Blistex®
e95439d3-f12b-4c1c-999a-79f2ddba4cfb
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Description
Drug Facts
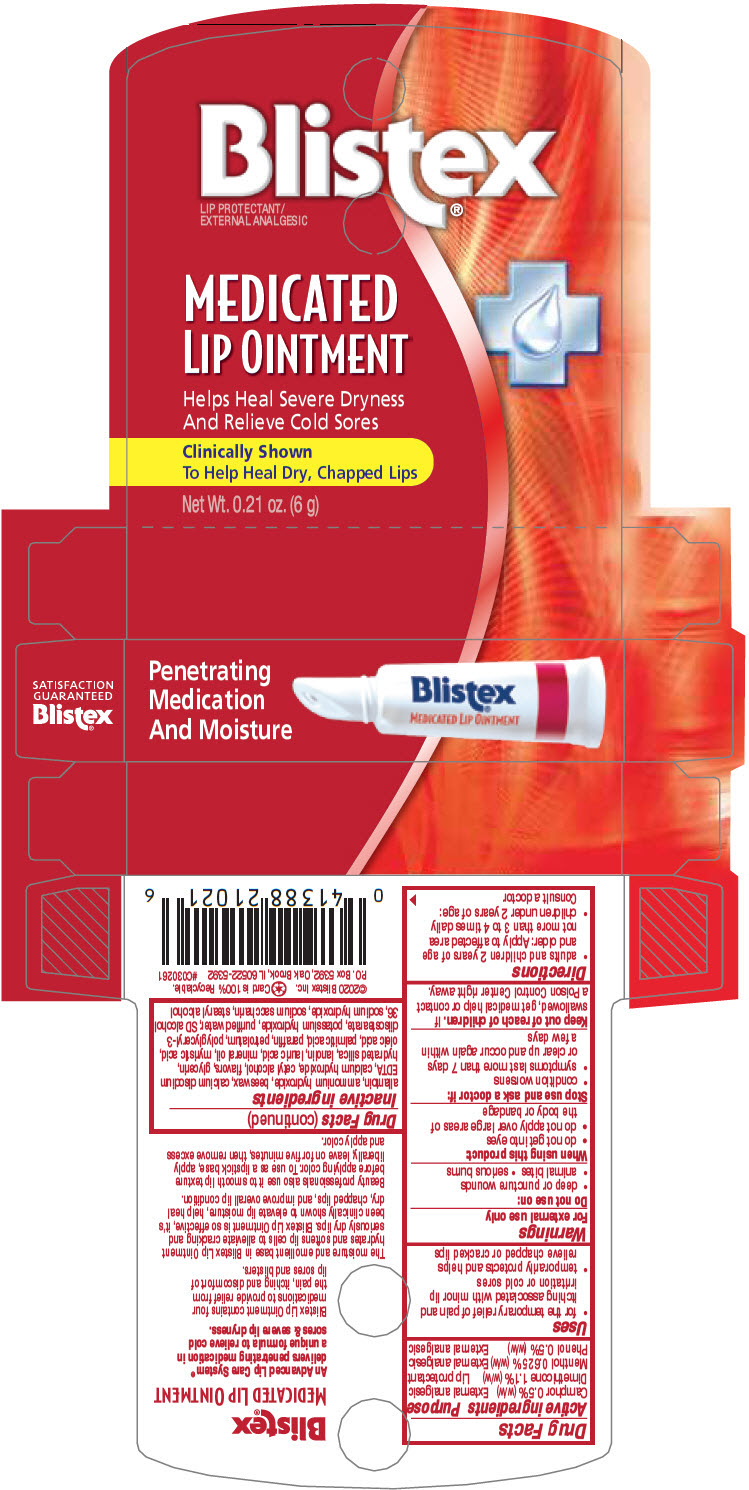
Uses
- for the temporary relief of pain and itching associated with minor lip irritation or cold sores
- temporarily protects and helps relieve chapped or cracked lips
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 55105-1
| Active ingredients | Purpose |
|---|---|
| Camphor 0.5% (w/w) | External analgesic |
| Dimethicone 1.1% (w/w) | Lip protectant |
| Menthol 0.625% (w/w) | External analgesic |
| Phenol 0.5% (w/w) | External analgesic |
Warnings
For external use only
Directions
- adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: Consult a doctor
Do Not Use On
- deep or puncture wounds
- animal bites
- serious burns
Inactive Ingredients
allantoin, ammonium hydroxide, beeswax, calcium disodium EDTA, calcium hydroxide, cetyl alcohol, flavors, glycerin, hydrated silica, lanolin, lauric acid, mineral oil, myristic acid, oleic acid, palmitic acid, paraffin, petrolatum, polyglyceryl-3 diisostearate, potassium hydroxide, purified water, SD alcohol 36, sodium hydroxide, sodium saccharin, stearyl alcohol
When Using This Product
- do not get into eyes
- do not apply over large areas of the body or bandage
Stop Use and Ask A Doctor If
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Principal Display Panel 6 G Tube Carton
Penetrating
Medication
And Moisture
Blistex®
MEDICATED LIP OINTMENT
Structured Label Content
Uses
- for the temporary relief of pain and itching associated with minor lip irritation or cold sores
- temporarily protects and helps relieve chapped or cracked lips
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 55105-1 (55105-1)
| Active ingredients | Purpose |
|---|---|
| Camphor 0.5% (w/w) | External analgesic |
| Dimethicone 1.1% (w/w) | Lip protectant |
| Menthol 0.625% (w/w) | External analgesic |
| Phenol 0.5% (w/w) | External analgesic |
Warnings
For external use only
Directions
- adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: Consult a doctor
Do Not Use On (Do not use on)
- deep or puncture wounds
- animal bites
- serious burns
Inactive Ingredients (Inactive ingredients)
allantoin, ammonium hydroxide, beeswax, calcium disodium EDTA, calcium hydroxide, cetyl alcohol, flavors, glycerin, hydrated silica, lanolin, lauric acid, mineral oil, myristic acid, oleic acid, palmitic acid, paraffin, petrolatum, polyglyceryl-3 diisostearate, potassium hydroxide, purified water, SD alcohol 36, sodium hydroxide, sodium saccharin, stearyl alcohol
When Using This Product (When using this product)
- do not get into eyes
- do not apply over large areas of the body or bandage
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Principal Display Panel 6 G Tube Carton (PRINCIPAL DISPLAY PANEL - 6 g Tube Carton)
Penetrating
Medication
And Moisture
Blistex®
MEDICATED LIP OINTMENT
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:45.923800 · Updated: 2026-03-14T23:05:42.419463