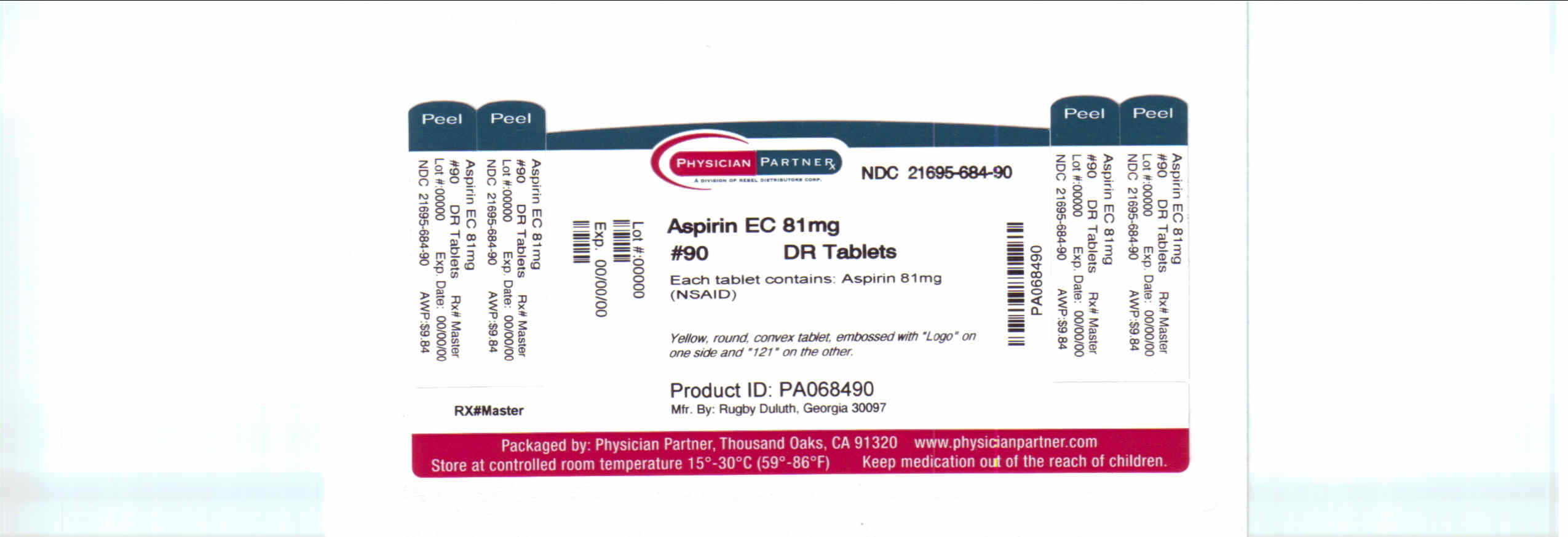
aspirin ec tablets, 81 mg
ec6b0a04-3fb3-46ab-bca6-1415d2f4e72b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Pain reliever
Description
Drug Facts
Medication Information
Active Ingredient
Aspirin 81 mg (NSAID
nonsteroidal anti-inflammatory drug
Uses
- temporarily relieves minor aches and pains
- for other uses, see your doctor, but do not use for more than 10 days without consulting your doctor because serious side effects may occur
Purpose
Pain reliever
Directions
- drink a full glass of water with each dose
- adults and children 12 years and over: take 4 to 8 tablets every 4 hours not to exceed 48 tablets in 24 hours unless directed by a doctor
- children under 12 years: consult a doctor
Other Information
- store at 25°C (77°F) excursions permitted between 15°-30°C (59°-86°F)
- use by expiration date on package
Inactive Ingredients
anhydrous lactose, carnauba wax, colloidal silicon dioxide, croscarmellose sodium, D&C yellow #10 aluminum lake, iron oxide ochre, methacrylic acid copolymer, microcrystalline cellulose, polysorbate 80, simethicone, sodium hydroxide, sodium lauryl sulfate, talc, titanium dioxide, triethyl citrate
Description
Drug Facts
Questions?
call 1-888-838-2872, weekdays, 8 AM-5 PM ET
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- have bloody or black stools
- vomit blood
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- any new symptoms appear
- ringing in the ears or a loss of hearing occurs
Section 50568-5
Ask a doctor or pharmacist before use if you are
- taking a prescription drug for diabetes, gout, or arthritis
- taking any other drug
- under a doctor's care for any serious condition
Section 50569-3
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- you are taking a diuretic
- you have asthma
- you have not been drinking fluids
- you have lost a lot of fluid due to vomiting or diarrhea
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Do Not Use
- if you are allergic to aspirin or any other pain reliever/fever reducer
Allergy Alert
Aspirin may cause a severe allergic reaction, which may include:
- hives
- facial swelling
- shock
- asthma (wheezing)
Reye's Syndrome
Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Stomach Bleeding Warning
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Principal Display Panel 81 Mg Bottle Label
Structured Label Content
Active Ingredient (Active ingredient (in each tablet))
Aspirin 81 mg (NSAID
nonsteroidal anti-inflammatory drug
Uses
- temporarily relieves minor aches and pains
- for other uses, see your doctor, but do not use for more than 10 days without consulting your doctor because serious side effects may occur
Purpose
Pain reliever
Directions
- drink a full glass of water with each dose
- adults and children 12 years and over: take 4 to 8 tablets every 4 hours not to exceed 48 tablets in 24 hours unless directed by a doctor
- children under 12 years: consult a doctor
Other Information (Other information)
- store at 25°C (77°F) excursions permitted between 15°-30°C (59°-86°F)
- use by expiration date on package
Inactive Ingredients (Inactive ingredients)
anhydrous lactose, carnauba wax, colloidal silicon dioxide, croscarmellose sodium, D&C yellow #10 aluminum lake, iron oxide ochre, methacrylic acid copolymer, microcrystalline cellulose, polysorbate 80, simethicone, sodium hydroxide, sodium lauryl sulfate, talc, titanium dioxide, triethyl citrate
Questions?
call 1-888-838-2872, weekdays, 8 AM-5 PM ET
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- have bloody or black stools
- vomit blood
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- any new symptoms appear
- ringing in the ears or a loss of hearing occurs
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are
- taking a prescription drug for diabetes, gout, or arthritis
- taking any other drug
- under a doctor's care for any serious condition
Section 50569-3 (50569-3)
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- you are taking a diuretic
- you have asthma
- you have not been drinking fluids
- you have lost a lot of fluid due to vomiting or diarrhea
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Do Not Use (Do not use)
- if you are allergic to aspirin or any other pain reliever/fever reducer
Allergy Alert (Allergy alert)
Aspirin may cause a severe allergic reaction, which may include:
- hives
- facial swelling
- shock
- asthma (wheezing)
Reye's Syndrome (Reye's syndrome)
Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Stomach Bleeding Warning (Stomach bleeding warning)
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Principal Display Panel 81 Mg Bottle Label (PRINCIPAL DISPLAY PANEL - 81 mg Bottle Label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:12.751372 · Updated: 2026-03-14T22:52:35.540850