ec6f229a-331e-4b0d-91f1-056eef2eef8b
34390-5
HUMAN OTC DRUG LABEL
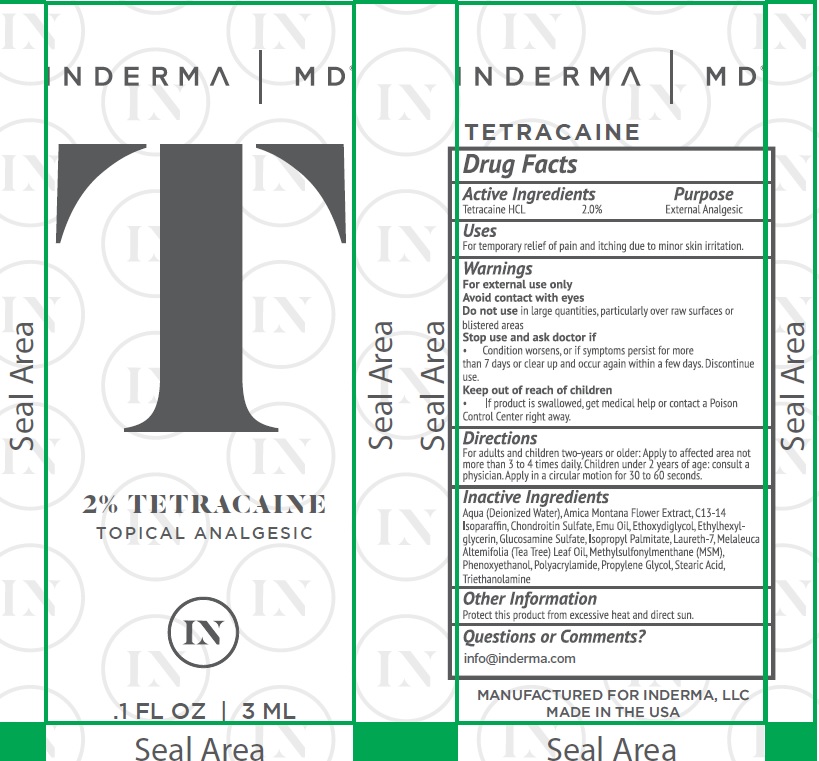
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Tetracaine 2%
Purpose
External Analgesic
Medication Information
Purpose
External Analgesic
Description
Tetracaine 2%
Uses
For temporary relief of pain and itching due to minor skin irritation.
Warnings
For external use only
Avoid contact with eyes
Do not use in large quantities, particularly over raw surfaces or blistered areas
Stop use and ask a doctor if
Condition worsens or, if symptoms persists for more than 7 days or clear up and occur again within a few days. Discontinue use
Directions
For adults and children two-years or older: Apply to affected area not more than 3 or 4 times
daily. Children under 2 years of age: consult a physician. Apply in a circular motion for 30 to 60 seconds.
Drug Facts
Product Label
Active Ingredient
Tetracaine 2%
Other Information
Protect this product from excessive heat and direct sun.
Inactive Ingredients
Aqua (Deionized Water), Arnica Montana Flower Extract, C13-14 Isoparaffin, Chrondroitin Sulfate,
Emu Oil, Ethoxydiglycol, Ethylhexyglycerin, Glucosamine Sulfate, Isopropyl Palmitate, Laureth-7, Melaleuca Alternifolia (Tea Tree) Oil, Methylsulfonylmethane (MSM), Phenoxyethanol, Polyacrylamide, Propylene Glycol, Stearic Acid, Triethanolamine.
Questions and Comments?
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
For temporary relief of pain and itching due to minor skin irritation.
Purpose
External Analgesic
Warnings
For external use only
Avoid contact with eyes
Do not use in large quantities, particularly over raw surfaces or blistered areas
Stop use and ask a doctor if
Condition worsens or, if symptoms persists for more than 7 days or clear up and occur again within a few days. Discontinue use
Directions
For adults and children two-years or older: Apply to affected area not more than 3 or 4 times
daily. Children under 2 years of age: consult a physician. Apply in a circular motion for 30 to 60 seconds.
Drug Facts
Product Label (Product label)
Active Ingredient (Active ingredient)
Tetracaine 2%
Other Information (Other information)
Protect this product from excessive heat and direct sun.
Inactive Ingredients (Inactive ingredients)
Aqua (Deionized Water), Arnica Montana Flower Extract, C13-14 Isoparaffin, Chrondroitin Sulfate,
Emu Oil, Ethoxydiglycol, Ethylhexyglycerin, Glucosamine Sulfate, Isopropyl Palmitate, Laureth-7, Melaleuca Alternifolia (Tea Tree) Oil, Methylsulfonylmethane (MSM), Phenoxyethanol, Polyacrylamide, Propylene Glycol, Stearic Acid, Triethanolamine.
Questions and Comments?
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:39.198167 · Updated: 2026-03-14T23:01:08.226420