Moisture Surge Cc
ea4d564f-ac64-abac-e053-2a95a90a8abb
34390-5
HUMAN OTC DRUG LABEL
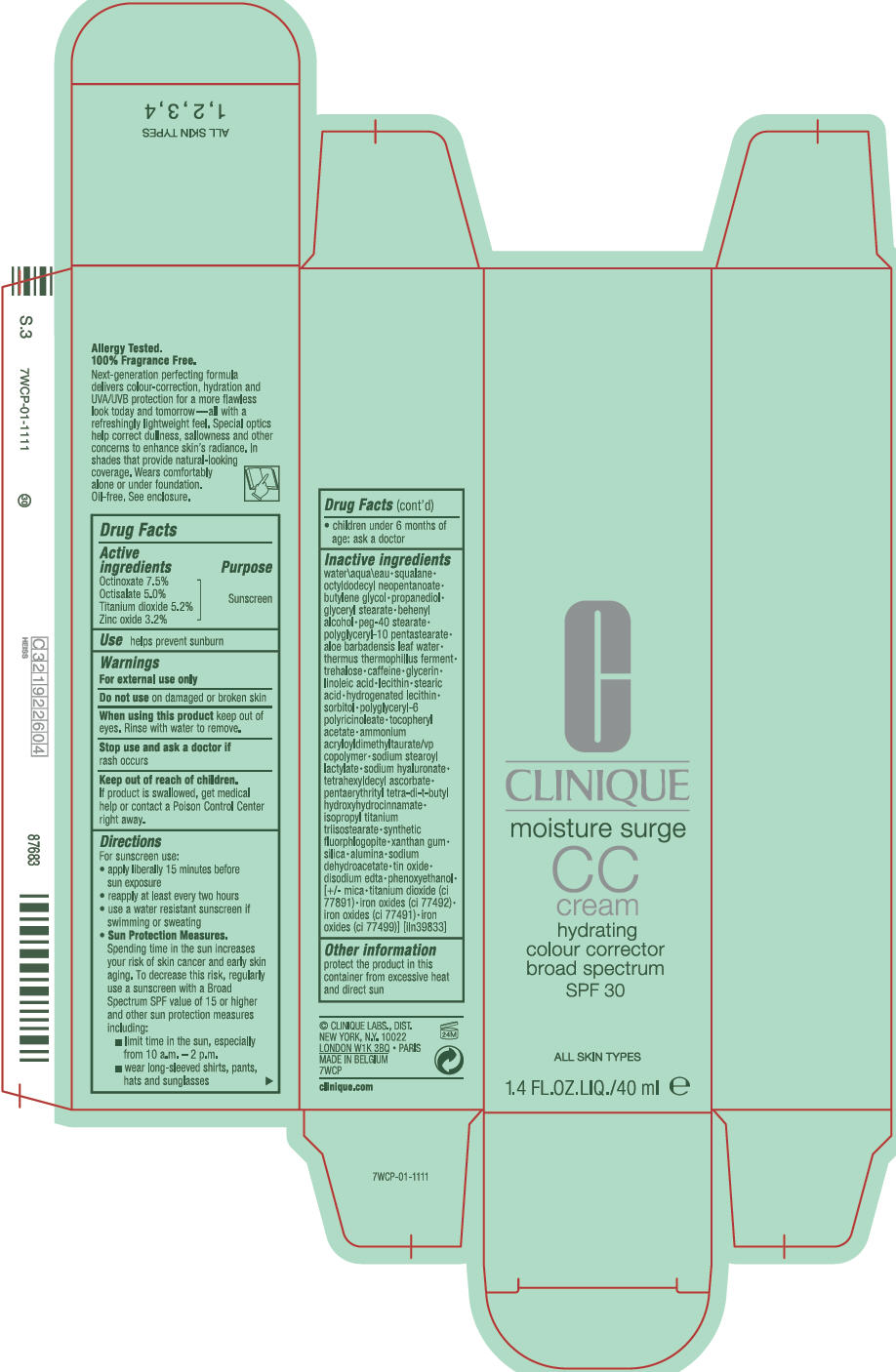
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Sunscreen
Medication Information
Purpose
Sunscreen
Description
Drug Facts
Use
helps prevent sunburn
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if rash occurs
Section 50567-7
When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1
Do not use on damaged or broken skin
Warnings
For external use only
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
Other Information
protect the product in this container from excessive heat and direct sun
Active Ingredients
Octinoxate 7.5%
Octisalate 5.0%
Titanium dioxide 5.2%
Zinc oxide 3.2%
Inactive Ingredients
water\aqua\eau • squalane • octyldodecyl neopentanoate • butylene glycol • propanediol • glyceryl stearate • behenyl alcohol • peg-40 stearate • polyglyceryl-10 pentastearate • aloe barbadensis leaf water • thermus thermophillus ferment • trehalose • caffeine • glycerin • linoleic acid • lecithin • stearic acid • hydrogenated lecithin • sorbitol • polyglyceryl-6 polyricinoleate • tocopheryl acetate • ammonium acryloyldimethyltaurate/vp copolymer • sodium stearoyl lactylate • sodium hyaluronate • tetrahexyldecyl ascorbate • pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate • isopropyl titanium triisostearate • synthetic fluorphlogopite • xanthan gum • silica • alumina • sodium dehydroacetate • tin oxide • disodium edta • phenoxyethanol • [+/- mica • titanium dioxide (ci 77891) • iron oxides (ci 77492) • iron oxides (ci 77491) • iron oxides (ci 7799)] [iln39833]
Principal Display Panel 40 Ml Bottle Carton
C
CLINIQUE
moisture surge
CC
cream
hydrating
colour corrector
broad spectrum
SPF 30
ALL SKIN TYPES
1.4 FL.OZ.LIQ./40 ml e
Structured Label Content
Use
helps prevent sunburn
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if rash occurs
Section 50567-7 (50567-7)
When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1 (50570-1)
Do not use on damaged or broken skin
Purpose
Sunscreen
Warnings
For external use only
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
Other Information (Other information)
protect the product in this container from excessive heat and direct sun
Active Ingredients (Active ingredients)
Octinoxate 7.5%
Octisalate 5.0%
Titanium dioxide 5.2%
Zinc oxide 3.2%
Inactive Ingredients (Inactive ingredients)
water\aqua\eau • squalane • octyldodecyl neopentanoate • butylene glycol • propanediol • glyceryl stearate • behenyl alcohol • peg-40 stearate • polyglyceryl-10 pentastearate • aloe barbadensis leaf water • thermus thermophillus ferment • trehalose • caffeine • glycerin • linoleic acid • lecithin • stearic acid • hydrogenated lecithin • sorbitol • polyglyceryl-6 polyricinoleate • tocopheryl acetate • ammonium acryloyldimethyltaurate/vp copolymer • sodium stearoyl lactylate • sodium hyaluronate • tetrahexyldecyl ascorbate • pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate • isopropyl titanium triisostearate • synthetic fluorphlogopite • xanthan gum • silica • alumina • sodium dehydroacetate • tin oxide • disodium edta • phenoxyethanol • [+/- mica • titanium dioxide (ci 77891) • iron oxides (ci 77492) • iron oxides (ci 77491) • iron oxides (ci 7799)] [iln39833]
Principal Display Panel 40 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 40 ml Bottle Carton)
C
CLINIQUE
moisture surge
CC
cream
hydrating
colour corrector
broad spectrum
SPF 30
ALL SKIN TYPES
1.4 FL.OZ.LIQ./40 ml e
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:47.135363 · Updated: 2026-03-14T23:09:45.619768