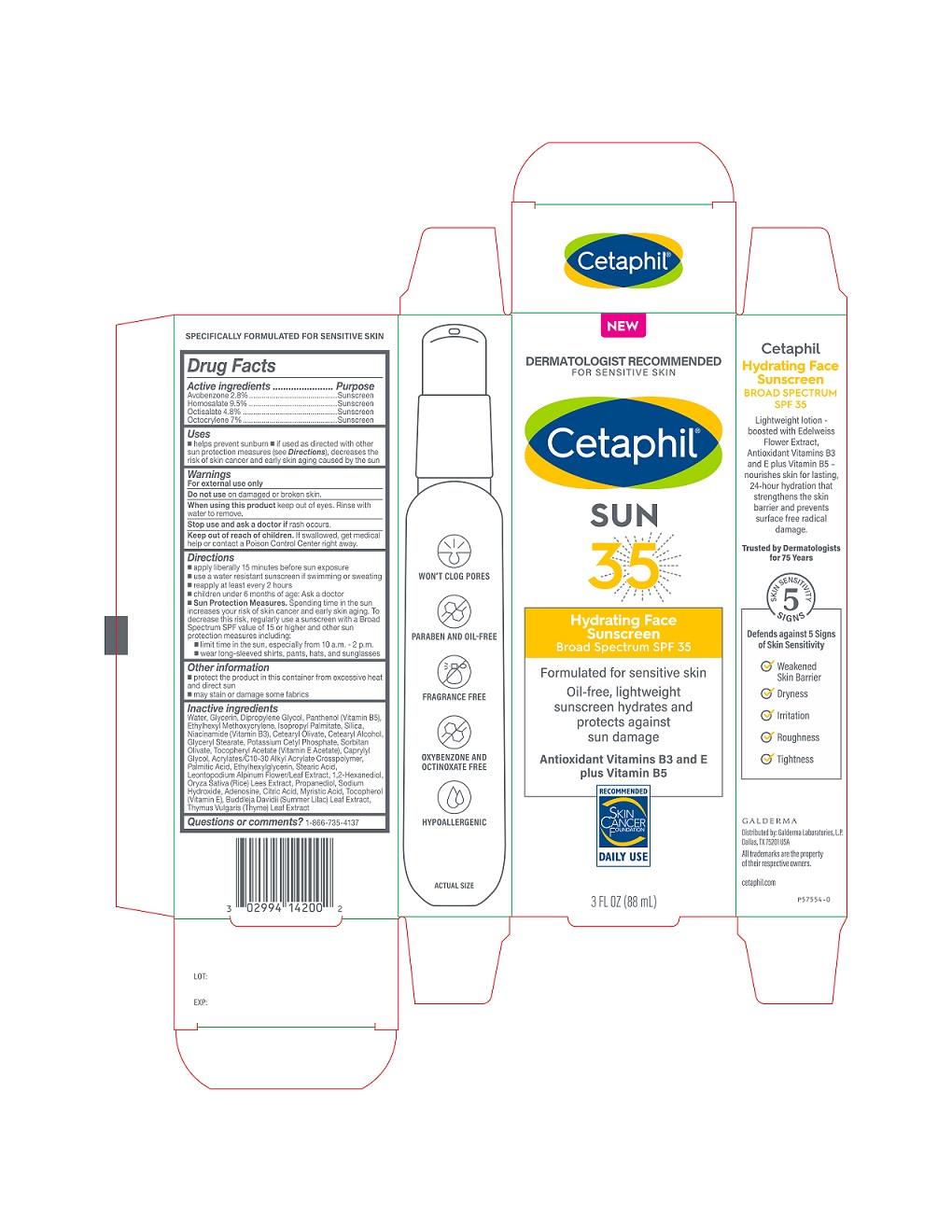
Drug Fact
e924bfd5-d64d-4918-b57b-7d4da28bd013
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Avobenzone 2.8% .................Sunscreen Homosalate 9.5% .....................Sunscreen Octisalate 4.8% ..................Sunscreen Octocrylene 7% .................Sunscreen
Purpose
Sunscreen
Medication Information
Purpose
Sunscreen
Description
Avobenzone 2.8% .................Sunscreen Homosalate 9.5% .....................Sunscreen Octisalate 4.8% ..................Sunscreen Octocrylene 7% .................Sunscreen
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Warnings
For external use only
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months of age: Ask a doctor
-
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
Do Not Use
on damaged or broken skin
Other Information
- protect the product in this container from excessive heat and direct sun
- may stain or damage some fabrics
Active Ingredients
Avobenzone 2.8% .................Sunscreen
Homosalate 9.5% .....................Sunscreen
Octisalate 4.8% ..................Sunscreen
Octocrylene 7% .................Sunscreen
Inactive Ingredients
Water, Glycerin, Dipropylene Glycol, Panthenol (Vitamin B5), Ethylhexyl Methoxycrylene, lsopropyl Palmitate, Silica, Niacinamide, Cetearyl Olivate, Cetearyl Alcohol, Glyceryl Stearate, Potassium Cetyl Phosphate, Sorbitan Olivate, Tocopheryl Acetate (Vitamin E Acetate), Caprylyl Glycol, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Palmitic Acid, Ethylhexylglycerin, Stearic Acid, Leontopodium Alpinum Flower/Leaf Extract, 1,2-Hexanediol, Oryza Saliva (Rice) Lees Extract, Propanediol, Sodium Hydroxide, Adenosine, Citric Acid, Myristic Acid, Tocopherol (Vitamin E), Buddleja Davidii (Summer Lilac) Leaf Extract, Thymus Vulgaris (Thyme) Leaf Extract
Otc Purpose Section
Sunscreen
Questions Or Comments?
1-866-735-4137
When Using This Product
keep out of eyes. Rinse with water to remove.
Stop Use and Ask A Doctor If
rash occurs.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Principal Display Panel 3 Fl Oz Carton
NEW
DERMATOLOGIST RECOMMENDED
FOR SENSITIVE SKIN
Cetaphil
®
SUN
35
Hydrating Face
Sunscreen
Broad Spectrum SPF 35
Formulated for sensitive skin
Oil-free, lightweight
sunscreen hydrates and
protects against
sun damage
Antioxidant Vitamins B3 and E
plus Vitamin B5
Skin Cancer Foundation logo
3 FL OZ (88 mL)
Distributed by:
Galderma Laboratories, L.P.
Dallas, Texas 75201 USA
All trademarks are the property of their respective owners.
cetaphil.com
P57554-0
Structured Label Content
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Warnings
For external use only
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months of age: Ask a doctor
-
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
Do Not Use (Do not use)
on damaged or broken skin
Other Information (Other information)
- protect the product in this container from excessive heat and direct sun
- may stain or damage some fabrics
Active Ingredients
Avobenzone 2.8% .................Sunscreen
Homosalate 9.5% .....................Sunscreen
Octisalate 4.8% ..................Sunscreen
Octocrylene 7% .................Sunscreen
Inactive Ingredients
Water, Glycerin, Dipropylene Glycol, Panthenol (Vitamin B5), Ethylhexyl Methoxycrylene, lsopropyl Palmitate, Silica, Niacinamide, Cetearyl Olivate, Cetearyl Alcohol, Glyceryl Stearate, Potassium Cetyl Phosphate, Sorbitan Olivate, Tocopheryl Acetate (Vitamin E Acetate), Caprylyl Glycol, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Palmitic Acid, Ethylhexylglycerin, Stearic Acid, Leontopodium Alpinum Flower/Leaf Extract, 1,2-Hexanediol, Oryza Saliva (Rice) Lees Extract, Propanediol, Sodium Hydroxide, Adenosine, Citric Acid, Myristic Acid, Tocopherol (Vitamin E), Buddleja Davidii (Summer Lilac) Leaf Extract, Thymus Vulgaris (Thyme) Leaf Extract
Otc Purpose Section (OTC - PURPOSE SECTION)
Sunscreen
Questions Or Comments? (Questions or comments?)
1-866-735-4137
When Using This Product (When using this product)
keep out of eyes. Rinse with water to remove.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
rash occurs.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Principal Display Panel 3 Fl Oz Carton (Principal Display Panel - 3 FL OZ Carton)
NEW
DERMATOLOGIST RECOMMENDED
FOR SENSITIVE SKIN
Cetaphil
®
SUN
35
Hydrating Face
Sunscreen
Broad Spectrum SPF 35
Formulated for sensitive skin
Oil-free, lightweight
sunscreen hydrates and
protects against
sun damage
Antioxidant Vitamins B3 and E
plus Vitamin B5
Skin Cancer Foundation logo
3 FL OZ (88 mL)
Distributed by:
Galderma Laboratories, L.P.
Dallas, Texas 75201 USA
All trademarks are the property of their respective owners.
cetaphil.com
P57554-0
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:11.539822 · Updated: 2026-03-14T23:01:36.166619