These Highlights Do Not Include All The Information Needed To Use Zirgan® Safely And Effectively. See Full Prescribing Information For Zirgan.
ea25e7ac-7a93-4ec8-975b-2cec197e1c27
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ZIRGAN is indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in adults and pediatric patients aged 2 years and older.
Indications and Usage
ZIRGAN is indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in adults and pediatric patients aged 2 years and older.
Dosage and Administration
The recommended dosage is 1 drop in the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals, and then 1 drop 3 times per day for 7 days.
Warnings and Precautions
ZIRGAN is indicated for topical ophthalmic use only. ( 5.1 ) Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with ZIRGAN. ( 5.2 )
Contraindications
None.
Adverse Reactions
Most common adverse reactions reported in patients were blurred vision (60%), eye irritation (20%), punctate keratitis (5%), and conjunctival hyperemia (5%).
Storage and Handling
ZIRGAN (ganciclovir ophthalmic gel) 0.15% is supplied as 5 grams of a sterile, preserved, clear, colorless, topical ophthalmic gel containing 0.15% of ganciclovir in a polycoated aluminum tube with a white polyethylene tip and cap and protective band (NDC 24208-535-35). Storage Store at 15°C to 25°C (59°F to 77°F). Do not freeze.
How Supplied
ZIRGAN (ganciclovir ophthalmic gel) 0.15% is supplied as 5 grams of a sterile, preserved, clear, colorless, topical ophthalmic gel containing 0.15% of ganciclovir in a polycoated aluminum tube with a white polyethylene tip and cap and protective band (NDC 24208-535-35). Storage Store at 15°C to 25°C (59°F to 77°F). Do not freeze.
Medication Information
Warnings and Precautions
ZIRGAN is indicated for topical ophthalmic use only. ( 5.1 ) Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with ZIRGAN. ( 5.2 )
Indications and Usage
ZIRGAN is indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in adults and pediatric patients aged 2 years and older.
Dosage and Administration
The recommended dosage is 1 drop in the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals, and then 1 drop 3 times per day for 7 days.
Contraindications
None.
Adverse Reactions
Most common adverse reactions reported in patients were blurred vision (60%), eye irritation (20%), punctate keratitis (5%), and conjunctival hyperemia (5%).
Storage and Handling
ZIRGAN (ganciclovir ophthalmic gel) 0.15% is supplied as 5 grams of a sterile, preserved, clear, colorless, topical ophthalmic gel containing 0.15% of ganciclovir in a polycoated aluminum tube with a white polyethylene tip and cap and protective band (NDC 24208-535-35). Storage Store at 15°C to 25°C (59°F to 77°F). Do not freeze.
How Supplied
ZIRGAN (ganciclovir ophthalmic gel) 0.15% is supplied as 5 grams of a sterile, preserved, clear, colorless, topical ophthalmic gel containing 0.15% of ganciclovir in a polycoated aluminum tube with a white polyethylene tip and cap and protective band (NDC 24208-535-35). Storage Store at 15°C to 25°C (59°F to 77°F). Do not freeze.
Description
ZIRGAN is indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in adults and pediatric patients aged 2 years and older.
8.1 Pregnancy
Risk Summary
There are no available human data on use of ZIRGAN or ganciclovir during pregnancy to inform any drug-associated risk. Intravenous administration of ganciclovir to pregnant mice or rabbits during organogenesis or during the pre/postnatal period did not produce adverse embryofetal or offspring effects at clinically relevant doses (see Data) .
The background risk in the U.S. general population of major birth defects is 2 to 4% and the risk of miscarriage is 15 to 20% of clinically recognized pregnancies.
Data
Animal Data
Daily intravenous doses of ganciclovir were administered to pregnant mice [up to 108 mg/kg/day, approximately 1400 times the maximum recommended human ocular dose (RHOD) of 0.375 mg] and rabbits [up to 60 mg/kg/day, approximately 2400 times the maximum RHOD], and also to female mice [up to 90 mg/kg, approximately 1174 times the maximum RHOD] prior to mating, during gestation, and during lactation. Fetal resorptions were present in at least 85% of rabbits and mice. Additional effects observed in rabbits included fetal growth retardation, embryolethality, teratogenicity, and/or maternal toxicity. Teratogenic changes included cleft palate, anophthalmia/microphthalmia, aplastic organs (kidney and pancreas), hydrocephaly and brachygnathia. A maternal no observed adverse effect level (NOAEL) was observed at 36 mg/kg/day (approximately 470 times higher than the maximum RHOD, based on body surface area) in mice and at 6 mg/kg/day (approximately 240 times higher than the maximum RHOD, based on body surface area) in rabbits.
In pre/postnatal development studies in mice, there were maternal/fetal toxicity and embryolethality which included fetal effects of hypoplasia of the testes and seminal vesicles in the male offspring, as well as pathologic changes in the nonglandular region of the stomach. A maternal no observed adverse effect level (NOAEL) was observed at 20 mg/kg/day (approximately 261 times higher than the maximum RHOD, based on body surface area).
8.2 Lactation
Risk Summary
No data are available regarding the presence of ganciclovir in human milk, the effects on the breastfed infant, or the effects on milk production. It is not known whether topical ophthalmic ganciclovir administration could result in sufficient systemic absorption to produce detectable quantities in breast milk. Animal data indicate that ganciclovir is excreted in the milk of lactating rats. Caution should be exercised when ZIRGAN is administered to nursing mothers.
11 Description
ZIRGAN (ganciclovir ophthalmic gel) 0.15% contains ganciclovir, a nucleoside analog antiviral. ZIRGAN is a sterile, preserved, clear, colorless, ophthalmic gel for topical ophthalmic use.
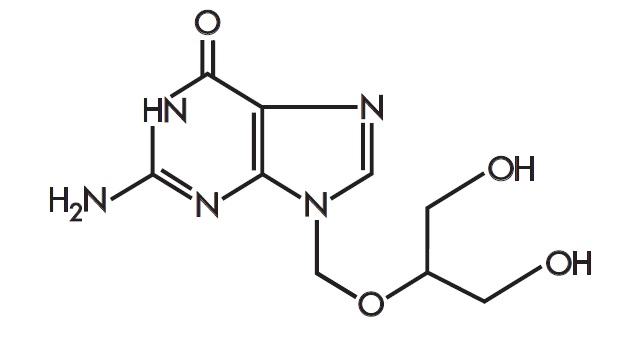
The chemical name is 9-[[2-hydroxy-1-(hydroxymethyl)ethoxy]methyl]guanine (CAS number 82410-32-0). Ganciclovir is represented by the following structural formula:
Ganciclovir has a molecular weight of 255.23, and the empirical formula is C 9H 13N 5O 4.
Each gram of gel contains: ACTIVE: ganciclovir 1.5 mg (0.15%). INACTIVES: carbomer homopolymer, water for injection, sodium hydroxide (to adjust the pH to 7.2-7.6), mannitol. PRESERVATIVE: benzalkonium chloride 0.075 mg (0.0075%).
12.4 Microbiology
Mechanism of Action
ZIRGAN contains ganciclovir, a guanosine derivative that upon phosphorylation inhibits DNA replication by herpes simplex viruses (HSV). Ganciclovir is transformed by viral and cellular thymidine kinases (TK) to ganciclovir triphosphate, which works as an antiviral agent by inhibiting the synthesis of viral DNA in 2 ways: competitive inhibition of viral DNA-polymerase and direct incorporation into viral primer strand DNA, resulting in DNA chain termination and prevention of replication.
8.4 Pediatric Use
The safety and effectiveness of ZIRGAN for the treatment of acute herpetic keratitis (dendritic ulcers) have been established in pediatric patients aged 2 years and older. Use of ZIRGAN for this indication is supported by evidence from adequate and well-controlled studies.
The safety and effectiveness of ZIRGAN have not been established in pediatric patients below the age of 2 years.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
14 Clinical Studies
In one open-label, randomized, controlled, multicenter clinical trial which enrolled 164 patients with herpetic keratitis, ZIRGAN was non-inferior to acyclovir ophthalmic ointment, 3% in patients with dendritic ulcers. Clinical resolution (healed ulcers) at Day 7 was achieved in 77% (55/71) for ZIRGAN versus 72% (48/67) for acyclovir, 3% (difference 5.8%, 95% CI - 9.6%-18.3%). In three randomized, single-masked, controlled, multicenter clinical trials which enrolled 213 total patients, ZIRGAN was non-inferior to acyclovir ophthalmic ointment, 3% in patients with dendritic ulcers. Clinical resolution at Day 7 was achieved in 72% (41/57) for ZIRGAN versus 69% (34/49) for acyclovir (difference 2.5%, 95% CI - 15.6%-20.9%).
4 Contraindications
None.
6 Adverse Reactions
Most common adverse reactions reported in patients were blurred vision (60%), eye irritation (20%), punctate keratitis (5%), and conjunctival hyperemia (5%).
12.3 Pharmacokinetics
The estimated maximum daily dose of ganciclovir administered as 1 drop, 5 times per day is 0.375 mg. Compared to maintenance doses of systemically administered ganciclovir of 900 mg (oral valganciclovir) and 5 mg/kg (IV ganciclovir), the ophthalmically administered daily dose is approximately 0.04% and 0.1% of the oral dose and IV doses, respectively, thus minimal systemic exposure is expected.
1 Indications and Usage
ZIRGAN is indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in adults and pediatric patients aged 2 years and older.
12.1 Mechanism of Action
ZIRGAN is an antiviral drug [ see Microbiology (12.4) ].
5.3 Risk of Contamination
Do not allow the tip of the container to touch any surface, as this may contaminate the ointment. If pain develops, or if redness, itching, or inflammation becomes aggravated, the patient should be advised to consult a physician.
5 Warnings and Precautions
2 Dosage and Administration
The recommended dosage is 1 drop in the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals, and then 1 drop 3 times per day for 7 days.
3 Dosage Forms and Strengths
Ophthalmic gel: 0.15% ganciclovir.
5.1 Topical Ophthalmic Use Only
ZIRGAN is indicated for topical ophthalmic use only.
5.2 Avoidance of Contact Lenses
Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with ZIRGAN.
17 Patient Counseling Information
When to Consult a Physician
Advise patients to consult a physician if pain develops, or if redness, itching, or inflammation becomes aggravated.
Risk of Contamination
ZIRGAN is sterile when packaged. Advise patients to not allow the dropper tip to touch any surface, as this may contaminate the gel.
Avoidance of Contact Lenses
Advise patients to not wear contact lenses when using ZIRGAN.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
Zirgan is a trademark of Laboratoires Théa Corporation used under license.
© 2025 Bausch & Lomb Incorporated or its affiliates
9224806
16 How Supplied/storage and Handling
ZIRGAN (ganciclovir ophthalmic gel) 0.15% is supplied as 5 grams of a sterile, preserved, clear, colorless, topical ophthalmic gel containing 0.15% of ganciclovir in a polycoated aluminum tube with a white polyethylene tip and cap and protective band (NDC 24208-535-35).
Storage
Store at 15°C to 25°C (59°F to 77°F). Do not freeze.
Package/label Principal Display Panel
NDC24208-535-35
BAUSCH + LOMB
Zirgan
(ganciclovir
ophthalmic gel)
0.15%
Sterile
FOR TOPICAL
OPHTHALMIC
USE ONLY
Rx only
5 g
AB48835
9691901
8.3 Females and Males of Reproductive Potential
Infertility
It is not known whether topical ophthalmic ganciclovir administration could result in sufficient absorption to impair fertility in humans. Animal data indicate decreased fertility in female mice following intravenous doses at 90 mg/kg/day, approximately 1174 times the maximum RHOD (based on body surface area), and decreased fertility and hypospermatogenesis in male mice and dogs following intravenous doses at 10 mg/kg/day for mice, approximately 130 times the maximum RHOD (based on body surface area), and 3.6 mg/kg/day for dogs, approximately 240 times the maximum RHOD (based on body surface area).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Ganciclovir was carcinogenic in the mouse at oral doses of 20 and 1,000 mg/kg/day (approximately 3,000 and 160,000 times the human ocular dose of 6.25 mcg/kg/day, assuming complete absorption). At the dose of 1,000 mg/kg/day there was a significant increase in the incidence of tumors of the preputial gland in males, forestomach (nonglandular mucosa) in males and females, and reproductive tissues (ovaries, uterus, mammary gland, clitoral gland, and vagina) and liver in females. At the dose of 20 mg/kg/day, a slightly increased incidence of tumors was noted in the preputial and harderian glands in males, forestomach in males and females, and liver in females. No carcinogenic effect was observed in mice administered ganciclovir at 1 mg/kg/day (160 times the human ocular dose). Except for histocytic sarcoma of the liver, ganciclovir-induced tumors were generally of epithelial or vascular origin. Although the preputial and clitoral glands, forestomach and harderian glands of mice do not have human counterparts, ganciclovir should be considered a potential carcinogen in humans.
Mutagenesis
Ganciclovir increased mutations in mouse lymphoma cells and DNA damage in human lymphocytes in vitroat concentrations between 50 to 500 and 250 to 2,000 mcg/mL, respectively.
In the mouse micronucleus assay, ganciclovir was clastogenic at doses of 150 and 500 mg/kg (IV) (24,000 to 80,000 times the human ocular dose) but not 50 mg/kg (8,000 times the human ocular dose). Ganciclovir was not mutagenic in the Ames Salmonella assay at concentrations of 500 to 5,000 mcg/mL.
Impairment of Fertility
Ganciclovir caused decreased mating behavior, decreased fertility, and an increased incidence of embryolethality in female mice following intravenous doses of 90 mg/kg/day (approximately 14,000 times the human ocular dose of 6.25 mcg/kg/day). Ganciclovir caused decreased fertility in male mice and hypospermatogenesis in mice and dogs following daily oral or intravenous administration of doses ranging from 0.2 to 10 mg/kg (30 to 1,600 times the human ocular dose).
Structured Label Content
8.1 Pregnancy
Risk Summary
There are no available human data on use of ZIRGAN or ganciclovir during pregnancy to inform any drug-associated risk. Intravenous administration of ganciclovir to pregnant mice or rabbits during organogenesis or during the pre/postnatal period did not produce adverse embryofetal or offspring effects at clinically relevant doses (see Data) .
The background risk in the U.S. general population of major birth defects is 2 to 4% and the risk of miscarriage is 15 to 20% of clinically recognized pregnancies.
Data
Animal Data
Daily intravenous doses of ganciclovir were administered to pregnant mice [up to 108 mg/kg/day, approximately 1400 times the maximum recommended human ocular dose (RHOD) of 0.375 mg] and rabbits [up to 60 mg/kg/day, approximately 2400 times the maximum RHOD], and also to female mice [up to 90 mg/kg, approximately 1174 times the maximum RHOD] prior to mating, during gestation, and during lactation. Fetal resorptions were present in at least 85% of rabbits and mice. Additional effects observed in rabbits included fetal growth retardation, embryolethality, teratogenicity, and/or maternal toxicity. Teratogenic changes included cleft palate, anophthalmia/microphthalmia, aplastic organs (kidney and pancreas), hydrocephaly and brachygnathia. A maternal no observed adverse effect level (NOAEL) was observed at 36 mg/kg/day (approximately 470 times higher than the maximum RHOD, based on body surface area) in mice and at 6 mg/kg/day (approximately 240 times higher than the maximum RHOD, based on body surface area) in rabbits.
In pre/postnatal development studies in mice, there were maternal/fetal toxicity and embryolethality which included fetal effects of hypoplasia of the testes and seminal vesicles in the male offspring, as well as pathologic changes in the nonglandular region of the stomach. A maternal no observed adverse effect level (NOAEL) was observed at 20 mg/kg/day (approximately 261 times higher than the maximum RHOD, based on body surface area).
8.2 Lactation
Risk Summary
No data are available regarding the presence of ganciclovir in human milk, the effects on the breastfed infant, or the effects on milk production. It is not known whether topical ophthalmic ganciclovir administration could result in sufficient systemic absorption to produce detectable quantities in breast milk. Animal data indicate that ganciclovir is excreted in the milk of lactating rats. Caution should be exercised when ZIRGAN is administered to nursing mothers.
11 Description (11 DESCRIPTION)
ZIRGAN (ganciclovir ophthalmic gel) 0.15% contains ganciclovir, a nucleoside analog antiviral. ZIRGAN is a sterile, preserved, clear, colorless, ophthalmic gel for topical ophthalmic use.
The chemical name is 9-[[2-hydroxy-1-(hydroxymethyl)ethoxy]methyl]guanine (CAS number 82410-32-0). Ganciclovir is represented by the following structural formula:
Ganciclovir has a molecular weight of 255.23, and the empirical formula is C 9H 13N 5O 4.
Each gram of gel contains: ACTIVE: ganciclovir 1.5 mg (0.15%). INACTIVES: carbomer homopolymer, water for injection, sodium hydroxide (to adjust the pH to 7.2-7.6), mannitol. PRESERVATIVE: benzalkonium chloride 0.075 mg (0.0075%).
12.4 Microbiology
Mechanism of Action
ZIRGAN contains ganciclovir, a guanosine derivative that upon phosphorylation inhibits DNA replication by herpes simplex viruses (HSV). Ganciclovir is transformed by viral and cellular thymidine kinases (TK) to ganciclovir triphosphate, which works as an antiviral agent by inhibiting the synthesis of viral DNA in 2 ways: competitive inhibition of viral DNA-polymerase and direct incorporation into viral primer strand DNA, resulting in DNA chain termination and prevention of replication.
8.4 Pediatric Use
The safety and effectiveness of ZIRGAN for the treatment of acute herpetic keratitis (dendritic ulcers) have been established in pediatric patients aged 2 years and older. Use of ZIRGAN for this indication is supported by evidence from adequate and well-controlled studies.
The safety and effectiveness of ZIRGAN have not been established in pediatric patients below the age of 2 years.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
14 Clinical Studies (14 CLINICAL STUDIES)
In one open-label, randomized, controlled, multicenter clinical trial which enrolled 164 patients with herpetic keratitis, ZIRGAN was non-inferior to acyclovir ophthalmic ointment, 3% in patients with dendritic ulcers. Clinical resolution (healed ulcers) at Day 7 was achieved in 77% (55/71) for ZIRGAN versus 72% (48/67) for acyclovir, 3% (difference 5.8%, 95% CI - 9.6%-18.3%). In three randomized, single-masked, controlled, multicenter clinical trials which enrolled 213 total patients, ZIRGAN was non-inferior to acyclovir ophthalmic ointment, 3% in patients with dendritic ulcers. Clinical resolution at Day 7 was achieved in 72% (41/57) for ZIRGAN versus 69% (34/49) for acyclovir (difference 2.5%, 95% CI - 15.6%-20.9%).
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions reported in patients were blurred vision (60%), eye irritation (20%), punctate keratitis (5%), and conjunctival hyperemia (5%).
12.3 Pharmacokinetics
The estimated maximum daily dose of ganciclovir administered as 1 drop, 5 times per day is 0.375 mg. Compared to maintenance doses of systemically administered ganciclovir of 900 mg (oral valganciclovir) and 5 mg/kg (IV ganciclovir), the ophthalmically administered daily dose is approximately 0.04% and 0.1% of the oral dose and IV doses, respectively, thus minimal systemic exposure is expected.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZIRGAN is indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in adults and pediatric patients aged 2 years and older.
12.1 Mechanism of Action
ZIRGAN is an antiviral drug [ see Microbiology (12.4) ].
5.3 Risk of Contamination
Do not allow the tip of the container to touch any surface, as this may contaminate the ointment. If pain develops, or if redness, itching, or inflammation becomes aggravated, the patient should be advised to consult a physician.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage is 1 drop in the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals, and then 1 drop 3 times per day for 7 days.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Ophthalmic gel: 0.15% ganciclovir.
5.1 Topical Ophthalmic Use Only
ZIRGAN is indicated for topical ophthalmic use only.
5.2 Avoidance of Contact Lenses
Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with ZIRGAN.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
When to Consult a Physician
Advise patients to consult a physician if pain develops, or if redness, itching, or inflammation becomes aggravated.
Risk of Contamination
ZIRGAN is sterile when packaged. Advise patients to not allow the dropper tip to touch any surface, as this may contaminate the gel.
Avoidance of Contact Lenses
Advise patients to not wear contact lenses when using ZIRGAN.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
Zirgan is a trademark of Laboratoires Théa Corporation used under license.
© 2025 Bausch & Lomb Incorporated or its affiliates
9224806
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ZIRGAN (ganciclovir ophthalmic gel) 0.15% is supplied as 5 grams of a sterile, preserved, clear, colorless, topical ophthalmic gel containing 0.15% of ganciclovir in a polycoated aluminum tube with a white polyethylene tip and cap and protective band (NDC 24208-535-35).
Storage
Store at 15°C to 25°C (59°F to 77°F). Do not freeze.
Package/label Principal Display Panel (PACKAGE/LABEL PRINCIPAL DISPLAY PANEL)
NDC24208-535-35
BAUSCH + LOMB
Zirgan
(ganciclovir
ophthalmic gel)
0.15%
Sterile
FOR TOPICAL
OPHTHALMIC
USE ONLY
Rx only
5 g
AB48835
9691901
8.3 Females and Males of Reproductive Potential
Infertility
It is not known whether topical ophthalmic ganciclovir administration could result in sufficient absorption to impair fertility in humans. Animal data indicate decreased fertility in female mice following intravenous doses at 90 mg/kg/day, approximately 1174 times the maximum RHOD (based on body surface area), and decreased fertility and hypospermatogenesis in male mice and dogs following intravenous doses at 10 mg/kg/day for mice, approximately 130 times the maximum RHOD (based on body surface area), and 3.6 mg/kg/day for dogs, approximately 240 times the maximum RHOD (based on body surface area).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Ganciclovir was carcinogenic in the mouse at oral doses of 20 and 1,000 mg/kg/day (approximately 3,000 and 160,000 times the human ocular dose of 6.25 mcg/kg/day, assuming complete absorption). At the dose of 1,000 mg/kg/day there was a significant increase in the incidence of tumors of the preputial gland in males, forestomach (nonglandular mucosa) in males and females, and reproductive tissues (ovaries, uterus, mammary gland, clitoral gland, and vagina) and liver in females. At the dose of 20 mg/kg/day, a slightly increased incidence of tumors was noted in the preputial and harderian glands in males, forestomach in males and females, and liver in females. No carcinogenic effect was observed in mice administered ganciclovir at 1 mg/kg/day (160 times the human ocular dose). Except for histocytic sarcoma of the liver, ganciclovir-induced tumors were generally of epithelial or vascular origin. Although the preputial and clitoral glands, forestomach and harderian glands of mice do not have human counterparts, ganciclovir should be considered a potential carcinogen in humans.
Mutagenesis
Ganciclovir increased mutations in mouse lymphoma cells and DNA damage in human lymphocytes in vitroat concentrations between 50 to 500 and 250 to 2,000 mcg/mL, respectively.
In the mouse micronucleus assay, ganciclovir was clastogenic at doses of 150 and 500 mg/kg (IV) (24,000 to 80,000 times the human ocular dose) but not 50 mg/kg (8,000 times the human ocular dose). Ganciclovir was not mutagenic in the Ames Salmonella assay at concentrations of 500 to 5,000 mcg/mL.
Impairment of Fertility
Ganciclovir caused decreased mating behavior, decreased fertility, and an increased incidence of embryolethality in female mice following intravenous doses of 90 mg/kg/day (approximately 14,000 times the human ocular dose of 6.25 mcg/kg/day). Ganciclovir caused decreased fertility in male mice and hypospermatogenesis in mice and dogs following daily oral or intravenous administration of doses ranging from 0.2 to 10 mg/kg (30 to 1,600 times the human ocular dose).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:16.077680 · Updated: 2026-03-14T22:45:57.416825