Altalube Ointment
eb65004c-c3a7-4079-bd75-f2857d30c68a
34390-5
HUMAN OTC DRUG LABEL
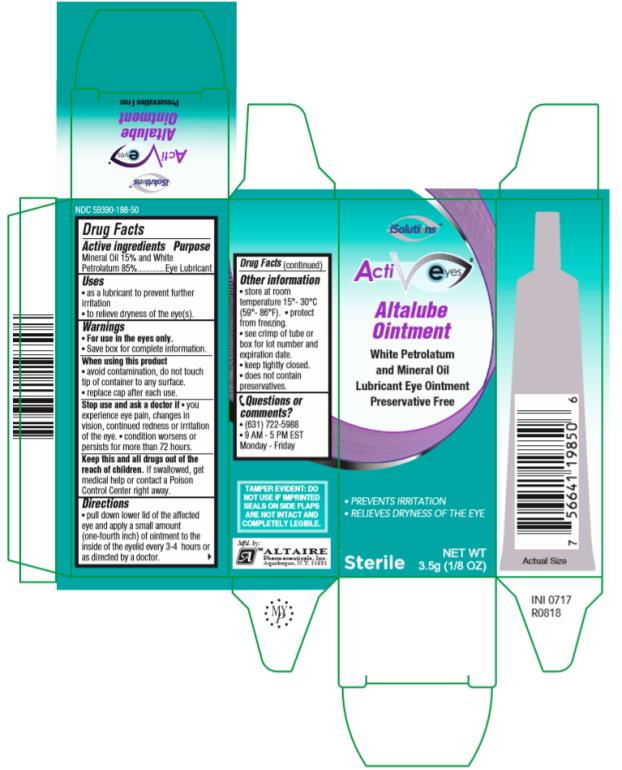
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ActivEyes Altalube Oin t ment 3.5g NDC 59390-198-50 Drug Facts
Purpose
Eye Lubricant
Medication Information
Purpose
Eye Lubricant
Description
ActivEyes Altalube Oin t ment 3.5g NDC 59390-198-50 Drug Facts
Uses
• as a lubricant to prevent further irritation • to relieve dryness of the eye(s).
Section 42229-5
ActivEyes
Altalube Oin t ment 3.5g
NDC 59390-198-50
Drug Facts
Warnings
• Save box for complete information. • For use in the eyes only.
Directions
• pull down lower lid of the affected eye and apply a small amount (one-fourth inch) of ointment to the inside of the eyelid every 3-4 hours or as directed by a doctor.
Other Information
• store at room temperature 15°-30°C (59°-86°F). • protect from freezing.
• see crimp of tube or box for lot number and expiration date.
• keep tightly closed.
• does not contain preservatives.
Active Ingredients
Mineral Oil 15% and White Petrolatum 85%
Inactive Ingredients
None remove
Questions Or Comments?
• (631) 722-5988• 9AM – 5 PM EST Monday- Friday
Principal Display Panel
ActiEyes Altalube OintmentWhite Petrolatum and Mineral OilLubricant Eye Ointment Preservative FreeSterileNET WT3.5g (1/8 OZ)
When Using This Product
• avoid contamination, do not touch tip of container to any surface.
• replace cap after each use.
Stop Use and Ask A Doctor If
• you experience eye pain, changes in vision, continued redness or irritation of the eye. • condition worsens or persists for more than 72 hours.
Keep This and All Drugs Out of the Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
• as a lubricant to prevent further irritation • to relieve dryness of the eye(s).
Section 42229-5 (42229-5)
ActivEyes
Altalube Oin t ment 3.5g
NDC 59390-198-50
Drug Facts
Purpose
Eye Lubricant
Warnings
• Save box for complete information. • For use in the eyes only.
Directions
• pull down lower lid of the affected eye and apply a small amount (one-fourth inch) of ointment to the inside of the eyelid every 3-4 hours or as directed by a doctor.
Other Information (Other information)
• store at room temperature 15°-30°C (59°-86°F). • protect from freezing.
• see crimp of tube or box for lot number and expiration date.
• keep tightly closed.
• does not contain preservatives.
Active Ingredients (Active ingredients)
Mineral Oil 15% and White Petrolatum 85%
Inactive Ingredients
None remove
Questions Or Comments? (Questions or comments?)
• (631) 722-5988• 9AM – 5 PM EST Monday- Friday
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
ActiEyes Altalube OintmentWhite Petrolatum and Mineral OilLubricant Eye Ointment Preservative FreeSterileNET WT3.5g (1/8 OZ)
When Using This Product (When using this product)
• avoid contamination, do not touch tip of container to any surface.
• replace cap after each use.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
• you experience eye pain, changes in vision, continued redness or irritation of the eye. • condition worsens or persists for more than 72 hours.
Keep This and All Drugs Out of the Reach of Children. (Keep this and all drugs out of the reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:30.292306 · Updated: 2026-03-14T23:02:05.629064