Diphenhydramine Hydrochloride And Zinc Acetate
ec0c9602-35cc-4bb6-b853-794a8b5bf42e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Description
Drug Facts
Uses
- temporarily relieves pain and itching associated with:
- insect bites
- minor burns
- sunburn
- minor skin irritations
- minor cuts
- scrapes
- rashes due to poison ivy, poison oak, and poison sumac
- dries the oozing and weeping of poison ivy, poison oak, and poison sumac
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7
When using this productavoid contact with eyes
Section 55105-1
| Active ingredients | Purpose |
|---|---|
| Diphenhydramine hydrochloride 2% | Topical analgesic |
| Zinc acetate 0.1% | Skin protectant |
Warnings
For external use only
Directions
- do not use more often than directed
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
Do Not Use
- on large areas of the body
- with any other product containing diphenhydramine, even one taken by mouth
Questions?
call 833-358-6431Monday to Friday 9:00am to 7:00pm EST
Other Information
- to open: unscrew cap, use pointed end of cap to puncture seal.
- store at 20° to 25°C (68° to 77°F)
- see carton or tube crimp for lot number and expiration date
Inactive Ingredients
cetyl alcohol, glyceryl stearate, glyceryl stearate/PEG-100 stearate, methylparaben, propylene glycol, propylparaben and purified water
Ask A Doctor Before Use
- on chicken pox
- on measles
Stop Use and Ask A Doctor If
- condition worsens or does not improve within 7 days
- symptoms persist for more than 7 days or clear up and occur again within a few days
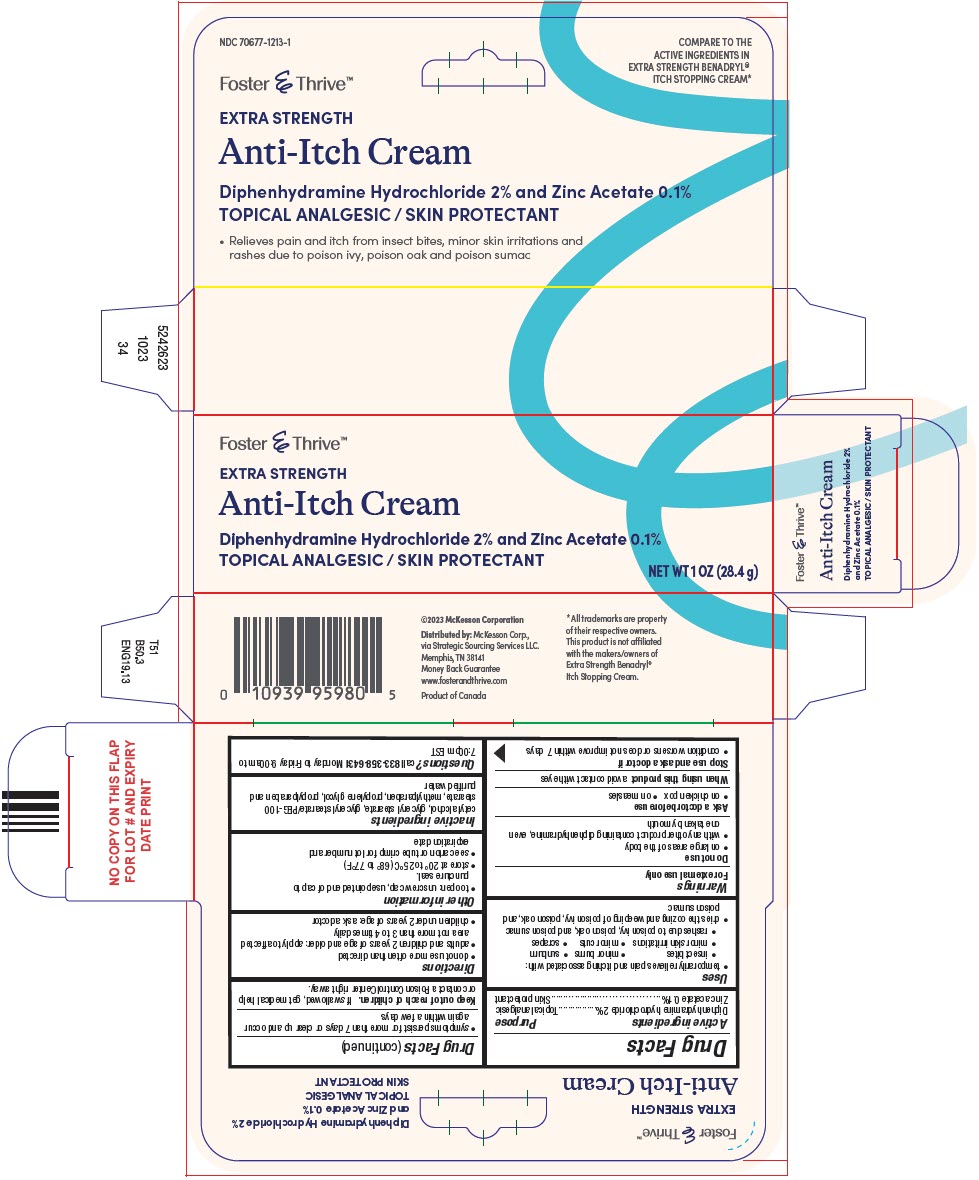
Principal Display Panel 28.4 G Tube Carton
Foster & Thrive™
EXTRA STRENGTH
Anti-Itch Cream
Diphenhydramine Hydrochloride 2% and Zinc Acetate 0.1%
TOPICAL ANALGESIC / SKIN PROTECTANT
NET WT 1 OZ (28.4 g)
Structured Label Content
Uses
- temporarily relieves pain and itching associated with:
- insect bites
- minor burns
- sunburn
- minor skin irritations
- minor cuts
- scrapes
- rashes due to poison ivy, poison oak, and poison sumac
- dries the oozing and weeping of poison ivy, poison oak, and poison sumac
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7 (50567-7)
When using this productavoid contact with eyes
Section 55105-1 (55105-1)
| Active ingredients | Purpose |
|---|---|
| Diphenhydramine hydrochloride 2% | Topical analgesic |
| Zinc acetate 0.1% | Skin protectant |
Warnings
For external use only
Directions
- do not use more often than directed
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
Do Not Use (Do not use)
- on large areas of the body
- with any other product containing diphenhydramine, even one taken by mouth
Questions?
call 833-358-6431Monday to Friday 9:00am to 7:00pm EST
Other Information (Other information)
- to open: unscrew cap, use pointed end of cap to puncture seal.
- store at 20° to 25°C (68° to 77°F)
- see carton or tube crimp for lot number and expiration date
Inactive Ingredients (Inactive ingredients)
cetyl alcohol, glyceryl stearate, glyceryl stearate/PEG-100 stearate, methylparaben, propylene glycol, propylparaben and purified water
Ask A Doctor Before Use (Ask a doctor before use)
- on chicken pox
- on measles
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens or does not improve within 7 days
- symptoms persist for more than 7 days or clear up and occur again within a few days
Principal Display Panel 28.4 G Tube Carton (PRINCIPAL DISPLAY PANEL - 28.4 g Tube Carton)
Foster & Thrive™
EXTRA STRENGTH
Anti-Itch Cream
Diphenhydramine Hydrochloride 2% and Zinc Acetate 0.1%
TOPICAL ANALGESIC / SKIN PROTECTANT
NET WT 1 OZ (28.4 g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:31.618886 · Updated: 2026-03-14T23:14:04.878170