-label studies with levetiracetam tablets for periods of 6 months and longer. There is no evidence that doses greater than 3000 mg/day confer additional benefit.
Section 43683-2 (43683-2)
| Warnings and Precautions (5.5) | 3/2024 |
Section 51945-4 (51945-4)
Principal Display Panel – 5mg/mL Carton Label
NDC 14789-110-05
Levetiracetam
in 0.82% Sodium Chloride Injection
For Intravenous Infusion Only
Rx ONLY
10 Single Use Bags
500 mg/
100 mL
(5 mg/mL)
NEXUS
PHARMACEUTICALS
16.2 Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
11 Description (11 DESCRIPTION)
Levetiracetam in Sodium Chloride Injection is an antiepileptic drug available as a clear, colorless, sterile solution for intravenous administration.
The chemical name of levetiracetam, a single enantiomer, is (-)-(S)-α-ethyl-2-oxo-1-pyrrolidine acetamide, its molecular formula is C8H14N2O2 and its molecular weight is 170.21.
Levetiracetam is chemically unrelated to existing antiepileptic drugs (AEDs). It has the following structural formula:
Levetiracetam is a white to off-white crystalline powder with a faint odor and a bitter taste. It is very soluble in water (104.0 g/100 mL). It is freely soluble in chloroform (65.3 g/100 mL) and in methanol (53.6 g/100 mL), soluble in ethanol (16.5 g/100 mL), sparingly soluble in acetonitrile (5.7 g/100 mL) and practically insoluble in n-hexane. (Solubility limits are expressed as g/100 mL solvent.)
Levetiracetam in Sodium Chloride Injection is a clear, colorless, sterile solution that is available in a single-dose dual port bag with an aluminum over wrap. The container closure is not made with natural rubber latex.
500 mg/100 mL: One 100 mL bag contains 500 mg of levetiracetam (5 mg/mL), water for injection, 820 mg sodium chloride, 5.5 mg of glacial acetic acid and buffered at approximately pH 5.5 with glacial acetic acid and 164 mg sodium acetate trihydrate.
1000 mg/100 mL: One 100 mL bag contains 1000 mg of levetiracetam (10 mg/mL), water for injection, 750 mg sodium chloride, 6.5 mg of glacial acetic acid and buffered at approximately pH 5.5 with glacial acetic acid and 164 mg sodium acetate trihydrate.
1500 mg/100 mL: One 100 mL bag contains 1500 mg of levetiracetam (15 mg/mL), water for injection, 540 mg sodium chloride, 7.5 mg of glacial acetic acid and buffered at approximately pH 5.5 with glacial acetic acid and 164 mg sodium acetate trihydrate.
10.3 Hemodialysis
Standard hemodialysis procedures result in significant clearance of levetiracetam (approximately 50% in 4 hours) and should be considered in cases of overdose. Although hemodialysis has not been performed in the few known cases of overdose, it may be indicated by the patient's clinical state or in patients with significant renal impairment.
16.1 How Supplied
Levetiracetam in Sodium Chloride Injection is a clear, colorless, sterile solution that is available in a single-dose 100 mL dual port bag with an aluminum over wrap. The container closure is not made with natural rubber latex. It is available in the following presentations:
| Strength | Package | NDC Number |
| 500 mg (5 mg/mL) | 1 single-dose bag | 14789-110-08 |
| 10 bags per carton | 14789-110-05 | |
| 1000 mg (10 mg/mL) | 1 single-dose bag | 14789-220-08 |
| 10 bags per carton | 14789-220-10 | |
| 1500 mg (15 mg/mL) | 1 single-dose bag | 14789-330-08 |
| 10 bags per carton | 14789-330-15 |
8.4 Pediatric Use
Safety and effectiveness of levetiracetam injection in patients below the age of 16 years have not been established.
8.5 Geriatric Use
There were 347 subjects in clinical studies of levetiracetam that were 65 years old and over. No overall differences in safety were observed between these subjects and younger subjects. There were insufficient numbers of elderly subjects in controlled trials of epilepsy to adequately assess the effectiveness of levetiracetam in these patients.
Levetiracetam is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
All clinical studies supporting the efficacy of levetiracetam utilized oral formulations. The finding of efficacy of levetiracetam injection is based on the results of studies using an oral formulation of levetiracetam, and on the demonstration of comparable bioavailability of the oral and parenteral formulations [see Pharmacokinetics (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
Levetiracetam in Sodium Chloride Injection is contraindicated in patients with a hypersensitivity to levetiracetam. Reactions have included anaphylaxis and angioedema [see Warnings and Precautions (5.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in more details in other sections of labeling:
- Psychiatric Reactions [see Warnings and Precautions (5.1)]
- Somnolence and Fatigue [see Warnings and Precautions (5.2)]
- Anaphylaxis and Angioedema [see Warnings and Precautions (5.3)]
- Serious Dermatological Reactions [see Warnings and Precautions (5.4)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity [see Warnings and Precautions (5.5)]
- Coordination Difficulties [see Warnings and Precautions (5.6)]
- Withdrawal Seizures [see Warnings and Precautions (5.7)]
- Hematologic Abnormalities [see Warnings and Precautions (5.8)]
- Seizure Control During Pregnancy [see Warnings and Precautions (5.9)]
8.6 Renal Impairment
Clearance of levetiracetam is decreased in patients with renal impairment and is correlated with creatinine clearance [see Clinical Pharmacology (12.3)]. Dosage adjustment is recommended for patients with impaired renal function and supplemental doses should be given to patients after dialysis [see Dosage and Administration (2.5)].
12.3 Pharmacokinetics
Equivalent doses of intravenous (IV) levetiracetam and oral levetiracetam result in equivalent Cmax, Cmin, and total systemic exposure to levetiracetam when the IV levetiracetam is administered as a 15 minute infusion.
1.4 Limitations of Use
Levetiracetam in Sodium Chloride Injection is an antiepileptic drug indicated for adult patients (16 years and older) when oral administration is temporarily not feasible.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Levetiracetam in Sodium Chloride Injection is indicated for adjunct therapy in adults (≥16 years of age) with the following seizure types when oral administration is temporarily not feasible:
5.7 Withdrawal Seizures
As with most antiepileptic drugs, levetiracetam should be withdrawn gradually because of the risk of increased seizure frequency and status epilepticus. But if withdrawal is needed because of a serious reaction, rapid discontinuation can be considered.
12.1 Mechanism of Action
The precise mechanism(s) by which levetiracetam exerts its antiepileptic effect is unknown.
A saturable and stereoselective neuronal binding site in rat brain tissue has been described for levetiracetam. Experimental data indicate that this binding site is the synaptic vesicle protein SV2A, thought to be involved in the regulation of vesicle exocytosis. Although the molecular significance of levetiracetam binding to synaptic vesicle protein SV2A is not understood, levetiracetam and related analogs showed a rank order of affinity for SV2A which correlated with the potency of their antiseizure activity in audiogenic seizure-prone mice. These findings suggest that the interaction of levetiracetam with the SV2A protein may contribute to the antiepileptic mechanism of action of the drug.
5.1 Psychiatric Reactions
In some patients levetiracetam causes behavioral abnormalities. The incidences of behavioral abnormalities in the myoclonic and primary generalized tonic-clonic seizure studies were comparable to those of the adult partial-onset seizure studies.
A total of 13.3% of adult levetiracetam-treated patients compared to 6.2% of placebo patients experienced non-psychotic behavioral symptoms (reported as aggression, agitation, anger, anxiety, apathy, depersonalization, depression, emotional lability, hostility, irritability, and nervousness).
A total of 1.7% of adult levetiracetam-treated patients discontinued treatment due to behavioral adverse events, compared to 0.2% of placebo patients. The treatment dose was reduced in 0.8% of adult levetiracetam-treated patients and in 0.5% of placebo patients.
One percent of adult levetiracetam-treated patients experienced psychotic symptoms compared to 0.2% of placebo patients.
Two (0.3%) adult levetiracetam-treated patients were hospitalized and their treatment was discontinued due to psychosis. Both events, reported as psychosis, developed within the first week of treatment and resolved within 1 to 2 weeks following treatment discontinuation.
The above psychiatric signs and symptoms should be monitored.
1.1 Partial Onset Seizures (1.1 Partial-Onset Seizures)
Levetiracetam in Sodium Chloride Injection is indicated as adjunctive therapy in the treatment of partial-onset seizures in adults with epilepsy.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Psychiatric Reactions: Behavioral abnormalities including psychotic symptoms, suicidal ideation, irritability, and aggressive behavior have been observed. Monitor patients for psychiatric signs and symptoms (5.1)
- Somnolence and Fatigue: Monitor patients for these symptoms and advise patients not to drive or operate machinery until they have gained sufficient experience on levetiracetam (5.2)
- Serious Dermatological Reactions: Discontinue Levetiracetam at the first sign of rash unless clearly not drug related. (5.4)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: Discontinue if no alternative etiology (5.5)
- Coordination Difficulties: Monitor for ataxia, abnormal gait, and incoordination. (5.6)
- Withdrawal Seizures: Levetiracetam must be gradually withdrawn (5.7)
5.2 Somnolence and Fatigue
In some patients, levetiracetam causes somnolence and fatigue. The incidences of somnolence and fatigue provided below are from controlled adult partial-onset seizure studies. In general, the incidences of somnolence and fatigue in the myoclonic and primary generalized tonicclonic studies were comparable to those of the adult partial-onset seizure studies.
In controlled trials of adult patients with epilepsy experiencing partial-onset seizures, 14.8% of levetiracetam-treated patients reported somnolence, compared to 8.4% of placebo patients. There was no clear dose response up to 3000 mg/day. In a study where there was no titration, about 45% of patients receiving 4000 mg/day reported somnolence. The somnolence was considered serious in 0.3% of the treated patients, compared to 0% in the placebo group. About 3% of levetiracetam-treated patients discontinued treatment due to somnolence, compared to 0.7% of placebo patients. In 1.4% of treated patients and in 0.9% of placebo patients the dose was reduced, while 0.3% of the treated patients were hospitalized due to somnolence.
In controlled trials of adult patients with epilepsy experiencing partial-onset seizures, 14.7% of levetiracetam-treated patients reported asthenia, compared to 9.1% of placebo patients.
Treatment was discontinued due to asthenia in 0.8% of treated patients as compared to 0.5% of placebo patients. In 0.5% of treated patients and in 0.2% of placebo patients the dose was reduced due to asthenia.
Somnolence and asthenia occurred most frequently within the first 4 weeks of treatment.
Patients should be monitored for these signs and symptoms and advised not to drive or operate machinery until they have gained sufficient experience on levetiracetam to gauge whether it adversely affects their ability to drive or operate machinery.
10.2 Management of Overdose
There is no specific antidote for overdose with levetiracetam. If indicated, elimination of unabsorbed drug should be attempted by emesis or gastric lavage; usual precautions should be observed to maintain airway. General supportive care of the patient is indicated including monitoring of vital signs and observation of the patient's clinical status. A Certified Poison Control Center should be contacted for up to date information on the management of overdose with levetiracetam.
14.1 Partial Onset Seizures (14.1 Partial-Onset Seizures)
The effectiveness of levetiracetam as adjunctive therapy (added to other antiepileptic drugs) in adults was established in three multicenter, randomized, double-blind, placebo-controlled clinical studies in patients who had refractory partial-onset seizures with or without secondary generalization. The tablet formulation was used in all these studies. In these studies, 904 patients were randomized to placebo, 1000 mg, 2000 mg, or 3000 mg/day. Patients enrolled in Study 1 or Study 2 had refractory partial-onset seizures for at least two years and had taken two or more classical AEDs. Patients enrolled in Study 3 had refractory partial-onset seizures for at least 1 year and had taken one classical AED. At the time of the study, patients were taking a stable dose regimen of at least one and could take a maximum of two AEDs. During the baseline period, patients had to have experienced at least two partial-onset seizures during each 4-week period.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For intravenous infusion only (2.1)
- Do not dilute prior to its use (2.1)
- Administer dose-specific bag intravenously over 15-minutes (2.1)
Initial Exposure to Levetiracetam
- Partial-Onset Seizures: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to a maximum recommended dose of 1500 mg twice daily (2.2).
- Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy: Initial dose is 500 mg twice daily. Increase by 500 mg twice daily every 2 weeks to the recommended daily dose of 1500 mg twice daily (2.2)
- Primary Generalized Tonic-Clonic Seizures: Initial dose is 500 mg twice daily. Increase dose by 500 mg daily every 2 weeks to the recommended dose of 1500 mg twice daily. (2.2).
Switching from or to oral Levetiracetam: The total daily dosage/frequency of levetiracetam injection should be equivalent to those of oral levetiracetam (2.3, 2.4).
Renal Impairment: Dose adjustment necessary based on creatinine clearance (2.5).
2.4 Switching to Oral Dosing
At the end of the intravenous treatment period, the patient may be switched to levetiracetam oral administration at the equivalent daily dosage and frequency of the intravenous administration.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: Levetiracetam in Sodium Chloride Injection is a clear, colorless solution, packaged in a single-dose bag and available in three strengths:
- 500 mg Levetiracetam in 0.82 % sodium chloride injection (500 mg/100 mL)
- 1000 mg Levetiracetam in 0.75 % sodium chloride injection (1000 mg/100 mL)
- 1500 mg Levetiracetam in 0.54% sodium chloride injection (1500 mg/100 mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of levetiracetam. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In addition to the adverse reactions listed above [see Adverse Reactions (6.1)], the following adverse reactions have been reported in patients receiving marketed levetiracetam worldwide. The listing is alphabetized: abnormal liver function test, acute kidney injury, agranulocytosis, anaphylaxis, angioedema, choreoathetosis, drug reaction with eosinophilia and systemic symptoms (DRESS), dyskinesia, erythema multiforme, hepatic failure, hepatitis, hyponatremia, muscular weakness, pancreatitis, pancytopenia (with bone marrow suppression identified in some of these cases), panic attack, weight loss, and worsening of seizures Alopecia has been reported with levetiracetam use; recovery was observed in majority of cases where levetiracetam was discontinued.
5.6 Coordination Difficulties
Coordination difficulties were only observed in the adult partial-onset seizure studies. A total of 3.4% of adult levetiracetam-treated patients experienced coordination difficulties, (reported as either ataxia, abnormal gait, or incoordination) compared to 1.6% of placebo patients. A total of 0.4% of patients in controlled trials discontinued levetiracetam treatment due to ataxia, compared to 0% of placebo patients. In 0.7% of treated patients and in 0.2% of placebo patients the dose was reduced due to coordination difficulties, while one of the treated patients was hospitalized due to worsening of pre-existing ataxia. These events occurred most frequently within the first 4 weeks of treatment.
Patients should be monitored for these signs and symptoms and advised not to drive or operate machinery until they have gained sufficient experience on levetiracetam to gauge whether it adversely affects their ability to drive or operate machinery.
5.8 Hematologic Abnormalities
Levetiracetam can cause hematologic abnormalities. Hematologic abnormalities occurred in clinical trials and included decreases in white blood cell (WBC), neutrophil, and red blood cells counts (RBC); decreases in hemoglobin and hematocrit; and increases in eosinophil counts.
Cases of agranulocytosis, pancytopenia, and thrombocytopenia have been reported in the postmarketing setting. A complete blood count is recommended in patients experiencing significant weakness, pyrexia, recurrent infections, or coagulation disorders.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.3 Anaphylaxis and Angioedema
Levetiracetam can cause anaphylaxis or angioedema after the first dose or at any time during treatment. Signs and symptoms in cases reported in the postmarketing setting with levetiracetam have included hypotension, hives, rash, respiratory distress, and swelling of the face, lip, mouth, eye, tongue, throat, and feet. In some reported cases, reactions were lifethreatening and required emergency treatment. If a patient develops signs or symptoms of anaphylaxis or angioedema, levetiracetam should be discontinued and the patient should seek immediate medical attention.
Levetiracetam should be discontinued permanently if a clear alternative etiology for the reaction cannot be established [see Contraindications ( 4 )].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The adverse reactions that result from levetiracetam injection use include all of those reported for levetiracetam tablets and oral solution. Equivalent doses of intravenous (IV) levetiracetam and oral levetiracetam result in equivalent Cmax, Cmin, and total systemic exposure to levetiracetam when the IV levetiracetam is administered as a 15-minute infusion.
2.3 Switching to Intravenous Dosing
When switching from oral levetiracetam, the initial total daily intravenous dosage of levetiracetam should be equivalent to the total daily dosage and frequency of oral levetiracetam.
2.7 Discontinuation of Levetiracetam
Avoid abrupt withdrawal from levetiracetam in order to reduce the risk of increased seizure frequency and status epilepticus [see Warnings and Precautions (5.7)].
5.4 Serious Dermatological Reactions
Serious dermatological reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported in patients treated with levetiracetam. The median time of onset is reported to be 14 to 17 days, but cases have been reported at least four months after initiation of treatment. Recurrence of the serious skin reactions following rechallenge with levetiracetam has also been reported. Levetiracetam should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed and alternative therapy should be considered.
5.9 Seizure Control During Pregnancy
Physiological changes may gradually decrease plasma levels of levetiracetam throughout pregnancy. This decrease is more pronounced during the third trimester. It is recommended that patients be monitored carefully during pregnancy. Close monitoring should continue through the postpartum period especially if the dose was changed during pregnancy.
2.2 Initial Exposure to Levetiracetam
Levetiracetam can be initiated with either intravenous or oral administration.
2.1 General Information – Administration
Levetiracetam in Sodium Chloride Injection is for intravenous infusion only. It is available in the following concentrations: three single-dose 100 mL bags, each containing a different total dosage of levetiracetam (500 mg [5 mg/mL], 1000 mg [10 mg/mL], or 1500 mg [15 mg/mL]).
A single-dose bag should be administered intravenously over a 15-minute IV infusion period.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Levetiracetam in Sodium Chloride Injection should not be further diluted prior to use. Any unused portion of the Levetiracetam in Sodium Chloride Injection contents should be discarded.
1.3 Primary Generalized Tonic Clonic Seizures (1.3 Primary Generalized Tonic-Clonic Seizures)
Levetiracetam in Sodium Chloride Injection is indicated as adjunctive therapy in the treatment of primary generalized tonic-clonic seizures in adults with idiopathic generalized epilepsy.
2.5 Adult Patients With Impaired Renal Function (2.5 Adult Patients with Impaired Renal Function)
Levetiracetam dosing must be individualized according to the patient's renal function status. Recommended doses and adjustment for dose for adults are shown in Table 1. To use this dosing table, an estimate of the patient's creatinine clearance (CLcr) in mL/min is needed.
|
1 Following dialysis, a 250 to 500 mg supplemental dose is recommended. |
|||
| Group | Creatinine Clearance (mL/min) | Dosage (mg) | Frequency |
| Normal | greater than 80 | 500 to 1,500 | Every 12 hours |
| Mild | 50 to 80 | 500 to 1,000 | Every 12 hours |
| Moderate | 30 to 50 | 250 to 750 | Every 12 hours |
| Severe | less than 30 | 250 to 500 | Every 12 hours |
| ESRD patients using dialysis | --- | 500 to 1,000 | 1Every 24 hours |
14.3 Primary Generalized Tonic Clonic Seizures (14.3 Primary Generalized Tonic - Clonic Seizures)
The effectiveness of levetiracetam as adjunctive therapy in patients with idiopathic generalized epilepsy experiencing primary generalized tonic-clonic (PGTC) seizures was established in one multicenter, randomized, double-blind, placebo-controlled study, conducted at 50 sites in 8 countries. Eligible patients on a stable dose of 1 or 2 antiepileptic drugs (AEDs) experiencing at least 3 PGTC seizures during the 8-week combined baseline period (at least one PGTC seizure during the 4 weeks prior to the prospective baseline period and at least one PGTC seizure during the 4-week prospective baseline period) were randomized to either levetiracetam or placebo. The 8-week combined baseline period is referred to as “baseline” in the remainder of this section. The population included 164 patients (levetiracetam N=80, placebo N=84) with idiopathic generalized epilepsy (predominately juvenile myoclonic epilepsy, juvenile absence epilepsy, childhood absence epilepsy, or epilepsy with Grand Mal seizures on awakening) experiencing primary generalized tonic-clonic seizures. Each of these syndromes of idiopathic generalized epilepsy was well represented in this patient population. Patients were titrated over 4 weeks to a target dose of 3000 mg/day for adults or a pediatric target dose of 60 mg/kg/day and treated at a stable dose of 3000 mg/day (or 60 mg/kg/day for children) over 20 weeks (evaluation period). Study drug was given in 2 equally divided doses per day.
The primary measure of effectiveness was the percent reduction from baseline in weekly PGTC seizure frequency for levetiracetam and placebo treatment groups over the treatment period (titration + evaluation periods). There was a statistically significant decrease from baseline in PGTC frequency in the levetiracetam-treated patients compared to the placebo-treated patients.
|
*Statistically significant versus placebo |
||
| Placebo (N=84) | Levetiracetam (N=78) | |
| Percentage reduction in PGTC seizure frequency | 44.6% | 77.6%* |
The percentage of patients (y-axis) who achieved ≥50% reduction in weekly seizure rates from baseline in PGTC seizure frequency over the entire randomized treatment period (titration + evaluation period) within the two treatment groups (x-axis) is presented in Figure 4.
Figure 4: Responder Rate (≥50% Reduction from Baseline) in PGTC Seizure Frequency per Week
2.6 Compatibility With Other Antiepileptic Drugs
Levetiracetam in Sodium Chloride Injection is found to be physically compatible and chemically stable for at least 24 hours when mixed with lorazepam, diazepam, and valproate sodium and stored at controlled room temperature 15° to 30°C (59° to 86°F).
There are no data to support the physical compatibility of levetiracetam injection with antiepileptic drugs that are not listed above.
1.2 Myoclonic Seizures in Patients With Juvenile Myoclonic Epilepsy (1.2 Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy)
Levetiracetam in Sodium Chloride Injection is indicated as adjunctive therapy in the treatment of myoclonic seizures in adults with juvenile myoclonic epilepsy.
14.2 Myoclonic Seizures in Patients With Juvenile Myoclonic Epilepsy (14.2 Myoclonic Seizures in Patients with Juvenile Myoclonic Epilepsy)
The effectiveness of levetiracetam as adjunctive therapy in patients with juvenile myoclonic epilepsy (JME) experiencing myoclonic seizures was established in one multicenter, randomized, double-blind, placebo-controlled study, conducted at 37 sites in 14 countries. Of the 120 patients enrolled, 113 had a diagnosis of confirmed or suspected JME. Eligible patients on a stable dose of 1 antiepileptic drug (AED) experiencing one or more myoclonic seizures per day for at least 8 days during the prospective 8-week baseline period were randomized to either levetiracetam or placebo (levetiracetam N=60, placebo N=60). Patients were titrated over 4 weeks to a target dose of 3000 mg/day and treated at a stable dose of 3000 mg/day over 12 weeks (evaluation period). Study drug was given in 2 divided doses. The primary measure of effectiveness was the proportion of patients with at least 50% reduction in the number of days per week with one or more myoclonic seizures during the treatment period (titration + evaluation periods) as compared to baseline. Table 10 displays the results for the 113 patients with JME in this study. Of 120 patients enrolled, 113 had a diagnosis of confirmed or suspected JME. The results are displayed in Table 10.
|
*Statistically significant versus placebo |
||
| Placebo (N=59) | Levetiracetam (N=54) | |
| Percentage of responders | 23.7% | 60.4%* |
10.1 Signs, Symptoms and Laboratory Findings of Acute Overdosage in Humans
The highest known dose of oral levetiracetam received in the clinical development program was 6000 mg/day. Other than drowsiness, there were no adverse reactions in the few known cases of overdose in clinical trials. Cases of somnolence, agitation, aggression, depressed level of consciousness, respiratory depression and coma were observed with levetiracetam overdoses in postmarketing use.
5.5 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)/multiorgan Hypersensitivity (5.5 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as multiorgan hypersensitivity, has been reported in patients taking antiepileptic drugs, including levetiracetam. These events can be fatal or life-threatening, particularly if diagnosis and treatment do not occur as early as possible. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis, sometimes resembling an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its expression, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Levetiracetam should be discontinued if an alternative etiology for the signs or symptoms cannot be established [see Contraindications (4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:56.608852 · Updated: 2026-03-14T22:04:33.828592