79740-011
ebe98b71-1f34-4d40-e053-2a95a90a4c02
34390-5
HUMAN OTC DRUG LABEL
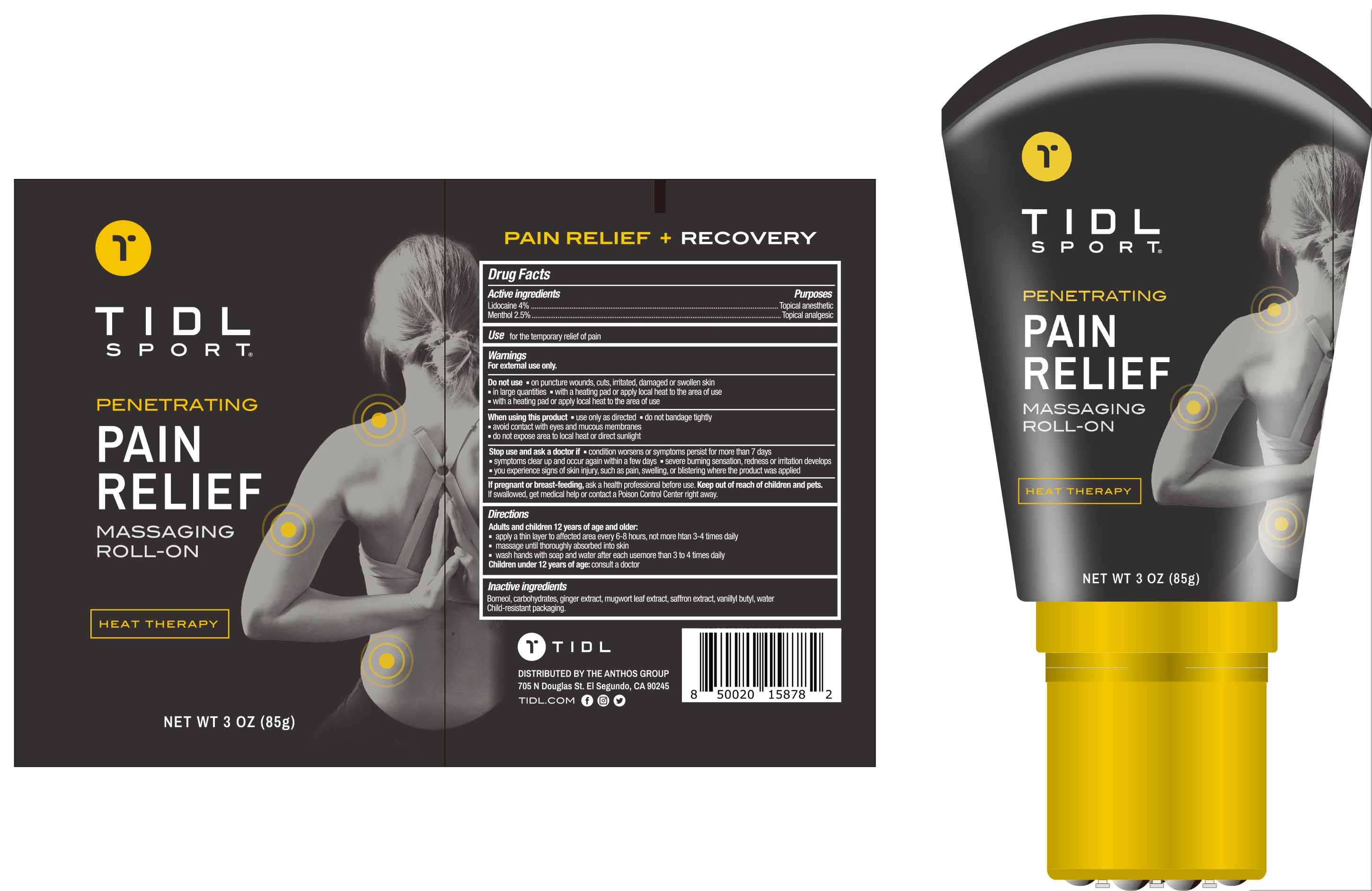
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%, Menthol 2.5%
Medication Information
Warnings and Precautions
Wamings
For extemal use only .
Indications and Usage
Use for the temporary relief of pain
Description
Lidocaine 4%, Menthol 2.5%
Section 50565-1
If pregnant or breast-feeding , ask a health professional before use . Keep out of reach of children and pets. If swallowed , get medical help or contact a Poison Control Center right away
Section 51727-6
Ginger Extract
Saffron Extract
Water
Mugwort Leaf Extract
Vanillyn Butyl
Borneol
Carbohydrates
Section 51945-4
79740-011-01
79740-011-02
Section 55105-1
Topical anesthetic
Section 55106-9
Lidocaine 4%,
Menthol 2.5%
Directions
Adults and children 12 years of age and older
apply a thin layer to affected area every 6-8 hours , not more han 3-4 times daily
massage until thoroughly absorbed into skin
wash hands with soap and water after each usemore than 3 to 4 times daily
Children under 12 years of age : consult a doctor
Structured Label Content
Indications and Usage (34067-9)
Use for the temporary relief of pain
Warnings and Precautions (34071-1)
Wamings
For extemal use only .
Section 50565-1 (50565-1)
If pregnant or breast-feeding , ask a health professional before use . Keep out of reach of children and pets. If swallowed , get medical help or contact a Poison Control Center right away
Section 51727-6 (51727-6)
Ginger Extract
Saffron Extract
Water
Mugwort Leaf Extract
Vanillyn Butyl
Borneol
Carbohydrates
Section 51945-4 (51945-4)
79740-011-01
79740-011-02
Section 55105-1 (55105-1)
Topical anesthetic
Section 55106-9 (55106-9)
Lidocaine 4%,
Menthol 2.5%
Directions
Adults and children 12 years of age and older
apply a thin layer to affected area every 6-8 hours , not more han 3-4 times daily
massage until thoroughly absorbed into skin
wash hands with soap and water after each usemore than 3 to 4 times daily
Children under 12 years of age : consult a doctor
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:21.191931 · Updated: 2026-03-14T23:00:44.685211