These Highlights Do Not Include All The Information Needed To Use Annovera Safely And Effectively.
eb18194f-2021-41fa-8bdf-31e0c0eb646b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cigarette smoking increases the risk of serious cardiovascular events from combination hormonal contraceptive (CHC) use. This risk increases with age, particularly in females over 35 years of age, and with the number of cigarettes smoked. For this reason, CHC should not be used by females who are over 35 years of age and smoke. [See Contraindications (4) and Warnings and Precautions (5.5) ].
Indications and Usage
ANNOVERA is indicated for use by females of reproductive potential to prevent pregnancy.
Dosage and Administration
One ANNOVERA is inserted in the vagina. The vaginal system must remain in place continuously for 3 weeks (21 days) followed by a 1-week (7-day) vaginal system-free interval. One vaginal system provides contraception for thirteen 28-day cycles (1 year). ( 2 )
Warnings and Precautions
Thrombotic Disorders and Other Vascular Problems: Stop ANNOVERA if a thrombotic or thromboembolic event occurs. Stop ANNOVERA at least 4 weeks before and through 2 weeks after major surgery. Start ANNOVERA no earlier than 4 weeks after delivery, in females who are not breastfeeding. Consider cardiovascular risk factors before initiating in all females, particularly those over 35 years. ( 5.1 , 5.5 ) Liver Disease: Discontinue if jaundice occurs. ( 5.2 ) Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment: Stop ANNOVERA prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir. ANNOVERA can be restarted 2 weeks following completion of this regimen. ( 5.3 ) Hypertension: Do not prescribe ANNOVERA for females with uncontrolled hypertension or hypertension with vascular disease. If used in females with well-controlled hypertension, monitor blood pressure and stop use if blood pressure rises significantly. ( 5.4 ) Carbohydrate and lipid metabolic effects: Monitor glucose in pre-diabetic and diabetic females taking ANNOVERA. Consider an alternate contraceptive method for females with uncontrolled dyslipidemias. ( 5.7 ) Headache: Evaluate significant change in headaches and discontinue ANNOVERA if indicated. ( 5.8 ) Bleeding Irregularities and Amenorrhea: May cause irregular bleeding or amenorrhea. Evaluate for other causes if irregular bleeding or amenorrhea persists. ( 5.9 )
Contraindications
ANNOVERA is contraindicated in females who are known to have or develop the following conditions: A high risk of arterial or venous thrombotic diseases. Examples include females who are known to: - Smoke, if over age 35 [see Boxed Warning and Warnings and Precautions (5.1) ]. - Have current or history of deep vein thrombosis or pulmonary embolism [see Warnings and Precautions (5.1) ] . - Have cerebrovascular disease [see Warnings and Precautions (5.1) ]. - Have coronary artery disease [see Warnings and Precautions (5.1) ]. - Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see Warnings and Precautions (5.1) ] . - Have inherited or acquired hypercoagulopathies [see Warnings and Precautions (5.1) ] . - Have uncontrolled hypertension or hypertension with vascular disease [see Warnings and Precautions (5.4) ] . - Have diabetes mellitus and are over age 35, diabetes mellitus with hypertension or vascular disease, or other end-organ damage, or diabetes mellitus of >20 years duration [see Warnings and Precautions (5.5) , (5.7) ] . - Have headaches with focal neurological symptoms, migraine headaches with aura, or are over age 35 with any migraine headaches [see Warnings and Precautions (5.8) ] . Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive [see Warnings and Precautions (5.11) ] . Liver tumors, acute hepatitis, or severe (decompensated) cirrhosis [see Warnings and Precautions (5.2) and Use in Specific Populations (8.6) ]. Undiagnosed abnormal uterine bleeding [see Warnings and Precautions (5.9) ] . Hypersensitivity to any of the components of ANNOVERA. Hypersensitivity reactions reported include: throat constriction, facial edema, urticaria, hives, and wheezing [see Adverse Reactions (6.1) ] . Use of Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine transaminase (ALT) elevations [see Warnings and Precautions (5.3) ].
Adverse Reactions
The following adverse reactions are described elsewhere in other sections of the labeling: Serious cardiovascular events and stroke [see Boxed Warning and Warnings and Precautions (5.1) ] Vascular events [see Warnings and Precautions (5.1) ] Liver disease [see Warnings and Precautions (5.2) ]
Drug Interactions
The sections below provide information on substances for which data on drug interactions with CHCs are available. There is little information available about the clinical effect of most drug interactions that may affect ANNOVERA. However, based on the known pharmacokinetic effects of these drugs, clinical strategies to minimize any potential adverse effect on contraceptive effectiveness or safety are suggested. Consult the approved product labeling of all concurrently used drugs to obtain further information about interactions with ANNOVERA or the potential for metabolic enzyme or transporter system alterations.
How Supplied
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white vaginal system. The vaginal system body has an overall diameter of 56 mm and a cross-sectional diameter of 8.4 mm. When placed inside the vagina, each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate and 0.013 mg/day of ethinyl estradiol over 21-day in-use period of each cycle for up to 13 cycles (total 273 days). Each cycle is 28 days, with 21 days in and 7 days out. Each ANNOVERA is individually packaged in an aluminum pouch. The pouch consists of a laminate from outside to inside of polyester, aluminum foil, and polyethylene. First insertion of ANNOVERA must be prior to the date of expiration. Discard one year after first insertions. A black compact case is provided with the drug product for storage of ANNOVERA by patients during each 7-day vaginal system-out interval. The vaginal system should be placed in the compact case after 13 cycles of use and discarded via a drug take-back option if one is available. If a take-back option is unavailable, then discard in the waste receptacle out of reach of children and pets. The vaginal system should NOT be flushed down the toilet. See www.fda.gov/drugdisposal for more information about disposal of medicines. Each box contains 1 ANNOVERA vaginal system in a pouch and 1 storage case. NDC 68308-752-01
Medication Information
Warnings and Precautions
Thrombotic Disorders and Other Vascular Problems: Stop ANNOVERA if a thrombotic or thromboembolic event occurs. Stop ANNOVERA at least 4 weeks before and through 2 weeks after major surgery. Start ANNOVERA no earlier than 4 weeks after delivery, in females who are not breastfeeding. Consider cardiovascular risk factors before initiating in all females, particularly those over 35 years. ( 5.1 , 5.5 ) Liver Disease: Discontinue if jaundice occurs. ( 5.2 ) Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment: Stop ANNOVERA prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir. ANNOVERA can be restarted 2 weeks following completion of this regimen. ( 5.3 ) Hypertension: Do not prescribe ANNOVERA for females with uncontrolled hypertension or hypertension with vascular disease. If used in females with well-controlled hypertension, monitor blood pressure and stop use if blood pressure rises significantly. ( 5.4 ) Carbohydrate and lipid metabolic effects: Monitor glucose in pre-diabetic and diabetic females taking ANNOVERA. Consider an alternate contraceptive method for females with uncontrolled dyslipidemias. ( 5.7 ) Headache: Evaluate significant change in headaches and discontinue ANNOVERA if indicated. ( 5.8 ) Bleeding Irregularities and Amenorrhea: May cause irregular bleeding or amenorrhea. Evaluate for other causes if irregular bleeding or amenorrhea persists. ( 5.9 )
Indications and Usage
ANNOVERA is indicated for use by females of reproductive potential to prevent pregnancy.
Dosage and Administration
One ANNOVERA is inserted in the vagina. The vaginal system must remain in place continuously for 3 weeks (21 days) followed by a 1-week (7-day) vaginal system-free interval. One vaginal system provides contraception for thirteen 28-day cycles (1 year). ( 2 )
Contraindications
ANNOVERA is contraindicated in females who are known to have or develop the following conditions: A high risk of arterial or venous thrombotic diseases. Examples include females who are known to: - Smoke, if over age 35 [see Boxed Warning and Warnings and Precautions (5.1) ]. - Have current or history of deep vein thrombosis or pulmonary embolism [see Warnings and Precautions (5.1) ] . - Have cerebrovascular disease [see Warnings and Precautions (5.1) ]. - Have coronary artery disease [see Warnings and Precautions (5.1) ]. - Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see Warnings and Precautions (5.1) ] . - Have inherited or acquired hypercoagulopathies [see Warnings and Precautions (5.1) ] . - Have uncontrolled hypertension or hypertension with vascular disease [see Warnings and Precautions (5.4) ] . - Have diabetes mellitus and are over age 35, diabetes mellitus with hypertension or vascular disease, or other end-organ damage, or diabetes mellitus of >20 years duration [see Warnings and Precautions (5.5) , (5.7) ] . - Have headaches with focal neurological symptoms, migraine headaches with aura, or are over age 35 with any migraine headaches [see Warnings and Precautions (5.8) ] . Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive [see Warnings and Precautions (5.11) ] . Liver tumors, acute hepatitis, or severe (decompensated) cirrhosis [see Warnings and Precautions (5.2) and Use in Specific Populations (8.6) ]. Undiagnosed abnormal uterine bleeding [see Warnings and Precautions (5.9) ] . Hypersensitivity to any of the components of ANNOVERA. Hypersensitivity reactions reported include: throat constriction, facial edema, urticaria, hives, and wheezing [see Adverse Reactions (6.1) ] . Use of Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine transaminase (ALT) elevations [see Warnings and Precautions (5.3) ].
Adverse Reactions
The following adverse reactions are described elsewhere in other sections of the labeling: Serious cardiovascular events and stroke [see Boxed Warning and Warnings and Precautions (5.1) ] Vascular events [see Warnings and Precautions (5.1) ] Liver disease [see Warnings and Precautions (5.2) ]
Drug Interactions
The sections below provide information on substances for which data on drug interactions with CHCs are available. There is little information available about the clinical effect of most drug interactions that may affect ANNOVERA. However, based on the known pharmacokinetic effects of these drugs, clinical strategies to minimize any potential adverse effect on contraceptive effectiveness or safety are suggested. Consult the approved product labeling of all concurrently used drugs to obtain further information about interactions with ANNOVERA or the potential for metabolic enzyme or transporter system alterations.
How Supplied
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white vaginal system. The vaginal system body has an overall diameter of 56 mm and a cross-sectional diameter of 8.4 mm. When placed inside the vagina, each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate and 0.013 mg/day of ethinyl estradiol over 21-day in-use period of each cycle for up to 13 cycles (total 273 days). Each cycle is 28 days, with 21 days in and 7 days out. Each ANNOVERA is individually packaged in an aluminum pouch. The pouch consists of a laminate from outside to inside of polyester, aluminum foil, and polyethylene. First insertion of ANNOVERA must be prior to the date of expiration. Discard one year after first insertions. A black compact case is provided with the drug product for storage of ANNOVERA by patients during each 7-day vaginal system-out interval. The vaginal system should be placed in the compact case after 13 cycles of use and discarded via a drug take-back option if one is available. If a take-back option is unavailable, then discard in the waste receptacle out of reach of children and pets. The vaginal system should NOT be flushed down the toilet. See www.fda.gov/drugdisposal for more information about disposal of medicines. Each box contains 1 ANNOVERA vaginal system in a pouch and 1 storage case. NDC 68308-752-01
Description
Cigarette smoking increases the risk of serious cardiovascular events from combination hormonal contraceptive (CHC) use. This risk increases with age, particularly in females over 35 years of age, and with the number of cigarettes smoked. For this reason, CHC should not be used by females who are over 35 years of age and smoke. [See Contraindications (4) and Warnings and Precautions (5.5) ].
Section 42229-5
Limitations of Use
ANNOVERA has not been adequately studied in females with a BMI >29 kg/m2.
Section 42230-3
| Patient Information ANNOVERA® (ann-o-VER-ah) (segesterone acetate and ethinyl estradiol vaginal system) |
|||||
|---|---|---|---|---|---|
| Read this Patient Information carefully before you decide if ANNOVERA is right for you. This information does not take the place of talking with your gynecologist or other healthcare provider who specializes in women's health. If you have any questions about ANNOVERA, ask your healthcare provider. You should also learn about other birth control methods to choose the one that is best for you. | |||||
|
What is the most important information I should know about ANNOVERA?
|
|||||
| ANNOVERA does not protect against HIV infection (AIDS) and other sexually transmitted infections (STIs). | |||||
|
What is ANNOVERA?
ANNOVERA is a hormone-releasing system used by females who are able to become pregnant to prevent pregnancy. You insert ANNOVERA into your vagina. ANNOVERA is in the shape of a ring that is reusable for 1 year. The vaginal system is made of silicone and contains two female hormones that are slowly released into your vagina and then enter your blood. One hormone is an estrogen called ethinyl estradiol. The other hormone is a progestin called segesterone acetate. |
|||||
|
How well does ANNOVERA work for contraception?
Your chance of getting pregnant depends on how well you follow the directions for using the vaginal system. The more carefully you follow the directions, the less chance you have of getting pregnant. Based on the results of two clinical studies that lasted 12 months, about 2 to 4 women out of 100 women may get pregnant during the first year they use ANNOVERA. ANNOVERA was designed to be reused for 1 year. Replace ANNOVERA at the end of 1 year if you choose to continue using ANNOVERA. There is not enough hormone left in the vaginal system to provide you effective birth control after 1 year of use. The following chart shows the chance of getting pregnant for women who use different methods of birth control. Each box on the chart contains a list of birth control methods that are similar in how well they work to prevent pregnancy. The most effective methods are at the top of the chart. The box at the bottom of the chart shows the chance of getting pregnant for women who do not use birth control and are trying to get pregnant. ANNOVERA, a vaginal system with hormones, is in the second box from the top of the chart.
|
|||||
|
Who should not use ANNOVERA? Do not use ANNOVERA if you:
|
|||||
|
What should I tell my healthcare provider before using ANNOVERA? Before you use ANNOVERA tell your healthcare provider if you:
Do not use any vaginal products such as oil-based suppositories, oil-based creams, or oil-based gels while the vaginal system is in your vagina. Do not use any vaginal lubricants that have silicone or oil in them. Water-based lubricants are ok to use. Be sure to read the ingredients on the label carefully before you buy a vaginal lubricant. Some medicines and grapefruit juice may increase the level of ethinyl estradiol in your blood if used together, including:
Females who take thyroid hormone replacement medicine or corticosteroid replacement medicine may need increased doses of their thyroid hormone or cortisol medicines. Ask your healthcare provider if you are not sure if you take any of the medicines listed above. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. Talk to your healthcare provider before you start taking a new medicine. |
|||||
How should I use ANNOVERA?
After you insert ANNOVERA for the first time, you will remove it at the end of week 3 and leave it out for 7 days. You will reinsert ANNOVERA at the end of week 4 of each 4- week cycle. You will repeat this pattern with ANNOVERA for up to 13 cycles. Scheduled insertions and removals of ANNOVERA should be at about the same time of the day and the same day of the week for each monthly cycle.
|
|||||
| Schedule | |||||
| Cycle 1 |
Put vaginal system in → (vaginal system change day) |
Day 1 |
Weeks 1, 2, and 3
Days 1-21 |
||
|
Take vaginal system out→ (vaginal system change day) |
Day 22 |
Week 4
Days 22-28 |
|||
|
Cycle 2 (repeat for Cycles 3-13) |
Put vaginal system in→ (vaginal system change day) |
Day 1 |
Weeks 1, 2, and 3
Days 1-21 |
||
|
Take vaginal system out→ (vaginal system change day) |
Day 22 |
Week 4
Days 22-28 |
|||
|
|||||
|
What happens if I am off schedule using the vaginal system?
It is very important to follow the schedule every cycle and remove and insert ANNOVERA on your vaginal system-in and vaginal system-out days at about the same time. If you take the vaginal system out too soon or put it back in too late, your chance of getting pregnant is higher. If you are more than 1 or 2 days off schedule for vaginal system insertion you will need to use new vaginal system-in and vaginal system-out days for the remaining cycles of use. You will also need to use back-up contraception, such as condoms or spermicide, for the first 7 days of the new schedule if you have sexual intercourse. |
|||||
| If you put the vaginal system back in: | Then: | ||||
| Too early, after it had been out for only 5 or 6 days. | Keep the vaginal system in for at least 3 weeks (21 days). You may keep it in up to your normal vaginal system-out day. | ||||
| Too late, after it had been out for more than 7 days. | Put the vaginal system back in right away. You will now have a new vaginal system-in day. You must use condoms or spermicide as back-up contraception for the next 7 days when you have sexual intercourse. | ||||
| If you took the vaginal system out: | Then: | ||||
| Too early, after it had been in for only 19 or 20 days. | Leave the vaginal system out for 1 week. Put it back in after the week is over, as you would normally do. You may then keep it in up to your normal vaginal system-out day, 22 or 23 days after you put the vaginal system in. | ||||
| Too late, after it had been in for 22 or 23 days. | Remove the vaginal system as soon as you realize this. Then reinsert the vaginal system 7 days later. | ||||
| During the 21 days of continuous use, if ANNOVERA is out of the vagina for more than 2 continuous hours or more than 2 cumulative hours (multiple inadvertent removals or expulsions adding up to 2 hours), then back-up contraception such as male condoms or spermicide should be used until the vaginal system has been in the vagina for 7 consecutive days. | |||||
|
What if I miss my menstrual period or if I think I am pregnant?
It is possible that you are pregnant if you miss your scheduled period (no bleeding on the 7 days that the vaginal system is out). Tell your healthcare provider right away that you have missed your period. Also, tell your healthcare provider if you have symptoms of pregnancy such as morning sickness or unusual breast tenderness. It is important that your healthcare provider check you to find out if you are pregnant. You may need a pregnancy test to determine if you are pregnant. Do not remove the vaginal system until you are certain you are pregnant. Stop using ANNOVERA if your healthcare provider tells you that you are pregnant. |
|||||
|
What are the possible side effects of ANNOVERA? ANNOVERA can cause serious side effects, including:
See: "What is the most important information I should know about ANNOVERA?"
|
|||||
|
|
||||
|
|||||
|
|
||||
| This is not a complete list of possible side effects. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Mayne Pharma at 1-844-825-8500. No serious problems have been reported from a hormonal contraceptive overdose. |
|||||
|
Does hormonal birth control cause cancer?
It is not known if hormonal birth control causes breast cancer. Some studies, but not all, suggest that there could be a slight increase in the risk of breast cancer among current users with longer duration of use. If you have breast cancer now, or have had it in the past, do not use hormonal birth control because some breast cancers are sensitive to hormones. Women who use hormonal contraceptives may have a slightly higher chance of getting cervical cancer. However, this may be due to other reasons such as having more sexual partners. |
|||||
|
What should I know about my period when taking ANNOVERA?
When you take ANNOVERA you should expect to have regular 28-day cycles. Each period is likely to last about 5 days. You may have bleeding or spotting between your scheduled periods especially during the first cycle. This bleeding or spotting tends to decrease after the first cycle. Do not stop taking ANNOVERA because of this bleeding or spotting. If the spotting continues for more than 7 consecutive days or if the bleeding is unusually heavy, call your healthcare provider. Tell your healthcare provider if you do not have your period. |
|||||
|
What if I want to become pregnant?
You may stop using ANNOVERA whenever you wish. Consider a visit with your healthcare provider for a pre-pregnancy checkup before you stop taking ANNOVERA. |
|||||
|
General information about the safe and effective use of ANNOVERA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ANNOVERA for a condition for which it was not prescribed. Do not give ANNOVERA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about ANNOVERA that is written for health professionals. |
|||||
|
What if I have other questions?
If you have concerns or questions, ask your healthcare provider. |
|||||
|
What are the ingredients in ANNOVERA?
Active ingredients: segesterone acetate and ethinyl estradiol. Inactive ingredients: dibutyltin dilaurate, silicone elastomers, silicone medical adhesive, and titanium dioxide. |
Section 43683-2
| Warnings and Precautions (5.11) | 04/2022 |
5.8 Headache
ANNOVERA is contraindicated in females who have headaches with focal neurological symptoms or have migraine headaches with aura, and in females over age 35 years who have migraine headaches with or without aura [see Contraindications (4)].
If a woman taking ANNOVERA develops new headaches that are recurrent, persistent, or severe, evaluate the cause and discontinue ANNOVERA if indicated.
Consider discontinuation of ANNOVERA in the case of increased frequency or severity of migraine during CHC use (which may be prodromal of a cerebrovascular event) [see Contraindications (4)].
10 Overdosage
There have been no reports of serious ill effects from overdose of CHCs. Overdosage may cause withdrawal bleeding in females and nausea. In case of suspected overdose, all ANNOVERA vaginal systems should be removed and symptomatic treatment given.
15 References
-
1.Dinger, J, Mohner S, Heinemann K. Cardiovascular risk associated with the use of an etonogestrel- containing vaginal ring. Obstetrics & Gynecology 2013; 122(4): 800-808.
-
2.Sidney, S, Cheetham TC, Connell FA, Oullet-Hellstrom R, Graham DJ, Davis D,, Recent combined hormonal contraceptives (CHCs) and the risk of thromboembolism and other cardiovascular events in new users. Contraception 2013; 87: 93–100.
-
3.Combined hormonal contraceptives (CHCs) and the risk of cardiovascular endpoints. Sidney, S. (primary author) http://www.fda.gov/downloads/Drugs/DrugSafety/UCM277384.pdf, accessed 04- April-2019.
5.14 Chloasma
Chloasma may occur with ANNOVERA use, especially in females with a history of chloasma gravidarum. Advise females who tend to develop chloasma to avoid exposure to the sun or ultraviolet radiation while using ANNOVERA.
11 Description
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white vaginal system containing two active components, a progestin, segesterone acetate, and an estrogen, ethinyl estradiol. When placed in the vagina, each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate and 0.013 mg/day of ethinyl estradiol over the 21 days in-use period of each cycle for up to 13 cycles (total of 273 days). Each cycle is 28 days, with 21 days in and 7 days out.
The inactive ingredients are dibutyltin dilaurate, silicone elastomers, silicone medical adhesive, and titanium dioxide. The elastomers are all methyl siloxane-based polymers.
The vaginal system body has an overall diameter of 56 mm and a cross-sectional diameter of 8.4 mm. It contains two channels of approximately 3.0 mm diameter and 27 mm length into which steroid-containing cores are inserted. Each ANNOVERA contains 103 mg of SA distributed throughout both cores and 17.4 mg of EE distributed throughout only one core. The core containing 40% SA and 12% EE of its mass is 3 mm in diameter and 18 mm in length. The core containing 50% SA of its mass is 3 mm in diameter and 11 mm in length. Contact between the cores and the vaginal system body is fixed by coating the channels with silicone medical adhesive before introducing the cores. After insertion of the cores, the channels are sealed with the silicone medical adhesive.
The structural formulas, and properties for the active components are shown below:
STRUCTURAL FORMULAS:
Segesterone Acetate (SA)
Ethinyl Estradiol (EE)
PROPERTIES:
Established Name: Segesterone Acetate
Chemical Name: 16-methylene-17α-acetoxy-19-nor-pregn-4-ene-3,20-dione
Molecular Formula: C23H30O4
Molecular Weight: 370.5
Physical Form: White, or yellowish white powder
Solubility: Slightly soluble in n-hexane, soluble in ethyl acetate and methanol, freely soluble in acetone (USP classification)
Melting Point: 173°C–177°C
Established Name: Ethinyl Estradiol
Chemical Name: 19-Nor-17α -pregna-1,3,5(10)-trien-20-yne-3,17-diol
Molecular Formula: C20H24O2
Molecular Weight: 296.4
Physical Form: White to slightly yellowish-white crystalline powder
Solubility: Practically insoluble in water, freely soluble in alcohol, it dissolves in alkaline solution
Melting Point: 181°C–185°C
The steroids diffuse out of the vaginal system with release rates that vary over time. Based on in vitro data, the daily release rates of SA and EE are higher during each initial 24–48 hours of use achieving a somewhat lower steady-state with continued use over the subsequent days in each cycle. The vaginal system is designed to be used for 13 cycles (1 year) on a 21/7 days in/out schedule. The total in-use time with the 21/7 days in/out schedule over 13 cycles is 273 days. Based on the residual amount of drug in vaginal systems used in clinical trials over 13 cycles, a total of 41.3 mg of SA and 3.4 mg of EE are released over this period. This translates to an approximate average daily dose of 0.15 mg of segesterone acetate and 0.013 mg of ethinyl estradiol with higher release rate expected at the beginning of dosing and a lower release rate toward the end.
5.10 Depression
Carefully observe females with a history of depression and discontinue ANNOVERA if depression recurs to a serious degree. Data on the association of CHCs with onset of depression or exacerbation of existing depression are limited.
5.16 Vaginal Use
Some females are aware of the vaginal system on occasion during the 21 days of use or during coitus, and partners may feel the vaginal system during coitus.
There is no information on the concomitant use of ANNOVERA with diaphragms, cervical caps, and female condoms.
ANNOVERA may not be suitable for females with conditions that make the vagina more susceptible to vaginal irritation or ulceration. Vaginal and cervical erosion and/or ulceration has been reported in females using other contraceptive vaginal devices. In some cases, the ring adhered to vaginal tissue, which necessitated removal by a healthcare provider.
5.4 Hypertension
ANNOVERA is contraindicated in females with uncontrolled hypertension or hypertension with vascular disease [see Contraindications (4)]. For all females, including those with well- controlled hypertension, monitor blood pressure at routine visits and stop ANNOVERA if blood pressure rises significantly.
An increase in blood pressure has been reported in females using CHCs, and this increase is more likely in older females and with extended duration of use. The effect of CHCs on blood pressure may vary according to the progestin in the CHC.
16.1 How Supplied
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white vaginal system. The vaginal system body has an overall diameter of 56 mm and a cross-sectional diameter of 8.4 mm. When placed inside the vagina, each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate and 0.013 mg/day of ethinyl estradiol over 21-day in-use period of each cycle for up to 13 cycles (total 273 days). Each cycle is 28 days, with 21 days in and 7 days out.
Each ANNOVERA is individually packaged in an aluminum pouch. The pouch consists of a laminate from outside to inside of polyester, aluminum foil, and polyethylene.
First insertion of ANNOVERA must be prior to the date of expiration. Discard one year after first insertions.
A black compact case is provided with the drug product for storage of ANNOVERA by patients during each 7-day vaginal system-out interval.
The vaginal system should be placed in the compact case after 13 cycles of use and discarded via a drug take-back option if one is available. If a take-back option is unavailable, then discard in the waste receptacle out of reach of children and pets. The vaginal system should NOT be flushed down the toilet. See www.fda.gov/drugdisposal for more information about disposal of medicines.
Each box contains 1 ANNOVERA vaginal system in a pouch and 1 storage case.
NDC 68308-752-01
8.4 Pediatric Use
Safety and efficacy of ANNOVERA have been established in women of reproductive age. Efficacy is expected to be the same for postpubertal adolescents under the age of 18 as for users 18 years and older. Use of ANNOVERA before menarche is not indicated.
8.5 Geriatric Use
ANNOVERA has not been studied in females who have reached menopause and is not indicated in this population.
14 Clinical Studies
The efficacy of ANNOVERA was evaluated in two 1-year multicenter trials enrolling 2,265 females, age 18–40 years, who were healthy and sexually active with regular menstrual cycles. The trials were conducted in the U.S., Dominican Republic, Brazil, Chile, Finland, Hungary, Sweden, and Australia, with 67.1% of females from the U.S. The racial/ethnic distribution was Caucasian (71.2%), African-American (14.1%), Asian (3.5%), other/multiple races (11.2%); 28.7% of the study population was Hispanic. The mean age was 26.7 years and the mean (range) BMI was 24.1 (16.0, 41.5) kg/m2. At approximately 50% enrollment, women with BMI >29.0 kg/m2 were no longer enrolled in the two trials and all women with a BMI >29.0 kg/m2 were discontinued from the trials.
Based on pooled data from the two trials, 2,111 females ≤35 years of age completed 17,427 evaluable 28-day cycles (cycles in which no back-up contraception was used). The pooled pregnancy rate, evaluated by the Pearl Index (PI), was 2.98 (95% Confidence Interval [2.13, 4.06]) per 100 woman-years of ANNOVERA use.
Return to fertility was assessed in 290 of the subjects in the two trials who either desired pregnancy or switched to a nonhormonal method after the trials, and all 290 subjects reported a return to fertility during the 6-month follow-up period (defined as a return of menses or pregnancy).
4 Contraindications
ANNOVERA is contraindicated in females who are known to have or develop the following conditions:
- A high risk of arterial or venous thrombotic diseases. Examples include females who are known to:
-
-Smoke, if over age 35 [see Boxed Warning and Warnings and Precautions (5.1)].
-
-Have current or history of deep vein thrombosis or pulmonary embolism [see Warnings and Precautions (5.1)].
-
-Have cerebrovascular disease [see Warnings and Precautions (5.1)].
-
-Have coronary artery disease [see Warnings and Precautions (5.1)].
-
-Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see Warnings and Precautions (5.1)].
-
-Have inherited or acquired hypercoagulopathies [see Warnings and Precautions (5.1)].
-
-Have uncontrolled hypertension or hypertension with vascular disease [see Warnings and Precautions (5.4)].
-
-Have diabetes mellitus and are over age 35, diabetes mellitus with hypertension or vascular disease, or other end-organ damage, or diabetes mellitus of >20 years duration [see Warnings and Precautions (5.5), (5.7)].
-
-Have headaches with focal neurological symptoms, migraine headaches with aura, or are over age 35 with any migraine headaches [see Warnings and Precautions (5.8)].
-
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive [see Warnings and Precautions (5.11)].
- Liver tumors, acute hepatitis, or severe (decompensated) cirrhosis [see Warnings and Precautions (5.2) and Use in Specific Populations (8.6)].
- Undiagnosed abnormal uterine bleeding [see Warnings and Precautions (5.9)].
- Hypersensitivity to any of the components of ANNOVERA. Hypersensitivity reactions reported include: throat constriction, facial edema, urticaria, hives, and wheezing [see Adverse Reactions (6.1)].
- Use of Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine transaminase (ALT) elevations [see Warnings and Precautions (5.3)].
6 Adverse Reactions
The following adverse reactions are described elsewhere in other sections of the labeling:
- Serious cardiovascular events and stroke [see Boxed Warning and Warnings and Precautions (5.1)]
- Vascular events [see Warnings and Precautions (5.1)]
- Liver disease [see Warnings and Precautions (5.2)]
7 Drug Interactions
The sections below provide information on substances for which data on drug interactions with CHCs are available. There is little information available about the clinical effect of most drug interactions that may affect ANNOVERA. However, based on the known pharmacokinetic effects of these drugs, clinical strategies to minimize any potential adverse effect on contraceptive effectiveness or safety are suggested.
Consult the approved product labeling of all concurrently used drugs to obtain further information about interactions with ANNOVERA or the potential for metabolic enzyme or transporter system alterations.
8.7 Renal Impairment
No studies were conducted in subjects with renal impairment; ANNOVERA is not recommended in patients with renal impairment.
Instructions for Use
ANNOVERA® (ann-o-VER-ah)
(segesterone acetate and ethinyl estradiol vaginal system)
Read this Instructions for Use carefully before you decide if ANNOVERA is right for you. This information does not take the place of talking with your gynecologist or other healthcare provider who specializes in women's health. If you have any questions about ANNOVERA, ask your healthcare provider. You should also learn about other birth control methods to choose the one that is best for you.
How should I start using ANNOVERA?
If you are not currently using hormonal birth control, start using ANNOVERA between Days 2 and 5 of your menstrual period.
- If your menstrual periods are not regular or if you start using ANNOVERA more than 5 days from when you started your menstrual period, you should use a barrier method of birth control, such as a male condom or spermicide during sexual intercourse for the first 7 days you use ANNOVERA.
If you are changing from a birth control pill or patch or monthly disposable contraceptive vaginal ring to ANNOVERA:
- If you have been using your birth control method correctly and are certain that you are not pregnant, you can change to ANNOVERA any day of your birth control cycle. Do not start ANNOVERA any later than the day you would start your next birth control pill, apply your next patch or insert your next monthly disposable contraceptive vaginal ring.
If you are changing from a progestin-only birth control method, such as a minipill, injection, implant or intrauterine system (IUS):
- You may switch from a minipill on any day. Start using ANNOVERA on the day that you would have taken your next minipill.
- You should switch from an injectable and start using ANNOVERA on the day when your next injection would be due.
- You should switch from an implant or the IUS and start using ANNOVERA at the time the implant or IUS is removed.
If you are changing from a minipill, injection, implant, or an intrauterine system to ANNOVERA, you should use a barrier method of birth control, such as a male condom or spermicide, during sexual intercourse for the first 7 days you use ANNOVERA.
If you start using ANNOVERA after an abortion or miscarriage:
-
Following a first trimester abortion or miscarriage: You may start ANNOVERA within 5 days following a first trimester abortion or miscarriage (the first 12 weeks of pregnancy). You do not need to use an additional birth control method.
- If you do not start ANNOVERA within 5 days after a first trimester abortion or miscarriage, use a non-hormonal birth control method, such as male condoms or spermicide, while you wait for your menstrual period to start. Start using ANNOVERA between days 2 and 5 of your menstrual period.
- If you start using ANNOVERA more than 5 days from when you started your menstrual period, you should use a barrier method of birth control, such as a male condom or spermicide during sexual intercourse for the first 7 days you use ANNOVERA.
- Following a second trimester abortion or miscarriage: You may start using ANNOVERA no sooner than 4 weeks (28 days) after a second trimester abortion (after the first 12 weeks of pregnancy).
If you are starting ANNOVERA after childbirth:
- You may start using ANNOVERA no sooner than 4 weeks (28 days) after having a baby if you are not breastfeeding.
- If you have not gotten your menstrual period after childbirth, you should talk to your healthcare provider. You may need a pregnancy test to make sure you are not pregnant before you start using ANNOVERA.
- Use another birth control method, such as a male condom or spermicide, during sexual intercourse for the first 7 days you use ANNOVERA if you have not yet had a period.
If you are breastfeeding, you should not use ANNOVERA. Use other birth control methods until you are no longer breastfeeding.
How do I use ANNOVERA?
|
|
Step 1. Open the package and remove ANNOVERA.
|
|
|
Step 2. Prepare to insert ANNOVERA.
|
|
|
Step 3. Choose a position for insertion of ANNOVERA.
|
|
|
Step 4. Insert ANNOVERA into your vagina.
|
|
|
Note:
|
|
|
Step 5. How do I remove ANNOVERA?
|
What else should I know about ANNOVERA?
- The day of the week when you first insert ANNOVERA (called "Day 1") is your vaginal system change day. This is described further in the Patient Information section entitled "How should I use ANNOVERA?"
- For each cycle, you put the vaginal system into your vagina and let it stay there 3 weeks (21 full days). Remember to keep the vaginal system in for the whole 3 weeks (21 full days).
- You take the vaginal system out on your vaginal system change day (Day 22) and let it stay out for 1 week (7 full days). Note that your vaginal system should be stored in the case provided, away from children, pets, and extreme temperatures.
- Then you start over again for another 4 weeks. You may not be bleeding when you put the vaginal system in.
- Always put the vaginal system in or take it out on your vaginal system change day at about the same time of day. For example, if you put your vaginal system in on Monday at 9:00 in the morning, always take it out or put it back in on Monday at about 9:00 in the morning.
- You do not have to take the vaginal system out when you have sex. If you decide to remove it, remember to reinsert it within 2 hours after removing it or you may not be protected from pregnancy. However, if ANNOVERA is out of your vagina for more than 2 hours at one time or if ANNOVERA is out of your vagina at different times that add up to more than a total of 2 hours over the first 21 days of your cycle, you will need to use another method of birth control until ANNOVERA has been in your vagina for 7 days in a row, such as male condoms or spermicides.
- Repeat the 4-week cycle for all 13 cycles of the use of the vaginal system.
- Do not use the vaginal system for more than 13 cycles (1 year). When you take the vaginal system out of your vagina at the end of the 13 cycles, dispose of it. See "How should I dispose of (throw away) ANNOVERA?" below.
- If you want to continue using ANNOVERA after 13 cycles, you will need to get a new prescription from your healthcare provider to obtain a new vaginal system.
- Do not to use any vaginal products such as oil-based suppositories, oil-based creams, or oil-based gels while the vaginal system is in your vagina. Do not use any vaginal lubricants that have silicone or oil in them. Water-based lubricants are ok to use. Be sure to read the ingredients on the labels carefully before you buy a vaginal lubricant.
- First insertion of ANNOVERA must be prior to the date of expiration. Discard one year after first insertion. See "How should I dispose of (throw away) ANNOVERA?" below.
- Do not take any medicines unless your healthcare provider says it is ok to take them.
Tell your healthcare provider when you start a new medicine.
How do I clean ANNOVERA?
- After removing ANNOVERA from your vagina, wash it with mild soap and warm water, rinse, and pat it dry with a clean cloth towel or paper towel before you store it. Only use mild soap and warm water to clean.
- Wash ANNOVERA with mild soap and warm water and pat it dry before you put it back into your vagina.
- Store ANNOVERA in the case provided.
How should I store ANNOVERA?
- Store ANNOVERA in the supplied case at room temperature between 68°F to 77°F (20°C to 25°C).
- Protect ANNOVERA from direct sunlight.
- Do not refrigerate or freeze ANNOVERA.
- Avoid storing ANNOVERA in extreme heat.
- Keep ANNOVERA and all medicines out of the reach of children and pets.
How should I dispose of (throw away) ANNOVERA?
- After 13 cycles of use, place ANNOVERA in the case that comes with it.
- Dispose of ANNOVERA at a drug take-back location, if available. If a drug take-back location is not available, dispose of ANNOVERA in the trash, out of the reach of children and pets.
- Do not throw away ANNOVERA in the toilet.
- See www.fda.gov/drugdisposal for more information about disposal of medicines.
When does ANNOVERA become effective?
ANNOVERA becomes effective on the day it is inserted (Day 1), if it is inserted between Days 2 and 5 of your menstrual period. If ANNOVERA is inserted more than 5 days from when you start your menstrual period, you should use a barrier method of birth control, such as a male condom or spermicide, during sexual intercourse for the first 7 days you use ANNOVERA.
Will ANNOVERA interfere during sexual intercourse?
- If ANNOVERA is placed as high as possible in your vagina, it will not interfere with sexual intercourse.
Can I take ANNOVERA out during the first 3 weeks of my cycle?
- You should leave ANNOVERA in the entire 21 days (3 weeks).
- If ANNOVERA comes out or if you remove it, put it back in as soon as possible. If you put ANNOVERA back into your vagina before 2 hours has passed, you do not need to use another method of birth control. However, if ANNOVERA is out of your vagina for more than 2 hours at one time or if ANNOVERA is out of your vagina at different times that add up to more than a total of 2 hours over the first 21 days of your cycle, you will need to use another method of birth control until ANNOVERA has been in your vagina for 7 days in a row, such as male condoms or spermicides.
What should I do if ANNOVERA comes out of my vagina?
ANNOVERA can slip or accidentally come out of your vagina (expelled), for example, during sexual intercourse, bowel movements, or use of tampons.
- If ANNOVERA slips out of your vagina, wash ANNOVERA with mild soap and warm water, rinse and pat dry with a clean cloth towel or paper towel and put it back in your vagina right away or within 2 hours. See "Can I take ANNOVERA out during the first 3 weeks of my cycle?" in the section above.
- ANNOVERA can move around and become visible at the opening of your vagina. If this happens follow "Step 4" above for directions on how to push ANNOVERA back to its right position.
What if I lose ANNOVERA?
- Call your healthcare provider right away if you lose ANNOVERA. You should use a back-up method of birth control such as a male condom or spermicide for preventing pregnancy until you get a new ANNOVERA.
If you have other questions call your healthcare provider.
For information, call Mayne Pharma at 1-844- 825-8500
Distributed by:
Mayne Pharma
Raleigh, NC 27609
ANNOVERA is a registered trademark of Population Council and licensed under license.
The Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Revised 08/2024
8.6 Hepatic Impairment
No studies have been conducted to evaluate the effect of hepatic impairment on the disposition of ANNOVERA. However, steroid hormones may be poorly metabolized in patients with hepatic impairment. Acute or chronic disturbances of liver function may necessitate the discontinuation of CHC use until markers of liver function return to normal and CHC causation has been excluded [see Contraindications (4) and Warnings and Precautions (5.2)].
1 Indications and Usage
ANNOVERA is indicated for use by females of reproductive potential to prevent pregnancy.
16.2 Storage Conditions
Prior to dispensing ANNOVERA to the user, store ANNOVERA at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Protect ANNOVERA from direct sunlight.
Do not refrigerate or freeze and avoid excessive heat.
2.1 How to Use Annovera
Instruct patients that ANNOVERA should be used as directed [see How to Start ANNOVERA (2.2)]. One ANNOVERA should be placed in the vagina. For maximum contraceptive effectiveness, ANNOVERA is to remain in the vagina continuously for 21 days (3 complete weeks). It is removed for a 1-week dose-free interval, and during this time a withdrawal bleed usually occurs. The removed vaginal system should be cleaned with mild soap and warm water, patted dry with a clean cloth towel or paper towel, and placed in its case during the 1-week dose- free interval. At the end of the dose-free interval, the vaginal system should be cleaned prior to being placed back in the vagina for another 21 continuous days (3 complete weeks). This pattern of ANNOVERA use made up of 3-weeks in and 1-week out is a cycle of use; one ANNOVERA vaginal system will provide contraception for 13 cycles.
With clean hands, the user can choose an insertion position that is comfortable, such as lying down, squatting, or standing. The sides of the vaginal system are pressed together for insertion into the vagina. When properly inserted, the vaginal system should be entirely in the vagina and behind the pelvic bone. The day and time of insertion should be noted so that the vaginal system can be removed 3 weeks later on the same day and at about the same time.
ANNOVERA can be removed by hooking an index finger into the vaginal system inside the vagina and gently pulling the vaginal system.
For patient instructions regarding cleaning the vaginal system, see FDA-approved Patient Information.
5.6 Gallbladder Disease
Studies suggest a small increased relative risk of developing gallbladder disease among CHC users. Use of CHCs may also worsen existing gallbladder disease.
A past history of CHC-related cholestasis predicts an increased risk with subsequent CHC use. Females with a history of pregnancy-related cholestasis may be at an increased risk for CHC- related cholestasis.
12.1 Mechanism of Action
CHCs lower the risk of becoming pregnant primarily by suppressing ovulation.
2.2 How to Start Annovera
IMPORTANT: Consider the possibility of ovulation and conception prior to the first use of ANNOVERA.
5 Warnings and Precautions
- Thrombotic Disorders and Other Vascular Problems: Stop ANNOVERA if a thrombotic or thromboembolic event occurs. Stop ANNOVERA at least 4 weeks before and through 2 weeks after major surgery. Start ANNOVERA no earlier than 4 weeks after delivery, in females who are not breastfeeding. Consider cardiovascular risk factors before initiating in all females, particularly those over 35 years. (5.1, 5.5)
- Liver Disease: Discontinue if jaundice occurs. (5.2)
- Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment: Stop ANNOVERA prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir. ANNOVERA can be restarted 2 weeks following completion of this regimen. (5.3)
- Hypertension: Do not prescribe ANNOVERA for females with uncontrolled hypertension or hypertension with vascular disease. If used in females with well-controlled hypertension, monitor blood pressure and stop use if blood pressure rises significantly. (5.4)
- Carbohydrate and lipid metabolic effects: Monitor glucose in pre-diabetic and diabetic females taking ANNOVERA. Consider an alternate contraceptive method for females with uncontrolled dyslipidemias. (5.7)
- Headache: Evaluate significant change in headaches and discontinue ANNOVERA if indicated. (5.8)
- Bleeding Irregularities and Amenorrhea: May cause irregular bleeding or amenorrhea. Evaluate for other causes if irregular bleeding or amenorrhea persists. (5.9)
5.13 Hereditary Angioedema
In females with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema.
2 Dosage and Administration
One ANNOVERA is inserted in the vagina. The vaginal system must remain in place continuously for 3 weeks (21 days) followed by a 1-week (7-day) vaginal system-free interval. One vaginal system provides contraception for thirteen 28-day cycles (1 year). (2)
3 Dosage Forms and Strengths
Vaginal system: Each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate (SA) and 0.013 mg/day of ethinyl estradiol (EE) when placed in the vagina over a period of 21 days of each cycle for up to 13 cycles (total of 273 days). Each cycle of use is 28 days with 21 days in and 7 days out.
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white, contraceptive vaginal system containing 103 mg of SA and 17.4 mg EE.
ANNOVERA is 56 mm in overall diameter and 8.4 mm in cross-sectional diameter. It contains 2 channels of approximately 3.0 mm diameter and 27 mm length into which steroid-containing cores are inserted. The cores are 3 mm in diameter with one releasing SA alone and the other releasing both SA and EE.
Each ANNOVERA is designed to be used for up to 13 cycles (1 year).
ANNOVERA is not made with natural rubber latex.
6.2 Postmarketing Experience
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 2).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 2). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
Figure 2: Relevant Studies of Risk of Breast Cancer with Combined Oral Contraceptives
RR = relative risk; OR = odds ratio; HR = hazard ratio. "ever COC" are females with current or past COC use; "never COC use" are females who never used COCs.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The clinical trials that evaluated the safety of ANNOVERA were obtained from three 13- cycle trials. One trial was conducted entirely in the U.S. (15 sites), and the other two were global studies that included 5 U.S. sites and 7 international sites (Australia, Brazil, Chile, Dominican Republic, Finland, Hungary, Sweden). All three trials were open label and enrolled healthy females, desiring contraception, 18 to 40 years of age. At about 50% enrollment, females with BMI >29 kg/m2 were excluded due to the occurrence of two VTEs in this subgroup. In total, 2,308 females contributed 21,590 cycles of exposure for safety evaluation and 999 completed 13 cycles; there were 209 subjects with BMI >29 kg/m2 who contributed 1,254 cycles of exposure with 36 subjects completing 13 cycles. The demographic profile for subjects was: mean age 26.7 years, mean BMI 24.1 (16.0- 41.5) kg/m2; 67% were from the U.S. The racial distribution was 71% Caucasian, 14% African American, 4% Asian, and 11% Other; 30% of the population was Hispanic.
8 Use in Specific Populations
5.5 Age Related Considerations
The risk for cardiovascular disease and prevalence of risk factors for cardiovascular disease increase with age. Certain conditions, such as smoking and migraine headache without aura, that do not contraindicate CHC use in younger females, are contraindications to use in women over 35 years of age [see Contraindications (4) and Warnings and Precautions (5.1)]. Consider the presence of underlying risk factors that may increase the risk of cardiovascular disease or VTE, particularly before initiating ANNOVERA for women over 35 years, such as:
- Hypertension
- Diabetes
- Dyslipidemia
- Obesity
5.15 Toxic Shock Syndrome (tss)
Cases of TSS have been reported by vaginal ring users. TSS has been associated with tampons and certain barrier contraceptives, and in some TSS cases ring users were also using tampons. Causal relationship between the use of a vaginal ring and TSS has not been established. No cases of TSS occurred in clinical trials with ANNOVERA. If a patient exhibits signs or symptoms of TSS, consider the possibility of this diagnosis, remove ANNOVERA, and initiate appropriate medical evaluation and treatment.
5.12 Effect On Binding Globulins
The estrogen component of ANNOVERA may raise the serum concentrations of thyroxine- binding globulin, sex hormone-binding globulin, and cortisol-binding globulin. The dose of replacement thyroid hormone or cortisol therapy may need to be increased.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
8.8 Body Mass Index (bmi)/body Weight
The safety and efficacy of ANNOVERA in females with a BMI >29 kg/m2 have not been adequately evaluated because this subpopulation was excluded from the clinical trials after 2 VTEs occurred in females with a BMI > 29 kg/m2 [see Adverse Reactions (6.1) and Clinical Studies (14)]. Higher body weight is associated with lower systemic exposure of SA and EE [see Clinical Pharmacology (12.3)].
7.5 Interference With Laboratory Tests
The use of contraceptive steroids may influence the results of certain laboratory tests, such as coagulation factors, lipids, glucose tolerance, and binding proteins.
7.3 Use of Vaginal Products With Annovera
In a drug-drug interaction study with ANNOVERA and the concurrent use of three different formulations of vaginal miconazole, the use of water-based vaginal miconazole cream resulted in no change in exposure to EE or SA from the vaginal system. However, the use of either the 1- day or the 3-day oil-based miconazole suppository was associated with an overall increase in exposure up to 67% for EE and 32% for SA. Considering the potential long-term effect on vaginal system performance, concurrent use of oil-based vaginal suppositories should not occur with ANNOVERA use. If there is a need to treat a vaginal condition, water-based vaginal cream or oral therapy may be used concurrently with the vaginal system [see Clinical Pharmacology (12.3)].
5.9 Bleeding Irregularities and Amenorrhea
Bleeding and/or spotting that occurs at any time while the vaginal system is inserted is considered "unscheduled" bleeding/spotting. Bleeding/spotting that occurs during the dose-free week when the vaginal system is out is considered "scheduled" bleeding.
2.3 Deviations From the Recommended Regimen
Contraceptive efficacy of ANNOVERA may be reduced if a woman deviates from the recommended use. ANNOVERA should remain in the vagina for a continuous period of 21 days (3 weeks); then ANNOVERA should be taken out of the vagina for 7 days. In a 28-day cycle, a deviation that involves ANNOVERA being out of the vagina for less than 7 days will not increase pregnancy risk. In a 28-day cycle, a deviation that involves ANNOVERA being out of the vagina for more than 7 days will increase pregnancy risk and back-up contraception is recommended in these instances. Deviations from the recommended regimen may result in a new vaginal system change day [See FDA- approved Patient Information].
Principal Display Panel 30 Ring Pouch Carton
NDC 68308-752-01
Rx only
Annovera®
(segesterone acetate and
ethinyl estradiol vaginal system)
Delivers 0.15 mg/0.013 mg per day
This product is intended to prevent pregnancy.
It does not protect against HIV infection (AIDS)
and other sexually transmitted diseases.
Keep out of reach of children and pets
For Vaginal Use
CONTAINS 1 VAGINAL SYSTEM
mayne pharma
5.1 Thromboembolic Disorders and Other Vascular Conditions
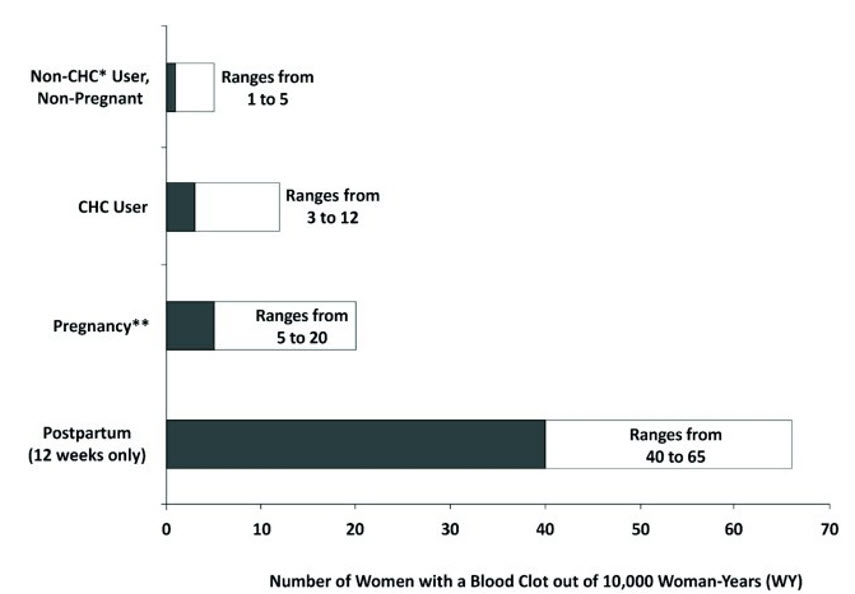
Females are at increased risk for a venous thrombotic event (VTE) when using ANNOVERA. Limited data are available in females with a BMI >29.0 kg/m2 because this subpopulation was excluded from the clinical trials after VTEs were reported.
- Stop ANNOVERA if an arterial or deep venous thrombotic event occurs.
- Stop ANNOVERA if there is unexplained loss of vision, proptosis, diplopia, papilledema, or retinal vascular lesions and evaluate for retinal vein thrombosis immediately.
- Discontinue ANNOVERA during prolonged immobilization. If feasible, stop ANNOVERA at least 4 weeks before and through 2 weeks after major surgery or other surgeries known to have an elevated risk of VTE.
- Start ANNOVERA no earlier than 4 weeks after delivery in females who are not breastfeeding. The risk of postpartum VTE decreases after the third postpartum week, whereas the risk of ovulation increases after the third postpartum week.
- Before starting ANNOVERA, evaluate any past medical history or family history of thrombotic or thromboembolic disorders and consider whether the history suggests an inherited or acquired hypercoagulopathy. ANNOVERA is contraindicated in females with a high risk of arterial or venous thrombotic/thromboembolic diseases [see Contraindications (4)].
Warning: Cigarette Smoking and Serious Cardiovascular Events
Cigarette smoking increases the risk of serious cardiovascular events from combination hormonal contraceptive (CHC) use. This risk increases with age, particularly in females over 35 years of age, and with the number of cigarettes smoked. For this reason, CHC should not be used by females who are over 35 years of age and smoke. [See Contraindications (4) and Warnings and Precautions (5.5)].
7.2 Effects of Combined Hormonal Contraceptives On Other Drugs
Table 4 provides significant drug interaction information for drugs co-administered with CHCs.
| Lamotrigine | |
| Clinical effect | Concomitant use of CHCs with lamotrigine may significantly decrease systemic exposure of lamotrigine due to induction of lamotrigine glucuronidation. |
| Prevention or management | Dose adjustment for lamotrigine may be necessary. Consult the approved product labeling for lamotrigine. |
| Thyroid Hormone Replacement Therapy or Corticosteroid Replacement Therapy | |
| Clinical effect | Concomitant use of CHCs with thyroid hormone replacement therapy or corticosteroid replacement therapy may increase systemic exposure of thyroid-binding and cortisol-binding globulin [see Warnings and Precautions (5.12)]. |
| Prevention or management | The dose of replacement thyroid hormone or cortisol therapy may need to be increased. Consult the approved product labeling for the therapy in use [see Warnings and Precautions (5.12)]. |
| Other Drugs | |
| Clinical effect | Concomitant use of CHCs may decrease systemic exposure of acetaminophen, morphine, salicylic acid, and temazepam. Concomitant use with ethinyl estradiol-containing CHCs may increase systemic exposure of other drugs (eg, cyclosporine, prednisolone, theophylline, tizanidine, and voriconazole). |
| Prevention or management | The dosage of drugs that can be affected by this interaction may need to be increased or decreased. Consult the approved product labeling for the concomitantly used drug. |
5.3 Risk of Liver Enzyme Elevations With Concomitant Hepatitis C Treatment
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with and without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in females using ethinyl estradiol-containing medications, such as ANNOVERA. Discontinue ANNOVERA prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir [see Contraindications (4)]. ANNOVERA can be restarted approximately 2 weeks following completion of treatment with the Hepatitis C combination drug regimen.
7.4 Concomitant Use With Hcv Combination Therapy – Liver Enzyme Elevation
Do not co-administer ANNOVERA with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations [see Contraindications (4) and Warnings and Precautions (5.3)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
ANNOVERA has not been adequately studied in females with a BMI >29 kg/m2.
Section 42230-3 (42230-3)
| Patient Information ANNOVERA® (ann-o-VER-ah) (segesterone acetate and ethinyl estradiol vaginal system) |
|||||
|---|---|---|---|---|---|
| Read this Patient Information carefully before you decide if ANNOVERA is right for you. This information does not take the place of talking with your gynecologist or other healthcare provider who specializes in women's health. If you have any questions about ANNOVERA, ask your healthcare provider. You should also learn about other birth control methods to choose the one that is best for you. | |||||
|
What is the most important information I should know about ANNOVERA?
|
|||||
| ANNOVERA does not protect against HIV infection (AIDS) and other sexually transmitted infections (STIs). | |||||
|
What is ANNOVERA?
ANNOVERA is a hormone-releasing system used by females who are able to become pregnant to prevent pregnancy. You insert ANNOVERA into your vagina. ANNOVERA is in the shape of a ring that is reusable for 1 year. The vaginal system is made of silicone and contains two female hormones that are slowly released into your vagina and then enter your blood. One hormone is an estrogen called ethinyl estradiol. The other hormone is a progestin called segesterone acetate. |
|||||
|
How well does ANNOVERA work for contraception?
Your chance of getting pregnant depends on how well you follow the directions for using the vaginal system. The more carefully you follow the directions, the less chance you have of getting pregnant. Based on the results of two clinical studies that lasted 12 months, about 2 to 4 women out of 100 women may get pregnant during the first year they use ANNOVERA. ANNOVERA was designed to be reused for 1 year. Replace ANNOVERA at the end of 1 year if you choose to continue using ANNOVERA. There is not enough hormone left in the vaginal system to provide you effective birth control after 1 year of use. The following chart shows the chance of getting pregnant for women who use different methods of birth control. Each box on the chart contains a list of birth control methods that are similar in how well they work to prevent pregnancy. The most effective methods are at the top of the chart. The box at the bottom of the chart shows the chance of getting pregnant for women who do not use birth control and are trying to get pregnant. ANNOVERA, a vaginal system with hormones, is in the second box from the top of the chart.
|
|||||
|
Who should not use ANNOVERA? Do not use ANNOVERA if you:
|
|||||
|
What should I tell my healthcare provider before using ANNOVERA? Before you use ANNOVERA tell your healthcare provider if you:
Do not use any vaginal products such as oil-based suppositories, oil-based creams, or oil-based gels while the vaginal system is in your vagina. Do not use any vaginal lubricants that have silicone or oil in them. Water-based lubricants are ok to use. Be sure to read the ingredients on the label carefully before you buy a vaginal lubricant. Some medicines and grapefruit juice may increase the level of ethinyl estradiol in your blood if used together, including:
Females who take thyroid hormone replacement medicine or corticosteroid replacement medicine may need increased doses of their thyroid hormone or cortisol medicines. Ask your healthcare provider if you are not sure if you take any of the medicines listed above. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. Talk to your healthcare provider before you start taking a new medicine. |
|||||
How should I use ANNOVERA?
After you insert ANNOVERA for the first time, you will remove it at the end of week 3 and leave it out for 7 days. You will reinsert ANNOVERA at the end of week 4 of each 4- week cycle. You will repeat this pattern with ANNOVERA for up to 13 cycles. Scheduled insertions and removals of ANNOVERA should be at about the same time of the day and the same day of the week for each monthly cycle.
|
|||||
| Schedule | |||||
| Cycle 1 |
Put vaginal system in → (vaginal system change day) |
Day 1 |
Weeks 1, 2, and 3
Days 1-21 |
||
|
Take vaginal system out→ (vaginal system change day) |
Day 22 |
Week 4
Days 22-28 |
|||
|
Cycle 2 (repeat for Cycles 3-13) |
Put vaginal system in→ (vaginal system change day) |
Day 1 |
Weeks 1, 2, and 3
Days 1-21 |
||
|
Take vaginal system out→ (vaginal system change day) |
Day 22 |
Week 4
Days 22-28 |
|||
|
|||||
|
What happens if I am off schedule using the vaginal system?
It is very important to follow the schedule every cycle and remove and insert ANNOVERA on your vaginal system-in and vaginal system-out days at about the same time. If you take the vaginal system out too soon or put it back in too late, your chance of getting pregnant is higher. If you are more than 1 or 2 days off schedule for vaginal system insertion you will need to use new vaginal system-in and vaginal system-out days for the remaining cycles of use. You will also need to use back-up contraception, such as condoms or spermicide, for the first 7 days of the new schedule if you have sexual intercourse. |
|||||
| If you put the vaginal system back in: | Then: | ||||
| Too early, after it had been out for only 5 or 6 days. | Keep the vaginal system in for at least 3 weeks (21 days). You may keep it in up to your normal vaginal system-out day. | ||||
| Too late, after it had been out for more than 7 days. | Put the vaginal system back in right away. You will now have a new vaginal system-in day. You must use condoms or spermicide as back-up contraception for the next 7 days when you have sexual intercourse. | ||||
| If you took the vaginal system out: | Then: | ||||
| Too early, after it had been in for only 19 or 20 days. | Leave the vaginal system out for 1 week. Put it back in after the week is over, as you would normally do. You may then keep it in up to your normal vaginal system-out day, 22 or 23 days after you put the vaginal system in. | ||||
| Too late, after it had been in for 22 or 23 days. | Remove the vaginal system as soon as you realize this. Then reinsert the vaginal system 7 days later. | ||||
| During the 21 days of continuous use, if ANNOVERA is out of the vagina for more than 2 continuous hours or more than 2 cumulative hours (multiple inadvertent removals or expulsions adding up to 2 hours), then back-up contraception such as male condoms or spermicide should be used until the vaginal system has been in the vagina for 7 consecutive days. | |||||
|
What if I miss my menstrual period or if I think I am pregnant?
It is possible that you are pregnant if you miss your scheduled period (no bleeding on the 7 days that the vaginal system is out). Tell your healthcare provider right away that you have missed your period. Also, tell your healthcare provider if you have symptoms of pregnancy such as morning sickness or unusual breast tenderness. It is important that your healthcare provider check you to find out if you are pregnant. You may need a pregnancy test to determine if you are pregnant. Do not remove the vaginal system until you are certain you are pregnant. Stop using ANNOVERA if your healthcare provider tells you that you are pregnant. |
|||||
|
What are the possible side effects of ANNOVERA? ANNOVERA can cause serious side effects, including:
See: "What is the most important information I should know about ANNOVERA?"
|
|||||
|
|
||||
|
|||||
|
|
||||
| This is not a complete list of possible side effects. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Mayne Pharma at 1-844-825-8500. No serious problems have been reported from a hormonal contraceptive overdose. |
|||||
|
Does hormonal birth control cause cancer?
It is not known if hormonal birth control causes breast cancer. Some studies, but not all, suggest that there could be a slight increase in the risk of breast cancer among current users with longer duration of use. If you have breast cancer now, or have had it in the past, do not use hormonal birth control because some breast cancers are sensitive to hormones. Women who use hormonal contraceptives may have a slightly higher chance of getting cervical cancer. However, this may be due to other reasons such as having more sexual partners. |
|||||
|
What should I know about my period when taking ANNOVERA?
When you take ANNOVERA you should expect to have regular 28-day cycles. Each period is likely to last about 5 days. You may have bleeding or spotting between your scheduled periods especially during the first cycle. This bleeding or spotting tends to decrease after the first cycle. Do not stop taking ANNOVERA because of this bleeding or spotting. If the spotting continues for more than 7 consecutive days or if the bleeding is unusually heavy, call your healthcare provider. Tell your healthcare provider if you do not have your period. |
|||||
|
What if I want to become pregnant?
You may stop using ANNOVERA whenever you wish. Consider a visit with your healthcare provider for a pre-pregnancy checkup before you stop taking ANNOVERA. |
|||||
|
General information about the safe and effective use of ANNOVERA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ANNOVERA for a condition for which it was not prescribed. Do not give ANNOVERA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about ANNOVERA that is written for health professionals. |
|||||
|
What if I have other questions?
If you have concerns or questions, ask your healthcare provider. |
|||||
|
What are the ingredients in ANNOVERA?
Active ingredients: segesterone acetate and ethinyl estradiol. Inactive ingredients: dibutyltin dilaurate, silicone elastomers, silicone medical adhesive, and titanium dioxide. |
Section 43683-2 (43683-2)
| Warnings and Precautions (5.11) | 04/2022 |
5.8 Headache
ANNOVERA is contraindicated in females who have headaches with focal neurological symptoms or have migraine headaches with aura, and in females over age 35 years who have migraine headaches with or without aura [see Contraindications (4)].
If a woman taking ANNOVERA develops new headaches that are recurrent, persistent, or severe, evaluate the cause and discontinue ANNOVERA if indicated.
Consider discontinuation of ANNOVERA in the case of increased frequency or severity of migraine during CHC use (which may be prodromal of a cerebrovascular event) [see Contraindications (4)].
10 Overdosage (10 OVERDOSAGE)
There have been no reports of serious ill effects from overdose of CHCs. Overdosage may cause withdrawal bleeding in females and nausea. In case of suspected overdose, all ANNOVERA vaginal systems should be removed and symptomatic treatment given.
15 References (15 REFERENCES)
-
1.Dinger, J, Mohner S, Heinemann K. Cardiovascular risk associated with the use of an etonogestrel- containing vaginal ring. Obstetrics & Gynecology 2013; 122(4): 800-808.
-
2.Sidney, S, Cheetham TC, Connell FA, Oullet-Hellstrom R, Graham DJ, Davis D,, Recent combined hormonal contraceptives (CHCs) and the risk of thromboembolism and other cardiovascular events in new users. Contraception 2013; 87: 93–100.
-
3.Combined hormonal contraceptives (CHCs) and the risk of cardiovascular endpoints. Sidney, S. (primary author) http://www.fda.gov/downloads/Drugs/DrugSafety/UCM277384.pdf, accessed 04- April-2019.
5.14 Chloasma
Chloasma may occur with ANNOVERA use, especially in females with a history of chloasma gravidarum. Advise females who tend to develop chloasma to avoid exposure to the sun or ultraviolet radiation while using ANNOVERA.
11 Description (11 DESCRIPTION)
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white vaginal system containing two active components, a progestin, segesterone acetate, and an estrogen, ethinyl estradiol. When placed in the vagina, each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate and 0.013 mg/day of ethinyl estradiol over the 21 days in-use period of each cycle for up to 13 cycles (total of 273 days). Each cycle is 28 days, with 21 days in and 7 days out.
The inactive ingredients are dibutyltin dilaurate, silicone elastomers, silicone medical adhesive, and titanium dioxide. The elastomers are all methyl siloxane-based polymers.
The vaginal system body has an overall diameter of 56 mm and a cross-sectional diameter of 8.4 mm. It contains two channels of approximately 3.0 mm diameter and 27 mm length into which steroid-containing cores are inserted. Each ANNOVERA contains 103 mg of SA distributed throughout both cores and 17.4 mg of EE distributed throughout only one core. The core containing 40% SA and 12% EE of its mass is 3 mm in diameter and 18 mm in length. The core containing 50% SA of its mass is 3 mm in diameter and 11 mm in length. Contact between the cores and the vaginal system body is fixed by coating the channels with silicone medical adhesive before introducing the cores. After insertion of the cores, the channels are sealed with the silicone medical adhesive.
The structural formulas, and properties for the active components are shown below:
STRUCTURAL FORMULAS:
Segesterone Acetate (SA)
Ethinyl Estradiol (EE)
PROPERTIES:
Established Name: Segesterone Acetate
Chemical Name: 16-methylene-17α-acetoxy-19-nor-pregn-4-ene-3,20-dione
Molecular Formula: C23H30O4
Molecular Weight: 370.5
Physical Form: White, or yellowish white powder
Solubility: Slightly soluble in n-hexane, soluble in ethyl acetate and methanol, freely soluble in acetone (USP classification)
Melting Point: 173°C–177°C
Established Name: Ethinyl Estradiol
Chemical Name: 19-Nor-17α -pregna-1,3,5(10)-trien-20-yne-3,17-diol
Molecular Formula: C20H24O2
Molecular Weight: 296.4
Physical Form: White to slightly yellowish-white crystalline powder
Solubility: Practically insoluble in water, freely soluble in alcohol, it dissolves in alkaline solution
Melting Point: 181°C–185°C
The steroids diffuse out of the vaginal system with release rates that vary over time. Based on in vitro data, the daily release rates of SA and EE are higher during each initial 24–48 hours of use achieving a somewhat lower steady-state with continued use over the subsequent days in each cycle. The vaginal system is designed to be used for 13 cycles (1 year) on a 21/7 days in/out schedule. The total in-use time with the 21/7 days in/out schedule over 13 cycles is 273 days. Based on the residual amount of drug in vaginal systems used in clinical trials over 13 cycles, a total of 41.3 mg of SA and 3.4 mg of EE are released over this period. This translates to an approximate average daily dose of 0.15 mg of segesterone acetate and 0.013 mg of ethinyl estradiol with higher release rate expected at the beginning of dosing and a lower release rate toward the end.
5.10 Depression
Carefully observe females with a history of depression and discontinue ANNOVERA if depression recurs to a serious degree. Data on the association of CHCs with onset of depression or exacerbation of existing depression are limited.
5.16 Vaginal Use
Some females are aware of the vaginal system on occasion during the 21 days of use or during coitus, and partners may feel the vaginal system during coitus.
There is no information on the concomitant use of ANNOVERA with diaphragms, cervical caps, and female condoms.
ANNOVERA may not be suitable for females with conditions that make the vagina more susceptible to vaginal irritation or ulceration. Vaginal and cervical erosion and/or ulceration has been reported in females using other contraceptive vaginal devices. In some cases, the ring adhered to vaginal tissue, which necessitated removal by a healthcare provider.
5.4 Hypertension
ANNOVERA is contraindicated in females with uncontrolled hypertension or hypertension with vascular disease [see Contraindications (4)]. For all females, including those with well- controlled hypertension, monitor blood pressure at routine visits and stop ANNOVERA if blood pressure rises significantly.
An increase in blood pressure has been reported in females using CHCs, and this increase is more likely in older females and with extended duration of use. The effect of CHCs on blood pressure may vary according to the progestin in the CHC.
16.1 How Supplied
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white vaginal system. The vaginal system body has an overall diameter of 56 mm and a cross-sectional diameter of 8.4 mm. When placed inside the vagina, each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate and 0.013 mg/day of ethinyl estradiol over 21-day in-use period of each cycle for up to 13 cycles (total 273 days). Each cycle is 28 days, with 21 days in and 7 days out.
Each ANNOVERA is individually packaged in an aluminum pouch. The pouch consists of a laminate from outside to inside of polyester, aluminum foil, and polyethylene.
First insertion of ANNOVERA must be prior to the date of expiration. Discard one year after first insertions.
A black compact case is provided with the drug product for storage of ANNOVERA by patients during each 7-day vaginal system-out interval.
The vaginal system should be placed in the compact case after 13 cycles of use and discarded via a drug take-back option if one is available. If a take-back option is unavailable, then discard in the waste receptacle out of reach of children and pets. The vaginal system should NOT be flushed down the toilet. See www.fda.gov/drugdisposal for more information about disposal of medicines.
Each box contains 1 ANNOVERA vaginal system in a pouch and 1 storage case.
NDC 68308-752-01
8.4 Pediatric Use
Safety and efficacy of ANNOVERA have been established in women of reproductive age. Efficacy is expected to be the same for postpubertal adolescents under the age of 18 as for users 18 years and older. Use of ANNOVERA before menarche is not indicated.
8.5 Geriatric Use
ANNOVERA has not been studied in females who have reached menopause and is not indicated in this population.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of ANNOVERA was evaluated in two 1-year multicenter trials enrolling 2,265 females, age 18–40 years, who were healthy and sexually active with regular menstrual cycles. The trials were conducted in the U.S., Dominican Republic, Brazil, Chile, Finland, Hungary, Sweden, and Australia, with 67.1% of females from the U.S. The racial/ethnic distribution was Caucasian (71.2%), African-American (14.1%), Asian (3.5%), other/multiple races (11.2%); 28.7% of the study population was Hispanic. The mean age was 26.7 years and the mean (range) BMI was 24.1 (16.0, 41.5) kg/m2. At approximately 50% enrollment, women with BMI >29.0 kg/m2 were no longer enrolled in the two trials and all women with a BMI >29.0 kg/m2 were discontinued from the trials.
Based on pooled data from the two trials, 2,111 females ≤35 years of age completed 17,427 evaluable 28-day cycles (cycles in which no back-up contraception was used). The pooled pregnancy rate, evaluated by the Pearl Index (PI), was 2.98 (95% Confidence Interval [2.13, 4.06]) per 100 woman-years of ANNOVERA use.
Return to fertility was assessed in 290 of the subjects in the two trials who either desired pregnancy or switched to a nonhormonal method after the trials, and all 290 subjects reported a return to fertility during the 6-month follow-up period (defined as a return of menses or pregnancy).
4 Contraindications (4 CONTRAINDICATIONS)
ANNOVERA is contraindicated in females who are known to have or develop the following conditions:
- A high risk of arterial or venous thrombotic diseases. Examples include females who are known to:
-
-Smoke, if over age 35 [see Boxed Warning and Warnings and Precautions (5.1)].
-
-Have current or history of deep vein thrombosis or pulmonary embolism [see Warnings and Precautions (5.1)].
-
-Have cerebrovascular disease [see Warnings and Precautions (5.1)].
-
-Have coronary artery disease [see Warnings and Precautions (5.1)].
-
-Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see Warnings and Precautions (5.1)].
-
-Have inherited or acquired hypercoagulopathies [see Warnings and Precautions (5.1)].
-
-Have uncontrolled hypertension or hypertension with vascular disease [see Warnings and Precautions (5.4)].
-
-Have diabetes mellitus and are over age 35, diabetes mellitus with hypertension or vascular disease, or other end-organ damage, or diabetes mellitus of >20 years duration [see Warnings and Precautions (5.5), (5.7)].
-
-Have headaches with focal neurological symptoms, migraine headaches with aura, or are over age 35 with any migraine headaches [see Warnings and Precautions (5.8)].
-
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive [see Warnings and Precautions (5.11)].
- Liver tumors, acute hepatitis, or severe (decompensated) cirrhosis [see Warnings and Precautions (5.2) and Use in Specific Populations (8.6)].
- Undiagnosed abnormal uterine bleeding [see Warnings and Precautions (5.9)].
- Hypersensitivity to any of the components of ANNOVERA. Hypersensitivity reactions reported include: throat constriction, facial edema, urticaria, hives, and wheezing [see Adverse Reactions (6.1)].
- Use of Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine transaminase (ALT) elevations [see Warnings and Precautions (5.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described elsewhere in other sections of the labeling:
- Serious cardiovascular events and stroke [see Boxed Warning and Warnings and Precautions (5.1)]
- Vascular events [see Warnings and Precautions (5.1)]
- Liver disease [see Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
The sections below provide information on substances for which data on drug interactions with CHCs are available. There is little information available about the clinical effect of most drug interactions that may affect ANNOVERA. However, based on the known pharmacokinetic effects of these drugs, clinical strategies to minimize any potential adverse effect on contraceptive effectiveness or safety are suggested.
Consult the approved product labeling of all concurrently used drugs to obtain further information about interactions with ANNOVERA or the potential for metabolic enzyme or transporter system alterations.
8.7 Renal Impairment
No studies were conducted in subjects with renal impairment; ANNOVERA is not recommended in patients with renal impairment.
Instructions for Use
ANNOVERA® (ann-o-VER-ah)
(segesterone acetate and ethinyl estradiol vaginal system)
Read this Instructions for Use carefully before you decide if ANNOVERA is right for you. This information does not take the place of talking with your gynecologist or other healthcare provider who specializes in women's health. If you have any questions about ANNOVERA, ask your healthcare provider. You should also learn about other birth control methods to choose the one that is best for you.
How should I start using ANNOVERA?
If you are not currently using hormonal birth control, start using ANNOVERA between Days 2 and 5 of your menstrual period.
- If your menstrual periods are not regular or if you start using ANNOVERA more than 5 days from when you started your menstrual period, you should use a barrier method of birth control, such as a male condom or spermicide during sexual intercourse for the first 7 days you use ANNOVERA.
If you are changing from a birth control pill or patch or monthly disposable contraceptive vaginal ring to ANNOVERA:
- If you have been using your birth control method correctly and are certain that you are not pregnant, you can change to ANNOVERA any day of your birth control cycle. Do not start ANNOVERA any later than the day you would start your next birth control pill, apply your next patch or insert your next monthly disposable contraceptive vaginal ring.
If you are changing from a progestin-only birth control method, such as a minipill, injection, implant or intrauterine system (IUS):
- You may switch from a minipill on any day. Start using ANNOVERA on the day that you would have taken your next minipill.
- You should switch from an injectable and start using ANNOVERA on the day when your next injection would be due.
- You should switch from an implant or the IUS and start using ANNOVERA at the time the implant or IUS is removed.
If you are changing from a minipill, injection, implant, or an intrauterine system to ANNOVERA, you should use a barrier method of birth control, such as a male condom or spermicide, during sexual intercourse for the first 7 days you use ANNOVERA.
If you start using ANNOVERA after an abortion or miscarriage:
-
Following a first trimester abortion or miscarriage: You may start ANNOVERA within 5 days following a first trimester abortion or miscarriage (the first 12 weeks of pregnancy). You do not need to use an additional birth control method.
- If you do not start ANNOVERA within 5 days after a first trimester abortion or miscarriage, use a non-hormonal birth control method, such as male condoms or spermicide, while you wait for your menstrual period to start. Start using ANNOVERA between days 2 and 5 of your menstrual period.
- If you start using ANNOVERA more than 5 days from when you started your menstrual period, you should use a barrier method of birth control, such as a male condom or spermicide during sexual intercourse for the first 7 days you use ANNOVERA.
- Following a second trimester abortion or miscarriage: You may start using ANNOVERA no sooner than 4 weeks (28 days) after a second trimester abortion (after the first 12 weeks of pregnancy).
If you are starting ANNOVERA after childbirth:
- You may start using ANNOVERA no sooner than 4 weeks (28 days) after having a baby if you are not breastfeeding.
- If you have not gotten your menstrual period after childbirth, you should talk to your healthcare provider. You may need a pregnancy test to make sure you are not pregnant before you start using ANNOVERA.
- Use another birth control method, such as a male condom or spermicide, during sexual intercourse for the first 7 days you use ANNOVERA if you have not yet had a period.
If you are breastfeeding, you should not use ANNOVERA. Use other birth control methods until you are no longer breastfeeding.
How do I use ANNOVERA?
|
|
Step 1. Open the package and remove ANNOVERA.
|
|
|
Step 2. Prepare to insert ANNOVERA.
|
|
|
Step 3. Choose a position for insertion of ANNOVERA.
|
|
|
Step 4. Insert ANNOVERA into your vagina.
|
|
|
Note:
|
|
|
Step 5. How do I remove ANNOVERA?
|
What else should I know about ANNOVERA?
- The day of the week when you first insert ANNOVERA (called "Day 1") is your vaginal system change day. This is described further in the Patient Information section entitled "How should I use ANNOVERA?"
- For each cycle, you put the vaginal system into your vagina and let it stay there 3 weeks (21 full days). Remember to keep the vaginal system in for the whole 3 weeks (21 full days).
- You take the vaginal system out on your vaginal system change day (Day 22) and let it stay out for 1 week (7 full days). Note that your vaginal system should be stored in the case provided, away from children, pets, and extreme temperatures.
- Then you start over again for another 4 weeks. You may not be bleeding when you put the vaginal system in.
- Always put the vaginal system in or take it out on your vaginal system change day at about the same time of day. For example, if you put your vaginal system in on Monday at 9:00 in the morning, always take it out or put it back in on Monday at about 9:00 in the morning.
- You do not have to take the vaginal system out when you have sex. If you decide to remove it, remember to reinsert it within 2 hours after removing it or you may not be protected from pregnancy. However, if ANNOVERA is out of your vagina for more than 2 hours at one time or if ANNOVERA is out of your vagina at different times that add up to more than a total of 2 hours over the first 21 days of your cycle, you will need to use another method of birth control until ANNOVERA has been in your vagina for 7 days in a row, such as male condoms or spermicides.
- Repeat the 4-week cycle for all 13 cycles of the use of the vaginal system.
- Do not use the vaginal system for more than 13 cycles (1 year). When you take the vaginal system out of your vagina at the end of the 13 cycles, dispose of it. See "How should I dispose of (throw away) ANNOVERA?" below.
- If you want to continue using ANNOVERA after 13 cycles, you will need to get a new prescription from your healthcare provider to obtain a new vaginal system.
- Do not to use any vaginal products such as oil-based suppositories, oil-based creams, or oil-based gels while the vaginal system is in your vagina. Do not use any vaginal lubricants that have silicone or oil in them. Water-based lubricants are ok to use. Be sure to read the ingredients on the labels carefully before you buy a vaginal lubricant.
- First insertion of ANNOVERA must be prior to the date of expiration. Discard one year after first insertion. See "How should I dispose of (throw away) ANNOVERA?" below.
- Do not take any medicines unless your healthcare provider says it is ok to take them.
Tell your healthcare provider when you start a new medicine.
How do I clean ANNOVERA?
- After removing ANNOVERA from your vagina, wash it with mild soap and warm water, rinse, and pat it dry with a clean cloth towel or paper towel before you store it. Only use mild soap and warm water to clean.
- Wash ANNOVERA with mild soap and warm water and pat it dry before you put it back into your vagina.
- Store ANNOVERA in the case provided.
How should I store ANNOVERA?
- Store ANNOVERA in the supplied case at room temperature between 68°F to 77°F (20°C to 25°C).
- Protect ANNOVERA from direct sunlight.
- Do not refrigerate or freeze ANNOVERA.
- Avoid storing ANNOVERA in extreme heat.
- Keep ANNOVERA and all medicines out of the reach of children and pets.
How should I dispose of (throw away) ANNOVERA?
- After 13 cycles of use, place ANNOVERA in the case that comes with it.
- Dispose of ANNOVERA at a drug take-back location, if available. If a drug take-back location is not available, dispose of ANNOVERA in the trash, out of the reach of children and pets.
- Do not throw away ANNOVERA in the toilet.
- See www.fda.gov/drugdisposal for more information about disposal of medicines.
When does ANNOVERA become effective?
ANNOVERA becomes effective on the day it is inserted (Day 1), if it is inserted between Days 2 and 5 of your menstrual period. If ANNOVERA is inserted more than 5 days from when you start your menstrual period, you should use a barrier method of birth control, such as a male condom or spermicide, during sexual intercourse for the first 7 days you use ANNOVERA.
Will ANNOVERA interfere during sexual intercourse?
- If ANNOVERA is placed as high as possible in your vagina, it will not interfere with sexual intercourse.
Can I take ANNOVERA out during the first 3 weeks of my cycle?
- You should leave ANNOVERA in the entire 21 days (3 weeks).
- If ANNOVERA comes out or if you remove it, put it back in as soon as possible. If you put ANNOVERA back into your vagina before 2 hours has passed, you do not need to use another method of birth control. However, if ANNOVERA is out of your vagina for more than 2 hours at one time or if ANNOVERA is out of your vagina at different times that add up to more than a total of 2 hours over the first 21 days of your cycle, you will need to use another method of birth control until ANNOVERA has been in your vagina for 7 days in a row, such as male condoms or spermicides.
What should I do if ANNOVERA comes out of my vagina?
ANNOVERA can slip or accidentally come out of your vagina (expelled), for example, during sexual intercourse, bowel movements, or use of tampons.
- If ANNOVERA slips out of your vagina, wash ANNOVERA with mild soap and warm water, rinse and pat dry with a clean cloth towel or paper towel and put it back in your vagina right away or within 2 hours. See "Can I take ANNOVERA out during the first 3 weeks of my cycle?" in the section above.
- ANNOVERA can move around and become visible at the opening of your vagina. If this happens follow "Step 4" above for directions on how to push ANNOVERA back to its right position.
What if I lose ANNOVERA?
- Call your healthcare provider right away if you lose ANNOVERA. You should use a back-up method of birth control such as a male condom or spermicide for preventing pregnancy until you get a new ANNOVERA.
If you have other questions call your healthcare provider.
For information, call Mayne Pharma at 1-844- 825-8500
Distributed by:
Mayne Pharma
Raleigh, NC 27609
ANNOVERA is a registered trademark of Population Council and licensed under license.
The Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Revised 08/2024
8.6 Hepatic Impairment
No studies have been conducted to evaluate the effect of hepatic impairment on the disposition of ANNOVERA. However, steroid hormones may be poorly metabolized in patients with hepatic impairment. Acute or chronic disturbances of liver function may necessitate the discontinuation of CHC use until markers of liver function return to normal and CHC causation has been excluded [see Contraindications (4) and Warnings and Precautions (5.2)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ANNOVERA is indicated for use by females of reproductive potential to prevent pregnancy.
16.2 Storage Conditions
Prior to dispensing ANNOVERA to the user, store ANNOVERA at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Protect ANNOVERA from direct sunlight.
Do not refrigerate or freeze and avoid excessive heat.
2.1 How to Use Annovera (2.1 How to Use ANNOVERA)
Instruct patients that ANNOVERA should be used as directed [see How to Start ANNOVERA (2.2)]. One ANNOVERA should be placed in the vagina. For maximum contraceptive effectiveness, ANNOVERA is to remain in the vagina continuously for 21 days (3 complete weeks). It is removed for a 1-week dose-free interval, and during this time a withdrawal bleed usually occurs. The removed vaginal system should be cleaned with mild soap and warm water, patted dry with a clean cloth towel or paper towel, and placed in its case during the 1-week dose- free interval. At the end of the dose-free interval, the vaginal system should be cleaned prior to being placed back in the vagina for another 21 continuous days (3 complete weeks). This pattern of ANNOVERA use made up of 3-weeks in and 1-week out is a cycle of use; one ANNOVERA vaginal system will provide contraception for 13 cycles.
With clean hands, the user can choose an insertion position that is comfortable, such as lying down, squatting, or standing. The sides of the vaginal system are pressed together for insertion into the vagina. When properly inserted, the vaginal system should be entirely in the vagina and behind the pelvic bone. The day and time of insertion should be noted so that the vaginal system can be removed 3 weeks later on the same day and at about the same time.
ANNOVERA can be removed by hooking an index finger into the vaginal system inside the vagina and gently pulling the vaginal system.
For patient instructions regarding cleaning the vaginal system, see FDA-approved Patient Information.
5.6 Gallbladder Disease
Studies suggest a small increased relative risk of developing gallbladder disease among CHC users. Use of CHCs may also worsen existing gallbladder disease.
A past history of CHC-related cholestasis predicts an increased risk with subsequent CHC use. Females with a history of pregnancy-related cholestasis may be at an increased risk for CHC- related cholestasis.
12.1 Mechanism of Action
CHCs lower the risk of becoming pregnant primarily by suppressing ovulation.
2.2 How to Start Annovera (2.2 How to Start ANNOVERA)
IMPORTANT: Consider the possibility of ovulation and conception prior to the first use of ANNOVERA.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Thrombotic Disorders and Other Vascular Problems: Stop ANNOVERA if a thrombotic or thromboembolic event occurs. Stop ANNOVERA at least 4 weeks before and through 2 weeks after major surgery. Start ANNOVERA no earlier than 4 weeks after delivery, in females who are not breastfeeding. Consider cardiovascular risk factors before initiating in all females, particularly those over 35 years. (5.1, 5.5)
- Liver Disease: Discontinue if jaundice occurs. (5.2)
- Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment: Stop ANNOVERA prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir. ANNOVERA can be restarted 2 weeks following completion of this regimen. (5.3)
- Hypertension: Do not prescribe ANNOVERA for females with uncontrolled hypertension or hypertension with vascular disease. If used in females with well-controlled hypertension, monitor blood pressure and stop use if blood pressure rises significantly. (5.4)
- Carbohydrate and lipid metabolic effects: Monitor glucose in pre-diabetic and diabetic females taking ANNOVERA. Consider an alternate contraceptive method for females with uncontrolled dyslipidemias. (5.7)
- Headache: Evaluate significant change in headaches and discontinue ANNOVERA if indicated. (5.8)
- Bleeding Irregularities and Amenorrhea: May cause irregular bleeding or amenorrhea. Evaluate for other causes if irregular bleeding or amenorrhea persists. (5.9)
5.13 Hereditary Angioedema
In females with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
One ANNOVERA is inserted in the vagina. The vaginal system must remain in place continuously for 3 weeks (21 days) followed by a 1-week (7-day) vaginal system-free interval. One vaginal system provides contraception for thirteen 28-day cycles (1 year). (2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Vaginal system: Each ANNOVERA releases an approximate average 0.15 mg/day of segesterone acetate (SA) and 0.013 mg/day of ethinyl estradiol (EE) when placed in the vagina over a period of 21 days of each cycle for up to 13 cycles (total of 273 days). Each cycle of use is 28 days with 21 days in and 7 days out.
ANNOVERA (segesterone acetate and ethinyl estradiol vaginal system) is a toroidal- shaped (ring), nonbiodegradable, flexible, opaque white, contraceptive vaginal system containing 103 mg of SA and 17.4 mg EE.
ANNOVERA is 56 mm in overall diameter and 8.4 mm in cross-sectional diameter. It contains 2 channels of approximately 3.0 mm diameter and 27 mm length into which steroid-containing cores are inserted. The cores are 3 mm in diameter with one releasing SA alone and the other releasing both SA and EE.
Each ANNOVERA is designed to be used for up to 13 cycles (1 year).
ANNOVERA is not made with natural rubber latex.
6.2 Postmarketing Experience
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 2).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 2). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
Figure 2: Relevant Studies of Risk of Breast Cancer with Combined Oral Contraceptives
RR = relative risk; OR = odds ratio; HR = hazard ratio. "ever COC" are females with current or past COC use; "never COC use" are females who never used COCs.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The clinical trials that evaluated the safety of ANNOVERA were obtained from three 13- cycle trials. One trial was conducted entirely in the U.S. (15 sites), and the other two were global studies that included 5 U.S. sites and 7 international sites (Australia, Brazil, Chile, Dominican Republic, Finland, Hungary, Sweden). All three trials were open label and enrolled healthy females, desiring contraception, 18 to 40 years of age. At about 50% enrollment, females with BMI >29 kg/m2 were excluded due to the occurrence of two VTEs in this subgroup. In total, 2,308 females contributed 21,590 cycles of exposure for safety evaluation and 999 completed 13 cycles; there were 209 subjects with BMI >29 kg/m2 who contributed 1,254 cycles of exposure with 36 subjects completing 13 cycles. The demographic profile for subjects was: mean age 26.7 years, mean BMI 24.1 (16.0- 41.5) kg/m2; 67% were from the U.S. The racial distribution was 71% Caucasian, 14% African American, 4% Asian, and 11% Other; 30% of the population was Hispanic.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.5 Age Related Considerations (5.5 Age-Related Considerations)
The risk for cardiovascular disease and prevalence of risk factors for cardiovascular disease increase with age. Certain conditions, such as smoking and migraine headache without aura, that do not contraindicate CHC use in younger females, are contraindications to use in women over 35 years of age [see Contraindications (4) and Warnings and Precautions (5.1)]. Consider the presence of underlying risk factors that may increase the risk of cardiovascular disease or VTE, particularly before initiating ANNOVERA for women over 35 years, such as:
- Hypertension
- Diabetes
- Dyslipidemia
- Obesity
5.15 Toxic Shock Syndrome (tss) (5.15 Toxic Shock Syndrome (TSS))
Cases of TSS have been reported by vaginal ring users. TSS has been associated with tampons and certain barrier contraceptives, and in some TSS cases ring users were also using tampons. Causal relationship between the use of a vaginal ring and TSS has not been established. No cases of TSS occurred in clinical trials with ANNOVERA. If a patient exhibits signs or symptoms of TSS, consider the possibility of this diagnosis, remove ANNOVERA, and initiate appropriate medical evaluation and treatment.
5.12 Effect On Binding Globulins (5.12 Effect on Binding Globulins)
The estrogen component of ANNOVERA may raise the serum concentrations of thyroxine- binding globulin, sex hormone-binding globulin, and cortisol-binding globulin. The dose of replacement thyroid hormone or cortisol therapy may need to be increased.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
8.8 Body Mass Index (bmi)/body Weight (8.8 Body Mass Index (BMI)/Body Weight)
The safety and efficacy of ANNOVERA in females with a BMI >29 kg/m2 have not been adequately evaluated because this subpopulation was excluded from the clinical trials after 2 VTEs occurred in females with a BMI > 29 kg/m2 [see Adverse Reactions (6.1) and Clinical Studies (14)]. Higher body weight is associated with lower systemic exposure of SA and EE [see Clinical Pharmacology (12.3)].
7.5 Interference With Laboratory Tests (7.5 Interference with Laboratory Tests)
The use of contraceptive steroids may influence the results of certain laboratory tests, such as coagulation factors, lipids, glucose tolerance, and binding proteins.
7.3 Use of Vaginal Products With Annovera (7.3 Use of Vaginal Products with ANNOVERA)
In a drug-drug interaction study with ANNOVERA and the concurrent use of three different formulations of vaginal miconazole, the use of water-based vaginal miconazole cream resulted in no change in exposure to EE or SA from the vaginal system. However, the use of either the 1- day or the 3-day oil-based miconazole suppository was associated with an overall increase in exposure up to 67% for EE and 32% for SA. Considering the potential long-term effect on vaginal system performance, concurrent use of oil-based vaginal suppositories should not occur with ANNOVERA use. If there is a need to treat a vaginal condition, water-based vaginal cream or oral therapy may be used concurrently with the vaginal system [see Clinical Pharmacology (12.3)].
5.9 Bleeding Irregularities and Amenorrhea
Bleeding and/or spotting that occurs at any time while the vaginal system is inserted is considered "unscheduled" bleeding/spotting. Bleeding/spotting that occurs during the dose-free week when the vaginal system is out is considered "scheduled" bleeding.
2.3 Deviations From the Recommended Regimen (2.3 Deviations from the Recommended Regimen)
Contraceptive efficacy of ANNOVERA may be reduced if a woman deviates from the recommended use. ANNOVERA should remain in the vagina for a continuous period of 21 days (3 weeks); then ANNOVERA should be taken out of the vagina for 7 days. In a 28-day cycle, a deviation that involves ANNOVERA being out of the vagina for less than 7 days will not increase pregnancy risk. In a 28-day cycle, a deviation that involves ANNOVERA being out of the vagina for more than 7 days will increase pregnancy risk and back-up contraception is recommended in these instances. Deviations from the recommended regimen may result in a new vaginal system change day [See FDA- approved Patient Information].
Principal Display Panel 30 Ring Pouch Carton (PRINCIPAL DISPLAY PANEL - 30 Ring Pouch Carton)
NDC 68308-752-01
Rx only
Annovera®
(segesterone acetate and
ethinyl estradiol vaginal system)
Delivers 0.15 mg/0.013 mg per day
This product is intended to prevent pregnancy.
It does not protect against HIV infection (AIDS)
and other sexually transmitted diseases.
Keep out of reach of children and pets
For Vaginal Use
CONTAINS 1 VAGINAL SYSTEM
mayne pharma
5.1 Thromboembolic Disorders and Other Vascular Conditions
Females are at increased risk for a venous thrombotic event (VTE) when using ANNOVERA. Limited data are available in females with a BMI >29.0 kg/m2 because this subpopulation was excluded from the clinical trials after VTEs were reported.
- Stop ANNOVERA if an arterial or deep venous thrombotic event occurs.
- Stop ANNOVERA if there is unexplained loss of vision, proptosis, diplopia, papilledema, or retinal vascular lesions and evaluate for retinal vein thrombosis immediately.
- Discontinue ANNOVERA during prolonged immobilization. If feasible, stop ANNOVERA at least 4 weeks before and through 2 weeks after major surgery or other surgeries known to have an elevated risk of VTE.
- Start ANNOVERA no earlier than 4 weeks after delivery in females who are not breastfeeding. The risk of postpartum VTE decreases after the third postpartum week, whereas the risk of ovulation increases after the third postpartum week.
- Before starting ANNOVERA, evaluate any past medical history or family history of thrombotic or thromboembolic disorders and consider whether the history suggests an inherited or acquired hypercoagulopathy. ANNOVERA is contraindicated in females with a high risk of arterial or venous thrombotic/thromboembolic diseases [see Contraindications (4)].
Warning: Cigarette Smoking and Serious Cardiovascular Events (WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS)
Cigarette smoking increases the risk of serious cardiovascular events from combination hormonal contraceptive (CHC) use. This risk increases with age, particularly in females over 35 years of age, and with the number of cigarettes smoked. For this reason, CHC should not be used by females who are over 35 years of age and smoke. [See Contraindications (4) and Warnings and Precautions (5.5)].
7.2 Effects of Combined Hormonal Contraceptives On Other Drugs (7.2 Effects of Combined Hormonal Contraceptives on Other Drugs)
Table 4 provides significant drug interaction information for drugs co-administered with CHCs.
| Lamotrigine | |
| Clinical effect | Concomitant use of CHCs with lamotrigine may significantly decrease systemic exposure of lamotrigine due to induction of lamotrigine glucuronidation. |
| Prevention or management | Dose adjustment for lamotrigine may be necessary. Consult the approved product labeling for lamotrigine. |
| Thyroid Hormone Replacement Therapy or Corticosteroid Replacement Therapy | |
| Clinical effect | Concomitant use of CHCs with thyroid hormone replacement therapy or corticosteroid replacement therapy may increase systemic exposure of thyroid-binding and cortisol-binding globulin [see Warnings and Precautions (5.12)]. |
| Prevention or management | The dose of replacement thyroid hormone or cortisol therapy may need to be increased. Consult the approved product labeling for the therapy in use [see Warnings and Precautions (5.12)]. |
| Other Drugs | |
| Clinical effect | Concomitant use of CHCs may decrease systemic exposure of acetaminophen, morphine, salicylic acid, and temazepam. Concomitant use with ethinyl estradiol-containing CHCs may increase systemic exposure of other drugs (eg, cyclosporine, prednisolone, theophylline, tizanidine, and voriconazole). |
| Prevention or management | The dosage of drugs that can be affected by this interaction may need to be increased or decreased. Consult the approved product labeling for the concomitantly used drug. |
5.3 Risk of Liver Enzyme Elevations With Concomitant Hepatitis C Treatment (5.3 Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment)
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with and without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in females using ethinyl estradiol-containing medications, such as ANNOVERA. Discontinue ANNOVERA prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir [see Contraindications (4)]. ANNOVERA can be restarted approximately 2 weeks following completion of treatment with the Hepatitis C combination drug regimen.
7.4 Concomitant Use With Hcv Combination Therapy – Liver Enzyme Elevation (7.4 Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation)
Do not co-administer ANNOVERA with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations [see Contraindications (4) and Warnings and Precautions (5.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:49.103037 · Updated: 2026-03-14T22:25:41.517877