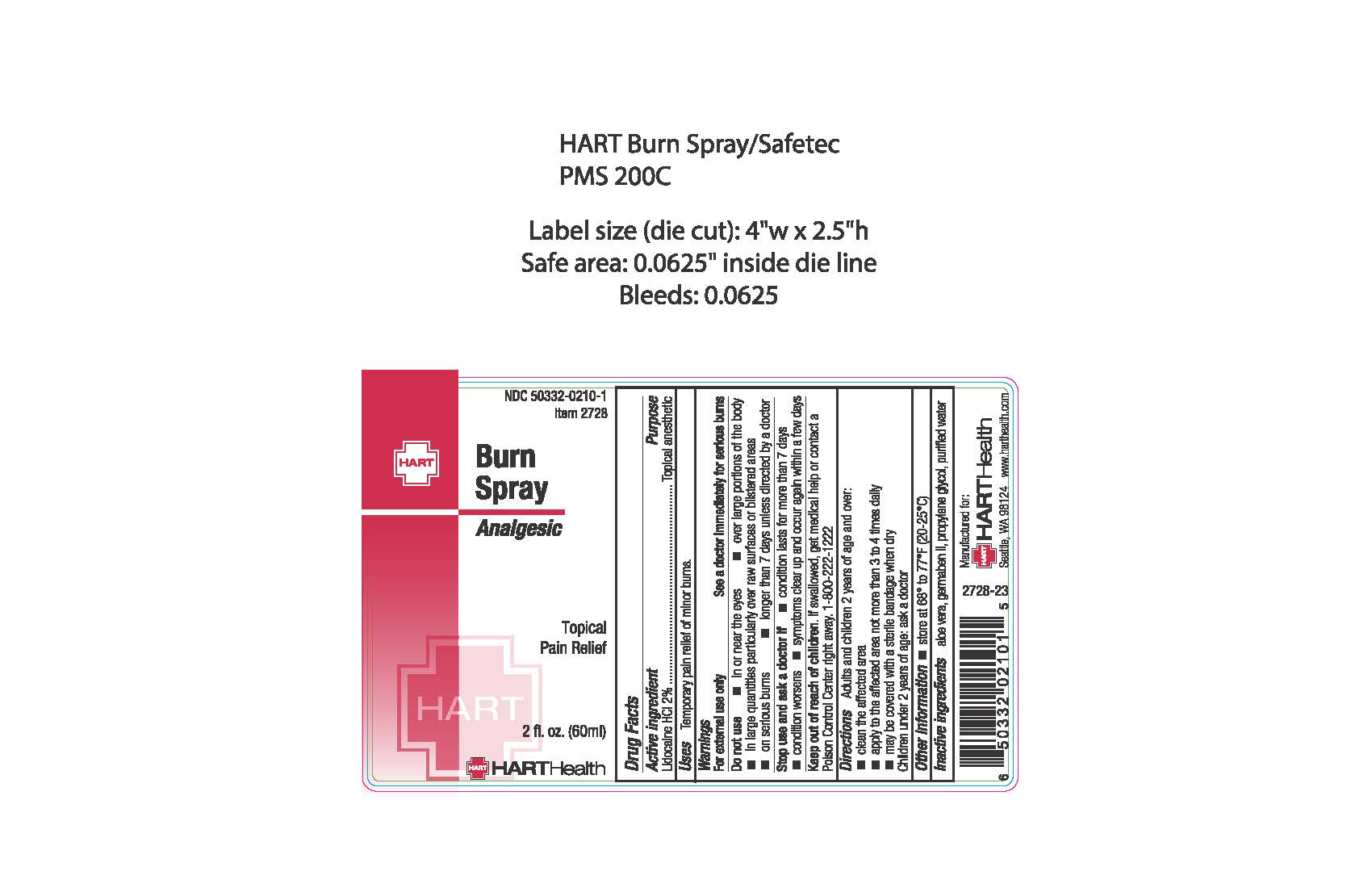
Burn Spray
e95ebdac-9789-41c2-97da-2307e4538794
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients: Lidocaine HCL 2%
Medication Information
Warnings and Precautions
Warnings
For external use only
See a doctor immediately or serious burns
Indications and Usage
Uses:
Temporary pain relief of minor burns
Dosage and Administration
Directions:
Adults and children 12 years of age and over:
- clean the affected area
- apply to the affected area not more than 3 to 4 times dailys
- may be covered with a sterile bandage when dry
Children under 12 years of age: ask a doctor
Description
Active Ingredients: Lidocaine HCL 2%
Section 50565-1
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9
Stop use and ask a doctor if
- condition worsens
- condition lasts for more than 7 days
- symptoms clear up and occur again within a few days
Section 50570-1
Do not use
- in or near the eyes
- over large portions of the body
- in large quantities particularly over raw surfaces or blistered areas
- on serious burns
- longer than 7 days unless directed by a doctor
Section 51727-6
Inactive Ingredients
aloe vera, germaben II, propylene glycol, purified water
Section 51945-4
Section 55105-1
Purpose:
Topical Anesthetic
Section 55106-9
Active Ingredients:
Lidocaine HCL 2%
Section 60561-8
store at 68° to 77°F (20-25°C)
Structured Label Content
Indications and Usage (34067-9)
Uses:
Temporary pain relief of minor burns
Dosage and Administration (34068-7)
Directions:
Adults and children 12 years of age and over:
- clean the affected area
- apply to the affected area not more than 3 to 4 times dailys
- may be covered with a sterile bandage when dry
Children under 12 years of age: ask a doctor
Warnings and Precautions (34071-1)
Warnings
For external use only
See a doctor immediately or serious burns
Section 50565-1 (50565-1)
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- condition worsens
- condition lasts for more than 7 days
- symptoms clear up and occur again within a few days
Section 50570-1 (50570-1)
Do not use
- in or near the eyes
- over large portions of the body
- in large quantities particularly over raw surfaces or blistered areas
- on serious burns
- longer than 7 days unless directed by a doctor
Section 51727-6 (51727-6)
Inactive Ingredients
aloe vera, germaben II, propylene glycol, purified water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose:
Topical Anesthetic
Section 55106-9 (55106-9)
Active Ingredients:
Lidocaine HCL 2%
Section 60561-8 (60561-8)
store at 68° to 77°F (20-25°C)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:43.452747 · Updated: 2026-03-14T23:02:25.522673