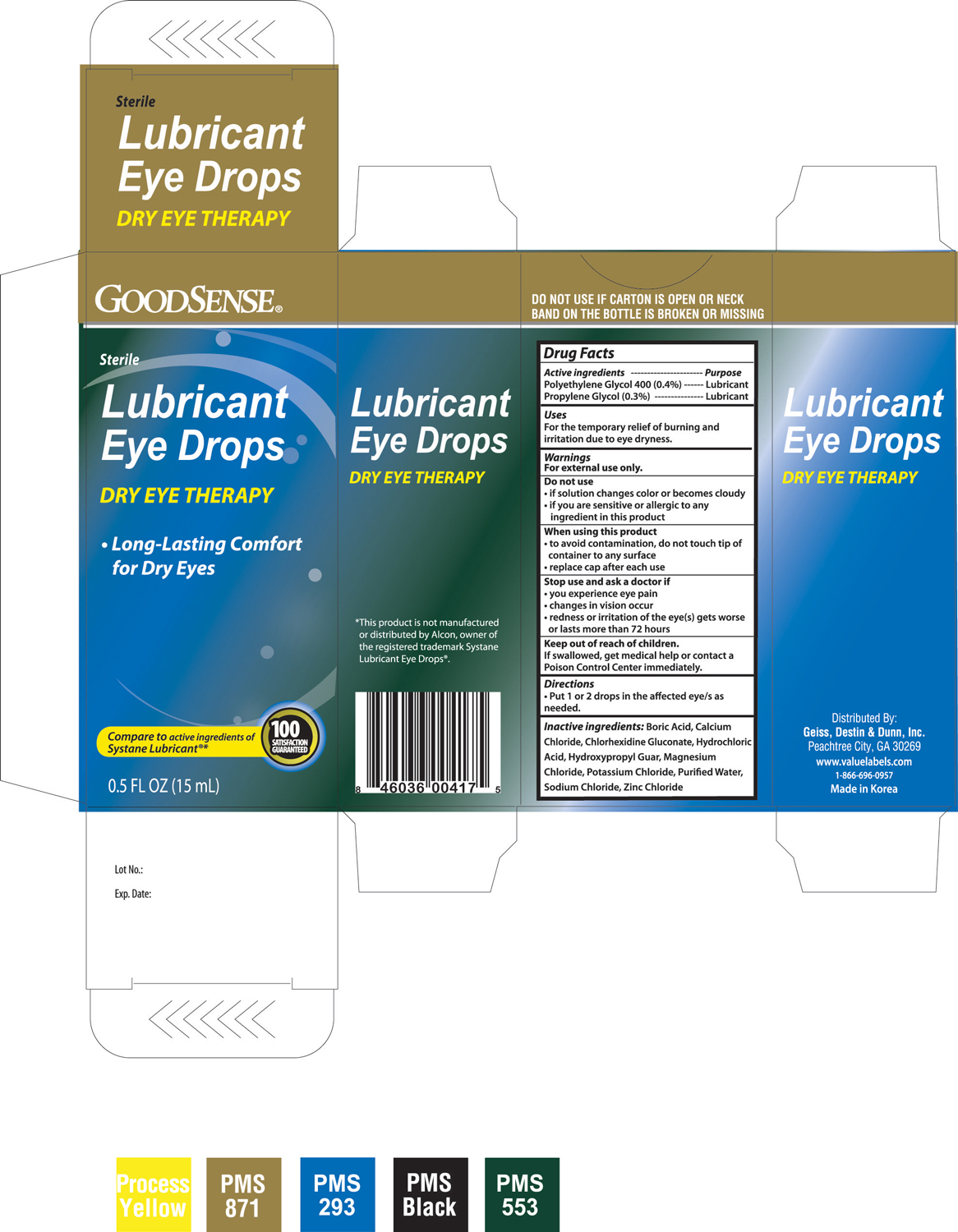
Drug Facts
eb0f7264-29d6-405d-8102-b7f902f2482d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients -----------------------------------------------------------------------Purpose Polyethylene Glycol 400 (0.4%) ---------------------------------------------------Lubricant Propylene Glycol (0.3%) --------------------------------------------------------------Lubricant
Medication Information
Warnings and Precautions
Warnings
For external use only.
Indications and Usage
Directions
- Put 1 or 2 drops in the affected eye/s as needed.
Description
Active ingredients -----------------------------------------------------------------------Purpose Polyethylene Glycol 400 (0.4%) ---------------------------------------------------Lubricant Propylene Glycol (0.3%) --------------------------------------------------------------Lubricant
Section 50565-1
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center immediately.
Section 50566-9
Stop use and ask a doctor if
- you experience eye pain
- changes in vision occur
- redness or irritation of the eye(s) gets worse or lasts more than 72 hours
Section 50567-7
When using this product
- to avoid contamination, do not touch tip of container to any surface
- replace cap after each use
Section 50570-1
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive or allergic to any ingredient in this product
Section 51727-6
Inactive ingredients: Boric Acid, Calcium Chloride, Chlorhexidine Gluconate, Hydrochloric Acid, Hydroxypropyl Guar, Magnesium Chloride, Potassium Chloride, Purified Water, Sodium Chloride, Zinc Chloride
Section 51945-4
Enter section text here
Section 55105-1
Uses
For the temporary relief of burning and irritation due to eye dryness.
Section 55106-9
Active ingredients -----------------------------------------------------------------------Purpose
Polyethylene Glycol 400 (0.4%) ---------------------------------------------------Lubricant
Propylene Glycol (0.3%) --------------------------------------------------------------Lubricant
Structured Label Content
Indications and Usage (34067-9)
Directions
- Put 1 or 2 drops in the affected eye/s as needed.
Warnings and Precautions (34071-1)
Warnings
For external use only.
Section 50565-1 (50565-1)
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center immediately.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you experience eye pain
- changes in vision occur
- redness or irritation of the eye(s) gets worse or lasts more than 72 hours
Section 50567-7 (50567-7)
When using this product
- to avoid contamination, do not touch tip of container to any surface
- replace cap after each use
Section 50570-1 (50570-1)
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive or allergic to any ingredient in this product
Section 51727-6 (51727-6)
Inactive ingredients: Boric Acid, Calcium Chloride, Chlorhexidine Gluconate, Hydrochloric Acid, Hydroxypropyl Guar, Magnesium Chloride, Potassium Chloride, Purified Water, Sodium Chloride, Zinc Chloride
Section 51945-4 (51945-4)
Enter section text here
Section 55105-1 (55105-1)
Uses
For the temporary relief of burning and irritation due to eye dryness.
Section 55106-9 (55106-9)
Active ingredients -----------------------------------------------------------------------Purpose
Polyethylene Glycol 400 (0.4%) ---------------------------------------------------Lubricant
Propylene Glycol (0.3%) --------------------------------------------------------------Lubricant
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:06.497712 · Updated: 2026-03-14T22:52:26.166879