Varenicline Tablets
e8a06e8e-1938-412c-a98a-28f0a29fe3ac
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Varenicline tablets are indicated for use as an aid to smoking cessation treatment.
Dosage and Administration
• Begin varenicline tablets dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin varenicline tablets dosing and then quit smoking between days 8 and 35 of treatment. ( 2.1 ) • Starting Week: 0.5 mg once daily on days 1-3 and 0.5 mg twice daily on days 4-7. ( 2.1 ) • Continuing Weeks: 1 mg twice daily for a total of 12 weeks. ( 2.1 ) • An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. ( 2.1 ) • Consider a gradual approach to quitting smoking with varenicline tablets for patients who are sure that they are not able or willing to quit abruptly. Patients should begin varenicline tablets dosing and reduce smoking by 50% from baseline within the first four weeks, by an additional 50% in the next four weeks, and continue reducing with the goal of reaching complete abstinence by 12 weeks. Continue treatment for an additional 12 weeks, for a total of 24 weeks. ( 2.1 ) • Severe Renal Impairment (estimated creatinine clearance less than 30 mL/min): Begin with 0.5 mg once daily and titrate to 0.5 mg twice daily. For patients with end-stage renal disease undergoing hemodialysis, a maximum of 0.5 mg daily may be given if tolerated. ( 2.2 ) • Consider dose reduction for patients who cannot tolerate adverse effects. ( 2.1 ) • Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. ( 2.1 ) • Provide patients with appropriate educational materials and counseling to support the quit attempt. ( 2.1 )
Contraindications
Varenicline tablets are contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to varenicline tablets.
Warnings and Precautions
• Neuropsychiatric Adverse Events : Postmarketing reports of serious or clinically significant neuropsychiatric adverse events have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Observe patients attempting to quit smoking with varenicline tablets for the occurrence of such symptoms and instruct them to discontinue varenicline tablets and contact a healthcare provider if they experience such adverse events. ( 5.1 ) • Seizures : New or worsening seizures have been observed in patients taking varenicline tablets. Varenicline tablets should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. ( 5.2 ) • Interaction with Alcohol : Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether varenicline tablets affects them. ( 5.3 ) • Accidental Injury : Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how varenicline tablets may affect them. ( 5.4 ) • Cardiovascular Events : Patients with underlying cardiovascular (CV) disease may be at increased risk of CV events; however, these concerns must be balanced with the health benefits of smoking cessation. Instruct patients to notify their healthcare providers of new or worsening CV symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction (MI) or stroke. ( 5.5 and 6.1 ) • Somnambulism : Cases of somnambulism have been reported in patients taking varenicline tablets. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue varenicline tablets and notify their healthcare provider if they experience somnambulism. ( 5.6 and 6.2 ) • Angioedema and Hypersensitivity Reactions : Such reactions, including angioedema, infrequently life-threatening, have been reported. Instruct patients to discontinue varenicline tablets and immediately seek medical care if symptoms occur. ( 5.7 and 6.2 ) • Serious Skin Reactions : Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue varenicline tablets and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. ( 5.8 and 6.2 ) • Nausea : Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. ( 5.9 )
Adverse Reactions
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling: • Neuropsychiatric Adverse Events including Suicidality [see Warnings and Precautions (5.1) ] • Seizures [see Warnings and Precautions (5.2) ] • Interaction with Alcohol [see Warnings and Precautions (5.3) ] • Accidental Injury [see Warnings and Precautions (5.4) ] • Cardiovascular Events [see Warnings and Precautions (5.5) ] • Somnambulism [see Warnings and Precautions (5.6) ] • Angioedema and Hypersensitivity Reactions [see Warnings and Precautions (5.7 )] • Serious Skin Reactions [see Warnings and Precautions (5.8) ] In the placebo-controlled premarketing studies, the most common adverse events associated with varenicline tablets (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting. The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for varenicline tablets, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in varenicline tablets-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo). Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
Drug Interactions
Based on varenicline characteristics and clinical experience to date, varenicline tablets has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3) ].
How Supplied
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg modified capsule shaped, white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg modified capsule shaped, light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side. Varenicline tablets are supplied in the following package configurations: Description NDC Packs Starting 4-week card: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing 4-week card: 1 mg x 56 tablets NDC 68462-894-04 Starting Month Box: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing Month Box: 1 mg x 56 tablets NDC 68462-894-04 Bottles 0.5 mg - bottle of 56 NDC 68462-893-56 1 mg - bottle of 56 NDC 68462-894-56 Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP. This package is child-resistant
Storage and Handling
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg modified capsule shaped, white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg modified capsule shaped, light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side. Varenicline tablets are supplied in the following package configurations: Description NDC Packs Starting 4-week card: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing 4-week card: 1 mg x 56 tablets NDC 68462-894-04 Starting Month Box: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing Month Box: 1 mg x 56 tablets NDC 68462-894-04 Bottles 0.5 mg - bottle of 56 NDC 68462-893-56 1 mg - bottle of 56 NDC 68462-894-56 Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP. This package is child-resistant
Description
Varenicline tablets are indicated for use as an aid to smoking cessation treatment.
Medication Information
Warnings and Precautions
• Neuropsychiatric Adverse Events : Postmarketing reports of serious or clinically significant neuropsychiatric adverse events have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Observe patients attempting to quit smoking with varenicline tablets for the occurrence of such symptoms and instruct them to discontinue varenicline tablets and contact a healthcare provider if they experience such adverse events. ( 5.1 ) • Seizures : New or worsening seizures have been observed in patients taking varenicline tablets. Varenicline tablets should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. ( 5.2 ) • Interaction with Alcohol : Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether varenicline tablets affects them. ( 5.3 ) • Accidental Injury : Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how varenicline tablets may affect them. ( 5.4 ) • Cardiovascular Events : Patients with underlying cardiovascular (CV) disease may be at increased risk of CV events; however, these concerns must be balanced with the health benefits of smoking cessation. Instruct patients to notify their healthcare providers of new or worsening CV symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction (MI) or stroke. ( 5.5 and 6.1 ) • Somnambulism : Cases of somnambulism have been reported in patients taking varenicline tablets. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue varenicline tablets and notify their healthcare provider if they experience somnambulism. ( 5.6 and 6.2 ) • Angioedema and Hypersensitivity Reactions : Such reactions, including angioedema, infrequently life-threatening, have been reported. Instruct patients to discontinue varenicline tablets and immediately seek medical care if symptoms occur. ( 5.7 and 6.2 ) • Serious Skin Reactions : Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue varenicline tablets and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. ( 5.8 and 6.2 ) • Nausea : Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. ( 5.9 )
Indications and Usage
Varenicline tablets are indicated for use as an aid to smoking cessation treatment.
Dosage and Administration
• Begin varenicline tablets dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin varenicline tablets dosing and then quit smoking between days 8 and 35 of treatment. ( 2.1 ) • Starting Week: 0.5 mg once daily on days 1-3 and 0.5 mg twice daily on days 4-7. ( 2.1 ) • Continuing Weeks: 1 mg twice daily for a total of 12 weeks. ( 2.1 ) • An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. ( 2.1 ) • Consider a gradual approach to quitting smoking with varenicline tablets for patients who are sure that they are not able or willing to quit abruptly. Patients should begin varenicline tablets dosing and reduce smoking by 50% from baseline within the first four weeks, by an additional 50% in the next four weeks, and continue reducing with the goal of reaching complete abstinence by 12 weeks. Continue treatment for an additional 12 weeks, for a total of 24 weeks. ( 2.1 ) • Severe Renal Impairment (estimated creatinine clearance less than 30 mL/min): Begin with 0.5 mg once daily and titrate to 0.5 mg twice daily. For patients with end-stage renal disease undergoing hemodialysis, a maximum of 0.5 mg daily may be given if tolerated. ( 2.2 ) • Consider dose reduction for patients who cannot tolerate adverse effects. ( 2.1 ) • Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. ( 2.1 ) • Provide patients with appropriate educational materials and counseling to support the quit attempt. ( 2.1 )
Contraindications
Varenicline tablets are contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to varenicline tablets.
Adverse Reactions
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling: • Neuropsychiatric Adverse Events including Suicidality [see Warnings and Precautions (5.1) ] • Seizures [see Warnings and Precautions (5.2) ] • Interaction with Alcohol [see Warnings and Precautions (5.3) ] • Accidental Injury [see Warnings and Precautions (5.4) ] • Cardiovascular Events [see Warnings and Precautions (5.5) ] • Somnambulism [see Warnings and Precautions (5.6) ] • Angioedema and Hypersensitivity Reactions [see Warnings and Precautions (5.7 )] • Serious Skin Reactions [see Warnings and Precautions (5.8) ] In the placebo-controlled premarketing studies, the most common adverse events associated with varenicline tablets (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting. The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for varenicline tablets, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in varenicline tablets-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo). Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
Drug Interactions
Based on varenicline characteristics and clinical experience to date, varenicline tablets has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3) ].
Storage and Handling
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg modified capsule shaped, white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg modified capsule shaped, light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side. Varenicline tablets are supplied in the following package configurations: Description NDC Packs Starting 4-week card: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing 4-week card: 1 mg x 56 tablets NDC 68462-894-04 Starting Month Box: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing Month Box: 1 mg x 56 tablets NDC 68462-894-04 Bottles 0.5 mg - bottle of 56 NDC 68462-893-56 1 mg - bottle of 56 NDC 68462-894-56 Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP. This package is child-resistant
How Supplied
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg modified capsule shaped, white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg modified capsule shaped, light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side. Varenicline tablets are supplied in the following package configurations: Description NDC Packs Starting 4-week card: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing 4-week card: 1 mg x 56 tablets NDC 68462-894-04 Starting Month Box: 0.5 mg x 11 tablets and 1 mg x 42 tablets NDC 68462-895-04 Continuing Month Box: 1 mg x 56 tablets NDC 68462-894-04 Bottles 0.5 mg - bottle of 56 NDC 68462-893-56 1 mg - bottle of 56 NDC 68462-894-56 Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP. This package is child-resistant
Description
Varenicline tablets are indicated for use as an aid to smoking cessation treatment.
Section 42229-5
Patients with Impaired Renal Function
No dosage adjustment is necessary for patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance less than 30 mL per min), the recommended starting dose of varenicline tablets is 0.5 mg once daily. The dose may then be titrated as needed to a maximum dose of 0.5 mg twice daily. For patients with end-stage renal disease undergoing hemodialysis, a maximum dose of 0.5 mg once daily may be administered if tolerated [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
Section 42231-1
|
MEDICATION GUIDE
|
|
|
What is the most important information I should know about varenicline tablets?
|
|
|
What is varenicline tablets?
|
|
|
Who should not take varenicline tablets?
|
|
|
How should I take varenicline tablets?
|
|
|
Weeks 1 through 4 |
Reduce your smoking to reach one-half of your starting daily number of cigarettes. |
|
Weeks 5 through 8 |
Reduce your smoking to reach one-quarter of your starting daily number of cigarettes. |
|
Weeks 9 through 12 |
Keep reducing your smoking until you are no longer smoking (you reach zero cigarettes each day). |
|
Aim to quit by the end of the 12th week of treatment, or sooner if you feel ready. Continue to take varenicline tablets for another 12 weeks, for a total of 24 weeks of treatment. |
|
|
Day 1 to Day 3 |
o White tablet (0.5 mg) |
|
Day 4 to Day 7 |
o White tablet (0.5 mg) |
|
Day 8 to end of treatment |
o Pink tablet (1 mg) |
|
|
|
What should I avoid while taking varenicline tablets?
|
|
|
What are the possible side effects of varenicline tablets?
|
|
|
How should I store varenicline tablets?
|
|
|
General information about the safe and effective use of varenicline tablets.
|
|
|
What are the ingredients in varenicline tablets?
Distributed by: |
Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: August 2025
5.9 Nausea
Nausea was the most common adverse reaction reported with varenicline tablets treatment. Nausea was generally described as mild or moderate and often transient; however, for some patients, it was persistent over several months. The incidence of nausea was dose-dependent. Initial dose-titration was beneficial in reducing the occurrence of nausea. For patients treated to the maximum recommended dose of 1 mg twice daily following initial dosage titration, the incidence of nausea was 30% compared with 10% in patients taking a comparable placebo regimen. In patients taking varenicline tablets 0.5 mg twice daily following initial titration, the incidence was 16% compared with 11% for placebo. Approximately 3% of patients treated with varenicline tablets 1 mg twice daily in studies involving 12 weeks of treatment discontinued treatment prematurely because of nausea. For patients with intolerable nausea, a dose reduction should be considered.
5.2 Seizures
During clinical trials and the postmarketing experience, there have been reports of seizures in patients treated with varenicline tablets. Some patients had no history of seizures, whereas others had a history of seizure disorder that was remote or well-controlled. In most cases, the seizure occurred within the first month of therapy. Weigh this potential risk against the potential benefits before prescribing varenicline tablets in patients with a history of seizures or other factors that can lower the seizure threshold. Advise patients to discontinue varenicline tablets and contact a healthcare provider immediately if they experience a seizure while on treatment [see Adverse Reactions (6.2)].
10 Overdosage
In case of overdose, standard supportive measures should be instituted as required.
Varenicline has been shown to be dialyzed in patients with end-stage renal disease [see Clinical Pharmacology (12.3)], however, there is no experience in dialysis following overdose.
11 Description
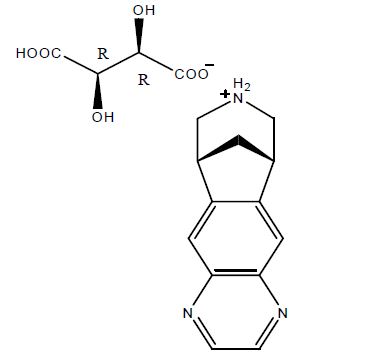
Varenicline tablets contain varenicline (as the tartrate salt), which is a partial nicotinic agonist selective for α4β2 nicotinic acetylcholine receptor subtypes.
Varenicline, as the tartrate salt, is a powder which is white to off white to slightly yellow solid with the following chemical name: 7,8,9,10-tetrahydro-6,10-methano-6H-pyrazino[2,3- h][3]benzazepine, (2R,3R)-2,3-dihydroxybutanedioate (1:1). It is freely soluble in water and very slightly soluble in methanol. Varenicline tartrate has a molecular weight of 361.35 Daltons, and a molecular formula of C13H13N3 • C4H6O6. The chemical structure is:
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg white to off-white, modified capsule shaped, film-coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg light pink to pink color, modified capsule shaped, film coated tablets debossed with 'L' on one side and '12' on other side. Each 0.5 mg varenicline tablets contains 0.85 mg of varenicline tartrate equivalent to 0.5 mg of varenicline free base; each 1 mg varenicline tablets contains 1.71 mg of varenicline tartrate equivalent to 1 mg of varenicline free base. The following inactive ingredients are included in the tablets: anhydrous dibasic calcium phosphate, butylated hydroxyanisole, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, maltodextrin, microcrystalline cellulose. The tablet film coating contains hydroxypropyl cellulose, hypromellose, and titanium dioxide. In addition to these, the 1 mg tablet film coating also includes D&C Red #27/phloxine aluminum lake, D&C yellow #10 aluminum lake, and FD&C blue #1/brilliant blue FCF aluminum lake and talc.
5.6 Somnambulism
Cases of somnambulism have been reported in patients taking varenicline tablets. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue varenicline tablets and notify their healthcare provider if they experience somnambulism [see Adverse Reactions (6.2)].
8.4 Pediatric Use
Varenicline tablets are not recommended for use in pediatric patients 16 years of age or younger because its efficacy in this population was not demonstrated.
Single and multiple-dose pharmacokinetics of varenicline have been investigated in pediatric patients aged 12 to 17 years old (inclusive) and were approximately dose-proportional over the 0.5 mg to 2 mg daily dose range studied. Steady-state systemic exposure in adolescent patients of bodyweight >55 kg, as assessed by AUC (0-24), was comparable to that noted for the same doses in the adult population. When 0.5 mg BID was given, steady-state daily exposure of varenicline was, on average, higher (by approximately 40%) in adolescent patients with bodyweight ≤55 kg compared to that noted in the adult population.
The efficacy and safety of varenicline was evaluated in a randomized, double-blind, placebo-controlled study of 312 patients aged 12 to 19 years, who smoked an average of at least 5 cigarettes per day during the 30 days prior to recruitment, had a score of at least 4 on the Fagerstrom Test for Nicotine Dependence scale, and at least one previous failed quit attempt. Patients were stratified by age (12 to 16 years of age, n = 216 and 17 to 19 years of age, n = 96) and by body weight (≤55 kg and >55 kg). Patients were randomized to one of two doses of varenicline, adjusted by weight to provide plasma levels in the efficacious range (based on adult studies) and placebo. Patients received treatment for 12 weeks, followed by a non-treatment period of 40 weeks, along with age-appropriate counseling throughout the study. Results from this study showed that varenicline, at either dose studied, did not improve continuous abstinence rates at weeks 9 through 12 of treatment compared with placebo in subjects 12 to 19 years of age. The varenicline safety profile in this study was consistent with that observed in adult studies.
8.5 Geriatric Use
A combined single- and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once daily or twice daily to 16 healthy elderly male and female smokers (aged 65–75 years) for 7 consecutive days was similar to that of younger subjects. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.2)].
No dosage adjustment is recommended for elderly patients.
14.2 Urge to Smoke
Based on responses to the Brief Questionnaire of Smoking Urges and the Minnesota Nicotine Withdrawal scale "urge to smoke" item, varenicline tablets reduced urge to smoke compared to placebo.
14 Clinical Studies
The efficacy of varenicline tablets in smoking cessation was demonstrated in six clinical trials in which a total of 3659 chronic cigarette smokers (≥10 cigarettes per day) were treated with varenicline tablets. In all clinical studies, abstinence from smoking was determined by patient self-report and verified by measurement of exhaled carbon monoxide (CO≤10 ppm) at weekly visits. Among the varenicline tablets-treated patients enrolled in these studies, the completion rate was 65%. Except for the dose-ranging study (Study 1) and the maintenance of abstinence study (Study 6), patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. Most patients enrolled in these trials were white (79-96%). All studies enrolled almost equal numbers of men and women. The average age of patients in these studies was 43 years. Patients on average had smoked about 21 cigarettes per day for an average of approximately 25 years. Patients set a date to stop smoking (target quit date) with dosing starting 1 week before this date.
Seven additional studies evaluated the efficacy of varenicline tablets in patients with cardiovascular disease, in patients with chronic obstructive pulmonary disease [see Clinical Studies (14.7)], in patients instructed to select their quit date within days 8 and 35 of treatment [see Clinical Studies (14.4)], patients with major depressive disorder [see Clinical Studies (14.9)], patients who had made a previous attempt to quit smoking with varenicline tablets, and either did not succeed in quitting or relapsed after treatment [see Clinical Studies (14.6)], in patients without or with a history of psychiatric disorder enrolled in a postmarketing neuropsychiatric safety outcome trial [see Warnings and Precautions (5.1), Clinical Studies (14.10) ], and in patients who were not able or willing to quit abruptly and were instructed to quit gradually [see Clinical studies (14.5)].
In all studies, patients were provided with an educational booklet on smoking cessation and received up to 10 minutes of smoking cessation counseling at each weekly treatment visit according to Agency for Healthcare Research and Quality guidelines.
4 Contraindications
Varenicline tablets are contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to varenicline tablets.
6 Adverse Reactions
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling:
-
•Neuropsychiatric Adverse Events including Suicidality [see Warnings and Precautions (5.1)]
-
•Seizures [see Warnings and Precautions (5.2)]
-
•Interaction with Alcohol [see Warnings and Precautions (5.3)]
-
•Accidental Injury [see Warnings and Precautions (5.4)]
-
•Cardiovascular Events [see Warnings and Precautions (5.5)]
-
•Somnambulism [see Warnings and Precautions (5.6)]
-
•Angioedema and Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
-
•Serious Skin Reactions [see Warnings and Precautions (5.8)]
In the placebo-controlled premarketing studies, the most common adverse events associated with varenicline tablets (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting.
The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for varenicline tablets, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in varenicline tablets-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo).
Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
7 Drug Interactions
Based on varenicline characteristics and clinical experience to date, varenicline tablets has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Varenicline is substantially eliminated by renal glomerular filtration along with active tubular secretion. Dose reduction is not required in patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30 mL/min), and for patients with end-stage renal disease undergoing hemodialysis, dosage adjustment is needed [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
5.4 Accidental Injury
There have been postmarketing reports of traffic accidents, near-miss incidents in traffic, or other accidental injuries in patients taking varenicline tablets. In some cases, the patients reported somnolence, dizziness, loss of consciousness or difficulty concentrating that resulted in impairment, or concern about potential impairment, in driving or operating machinery. Advise patients to use caution driving or operating machinery or engaging in other potentially hazardous activities until they know how varenicline tablets may affect them.
1 Indications and Usage
Varenicline tablets are indicated for use as an aid to smoking cessation treatment.
14.6 Re Treatment Study
Varenicline tablets was evaluated in a double-blind, placebo-controlled trial of patients who had made a previous attempt to quit smoking with varenicline tablets, and either did not succeed in quitting or relapsed after treatment. Subjects were randomized 1:1 to varenicline tablets 1 mg twice daily (N=249) or placebo (N=245) for 12 weeks of treatment and followed for 40 weeks post-treatment. Patients included in this study had taken varenicline tablets for a smoking-cessation attempt in the past (for a total treatment duration of a minimum of two weeks), at least three months prior to study entry, and had been smoking for at least four weeks.
Patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (45%) compared to patients treated with placebo (12%) and from weeks 9 through 52 (20%) compared to subjects treated with placebo (3%).
Table 9. Continuous Abstinence (95% confidence interval), Re-Treatment Study
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
Retreatment Study |
45% |
12% |
20% |
3% |
BID = twice daily
12.1 Mechanism of Action
Varenicline binds with high affinity and selectivity at α4β2 neuronal nicotinic acetylcholine receptors. The efficacy of varenicline tablets in smoking cessation is believed to be the result of varenicline's activity at α4β2 sub-type of the nicotinic receptor where its binding produces agonist activity, while simultaneously preventing nicotine binding to these receptors.
Electrophysiology studies in vitro and neurochemical studies in vivo have shown that varenicline binds to α4β2 neuronal nicotinic acetylcholine receptors and stimulates receptor-mediated activity, but at a significantly lower level than nicotine. Varenicline blocks the ability of nicotine to activate α4β2 receptors and thus to stimulate the central nervous mesolimbic dopamine system, believed to be the neuronal mechanism underlying reinforcement and reward experienced upon smoking. Varenicline is highly selective and binds more potently to α4β2 receptors than to other common nicotinic receptors (>500-fold α3β4, >3,500-fold α7, >20,000-fold α1βγδ), or to nonnicotinic receptors and transporters (>2,000-fold). Varenicline also binds with moderate affinity (Ki = 350 nM) to the 5-HT3 receptor.
9.1 Controlled Substance
Varenicline is not a controlled substance.
14.3 Long Term Abstinence
Studies 1 through 5 included 40 weeks of post-treatment follow-up. In each study, varenicline tablets-treated patients were more likely to maintain abstinence throughout the follow-up period than were patients treated with placebo (Figure 2, Table 8).
Table 8. Continuous Abstinence, Weeks 9 through 52 (95% confidence interval) Across Different Studies
|
Varenicline tablets
|
Varenicline tablets
|
Varenicline tablets
|
Bupropion SR
|
Placebo
|
|
|
Study 2 |
19% |
23% |
4% |
||
|
Study 3 |
22% |
8% |
|||
|
Study 4 |
21% |
16% |
8% |
||
|
Study 5 |
22% |
14% |
10% |
BID = twice daily
5.5 Cardiovascular Events
A comprehensive evaluation of cardiovascular (CV) risk with varenicline tablets suggests that patients with underlying CV disease may be at increased risk; however, these concerns must be balanced with the health benefits of smoking cessation. CV risk has been assessed for varenicline tablets in randomized controlled trials (RCT) and meta-analyses of RCTs. In a smoking cessation trial in patients with stable CV disease, CV events were infrequent overall; however, nonfatal myocardial infarction (MI) and nonfatal stroke occurred more frequently in patients treated with varenicline tablets compared to placebo. All-cause and CV mortality was lower in patients treated with varenicline tablets [see Clinical Studies (14.8)]. This study was included in a meta-analysis of 15 varenicline tablets efficacy trials in various clinical populations that showed an increased hazard ratio for Major Adverse Cardiovascular Events (MACE) of 1.95; however, the finding was not statistically significant (95% CI: 0.79, 4.82). In the large postmarketing neuropsychiatric safety outcome trial, an analysis of adjudicated MACE events was conducted for patients while in the trial and during a 28-week non-treatment extension period. Few MACE events occurred during the trial; therefore, the findings did not contribute substantively to the understanding of CV risk with varenicline tablets. Instruct patients to notify their healthcare providers of new or worsening CV symptoms and to seek immediate medical attention if they experience signs and symptoms of MI or stroke [see Clinical Studies (14.10)].
5 Warnings and Precautions
-
•Neuropsychiatric Adverse Events : Postmarketing reports of serious or clinically significant neuropsychiatric adverse events have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Observe patients attempting to quit smoking with varenicline tablets for the occurrence of such symptoms and instruct them to discontinue varenicline tablets and contact a healthcare provider if they experience such adverse events. (5.1)
-
•Seizures : New or worsening seizures have been observed in patients taking varenicline tablets. Varenicline tablets should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. (5.2)
-
•Interaction with Alcohol : Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether varenicline tablets affects them. (5.3)
-
•Accidental Injury : Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how varenicline tablets may affect them. (5.4)
-
•Cardiovascular Events : Patients with underlying cardiovascular (CV) disease may be at increased risk of CV events; however, these concerns must be balanced with the health benefits of smoking cessation. Instruct patients to notify their healthcare providers of new or worsening CV symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction (MI) or stroke. (5.5 and 6.1)
-
•Somnambulism : Cases of somnambulism have been reported in patients taking varenicline tablets. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue varenicline tablets and notify their healthcare provider if they experience somnambulism. (5.6 and 6.2)
-
•Angioedema and Hypersensitivity Reactions : Such reactions, including angioedema, infrequently life-threatening, have been reported. Instruct patients to discontinue varenicline tablets and immediately seek medical care if symptoms occur. (5.7 and 6.2)
-
•Serious Skin Reactions : Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue varenicline tablets and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. (5.8 and 6.2)
-
•Nausea : Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. (5.9)
5.8 Serious Skin Reactions
There have been postmarketing reports of rare but serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients using varenicline tablets [see Adverse Reactions (6.2)]. As these skin reactions can be life-threatening, instruct patients to stop taking varenicline tablets and contact a healthcare provider immediately at the first appearance of a skin rash with mucosal lesions or any other signs of hypersensitivity.
2 Dosage and Administration
-
•Begin varenicline tablets dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin varenicline tablets dosing and then quit smoking between days 8 and 35 of treatment. (2.1)
-
•Starting Week: 0.5 mg once daily on days 1-3 and 0.5 mg twice daily on days 4-7. (2.1)
-
•Continuing Weeks: 1 mg twice daily for a total of 12 weeks. (2.1)
-
•An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. (2.1)
-
•Consider a gradual approach to quitting smoking with varenicline tablets for patients who are sure that they are not able or willing to quit abruptly. Patients should begin varenicline tablets dosing and reduce smoking by 50% from baseline within the first four weeks, by an additional 50% in the next four weeks, and continue reducing with the goal of reaching complete abstinence by 12 weeks. Continue treatment for an additional 12 weeks, for a total of 24 weeks. (2.1)
-
•Severe Renal Impairment (estimated creatinine clearance less than 30 mL/min): Begin with 0.5 mg once daily and titrate to 0.5 mg twice daily. For patients with end-stage renal disease undergoing hemodialysis, a maximum of 0.5 mg daily may be given if tolerated. (2.2)
-
•Consider dose reduction for patients who cannot tolerate adverse effects. (2.1)
-
•Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. (2.1)
-
•Provide patients with appropriate educational materials and counseling to support the quit attempt. (2.1)
2.1 Usual Dosage for Adults
Smoking cessation therapies are more likely to succeed for patients who are motivated to stop smoking and who are provided additional advice and support. Provide patients with appropriate educational materials and counseling to support the quit attempt.
The patient should set a date to stop smoking. Begin varenicline tablets dosing one week before this date. Alternatively, the patient can begin varenicline tablets dosing and then quit smoking between days 8 and 35 of treatment.
Varenicline tablets should be taken orally after eating and with a full glass of water.
The recommended dose of varenicline tablets is 1 mg twice daily following a 1-week titration as follows:
|
Days 1 – 3: |
0.5 mg once daily |
|
Days 4 – 7: |
0.5 mg twice daily |
|
Day 8 – end of treatment: |
1 mg twice daily |
Patients should be treated with varenicline tablets for 12 weeks. For patients who have successfully stopped smoking at the end of 12 weeks, an additional course of 12 weeks treatment with varenicline tablets is recommended to further increase the likelihood of long-term abstinence.
For patients who are sure that they are not able or willing to quit abruptly, consider a gradual approach to quitting smoking with varenicline tablets. Patients should begin varenicline tablets dosing and reduce smoking by 50% from baseline within the first four weeks, by an additional 50% in the next four weeks, and continue reducing with the goal of reaching complete abstinence by 12 weeks. Continue varenicline tablets treatment for an additional 12 weeks, for a total of 24 weeks of treatment. Encourage patients to attempt quitting sooner if they feel ready [see Clinical Studies (14.5)].
Patients who are motivated to quit, and who did not succeed in stopping smoking during prior varenicline tablets therapy for reasons other than intolerability due to adverse events or who relapsed after treatment, should be encouraged to make another attempt with varenicline tablets once factors contributing to the failed attempt have been identified and addressed.
Consider a temporary or permanent dose reduction in patients who cannot tolerate the adverse effects of varenicline tablets.
3 Dosage Forms and Strengths
Modified capsule shaped tablets: 0.5 mg (white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side) and 1 mg (light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side).
5.3 Interaction With Alcohol
There have been postmarketing reports of patients experiencing increased intoxicating effects of alcohol while taking varenicline tablets. Some cases described unusual and sometimes aggressive behavior, and were often accompanied by amnesia for the events. Advise patients to reduce the amount of alcohol they consume while taking varenicline tablets until they know whether varenicline tablets affects their tolerance for alcohol [see Adverse Reactions (6.2)].
6.2 Postmarketing Experience
The following adverse events have been reported during post-approval use of varenicline tablets. Because these events are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
There have been reports of depression, mania, psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide in patients attempting to quit smoking while taking varenicline tablets [see Warnings and Precautions (5.1)].
There have been postmarketing reports of new or worsening seizures in patients treated with varenicline tablets [see Warnings and Precautions (5.2)].
There have been postmarketing reports of patients experiencing increased intoxicating effects of alcohol while taking varenicline tablets. Some reported neuropsychiatric events, including unusual and sometimes aggressive behavior [see Warnings and Precautions (5.1) and (5.3)].
There have been reports of hypersensitivity reactions, including angioedema [see Warnings and Precautions (5.7)].
There have also been reports of serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients taking varenicline tablets [see Warnings and Precautions (5.8)].
There have been reports of myocardial infarction (MI) and cerebrovascular accident (CVA) including ischemic and hemorrhagic events in patients taking varenicline tablets. In the majority of the reported cases, patients had pre-existing cardiovascular disease and/or other risk factors. Although smoking is a risk factor for MI and CVA, based on temporal relationship between medication use and events, a contributory role of varenicline cannot be ruled out [see Warnings and Precautions (5.5)].
There have been reports of hyperglycemia in patients following initiation of varenicline tablets.
There have been reports of somnambulism, some resulting in harmful behavior to self, others, or property in patients treated with varenicline tablets [see Warnings and Precautions (5.6)].
14.1 Initiation of Abstinence
Study 1
This was a six-week dose-ranging study comparing varenicline tablets to placebo. This study provided initial evidence that varenicline tablets at a total dose of 1 mg per day or 2 mg per day was effective as an aid to smoking cessation.
Study 2
This study of 627 patients compared varenicline tablets 1 mg per day and 2 mg per day with placebo. Patients were treated for 12 weeks (including one-week titration) and then were followed for 40 weeks post-treatment. Varenicline tablets was given in two divided doses daily. Each dose of varenicline tablets was given in two different regimens, with and without initial dose-titration, to explore the effect of different dosing regimens on tolerability. For the titrated groups, dosage was titrated up over the course of one week, with full dosage achieved starting with the second week of dosing. The titrated and nontitrated groups were pooled for efficacy analysis.
Forty-five percent of patients receiving varenicline tablets 1 mg per day (0.5 mg twice daily) and 51% of patients receiving 2 mg per day (1 mg twice daily) had CO-confirmed continuous abstinence during weeks 9 through 12 compared to 12% of patients in the placebo group (Figure 1). In addition, 31% of the 1 mg per day group and 31% of the 2 mg per day group were continuously abstinent from one week after TQD through the end of treatment as compared to 8% of the placebo group.
Study 3
This flexible-dosing study of 312 patients examined the effect of a patient-directed dosing strategy of varenicline tablets or placebo. After an initial one-week titration to a dose of 0.5 mg twice daily, patients could adjust their dosage as often as they wished between 0.5 mg once daily to 1 mg twice daily per day. Sixty-nine percent of patients titrated to the maximum allowable dose at any time during the study. For 44% of patients, the modal dose selected was 1 mg twice daily; for slightly over half of the study participants, the modal dose selected was 1 mg/day or less.
Of the patients treated with varenicline tablets, 40% had CO-confirmed continuous abstinence during weeks 9 through 12 compared to 12% in the placebo group. In addition, 29% of the varenicline tablets group were continuously abstinent from one week after TQD through the end of treatment as compared to 9% of the placebo group.
Study 4 and Study 5
These identical double-blind studies compared varenicline tablets 2 mg per day, bupropion sustained-release (SR) 150 mg twice daily, and placebo. Patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. The varenicline tablets dosage of 1 mg twice daily was achieved using a titration of 0.5 mg once daily for the initial 3 days followed by 0.5 mg twice daily for the next 4 days. The bupropion SR dosage of 150 mg twice daily was achieved using a 3-day titration of 150 mg once daily. Study 4 enrolled 1022 patients and Study 5 enrolled 1023 patients. Patients inappropriate for bupropion treatment or patients who had previously used bupropion were excluded.
In Study 4, patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (44%) compared to patients treated with bupropion SR (30%) or placebo (17%). The bupropion SR quit rate was also superior to placebo. In addition, 29% of the varenicline tablets group were continuously abstinent from one week after TQD through the end of treatment as compared to 12% of the placebo group and 23% of the bupropion SR group.
Similarly in Study 5, patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (44%) compared to patients treated with bupropion SR (30%) or placebo (18%). The bupropion SR quit rate was also superior to placebo. In addition, 29% of the varenicline tablets group were continuously abstinent from one week after TQD through the end of treatment as compared to 11% of the placebo group and 21% of the bupropion SR group.
Table 7. Continuous Abstinence, Weeks 9 through 12 (95% confidence interval)
|
Varenicline tablets
|
Varenicline tablets
|
Varenicline tablets Flexible
|
Bupropion SR
|
Placebo
|
|
|
Study 2 |
45% |
51% |
12% |
||
|
Study 3 |
40% |
12% |
|||
|
Study 4 |
44% |
30% |
17% |
||
|
Study 5 |
44% |
30% |
18% |
BID = twice daily
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reactions rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During the premarketing development of varenicline tablets, over 4500 subjects were exposed to varenicline tablets, with over 450 treated for at least 24 weeks and approximately 100 for a year. Most study participants were treated for 12 weeks or less.
The most common adverse event associated with varenicline tablets treatment is nausea, occurring in 30% of patients treated at the recommended dose, compared with 10% in patients taking a comparable placebo regimen [see Warnings and Precautions (5.9)].
Table 1 shows the adverse events for varenicline tablets and placebo in the 12- week fixed dose premarketing studies with titration in the first week [Studies 2 (titrated arm only), 4, and 5]. Adverse events were categorized using the Medical Dictionary for Regulatory Activities (MedDRA, Version 7.1).
MedDRA High Level Group Terms (HLGT) reported in ≥5% of patients in the varenicline tablets 1 mg twice daily dose group, and more commonly than in the placebo group, are listed, along with subordinate Preferred Terms (PT) reported in ≥1% of varenicline tablets patients (and at least 0.5% more frequent than placebo). Closely related Preferred Terms such as 'Insomnia', 'Initial insomnia', 'Middle insomnia', 'Early morning awakening' were grouped, but individual patients reporting two or more grouped events are only counted once.
Table 1. Common Treatment Emergent AEs (%) in the Fixed-Dose, Placebo-Controlled Studies (HLGTs >5% of Patients in the 1 mg BID Varenicline Tablets Group and More Commonly than Placebo and PT ≥1% in the 1 mg BID Varenicline Tablets Group, and 1 mg BID Varenicline Tablets at Least 0.5% More than Placebo)
|
SYSTEM ORGAN CLASS
|
Varenicline Tablets
|
Varenicline Tablets
|
Placebo
|
|
GASTROINTESTINAL (GI) |
|||
|
GI Signs and Symptoms |
|||
|
Nausea |
16 |
30 |
10 |
|
Abdominal Pain * |
5 |
7 |
5 |
|
Flatulence |
9 |
6 |
3 |
|
Dyspepsia |
5 |
5 |
3 |
|
Vomiting |
1 |
5 |
2 |
|
GI Motility/Defecation |
|||
|
Constipation |
5 |
8 |
3 |
|
Gastroesophageal reflux |
1 |
1 |
0 |
|
Salivary Gland Conditions |
|||
|
Dry mouth |
4 |
6 |
4 |
|
PSYCHIATRIC DISORDERS |
|||
|
Sleep Disorder/Disturbances |
|||
|
Insomnia ** |
19 |
18 |
13 |
|
Abnormal dreams |
9 |
13 |
5 |
|
Sleep disorder |
2 |
5 |
3 |
|
Nightmare |
2 |
1 |
0 |
|
NERVOUS SYSTEM |
|||
|
Headaches |
|||
|
Headache |
19 |
15 |
13 |
|
Neurological Disorders |
|||
|
NEC |
|||
|
Dysgeusia |
8 |
5 |
4 |
|
Somnolence |
3 |
3 |
2 |
|
Lethargy |
2 |
1 |
0 |
|
GENERAL DISORDERS |
|||
|
General Disorders NEC |
|||
|
Fatigue/Malaise/Asthenia |
4 |
7 |
6 |
|
RESPIR/THORACIC/MEDIAST |
|||
|
Respiratory Disorders NEC |
|||
|
Rhinorrhea |
0 |
1 |
0 |
|
Dyspnea |
2 |
1 |
1 |
|
Upper Respiratory Tract |
7 |
5 |
4 |
|
SKIN/SUBCUTANEOUS TISSUE |
|||
|
Epidermal and Dermal |
|||
|
Rash |
1 |
3 |
2 |
|
Pruritis |
0 |
1 |
1 |
|
METABOLISM and NUTRITION |
|||
|
Appetite/General Nutrition |
|||
|
Increased appetite |
4 |
3 |
2 |
|
Decreased appetite/ |
1 |
2 |
1 |
* Includes PTs Abdominal (pain, pain upper, pain lower, discomfort, tenderness, distension) and Stomach discomfort
** Includes PTs Insomnia/Initial insomnia/Middle insomnia/Early morning awakening
The overall pattern and frequency of adverse events during the longer-term premarketing trials was similar to those described in Table 1, though several of the most common events were reported by a greater proportion of patients with long-term use (e.g., nausea was reported in 40% of patients treated with varenicline tablets 1 mg twice daily in a one year study, compared to 8% of placebo-treated patients).
Following is a list of treatment-emergent adverse events reported by patients treated with varenicline tablets during all premarketing clinical trials and updated based on pooled data from 18 placebo-controlled pre- and postmarketing studies, including approximately 5,000 patients treated with varenicline. Adverse events were categorized using MedDRA, Version 16.0. The listing does not include those events already listed in the previous tables or elsewhere in labeling, those events for which a drug cause was remote, those events which were so general as to be uninformative, and those events reported only once which did not have a substantial probability of being acutely life-threatening.
Blood and Lymphatic System Disorders. Infrequent: anemia, lymphadenopathy. Rare: leukocytosis, splenomegaly, thrombocytopenia.
Cardiac Disorders. Infrequent: angina pectoris, myocardial infarction, palpitations, tachycardia. Rare: acute coronary syndrome, arrhythmia, atrial fibrillation, bradycardia, cardiac flutter, cor pulmonale, coronary artery disease, ventricular extrasystoles.
Ear and Labyrinth Disorders. Infrequent: tinnitus, vertigo. Rare: deafness, Meniere's disease.
Endocrine Disorders. Infrequent: thyroid gland disorders.
Eye Disorders. Infrequent: conjunctivitis, eye irritation, eye pain, vision blurred, visual impairment. Rare: blindness transient, cataract subcapsular, dry eye, night blindness, ocular vascular disorder, photophobia, vitreous floaters.
Gastrointestinal Disorders. Frequent: diarrhea, toothache. Infrequent: dysphagia, eructation, gastritis, gastrointestinal hemorrhage, mouth ulceration. Rare: enterocolitis, esophagitis, gastric ulcer, intestinal obstruction, pancreatitis acute.
General Disorders and Administration Site Conditions. Frequent: chest pain. Infrequent: chest discomfort, chills, edema, influenza-like illness, pyrexia.
Hepatobiliary Disorders. Rare: gall bladder disorder.
Investigations. Frequent: liver function test abnormal, weight increased. Infrequent: electrocardiogram abnormal. Rare: muscle enzyme increased, urine analysis abnormal.
Metabolism and Nutrition Disorders. Infrequent: diabetes mellitus, hypoglycemia. Rare: hyperlipidemia, hypokalemia.
Musculoskeletal and Connective Tissue Disorders. Frequent: arthralgia, back pain, myalgia. Infrequent: arthritis, muscle cramp, musculoskeletal pain. Rare: myositis, osteoporosis.
Nervous System Disorders. Frequent: disturbance in attention, dizziness. Infrequent: amnesia, convulsion, migraine, parosmia, syncope, tremor. Rare: balance disorder, cerebrovascular accident, dysarthria, mental impairment, multiple sclerosis, VIIth nerve paralysis, nystagmus, psychomotor hyperactivity, psychomotor skills impaired, restless legs syndrome, sensory disturbance, transient ischemic attack, visual field defect.
Psychiatric Disorders. Infrequent: dissociation, libido decreased, mood swings, thinking abnormal. Rare: bradyphrenia, disorientation, euphoric mood.
Renal and Urinary Disorders. Infrequent: nocturia, pollakiuria, urine abnormality. Rare: nephrolithiasis, polyuria, renal failure acute, urethral syndrome, urinary retention.
Reproductive System and Breast Disorders. Frequent: menstrual disorder. Infrequent: erectile dysfunction. Rare: sexual dysfunction.
Respiratory, Thoracic and Mediastinal Disorders. Frequent: respiratory disorders. Infrequent: asthma, epistaxis, rhinitis allergic, upper respiratory tract inflammation.
Rare: pleurisy, pulmonary embolism.
Skin and Subcutaneous Tissue Disorders. Infrequent: acne, dry skin, eczema, erythema, hyperhidrosis, urticaria. Rare: photosensitivity reaction, psoriasis.
Vascular Disorders. Infrequent: hot flush. Rare: thrombosis.
Varenicline tablets has also been studied in postmarketing trials including (1) a trial conducted in patients with chronic obstructive pulmonary disease (COPD), (2) a trial conducted in generally healthy patients (similar to those in the premarketing studies) in which they were allowed to select a quit date between days 8 and 35 of treatment ("alternative quit date instruction trial"), (3) a trial conducted in patients who did not succeed in stopping smoking during prior varenicline tablets therapy, or who relapsed after treatment ("re-treatment trial"), (4) a trial conducted in patients with stable cardiovascular disease, (5) a trial conducted in patients with stable schizophrenia or schizoaffective disorder, (6) a trial conducted in patients with major depressive disorder, (7) a postmarketing neuropsychiatric safety outcome trial in patients without or with a history of psychiatric disorder, (8) a non-treatment extension of the postmarketing neuropsychiatric safety outcome trial that assessed CV safety, (9) a trial in patients who were not able or willing to quit abruptly and who were instructed to quit gradually ("gradual approach to quitting smoking trial").
Adverse events in the trial of patients with COPD (1), in the alternative quit date instruction trial (2), and in the gradual approach to quitting smoking trial (9) were similar to those observed in premarketing studies. In the re-treatment trial (3), the profile of common adverse events was similar to that previously reported, but, in addition, varenicline-treated patients also commonly reported diarrhea (6% vs. 4% in placebo-treated patients), depressed mood disorders and disturbances (6% vs. 1%), and other mood disorders and disturbances (5% vs. 2%).
In the trial of patients with stable cardiovascular disease (4), more types and a greater number of cardiovascular events were reported compared to premarketing studies, as shown in Table 1 and in Table 2 below.
Table 2. Cardiovascular Mortality and Nonfatal Cardiovascular Events (%) with a Frequency >1% in Either Treatment Group in the Trial of Patients with Stable Cardiovascular Disease
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥1% in either treatment group
|
||
|
Up to 30 days after treatment
|
||
|
Angina pectoris |
3.7 |
2.0 |
|
Chest pain |
2.5 |
2.3 |
|
Peripheral edema |
2.0 |
1.1 |
|
Hypertension |
1.4 |
2.6 |
|
Palpitations |
0.6 |
1.1 |
|
Adjudicated Cardiovascular Mortality (up to 52 weeks)
|
0.3 |
0.6 |
|
Adjudicated Nonfatal Serious Cardiovascular Events ≥1% in either treatment group
|
||
|
Up to 30 days after treatment
|
||
|
Nonfatal MI |
1.1 |
0.3 |
|
Hospitalization for angina pectoris |
0.6 |
1.1 |
|
Beyond 30 days after treatment and up to 52 weeks
|
||
|
Need for coronary revascularization* |
2.0 |
0.6 |
|
Hospitalization for angina pectoris |
1.7 |
1.1 |
|
New diagnosis of peripheral vascular disease (PVD) or |
1.4 |
0.6 |
*some procedures were part of management of nonfatal MI and hospitalization for angina
In the trial of patients with stable schizophrenia or schizoaffective disorder (5), 128 smokers on antipsychotic medication were randomized 2:1 to varenicline (1 mg twice daily) or placebo for 12 weeks with 12-week non-drug follow-up. The most common treatment emergent adverse events reported in this trial are shown in Table 3 below.
Table 3. Common Treatment Emergent AEs (%) in the Trial of Patients with Stable Schizophrenia or Schizoaffective Disorder
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥10% in the varenicline group
|
||
|
Nausea |
24 |
14 |
|
Headache |
11 |
19 |
|
Vomiting |
11 |
9 |
|
Psychiatric Adverse Events ≥5% and at a higher rate than in the placebo group
|
||
|
Insomnia |
10 |
5 |
For the trial of patients with major depressive disorder (6), the most common treatment emergent adverse events reported are shown in Table 4 below. Additionally, in this trial, patients treated with varenicline were more likely than patients treated with placebo to report one of events related to hostility and aggression (3% vs. 1%).
Table 4. Common Treatment Emergent AEs (%) in the Trial of Patients with Major Depressive Disorder
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥10% in either treatment group
|
||
|
Nausea |
27 |
10 |
|
Headache |
17 |
11 |
|
Abnormal dreams |
11 |
8 |
|
Insomnia |
11 |
5 |
|
Irritability |
11 |
8 |
|
Psychiatric Adverse Events ≥2% in any treatment group and not included above
|
||
|
Depressed mood disorders and disturbances |
11 |
9 |
|
Anxiety |
7 |
9 |
|
Agitation |
7 |
4 |
|
Tension |
4 |
3 |
|
Hostility |
2 |
0.4 |
|
Restlessness |
2 |
2 |
In the trial of patients without or with a history of psychiatric disorder (7), the most common adverse events in subjects treated with varenicline were similar to those observed in premarketing studies. Most common treatment-emergent adverse events reported in this trial are shown in Table 5 below.
Table 5. Treatment Emergent Common AEs (%) in the Trial of Patients without or with a History of Psychiatric Disorder
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥10% in the varenicline group
|
||
|
Entire study population, N |
1982 |
1979 |
|
Nausea |
25 |
7 |
|
Headache |
12 |
10 |
|
Psychiatric Adverse Events ≥2% in any treatment group
|
||
|
Non-psychiatric cohort, N
|
975 |
982 |
|
Abnormal dreams |
8 |
4 |
|
Agitation |
3 |
3 |
|
Anxiety |
5 |
6 |
|
Depressed mood |
3 |
3 |
|
Insomnia |
10 |
7 |
|
Irritability |
3 |
4 |
|
Sleep disorder |
3 |
2 |
|
Psychiatric cohort, N
|
1007 |
997 |
|
Abnormal dreams |
12 |
5 |
|
Agitation |
5 |
4 |
|
Anxiety |
8 |
6 |
|
Depressed mood |
5 |
5 |
|
Depression |
5 |
5 |
|
Insomnia |
9 |
7 |
|
Irritability |
5 |
7 |
|
Nervousness |
2 |
3 |
|
Sleep disorder |
3 |
2 |
In the non-treatment extension of the postmarketing neuropsychiatric safety outcomes trial that assessed CV safety (8), the most common adverse events in subjects treated with varenicline and occurring up to 30 days after last dose of treatment were similar to those observed in premarketing studies.
17 Patient Counseling Information
See FDA-approved patient labeling (Medication Guide)
Initiate Treatment and Continue to Attempt to Quit if Lapse
Instruct patients to set a date to quit smoking and to initiate varenicline tablets treatment one week before the quit date. Alternatively, the patient can begin varenicline tablets dosing and then set a date to quit smoking between days 8 and 35 of treatment. Encourage patients to continue to attempt to quit if they have early lapses after quit day [see Dosage and Administration (2.1)].
For patients who are sure that they are not able or willing to quit abruptly, a gradual approach to quitting smoking with varenicline tablets may be considered. Patients should begin varenicline tablets dosing and reduce smoking during the first 12 weeks of treatment, then quit by the end of that period and continue treatment for an additional 12 weeks for a total of 24 weeks [see Dosage and Administration (2.1)].
Encourage patients who are motivated to quit and who did not succeed in stopping smoking during prior varenicline tablets therapy for reasons other than intolerability due to adverse events, or who relapsed after treatment to make another attempt with varenicline tablets once factors contributing to the failed attempt have been identified and addressed [see Dosage and Administration (2.1), Clinical Studies (14.6)].
How to Take
Advise patients that varenicline tablets should be taken orally after eating, and with a full glass of water [see Dosage and Administration (2.1)].
Starting Week Dosage
Instruct patients on how to titrate varenicline tablets, beginning at a dose of 0.5 mg/day. Explain that one 0.5 mg tablet should be taken daily for the first three days, and that for the next four days, one 0.5 mg tablet should be taken in the morning and one 0.5 mg tablet should be taken in the evening [see Dosage and Administration (2.1)].
Continuing Weeks Dosage
Advise patients that, after the first seven days, the dose should be increased to one 1 mg tablet in the morning and one 1 mg tablet in the evening [see Dosage and Administration (2.1)].
Dosage Adjustment for Varenicline Tablets or Other Drugs
Inform patients that nausea and insomnia are side effects of varenicline tablets and are usually transient; however, advise patients that if they are persistently troubled by these symptoms, they should notify the prescribing physician so that a dose reduction can be considered.
Inform patients that some drugs may require dose adjustment after quitting smoking [see Dosage and Administration (2.1)].
Counseling and Support
Provide patients with educational materials and necessary counseling to support an attempt at quitting smoking [see Dosage and Administration (2.1)].
Neuropsychiatric Adverse Events
Inform patients that some patients have experienced changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation and suicide when attempting to quit smoking while taking varenicline tablets. Instruct patients to discontinue varenicline tablets and contact a healthcare professional if they experience such symptoms [see Warnings and Precautions (5.1), Adverse Reactions (6.2)].
History of Psychiatric Illness
Encourage patients to reveal any history of psychiatric illness prior to initiating treatment.
Nicotine Withdrawal
Inform patients that quitting smoking, with or without varenicline tablets, may be associated with nicotine withdrawal symptoms (including depression or agitation) or exacerbation of pre-existing psychiatric illness.
Seizures
Encourage patients to report any history of seizures or other factors that can lower seizure threshold. Instruct patients to discontinue varenicline tablets and contact a healthcare provider immediately if they experience a seizure while on treatment [see Warnings and Precautions (5.2)].
Interaction with Alcohol
Advise patients to reduce the amount of alcohol they consume while taking varenicline tablets until they know whether varenicline tablets affects their tolerance for alcohol [see Warnings and Precautions (5.3), Adverse Reactions (6.2)].
Driving or Operating Machinery
Advise patients to use caution driving or operating machinery until they know how quitting smoking and/or varenicline may affect them [see Warnings and Precautions (5.4)].
Cardiovascular Events
Patients should be instructed to notify their healthcare providers of symptoms of new or worsening cardiovascular events and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke [see Warnings and Precautions (5.5), Adverse Reactions (6.1)].
Somnambulism
Patients should be instructed to discontinue varenicline tablets and notify their healthcare providers if they experience somnambulism [see Warnings and Precautions (5.6)].
Angioedema
Inform patients that there have been reports of angioedema, with swelling of the face, mouth (lip, gum, tongue) and neck (larynx and pharynx) that can lead to life-threatening respiratory compromise. Instruct patients to discontinue varenicline tablets and immediately seek medical care if they experience these symptoms [see Warnings and Precautions (5.7), Adverse Reactions (6.2)].
Serious Skin Reactions
Inform patients that serious skin reactions, such as Stevens-Johnson Syndrome and erythema multiforme, were reported by some patients taking varenicline tablets. Advise patients to stop taking varenicline tablets at the first sign of rash with mucosal lesions or skin reaction and contact a healthcare provider immediately [see Warnings and Precautions (5.8), Adverse Reactions (6.2)].
Vivid, Unusual, or Strange Dreams
Inform patients that they may experience vivid, unusual or strange dreams during treatment with varenicline tablets.
Pregnancy and Lactation
Patients who are pregnant or breastfeeding or planning to become pregnant should be advised of: the risks of smoking to a pregnant mother and her developing baby, the potential risks of varenicline tablets use during pregnancy and breastfeeding, and the benefits of smoking cessation with and without varenicline tablets. Advise breastfeeding women to monitor the infant for seizures and vomiting [see Use in Specific Populations (8.1 and 8.2)].
This product's label may have been updated. For full prescribing information, please visit www.glenmarkpharma-us.com
Distributed by:
Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
Code No.: HP/Drugs/N-MNB/17/194
Revised: August 2025
16 How Supplied/storage and Handling
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg modified capsule shaped, white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg modified capsule shaped, light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side.
Varenicline tablets are supplied in the following package configurations:
|
Description
|
NDC
|
|
|
Packs |
Starting 4-week card: 0.5 mg x 11 tablets and 1 mg x 42 tablets |
NDC 68462-895-04 |
|
Continuing 4-week card: 1 mg x 56 tablets |
NDC 68462-894-04 |
|
|
Starting Month Box: 0.5 mg x 11 tablets and 1 mg x 42 tablets |
NDC 68462-895-04 |
|
|
Continuing Month Box: 1 mg x 56 tablets |
NDC 68462-894-04 |
|
|
Bottles |
0.5 mg - bottle of 56 |
NDC 68462-893-56 |
|
1 mg - bottle of 56 |
NDC 68462-894-56 |
Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP.
This package is child-resistant
14.5 Gradual Approach to Quitting Smoking
Varenicline tablets was evaluated in a 52-week double-blind placebo-controlled study of 1,510 subjects who were not able or willing to quit smoking within four weeks, but were willing to gradually reduce their smoking over a 12 week period before quitting. Subjects were randomized to either varenicline tablets 1 mg twice daily (N=760) or placebo (N=750) for 24 weeks and followed up post-treatment through week 52. Subjects were instructed to reduce the number of cigarettes smoked by at least 50 percent by the end of the first four weeks of treatment, followed by a further 50 percent reduction from week four to week eight of treatment, with the goal of reaching complete abstinence by 12 weeks. After the initial 12-week reduction phase, subjects continued treatment for another 12 weeks. Subjects treated with varenicline tablets had a significantly higher Continuous Abstinence Rate compared with placebo at weeks 15 through 24 (32% vs. 7%) and weeks 15 through 52 (24% vs. 6%).
14.9 Subjects With Major Depressive Disorder
Varenicline tablets was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 18 to 75 years with major depressive disorder without psychotic features (DSM-IV TR). If on medication, subjects were to be on a stable antidepressant regimen for at least two months. If not on medication, subjects were to have experienced a major depressive episode in the past 2 years, which was successfully treated. Subjects were randomized to varenicline tablets 1 mg twice daily (N=256) or placebo (N=269) for a treatment of 12 weeks and then followed for 40 weeks post-treatment. Subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (36%) compared to subjects treated with placebo (16%) and from week 9 through 52 (20%) compared to subjects treated with placebo (10%).
Table 14. Continuous Abstinence (95% confidence interval), Study in Patients with Major Depressive Disorder (MDD)
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
MDD Study |
36% |
16% |
20% |
10% |
BID = twice daily
5.7 Angioedema and Hypersensitivity Reactions
There have been postmarketing reports of hypersensitivity reactions including angioedema in patients treated with varenicline tablets [see Adverse Reactions (6.2), Patient Counseling Information (17)]. Clinical signs included swelling of the face, mouth (tongue, lips, and gums), extremities, and neck (throat and larynx). There were infrequent reports of life-threatening angioedema requiring emergent medical attention due to respiratory compromise. Instruct patients to discontinue varenicline tablets and immediately seek medical care if they experience these symptoms.
7.1 Use With Other Drugs for Smoking Cessation
Safety and efficacy of varenicline tablets in combination with other smoking cessation therapies have not been studied.
7.2 Effect of Smoking Cessation On Other Drugs
Physiological changes resulting from smoking cessation, with or without treatment with varenicline tablets, may alter the pharmacokinetics or pharmacodynamics of certain drugs (e.g., theophylline, warfarin, insulin) for which dosage adjustment may be necessary.
14.4 Alternative Instructions for Setting A Quit Date
Varenicline tablets was evaluated in a double-blind, placebo-controlled trial where patients were instructed to select a target quit date between Day 8 and Day 35 of treatment. Subjects were randomized 3:1 to varenicline tablets 1 mg twice daily (N=486) or placebo (N=165) for 12 weeks of treatment and followed for another 12 weeks posttreatment. Patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (54%) compared to patients treated with placebo (19%) and from weeks 9 through 24 (35%) compared to subjects treated with placebo (13%).
14.7 Subjects With Chronic Obstructive Pulmonary Disease
Varenicline tablets was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged ≥ 35 years with mild-to-moderate COPD with post-bronchodilator FEV1/FVC <70% and FEV1 ≥ 50% of predicted normal value. Subjects were randomized to varenicline tablets 1 mg twice daily (N=223) or placebo (N=237) for a treatment of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (41%) compared to subjects treated with placebo (9%) and from week 9 through 52 (19%) compared to subjects treated with placebo (6%).
Table 10. Continuous Abstinence (95% confidence interval), Studies in Patients with Chronic Obstructive Pulmonary Disease (COPD)
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
COPD Study |
41% |
9% |
19% |
6% |
BID = twice daily
14.10 Postmarketing Neuropsychiatric Safety Outcome Trial
Varenicline tablets was evaluated in a randomized, double-blind, active and placebo-controlled trial that included subjects without a history of psychiatric disorder (non-psychiatric cohort, N=3912) and with a history of psychiatric disorder (psychiatric cohort, N=4003). Subjects aged 18-75 years, smoking 10 or more cigarettes per day were randomized 1:1:1:1 to varenicline tablets 1 mg BID, bupropion SR 150 mg BID, NRT patch 21 mg/day with taper or placebo for a treatment period of 12 weeks; they were then followed for another 12 weeks post-treatment. [See Warnings and Precautions (5.1)]
A composite safety endpoint intended to capture clinically significant neuropsychiatric (NPS) adverse events included the following NPS adverse events: anxiety, depression, feeling abnormal, hostility, agitation, aggression, delusions, hallucinations, homicidal ideation, mania, panic, paranoia, psychosis, irritability, suicidal ideation, suicidal behavior or completed suicide.
As shown in Table 15, the use of varenicline tablets, bupropion, and NRT in the non-psychiatric cohort was not associated with an increased risk of clinically significant NPS adverse events compared with placebo. Similarly, in the non-psychiatric cohort, the use of varenicline tablets was not associated with an increased risk of clinically significant NPS adverse events in the composite safety endpoint compared with bupropion or NRT.
Table 15. Number of Patients with Clinically Significant or Serious NPS Adverse Events by Treatment Group Among Patients without a History of Psychiatric Disorder
|
Varenicline tablets
|
Bupropion
|
NRT
|
Placebo
|
|
|
Clinically Significant NPS |
30 (3.1) |
34 (3.5) |
33 (3.3) |
40 (4.1) |
|
Serious NPS |
1 (0.1) |
5 (0.5) |
1 (0.1) |
4 (0.4) |
|
Psychiatric Hospitalizations |
1 (0.1) |
2 (0.2) |
0 (0.0) |
1 (0.1) |
As shown in Table 16, there were more clinically significant NPS adverse events reported in patients in the psychiatric cohort in each treatment group compared with the non-psychiatric cohort. The incidence of events in the composite endpoint was higher for each of the active treatments compared to placebo: Risk Differences (RDs) (95%CI) vs placebo were 2.7% (-0.05, 5.4) for varenicline tablets, 2.2% (-0.5, 4.9) for bupropion, and 0.4% (-2.2, 3.0) for NRT transdermal nicotine.
Table 16. Number of Patients with Clinically Significant or Serious NPS Adverse Events by Treatment Group Among Patients with a History of Psychiatric Disorder
|
Varenicline tablets
|
Bupropion
|
NRT
|
Placebo
|
|
|
Clinically Significant NPS |
123 (12.2) |
118 (11.8) |
98 (9.8) |
95 (9.5) |
|
Serious NPS |
6 (0.6) |
8 (0.8) |
4 (0.4) |
6 (0.6) |
|
Psychiatric hospitalizations |
5 (0.5) |
8 (0.8) |
4 (0.4) |
2 (0.2) |
There was one completed suicide, which occurred during treatment in a patient treated with placebo in the non-psychiatric cohort. There were no completed suicides reported in the psychiatric cohort.
In both cohorts, subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 and 9 through 24 compared to subjects treated with bupropion, nicotine patch and placebo.
Table 17 Continuous Abstinence (95% confidence interval), Study in Patients with or without a History of Psychiatric Disorder
|
Varenicline tablets
|
Bupropion SR
|
NRT
|
Placebo
|
|
|
Weeks 9 through 12
|
||||
|
Non- |
38% |
26% |
26% |
14% |
|
Psychiatric |
(35%, 41%) |
(23%, 29%) |
(24%, 29%) |
(12%, 16%) |
|
Cohort |
||||
|
Psychiatric |
29% |
19% |
20% |
11% |
|
Cohort |
(26%, 32%) |
(17%, 22%) |
(18%, 23%) |
(10%, 14%) |
|
Weeks 9 through 24
|
||||
|
Non- |
25% |
19% |
18% |
11% |
|
Psychiatric |
(23%, 28%) |
(16%, 21%) |
(16%, 21%) |
(9%, 13%) |
|
Cohort |
||||
|
Psychiatric |
18% |
14% |
13% |
8% |
|
Cohort |
(16%, 21%) |
(12%, 16%) |
(11%, 15%) |
(7%, 10%) |
BID = twice daily
5.1 Neuropsychiatric Adverse Events Including Suicidality
Serious neuropsychiatric adverse events have been reported in patients being treated with varenicline tablets [see Adverse Reactions (6.2)]. These postmarketing reports have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Some patients who stopped smoking may have been experiencing symptoms of nicotine withdrawal, including depressed mood. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these adverse events occurred in patients taking varenicline tablets who continued to smoke.
Neuropsychiatric adverse events occurred in patients without and with pre-existing psychiatric disease; some patients experienced worsening of their psychiatric illnesses. Some neuropsychiatric adverse events, including unusual and sometimes aggressive behavior directed to oneself or others, may have been worsened by concomitant use of alcohol [see Warnings and Precautions (5.3) , Adverse Reactions (6.2)]. Observe patients for the occurrence of neuropsychiatric adverse events. Advise patients and caregivers that the patient should stop taking varenicline tablets and contact a healthcare provider immediately if agitation, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. The healthcare provider should evaluate the severity of the symptoms and the extent to which the patient is benefiting from treatment, and consider options including dose reduction, continued treatment under closer monitoring, or discontinuing treatment. In many postmarketing cases, resolution of symptoms after discontinuation of varenicline tablets was reported. However, the symptoms persisted in some cases; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.
The neuropsychiatric safety of varenicline tablets was evaluated in a randomized, double-blind, active and placebo-controlled study that included patients without a history of psychiatric disorder (non-psychiatric cohort, N=3912) and patients with a history of psychiatric disorder (psychiatric cohort, N=4003). In the non-psychiatric cohort, varenicline tablets was not associated with an increased incidence of clinically significant neuropsychiatric adverse events in a composite endpoint comprising anxiety, depression, feeling abnormal, hostility, agitation, aggression, delusions, hallucinations, homicidal ideation, mania, panic, and irritability. In the psychiatric cohort, there were more events reported in each treatment group compared to the non-psychiatric cohort, and the incidence of events in the composite endpoint was higher for each of the active treatments compared to placebo: Risk Differences (RDs) (95%CI) vs. placebo were 2.7% (-0.05, 5.4) for varenicline tablets, 2.2% (-0.5, 4.9) for bupropion, and 0.4% (-2.2, 3.0) for transdermal nicotine. In the non-psychiatric cohort, neuropsychiatric adverse events of a serious nature were reported in 0.1% of varenicline tablets-treated patients and 0.4% of placebo-treated patients. In the psychiatric cohort, neuropsychiatric events of a serious nature were reported in 0.6% of varenicline tablets-treated patients, with 0.5% involving psychiatric hospitalization. In placebo-treated patients, serious neuropsychiatric events occurred in 0.6%, with 0.2% requiring psychiatric hospitalization [see Clinical Studies (14.10)].
Principal Display Panel 0.5 / 1 Mg Tablet Starting Pack Carton
NDC 68462-895-04
Varenicline Tablets
Dispense the Medication Guide provided separately to each patient.
STARTING MONTH BOX
(Your first 4 weeks)
Contains:
1 Starting Week (0.5 mg* x 11 tablets)
3 Continuing Weeks (1 mg† x 42 tablets)
Use the Starting Week (green) blister card first when beginning to take Varenicline Tablets.
Rx only
PRINCIPAL DISPLAY PANEL - 0.5 / 1 mg Tablet Starting Pack
NDC 68462-895-04
Varenicline Tablets
Dispense the Medication Guide provided separately to each patient.
STARTING PACK
Use the Starting Week (green) blister card first when beginning to take Varenicline Tablets.
Contains:
1 Starting Week (0.5 mg* x 11 tablets)
3 Continuing Weeks (1 mg† x 42 tablets)
Rx only
PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Bottle Label
NDC 68462-893-56
Varenicline Tablets
0.5 mg*
Dispense the Medication Guide provided separately to each patient.
Rx only 56 Tablets
PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label
NDC 68462-894-56
Varenicline Tablets
1 mg*
Dispense the Medication Guide provided separately to each patient.
56 Tablets
Rx only
14.8 Subjects With Cardiovascular Disease and Other Cardiovascular Analyses
Varenicline tablets was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 35 to 75 years with stable, documented cardiovascular disease (diagnoses other than, or in addition to, hypertension) that had been diagnosed for more than 2 months. Subjects were randomized to varenicline tablets 1 mg twice daily (N=353) or placebo (N=350) for a treatment period of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (47%) compared to subjects treated with placebo (14%) and from week 9 through 52 (20%) compared to subjects treated with placebo (7%).
Table 11. Continuous Abstinence (95% confidence interval), Studies in Patients with Cardiovascular Disease (CVD)
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
CVD Study |
47% |
14% |
20% |
7% |
BID = twice daily
In this study, all-cause and CV mortality was lower in patients treated with varenicline tablets, but certain nonfatal CV events occurred more frequently in patients treated with varenicline tablets than in patients treated with placebo [see Warnings and Precautions (5.5), Adverse Reactions (6.1)]. Table 12 below shows mortality and the incidence of selected nonfatal serious CV events occurring more frequently in the varenicline tablets arm compared to the placebo arm. These events were adjudicated by an independent blinded committee. Nonfatal serious CV events not listed occurred at the same incidence or more commonly in the placebo arm. Patients with more than one CV event of the same type are counted only once per row. Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina.
Table 12. Mortality and Adjudicated Nonfatal Serious Cardiovascular Events in the Placebo-Controlled Varenicline Tablets Trial in Patients with Stable Cardiovascular Disease
|
|
Varenicline tablets
|
Placebo
|
|
Mortality (Cardiovascular and All-cause up to 52 weeks)
|
||
|
Cardiovascular |
1 (0.3) |
2 (0.6) |
|
All-cause |
2 (0.6) |
5 (1.4) |
|
Nonfatal Cardiovascular Events (rate on varenicline tablets > Placebo)
|
||
|
Up to 30 days after treatment
|
||
|
Nonfatal myocardial infarction |
4 (1.1) |
1 (0.3) |
|
Nonfatal Stroke |
2 (0.6) |
0 (0) |
|
Beyond 30 days after treatment and
|
||
|
Nonfatal myocardial infarction |
3 (0.8) |
2 (0.6) |
|
Need for coronary |
7 (2.0) |
2 (0.6) |
|
Hospitalization for angina pectoris |
6 (1.7) |
4 (1.1) |
|
Transient ischemia attack |
1 (0.3) |
0 (0) |
|
New diagnosis of peripheral |
|
|
Following the CVD study, a meta-analysis of 15 clinical trials of ≥12 weeks treatment duration, including 7002 patients (4190 varenicline tablets, 2812 placebo), was conducted to systematically assess the CV safety of varenicline tablets. The study in patients with stable CV disease described above was included in the meta-analysis. There were lower rates of all-cause mortality (varenicline tablets 6 [0.14%]; placebo 7 [0.25%]) and CV mortality (varenicline tablets 2 [0.05%]; placebo 2 [0.07%]) in the varenicline tablets arms compared with the placebo arms in the meta-analysis.
The key CV safety analysis included occurrence and timing of a composite endpoint of Major Adverse Cardiovascular Events (MACE), defined as CV death, nonfatal MI, and nonfatal stroke. These events included in the endpoint were adjudicated by a blinded, independent committee. Overall, a small number of MACE occurred in the trials included in the meta-analysis, as described in Table 13. These events occurred primarily in patients with known CV disease.
Table 13. Number of MACE cases, Hazard Ratio and Rate Difference in a Meta-Analysis of 15 Clinical Trials Comparing Varenicline tablets to Placebo*
|
Varenicline tablets
|
Placebo N=2812
|
|
|
MACE cases, n (%)
|
13 (0.31%) |
6 (0.21%) |
|
Patient-years of exposure |
1316 |
839 |
|
Hazard Ratio (95% CI)
|
||
|
1.95 (0.79, 4.82) |
||
|
Rate Difference per 1,000 patient-years (95% CI)
|
||
|
6.30 (-2.40, 15.10) |
*Includes MACE occurring up to 30 days post-treatment.
The meta-analysis showed that exposure to varenicline tablets resulted in a hazard ratio for MACE of 1.95 (95% confidence interval from 0.79 to 4.82) for patients up to 30 days after treatment; this is equivalent to an estimated increase of 6.3 MACE events per 1,000 patient-years of exposure. The meta-analysis showed higher rates of CV endpoints in patients on varenicline tablets relative to placebo across different time frames and pre-specified sensitivity analyses, including various study groupings and CV outcomes. Although these findings were not statistically significant they were consistent. Because the number of events was small overall, the power for finding a statistically significant difference in a signal of this magnitude is low.
Additionally, a cardiovascular endpoint analysis was added to the postmarketing neuropsychiatric safety outcome study along with a non-treatment extension, [see Warnings and Precautions (5.5), Adverse Reactions (6.1), Clinical Studies (14.10)].
Structured Label Content
Section 42229-5 (42229-5)
Patients with Impaired Renal Function
No dosage adjustment is necessary for patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance less than 30 mL per min), the recommended starting dose of varenicline tablets is 0.5 mg once daily. The dose may then be titrated as needed to a maximum dose of 0.5 mg twice daily. For patients with end-stage renal disease undergoing hemodialysis, a maximum dose of 0.5 mg once daily may be administered if tolerated [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
Section 42231-1 (42231-1)
|
MEDICATION GUIDE
|
|
|
What is the most important information I should know about varenicline tablets?
|
|
|
What is varenicline tablets?
|
|
|
Who should not take varenicline tablets?
|
|
|
How should I take varenicline tablets?
|
|
|
Weeks 1 through 4 |
Reduce your smoking to reach one-half of your starting daily number of cigarettes. |
|
Weeks 5 through 8 |
Reduce your smoking to reach one-quarter of your starting daily number of cigarettes. |
|
Weeks 9 through 12 |
Keep reducing your smoking until you are no longer smoking (you reach zero cigarettes each day). |
|
Aim to quit by the end of the 12th week of treatment, or sooner if you feel ready. Continue to take varenicline tablets for another 12 weeks, for a total of 24 weeks of treatment. |
|
|
Day 1 to Day 3 |
o White tablet (0.5 mg) |
|
Day 4 to Day 7 |
o White tablet (0.5 mg) |
|
Day 8 to end of treatment |
o Pink tablet (1 mg) |
|
|
|
What should I avoid while taking varenicline tablets?
|
|
|
What are the possible side effects of varenicline tablets?
|
|
|
How should I store varenicline tablets?
|
|
|
General information about the safe and effective use of varenicline tablets.
|
|
|
What are the ingredients in varenicline tablets?
Distributed by: |
Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: August 2025
5.9 Nausea
Nausea was the most common adverse reaction reported with varenicline tablets treatment. Nausea was generally described as mild or moderate and often transient; however, for some patients, it was persistent over several months. The incidence of nausea was dose-dependent. Initial dose-titration was beneficial in reducing the occurrence of nausea. For patients treated to the maximum recommended dose of 1 mg twice daily following initial dosage titration, the incidence of nausea was 30% compared with 10% in patients taking a comparable placebo regimen. In patients taking varenicline tablets 0.5 mg twice daily following initial titration, the incidence was 16% compared with 11% for placebo. Approximately 3% of patients treated with varenicline tablets 1 mg twice daily in studies involving 12 weeks of treatment discontinued treatment prematurely because of nausea. For patients with intolerable nausea, a dose reduction should be considered.
5.2 Seizures
During clinical trials and the postmarketing experience, there have been reports of seizures in patients treated with varenicline tablets. Some patients had no history of seizures, whereas others had a history of seizure disorder that was remote or well-controlled. In most cases, the seizure occurred within the first month of therapy. Weigh this potential risk against the potential benefits before prescribing varenicline tablets in patients with a history of seizures or other factors that can lower the seizure threshold. Advise patients to discontinue varenicline tablets and contact a healthcare provider immediately if they experience a seizure while on treatment [see Adverse Reactions (6.2)].
10 Overdosage (10 OVERDOSAGE)
In case of overdose, standard supportive measures should be instituted as required.
Varenicline has been shown to be dialyzed in patients with end-stage renal disease [see Clinical Pharmacology (12.3)], however, there is no experience in dialysis following overdose.
11 Description (11 DESCRIPTION)
Varenicline tablets contain varenicline (as the tartrate salt), which is a partial nicotinic agonist selective for α4β2 nicotinic acetylcholine receptor subtypes.
Varenicline, as the tartrate salt, is a powder which is white to off white to slightly yellow solid with the following chemical name: 7,8,9,10-tetrahydro-6,10-methano-6H-pyrazino[2,3- h][3]benzazepine, (2R,3R)-2,3-dihydroxybutanedioate (1:1). It is freely soluble in water and very slightly soluble in methanol. Varenicline tartrate has a molecular weight of 361.35 Daltons, and a molecular formula of C13H13N3 • C4H6O6. The chemical structure is:
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg white to off-white, modified capsule shaped, film-coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg light pink to pink color, modified capsule shaped, film coated tablets debossed with 'L' on one side and '12' on other side. Each 0.5 mg varenicline tablets contains 0.85 mg of varenicline tartrate equivalent to 0.5 mg of varenicline free base; each 1 mg varenicline tablets contains 1.71 mg of varenicline tartrate equivalent to 1 mg of varenicline free base. The following inactive ingredients are included in the tablets: anhydrous dibasic calcium phosphate, butylated hydroxyanisole, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, maltodextrin, microcrystalline cellulose. The tablet film coating contains hydroxypropyl cellulose, hypromellose, and titanium dioxide. In addition to these, the 1 mg tablet film coating also includes D&C Red #27/phloxine aluminum lake, D&C yellow #10 aluminum lake, and FD&C blue #1/brilliant blue FCF aluminum lake and talc.
5.6 Somnambulism
Cases of somnambulism have been reported in patients taking varenicline tablets. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue varenicline tablets and notify their healthcare provider if they experience somnambulism [see Adverse Reactions (6.2)].
8.4 Pediatric Use
Varenicline tablets are not recommended for use in pediatric patients 16 years of age or younger because its efficacy in this population was not demonstrated.
Single and multiple-dose pharmacokinetics of varenicline have been investigated in pediatric patients aged 12 to 17 years old (inclusive) and were approximately dose-proportional over the 0.5 mg to 2 mg daily dose range studied. Steady-state systemic exposure in adolescent patients of bodyweight >55 kg, as assessed by AUC (0-24), was comparable to that noted for the same doses in the adult population. When 0.5 mg BID was given, steady-state daily exposure of varenicline was, on average, higher (by approximately 40%) in adolescent patients with bodyweight ≤55 kg compared to that noted in the adult population.
The efficacy and safety of varenicline was evaluated in a randomized, double-blind, placebo-controlled study of 312 patients aged 12 to 19 years, who smoked an average of at least 5 cigarettes per day during the 30 days prior to recruitment, had a score of at least 4 on the Fagerstrom Test for Nicotine Dependence scale, and at least one previous failed quit attempt. Patients were stratified by age (12 to 16 years of age, n = 216 and 17 to 19 years of age, n = 96) and by body weight (≤55 kg and >55 kg). Patients were randomized to one of two doses of varenicline, adjusted by weight to provide plasma levels in the efficacious range (based on adult studies) and placebo. Patients received treatment for 12 weeks, followed by a non-treatment period of 40 weeks, along with age-appropriate counseling throughout the study. Results from this study showed that varenicline, at either dose studied, did not improve continuous abstinence rates at weeks 9 through 12 of treatment compared with placebo in subjects 12 to 19 years of age. The varenicline safety profile in this study was consistent with that observed in adult studies.
8.5 Geriatric Use
A combined single- and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once daily or twice daily to 16 healthy elderly male and female smokers (aged 65–75 years) for 7 consecutive days was similar to that of younger subjects. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.2)].
No dosage adjustment is recommended for elderly patients.
14.2 Urge to Smoke
Based on responses to the Brief Questionnaire of Smoking Urges and the Minnesota Nicotine Withdrawal scale "urge to smoke" item, varenicline tablets reduced urge to smoke compared to placebo.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of varenicline tablets in smoking cessation was demonstrated in six clinical trials in which a total of 3659 chronic cigarette smokers (≥10 cigarettes per day) were treated with varenicline tablets. In all clinical studies, abstinence from smoking was determined by patient self-report and verified by measurement of exhaled carbon monoxide (CO≤10 ppm) at weekly visits. Among the varenicline tablets-treated patients enrolled in these studies, the completion rate was 65%. Except for the dose-ranging study (Study 1) and the maintenance of abstinence study (Study 6), patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. Most patients enrolled in these trials were white (79-96%). All studies enrolled almost equal numbers of men and women. The average age of patients in these studies was 43 years. Patients on average had smoked about 21 cigarettes per day for an average of approximately 25 years. Patients set a date to stop smoking (target quit date) with dosing starting 1 week before this date.
Seven additional studies evaluated the efficacy of varenicline tablets in patients with cardiovascular disease, in patients with chronic obstructive pulmonary disease [see Clinical Studies (14.7)], in patients instructed to select their quit date within days 8 and 35 of treatment [see Clinical Studies (14.4)], patients with major depressive disorder [see Clinical Studies (14.9)], patients who had made a previous attempt to quit smoking with varenicline tablets, and either did not succeed in quitting or relapsed after treatment [see Clinical Studies (14.6)], in patients without or with a history of psychiatric disorder enrolled in a postmarketing neuropsychiatric safety outcome trial [see Warnings and Precautions (5.1), Clinical Studies (14.10) ], and in patients who were not able or willing to quit abruptly and were instructed to quit gradually [see Clinical studies (14.5)].
In all studies, patients were provided with an educational booklet on smoking cessation and received up to 10 minutes of smoking cessation counseling at each weekly treatment visit according to Agency for Healthcare Research and Quality guidelines.
4 Contraindications (4 CONTRAINDICATIONS)
Varenicline tablets are contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to varenicline tablets.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling:
-
•Neuropsychiatric Adverse Events including Suicidality [see Warnings and Precautions (5.1)]
-
•Seizures [see Warnings and Precautions (5.2)]
-
•Interaction with Alcohol [see Warnings and Precautions (5.3)]
-
•Accidental Injury [see Warnings and Precautions (5.4)]
-
•Cardiovascular Events [see Warnings and Precautions (5.5)]
-
•Somnambulism [see Warnings and Precautions (5.6)]
-
•Angioedema and Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
-
•Serious Skin Reactions [see Warnings and Precautions (5.8)]
In the placebo-controlled premarketing studies, the most common adverse events associated with varenicline tablets (>5% and twice the rate seen in placebo-treated patients) were nausea, abnormal (vivid, unusual, or strange) dreams, constipation, flatulence, and vomiting.
The treatment discontinuation rate due to adverse events in patients dosed with 1 mg twice daily was 12% for varenicline tablets, compared to 10% for placebo in studies of three months' treatment. In this group, the discontinuation rates that are higher than placebo for the most common adverse events in varenicline tablets-treated patients were as follows: nausea (3% vs. 0.5% for placebo), insomnia (1.2% vs. 1.1% for placebo), and abnormal dreams (0.3% vs. 0.2% for placebo).
Smoking cessation, with or without treatment, is associated with nicotine withdrawal symptoms and has also been associated with the exacerbation of underlying psychiatric illness.
7 Drug Interactions (7 DRUG INTERACTIONS)
Based on varenicline characteristics and clinical experience to date, varenicline tablets has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Varenicline is substantially eliminated by renal glomerular filtration along with active tubular secretion. Dose reduction is not required in patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30 mL/min), and for patients with end-stage renal disease undergoing hemodialysis, dosage adjustment is needed [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
5.4 Accidental Injury
There have been postmarketing reports of traffic accidents, near-miss incidents in traffic, or other accidental injuries in patients taking varenicline tablets. In some cases, the patients reported somnolence, dizziness, loss of consciousness or difficulty concentrating that resulted in impairment, or concern about potential impairment, in driving or operating machinery. Advise patients to use caution driving or operating machinery or engaging in other potentially hazardous activities until they know how varenicline tablets may affect them.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Varenicline tablets are indicated for use as an aid to smoking cessation treatment.
14.6 Re Treatment Study (14.6 Re-Treatment Study)
Varenicline tablets was evaluated in a double-blind, placebo-controlled trial of patients who had made a previous attempt to quit smoking with varenicline tablets, and either did not succeed in quitting or relapsed after treatment. Subjects were randomized 1:1 to varenicline tablets 1 mg twice daily (N=249) or placebo (N=245) for 12 weeks of treatment and followed for 40 weeks post-treatment. Patients included in this study had taken varenicline tablets for a smoking-cessation attempt in the past (for a total treatment duration of a minimum of two weeks), at least three months prior to study entry, and had been smoking for at least four weeks.
Patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (45%) compared to patients treated with placebo (12%) and from weeks 9 through 52 (20%) compared to subjects treated with placebo (3%).
Table 9. Continuous Abstinence (95% confidence interval), Re-Treatment Study
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
Retreatment Study |
45% |
12% |
20% |
3% |
BID = twice daily
12.1 Mechanism of Action
Varenicline binds with high affinity and selectivity at α4β2 neuronal nicotinic acetylcholine receptors. The efficacy of varenicline tablets in smoking cessation is believed to be the result of varenicline's activity at α4β2 sub-type of the nicotinic receptor where its binding produces agonist activity, while simultaneously preventing nicotine binding to these receptors.
Electrophysiology studies in vitro and neurochemical studies in vivo have shown that varenicline binds to α4β2 neuronal nicotinic acetylcholine receptors and stimulates receptor-mediated activity, but at a significantly lower level than nicotine. Varenicline blocks the ability of nicotine to activate α4β2 receptors and thus to stimulate the central nervous mesolimbic dopamine system, believed to be the neuronal mechanism underlying reinforcement and reward experienced upon smoking. Varenicline is highly selective and binds more potently to α4β2 receptors than to other common nicotinic receptors (>500-fold α3β4, >3,500-fold α7, >20,000-fold α1βγδ), or to nonnicotinic receptors and transporters (>2,000-fold). Varenicline also binds with moderate affinity (Ki = 350 nM) to the 5-HT3 receptor.
9.1 Controlled Substance
Varenicline is not a controlled substance.
14.3 Long Term Abstinence (14.3 Long-Term Abstinence)
Studies 1 through 5 included 40 weeks of post-treatment follow-up. In each study, varenicline tablets-treated patients were more likely to maintain abstinence throughout the follow-up period than were patients treated with placebo (Figure 2, Table 8).
Table 8. Continuous Abstinence, Weeks 9 through 52 (95% confidence interval) Across Different Studies
|
Varenicline tablets
|
Varenicline tablets
|
Varenicline tablets
|
Bupropion SR
|
Placebo
|
|
|
Study 2 |
19% |
23% |
4% |
||
|
Study 3 |
22% |
8% |
|||
|
Study 4 |
21% |
16% |
8% |
||
|
Study 5 |
22% |
14% |
10% |
BID = twice daily
5.5 Cardiovascular Events
A comprehensive evaluation of cardiovascular (CV) risk with varenicline tablets suggests that patients with underlying CV disease may be at increased risk; however, these concerns must be balanced with the health benefits of smoking cessation. CV risk has been assessed for varenicline tablets in randomized controlled trials (RCT) and meta-analyses of RCTs. In a smoking cessation trial in patients with stable CV disease, CV events were infrequent overall; however, nonfatal myocardial infarction (MI) and nonfatal stroke occurred more frequently in patients treated with varenicline tablets compared to placebo. All-cause and CV mortality was lower in patients treated with varenicline tablets [see Clinical Studies (14.8)]. This study was included in a meta-analysis of 15 varenicline tablets efficacy trials in various clinical populations that showed an increased hazard ratio for Major Adverse Cardiovascular Events (MACE) of 1.95; however, the finding was not statistically significant (95% CI: 0.79, 4.82). In the large postmarketing neuropsychiatric safety outcome trial, an analysis of adjudicated MACE events was conducted for patients while in the trial and during a 28-week non-treatment extension period. Few MACE events occurred during the trial; therefore, the findings did not contribute substantively to the understanding of CV risk with varenicline tablets. Instruct patients to notify their healthcare providers of new or worsening CV symptoms and to seek immediate medical attention if they experience signs and symptoms of MI or stroke [see Clinical Studies (14.10)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Neuropsychiatric Adverse Events : Postmarketing reports of serious or clinically significant neuropsychiatric adverse events have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Observe patients attempting to quit smoking with varenicline tablets for the occurrence of such symptoms and instruct them to discontinue varenicline tablets and contact a healthcare provider if they experience such adverse events. (5.1)
-
•Seizures : New or worsening seizures have been observed in patients taking varenicline tablets. Varenicline tablets should be used cautiously in patients with a history of seizures or other factors that can lower the seizure threshold. (5.2)
-
•Interaction with Alcohol : Increased effects of alcohol have been reported. Instruct patients to reduce the amount of alcohol they consume until they know whether varenicline tablets affects them. (5.3)
-
•Accidental Injury : Accidental injuries (e.g., traffic accidents) have been reported. Instruct patients to use caution driving or operating machinery until they know how varenicline tablets may affect them. (5.4)
-
•Cardiovascular Events : Patients with underlying cardiovascular (CV) disease may be at increased risk of CV events; however, these concerns must be balanced with the health benefits of smoking cessation. Instruct patients to notify their healthcare providers of new or worsening CV symptoms and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction (MI) or stroke. (5.5 and 6.1)
-
•Somnambulism : Cases of somnambulism have been reported in patients taking varenicline tablets. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue varenicline tablets and notify their healthcare provider if they experience somnambulism. (5.6 and 6.2)
-
•Angioedema and Hypersensitivity Reactions : Such reactions, including angioedema, infrequently life-threatening, have been reported. Instruct patients to discontinue varenicline tablets and immediately seek medical care if symptoms occur. (5.7 and 6.2)
-
•Serious Skin Reactions : Rare, potentially life-threatening skin reactions have been reported. Instruct patients to discontinue varenicline tablets and contact a healthcare provider immediately at first appearance of skin rash with mucosal lesions. (5.8 and 6.2)
-
•Nausea : Nausea is the most common adverse reaction (up to 30% incidence rate). Dose reduction may be helpful. (5.9)
5.8 Serious Skin Reactions
There have been postmarketing reports of rare but serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients using varenicline tablets [see Adverse Reactions (6.2)]. As these skin reactions can be life-threatening, instruct patients to stop taking varenicline tablets and contact a healthcare provider immediately at the first appearance of a skin rash with mucosal lesions or any other signs of hypersensitivity.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Begin varenicline tablets dosing one week before the date set by the patient to stop smoking. Alternatively, the patient can begin varenicline tablets dosing and then quit smoking between days 8 and 35 of treatment. (2.1)
-
•Starting Week: 0.5 mg once daily on days 1-3 and 0.5 mg twice daily on days 4-7. (2.1)
-
•Continuing Weeks: 1 mg twice daily for a total of 12 weeks. (2.1)
-
•An additional 12 weeks of treatment is recommended for successful quitters to increase likelihood of long-term abstinence. (2.1)
-
•Consider a gradual approach to quitting smoking with varenicline tablets for patients who are sure that they are not able or willing to quit abruptly. Patients should begin varenicline tablets dosing and reduce smoking by 50% from baseline within the first four weeks, by an additional 50% in the next four weeks, and continue reducing with the goal of reaching complete abstinence by 12 weeks. Continue treatment for an additional 12 weeks, for a total of 24 weeks. (2.1)
-
•Severe Renal Impairment (estimated creatinine clearance less than 30 mL/min): Begin with 0.5 mg once daily and titrate to 0.5 mg twice daily. For patients with end-stage renal disease undergoing hemodialysis, a maximum of 0.5 mg daily may be given if tolerated. (2.2)
-
•Consider dose reduction for patients who cannot tolerate adverse effects. (2.1)
-
•Another attempt at treatment is recommended for those who fail to stop smoking or relapse when factors contributing to the failed attempt have been addressed. (2.1)
-
•Provide patients with appropriate educational materials and counseling to support the quit attempt. (2.1)
2.1 Usual Dosage for Adults
Smoking cessation therapies are more likely to succeed for patients who are motivated to stop smoking and who are provided additional advice and support. Provide patients with appropriate educational materials and counseling to support the quit attempt.
The patient should set a date to stop smoking. Begin varenicline tablets dosing one week before this date. Alternatively, the patient can begin varenicline tablets dosing and then quit smoking between days 8 and 35 of treatment.
Varenicline tablets should be taken orally after eating and with a full glass of water.
The recommended dose of varenicline tablets is 1 mg twice daily following a 1-week titration as follows:
|
Days 1 – 3: |
0.5 mg once daily |
|
Days 4 – 7: |
0.5 mg twice daily |
|
Day 8 – end of treatment: |
1 mg twice daily |
Patients should be treated with varenicline tablets for 12 weeks. For patients who have successfully stopped smoking at the end of 12 weeks, an additional course of 12 weeks treatment with varenicline tablets is recommended to further increase the likelihood of long-term abstinence.
For patients who are sure that they are not able or willing to quit abruptly, consider a gradual approach to quitting smoking with varenicline tablets. Patients should begin varenicline tablets dosing and reduce smoking by 50% from baseline within the first four weeks, by an additional 50% in the next four weeks, and continue reducing with the goal of reaching complete abstinence by 12 weeks. Continue varenicline tablets treatment for an additional 12 weeks, for a total of 24 weeks of treatment. Encourage patients to attempt quitting sooner if they feel ready [see Clinical Studies (14.5)].
Patients who are motivated to quit, and who did not succeed in stopping smoking during prior varenicline tablets therapy for reasons other than intolerability due to adverse events or who relapsed after treatment, should be encouraged to make another attempt with varenicline tablets once factors contributing to the failed attempt have been identified and addressed.
Consider a temporary or permanent dose reduction in patients who cannot tolerate the adverse effects of varenicline tablets.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Modified capsule shaped tablets: 0.5 mg (white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side) and 1 mg (light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side).
5.3 Interaction With Alcohol (5.3 Interaction with Alcohol)
There have been postmarketing reports of patients experiencing increased intoxicating effects of alcohol while taking varenicline tablets. Some cases described unusual and sometimes aggressive behavior, and were often accompanied by amnesia for the events. Advise patients to reduce the amount of alcohol they consume while taking varenicline tablets until they know whether varenicline tablets affects their tolerance for alcohol [see Adverse Reactions (6.2)].
6.2 Postmarketing Experience
The following adverse events have been reported during post-approval use of varenicline tablets. Because these events are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
There have been reports of depression, mania, psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide in patients attempting to quit smoking while taking varenicline tablets [see Warnings and Precautions (5.1)].
There have been postmarketing reports of new or worsening seizures in patients treated with varenicline tablets [see Warnings and Precautions (5.2)].
There have been postmarketing reports of patients experiencing increased intoxicating effects of alcohol while taking varenicline tablets. Some reported neuropsychiatric events, including unusual and sometimes aggressive behavior [see Warnings and Precautions (5.1) and (5.3)].
There have been reports of hypersensitivity reactions, including angioedema [see Warnings and Precautions (5.7)].
There have also been reports of serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients taking varenicline tablets [see Warnings and Precautions (5.8)].
There have been reports of myocardial infarction (MI) and cerebrovascular accident (CVA) including ischemic and hemorrhagic events in patients taking varenicline tablets. In the majority of the reported cases, patients had pre-existing cardiovascular disease and/or other risk factors. Although smoking is a risk factor for MI and CVA, based on temporal relationship between medication use and events, a contributory role of varenicline cannot be ruled out [see Warnings and Precautions (5.5)].
There have been reports of hyperglycemia in patients following initiation of varenicline tablets.
There have been reports of somnambulism, some resulting in harmful behavior to self, others, or property in patients treated with varenicline tablets [see Warnings and Precautions (5.6)].
14.1 Initiation of Abstinence
Study 1
This was a six-week dose-ranging study comparing varenicline tablets to placebo. This study provided initial evidence that varenicline tablets at a total dose of 1 mg per day or 2 mg per day was effective as an aid to smoking cessation.
Study 2
This study of 627 patients compared varenicline tablets 1 mg per day and 2 mg per day with placebo. Patients were treated for 12 weeks (including one-week titration) and then were followed for 40 weeks post-treatment. Varenicline tablets was given in two divided doses daily. Each dose of varenicline tablets was given in two different regimens, with and without initial dose-titration, to explore the effect of different dosing regimens on tolerability. For the titrated groups, dosage was titrated up over the course of one week, with full dosage achieved starting with the second week of dosing. The titrated and nontitrated groups were pooled for efficacy analysis.
Forty-five percent of patients receiving varenicline tablets 1 mg per day (0.5 mg twice daily) and 51% of patients receiving 2 mg per day (1 mg twice daily) had CO-confirmed continuous abstinence during weeks 9 through 12 compared to 12% of patients in the placebo group (Figure 1). In addition, 31% of the 1 mg per day group and 31% of the 2 mg per day group were continuously abstinent from one week after TQD through the end of treatment as compared to 8% of the placebo group.
Study 3
This flexible-dosing study of 312 patients examined the effect of a patient-directed dosing strategy of varenicline tablets or placebo. After an initial one-week titration to a dose of 0.5 mg twice daily, patients could adjust their dosage as often as they wished between 0.5 mg once daily to 1 mg twice daily per day. Sixty-nine percent of patients titrated to the maximum allowable dose at any time during the study. For 44% of patients, the modal dose selected was 1 mg twice daily; for slightly over half of the study participants, the modal dose selected was 1 mg/day or less.
Of the patients treated with varenicline tablets, 40% had CO-confirmed continuous abstinence during weeks 9 through 12 compared to 12% in the placebo group. In addition, 29% of the varenicline tablets group were continuously abstinent from one week after TQD through the end of treatment as compared to 9% of the placebo group.
Study 4 and Study 5
These identical double-blind studies compared varenicline tablets 2 mg per day, bupropion sustained-release (SR) 150 mg twice daily, and placebo. Patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. The varenicline tablets dosage of 1 mg twice daily was achieved using a titration of 0.5 mg once daily for the initial 3 days followed by 0.5 mg twice daily for the next 4 days. The bupropion SR dosage of 150 mg twice daily was achieved using a 3-day titration of 150 mg once daily. Study 4 enrolled 1022 patients and Study 5 enrolled 1023 patients. Patients inappropriate for bupropion treatment or patients who had previously used bupropion were excluded.
In Study 4, patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (44%) compared to patients treated with bupropion SR (30%) or placebo (17%). The bupropion SR quit rate was also superior to placebo. In addition, 29% of the varenicline tablets group were continuously abstinent from one week after TQD through the end of treatment as compared to 12% of the placebo group and 23% of the bupropion SR group.
Similarly in Study 5, patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (44%) compared to patients treated with bupropion SR (30%) or placebo (18%). The bupropion SR quit rate was also superior to placebo. In addition, 29% of the varenicline tablets group were continuously abstinent from one week after TQD through the end of treatment as compared to 11% of the placebo group and 21% of the bupropion SR group.
Table 7. Continuous Abstinence, Weeks 9 through 12 (95% confidence interval)
|
Varenicline tablets
|
Varenicline tablets
|
Varenicline tablets Flexible
|
Bupropion SR
|
Placebo
|
|
|
Study 2 |
45% |
51% |
12% |
||
|
Study 3 |
40% |
12% |
|||
|
Study 4 |
44% |
30% |
17% |
||
|
Study 5 |
44% |
30% |
18% |
BID = twice daily
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reactions rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During the premarketing development of varenicline tablets, over 4500 subjects were exposed to varenicline tablets, with over 450 treated for at least 24 weeks and approximately 100 for a year. Most study participants were treated for 12 weeks or less.
The most common adverse event associated with varenicline tablets treatment is nausea, occurring in 30% of patients treated at the recommended dose, compared with 10% in patients taking a comparable placebo regimen [see Warnings and Precautions (5.9)].
Table 1 shows the adverse events for varenicline tablets and placebo in the 12- week fixed dose premarketing studies with titration in the first week [Studies 2 (titrated arm only), 4, and 5]. Adverse events were categorized using the Medical Dictionary for Regulatory Activities (MedDRA, Version 7.1).
MedDRA High Level Group Terms (HLGT) reported in ≥5% of patients in the varenicline tablets 1 mg twice daily dose group, and more commonly than in the placebo group, are listed, along with subordinate Preferred Terms (PT) reported in ≥1% of varenicline tablets patients (and at least 0.5% more frequent than placebo). Closely related Preferred Terms such as 'Insomnia', 'Initial insomnia', 'Middle insomnia', 'Early morning awakening' were grouped, but individual patients reporting two or more grouped events are only counted once.
Table 1. Common Treatment Emergent AEs (%) in the Fixed-Dose, Placebo-Controlled Studies (HLGTs >5% of Patients in the 1 mg BID Varenicline Tablets Group and More Commonly than Placebo and PT ≥1% in the 1 mg BID Varenicline Tablets Group, and 1 mg BID Varenicline Tablets at Least 0.5% More than Placebo)
|
SYSTEM ORGAN CLASS
|
Varenicline Tablets
|
Varenicline Tablets
|
Placebo
|
|
GASTROINTESTINAL (GI) |
|||
|
GI Signs and Symptoms |
|||
|
Nausea |
16 |
30 |
10 |
|
Abdominal Pain * |
5 |
7 |
5 |
|
Flatulence |
9 |
6 |
3 |
|
Dyspepsia |
5 |
5 |
3 |
|
Vomiting |
1 |
5 |
2 |
|
GI Motility/Defecation |
|||
|
Constipation |
5 |
8 |
3 |
|
Gastroesophageal reflux |
1 |
1 |
0 |
|
Salivary Gland Conditions |
|||
|
Dry mouth |
4 |
6 |
4 |
|
PSYCHIATRIC DISORDERS |
|||
|
Sleep Disorder/Disturbances |
|||
|
Insomnia ** |
19 |
18 |
13 |
|
Abnormal dreams |
9 |
13 |
5 |
|
Sleep disorder |
2 |
5 |
3 |
|
Nightmare |
2 |
1 |
0 |
|
NERVOUS SYSTEM |
|||
|
Headaches |
|||
|
Headache |
19 |
15 |
13 |
|
Neurological Disorders |
|||
|
NEC |
|||
|
Dysgeusia |
8 |
5 |
4 |
|
Somnolence |
3 |
3 |
2 |
|
Lethargy |
2 |
1 |
0 |
|
GENERAL DISORDERS |
|||
|
General Disorders NEC |
|||
|
Fatigue/Malaise/Asthenia |
4 |
7 |
6 |
|
RESPIR/THORACIC/MEDIAST |
|||
|
Respiratory Disorders NEC |
|||
|
Rhinorrhea |
0 |
1 |
0 |
|
Dyspnea |
2 |
1 |
1 |
|
Upper Respiratory Tract |
7 |
5 |
4 |
|
SKIN/SUBCUTANEOUS TISSUE |
|||
|
Epidermal and Dermal |
|||
|
Rash |
1 |
3 |
2 |
|
Pruritis |
0 |
1 |
1 |
|
METABOLISM and NUTRITION |
|||
|
Appetite/General Nutrition |
|||
|
Increased appetite |
4 |
3 |
2 |
|
Decreased appetite/ |
1 |
2 |
1 |
* Includes PTs Abdominal (pain, pain upper, pain lower, discomfort, tenderness, distension) and Stomach discomfort
** Includes PTs Insomnia/Initial insomnia/Middle insomnia/Early morning awakening
The overall pattern and frequency of adverse events during the longer-term premarketing trials was similar to those described in Table 1, though several of the most common events were reported by a greater proportion of patients with long-term use (e.g., nausea was reported in 40% of patients treated with varenicline tablets 1 mg twice daily in a one year study, compared to 8% of placebo-treated patients).
Following is a list of treatment-emergent adverse events reported by patients treated with varenicline tablets during all premarketing clinical trials and updated based on pooled data from 18 placebo-controlled pre- and postmarketing studies, including approximately 5,000 patients treated with varenicline. Adverse events were categorized using MedDRA, Version 16.0. The listing does not include those events already listed in the previous tables or elsewhere in labeling, those events for which a drug cause was remote, those events which were so general as to be uninformative, and those events reported only once which did not have a substantial probability of being acutely life-threatening.
Blood and Lymphatic System Disorders. Infrequent: anemia, lymphadenopathy. Rare: leukocytosis, splenomegaly, thrombocytopenia.
Cardiac Disorders. Infrequent: angina pectoris, myocardial infarction, palpitations, tachycardia. Rare: acute coronary syndrome, arrhythmia, atrial fibrillation, bradycardia, cardiac flutter, cor pulmonale, coronary artery disease, ventricular extrasystoles.
Ear and Labyrinth Disorders. Infrequent: tinnitus, vertigo. Rare: deafness, Meniere's disease.
Endocrine Disorders. Infrequent: thyroid gland disorders.
Eye Disorders. Infrequent: conjunctivitis, eye irritation, eye pain, vision blurred, visual impairment. Rare: blindness transient, cataract subcapsular, dry eye, night blindness, ocular vascular disorder, photophobia, vitreous floaters.
Gastrointestinal Disorders. Frequent: diarrhea, toothache. Infrequent: dysphagia, eructation, gastritis, gastrointestinal hemorrhage, mouth ulceration. Rare: enterocolitis, esophagitis, gastric ulcer, intestinal obstruction, pancreatitis acute.
General Disorders and Administration Site Conditions. Frequent: chest pain. Infrequent: chest discomfort, chills, edema, influenza-like illness, pyrexia.
Hepatobiliary Disorders. Rare: gall bladder disorder.
Investigations. Frequent: liver function test abnormal, weight increased. Infrequent: electrocardiogram abnormal. Rare: muscle enzyme increased, urine analysis abnormal.
Metabolism and Nutrition Disorders. Infrequent: diabetes mellitus, hypoglycemia. Rare: hyperlipidemia, hypokalemia.
Musculoskeletal and Connective Tissue Disorders. Frequent: arthralgia, back pain, myalgia. Infrequent: arthritis, muscle cramp, musculoskeletal pain. Rare: myositis, osteoporosis.
Nervous System Disorders. Frequent: disturbance in attention, dizziness. Infrequent: amnesia, convulsion, migraine, parosmia, syncope, tremor. Rare: balance disorder, cerebrovascular accident, dysarthria, mental impairment, multiple sclerosis, VIIth nerve paralysis, nystagmus, psychomotor hyperactivity, psychomotor skills impaired, restless legs syndrome, sensory disturbance, transient ischemic attack, visual field defect.
Psychiatric Disorders. Infrequent: dissociation, libido decreased, mood swings, thinking abnormal. Rare: bradyphrenia, disorientation, euphoric mood.
Renal and Urinary Disorders. Infrequent: nocturia, pollakiuria, urine abnormality. Rare: nephrolithiasis, polyuria, renal failure acute, urethral syndrome, urinary retention.
Reproductive System and Breast Disorders. Frequent: menstrual disorder. Infrequent: erectile dysfunction. Rare: sexual dysfunction.
Respiratory, Thoracic and Mediastinal Disorders. Frequent: respiratory disorders. Infrequent: asthma, epistaxis, rhinitis allergic, upper respiratory tract inflammation.
Rare: pleurisy, pulmonary embolism.
Skin and Subcutaneous Tissue Disorders. Infrequent: acne, dry skin, eczema, erythema, hyperhidrosis, urticaria. Rare: photosensitivity reaction, psoriasis.
Vascular Disorders. Infrequent: hot flush. Rare: thrombosis.
Varenicline tablets has also been studied in postmarketing trials including (1) a trial conducted in patients with chronic obstructive pulmonary disease (COPD), (2) a trial conducted in generally healthy patients (similar to those in the premarketing studies) in which they were allowed to select a quit date between days 8 and 35 of treatment ("alternative quit date instruction trial"), (3) a trial conducted in patients who did not succeed in stopping smoking during prior varenicline tablets therapy, or who relapsed after treatment ("re-treatment trial"), (4) a trial conducted in patients with stable cardiovascular disease, (5) a trial conducted in patients with stable schizophrenia or schizoaffective disorder, (6) a trial conducted in patients with major depressive disorder, (7) a postmarketing neuropsychiatric safety outcome trial in patients without or with a history of psychiatric disorder, (8) a non-treatment extension of the postmarketing neuropsychiatric safety outcome trial that assessed CV safety, (9) a trial in patients who were not able or willing to quit abruptly and who were instructed to quit gradually ("gradual approach to quitting smoking trial").
Adverse events in the trial of patients with COPD (1), in the alternative quit date instruction trial (2), and in the gradual approach to quitting smoking trial (9) were similar to those observed in premarketing studies. In the re-treatment trial (3), the profile of common adverse events was similar to that previously reported, but, in addition, varenicline-treated patients also commonly reported diarrhea (6% vs. 4% in placebo-treated patients), depressed mood disorders and disturbances (6% vs. 1%), and other mood disorders and disturbances (5% vs. 2%).
In the trial of patients with stable cardiovascular disease (4), more types and a greater number of cardiovascular events were reported compared to premarketing studies, as shown in Table 1 and in Table 2 below.
Table 2. Cardiovascular Mortality and Nonfatal Cardiovascular Events (%) with a Frequency >1% in Either Treatment Group in the Trial of Patients with Stable Cardiovascular Disease
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥1% in either treatment group
|
||
|
Up to 30 days after treatment
|
||
|
Angina pectoris |
3.7 |
2.0 |
|
Chest pain |
2.5 |
2.3 |
|
Peripheral edema |
2.0 |
1.1 |
|
Hypertension |
1.4 |
2.6 |
|
Palpitations |
0.6 |
1.1 |
|
Adjudicated Cardiovascular Mortality (up to 52 weeks)
|
0.3 |
0.6 |
|
Adjudicated Nonfatal Serious Cardiovascular Events ≥1% in either treatment group
|
||
|
Up to 30 days after treatment
|
||
|
Nonfatal MI |
1.1 |
0.3 |
|
Hospitalization for angina pectoris |
0.6 |
1.1 |
|
Beyond 30 days after treatment and up to 52 weeks
|
||
|
Need for coronary revascularization* |
2.0 |
0.6 |
|
Hospitalization for angina pectoris |
1.7 |
1.1 |
|
New diagnosis of peripheral vascular disease (PVD) or |
1.4 |
0.6 |
*some procedures were part of management of nonfatal MI and hospitalization for angina
In the trial of patients with stable schizophrenia or schizoaffective disorder (5), 128 smokers on antipsychotic medication were randomized 2:1 to varenicline (1 mg twice daily) or placebo for 12 weeks with 12-week non-drug follow-up. The most common treatment emergent adverse events reported in this trial are shown in Table 3 below.
Table 3. Common Treatment Emergent AEs (%) in the Trial of Patients with Stable Schizophrenia or Schizoaffective Disorder
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥10% in the varenicline group
|
||
|
Nausea |
24 |
14 |
|
Headache |
11 |
19 |
|
Vomiting |
11 |
9 |
|
Psychiatric Adverse Events ≥5% and at a higher rate than in the placebo group
|
||
|
Insomnia |
10 |
5 |
For the trial of patients with major depressive disorder (6), the most common treatment emergent adverse events reported are shown in Table 4 below. Additionally, in this trial, patients treated with varenicline were more likely than patients treated with placebo to report one of events related to hostility and aggression (3% vs. 1%).
Table 4. Common Treatment Emergent AEs (%) in the Trial of Patients with Major Depressive Disorder
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥10% in either treatment group
|
||
|
Nausea |
27 |
10 |
|
Headache |
17 |
11 |
|
Abnormal dreams |
11 |
8 |
|
Insomnia |
11 |
5 |
|
Irritability |
11 |
8 |
|
Psychiatric Adverse Events ≥2% in any treatment group and not included above
|
||
|
Depressed mood disorders and disturbances |
11 |
9 |
|
Anxiety |
7 |
9 |
|
Agitation |
7 |
4 |
|
Tension |
4 |
3 |
|
Hostility |
2 |
0.4 |
|
Restlessness |
2 |
2 |
In the trial of patients without or with a history of psychiatric disorder (7), the most common adverse events in subjects treated with varenicline were similar to those observed in premarketing studies. Most common treatment-emergent adverse events reported in this trial are shown in Table 5 below.
Table 5. Treatment Emergent Common AEs (%) in the Trial of Patients without or with a History of Psychiatric Disorder
|
Varenicline tablets
|
Placebo
|
|
|
Adverse Events ≥10% in the varenicline group
|
||
|
Entire study population, N |
1982 |
1979 |
|
Nausea |
25 |
7 |
|
Headache |
12 |
10 |
|
Psychiatric Adverse Events ≥2% in any treatment group
|
||
|
Non-psychiatric cohort, N
|
975 |
982 |
|
Abnormal dreams |
8 |
4 |
|
Agitation |
3 |
3 |
|
Anxiety |
5 |
6 |
|
Depressed mood |
3 |
3 |
|
Insomnia |
10 |
7 |
|
Irritability |
3 |
4 |
|
Sleep disorder |
3 |
2 |
|
Psychiatric cohort, N
|
1007 |
997 |
|
Abnormal dreams |
12 |
5 |
|
Agitation |
5 |
4 |
|
Anxiety |
8 |
6 |
|
Depressed mood |
5 |
5 |
|
Depression |
5 |
5 |
|
Insomnia |
9 |
7 |
|
Irritability |
5 |
7 |
|
Nervousness |
2 |
3 |
|
Sleep disorder |
3 |
2 |
In the non-treatment extension of the postmarketing neuropsychiatric safety outcomes trial that assessed CV safety (8), the most common adverse events in subjects treated with varenicline and occurring up to 30 days after last dose of treatment were similar to those observed in premarketing studies.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Medication Guide)
Initiate Treatment and Continue to Attempt to Quit if Lapse
Instruct patients to set a date to quit smoking and to initiate varenicline tablets treatment one week before the quit date. Alternatively, the patient can begin varenicline tablets dosing and then set a date to quit smoking between days 8 and 35 of treatment. Encourage patients to continue to attempt to quit if they have early lapses after quit day [see Dosage and Administration (2.1)].
For patients who are sure that they are not able or willing to quit abruptly, a gradual approach to quitting smoking with varenicline tablets may be considered. Patients should begin varenicline tablets dosing and reduce smoking during the first 12 weeks of treatment, then quit by the end of that period and continue treatment for an additional 12 weeks for a total of 24 weeks [see Dosage and Administration (2.1)].
Encourage patients who are motivated to quit and who did not succeed in stopping smoking during prior varenicline tablets therapy for reasons other than intolerability due to adverse events, or who relapsed after treatment to make another attempt with varenicline tablets once factors contributing to the failed attempt have been identified and addressed [see Dosage and Administration (2.1), Clinical Studies (14.6)].
How to Take
Advise patients that varenicline tablets should be taken orally after eating, and with a full glass of water [see Dosage and Administration (2.1)].
Starting Week Dosage
Instruct patients on how to titrate varenicline tablets, beginning at a dose of 0.5 mg/day. Explain that one 0.5 mg tablet should be taken daily for the first three days, and that for the next four days, one 0.5 mg tablet should be taken in the morning and one 0.5 mg tablet should be taken in the evening [see Dosage and Administration (2.1)].
Continuing Weeks Dosage
Advise patients that, after the first seven days, the dose should be increased to one 1 mg tablet in the morning and one 1 mg tablet in the evening [see Dosage and Administration (2.1)].
Dosage Adjustment for Varenicline Tablets or Other Drugs
Inform patients that nausea and insomnia are side effects of varenicline tablets and are usually transient; however, advise patients that if they are persistently troubled by these symptoms, they should notify the prescribing physician so that a dose reduction can be considered.
Inform patients that some drugs may require dose adjustment after quitting smoking [see Dosage and Administration (2.1)].
Counseling and Support
Provide patients with educational materials and necessary counseling to support an attempt at quitting smoking [see Dosage and Administration (2.1)].
Neuropsychiatric Adverse Events
Inform patients that some patients have experienced changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation and suicide when attempting to quit smoking while taking varenicline tablets. Instruct patients to discontinue varenicline tablets and contact a healthcare professional if they experience such symptoms [see Warnings and Precautions (5.1), Adverse Reactions (6.2)].
History of Psychiatric Illness
Encourage patients to reveal any history of psychiatric illness prior to initiating treatment.
Nicotine Withdrawal
Inform patients that quitting smoking, with or without varenicline tablets, may be associated with nicotine withdrawal symptoms (including depression or agitation) or exacerbation of pre-existing psychiatric illness.
Seizures
Encourage patients to report any history of seizures or other factors that can lower seizure threshold. Instruct patients to discontinue varenicline tablets and contact a healthcare provider immediately if they experience a seizure while on treatment [see Warnings and Precautions (5.2)].
Interaction with Alcohol
Advise patients to reduce the amount of alcohol they consume while taking varenicline tablets until they know whether varenicline tablets affects their tolerance for alcohol [see Warnings and Precautions (5.3), Adverse Reactions (6.2)].
Driving or Operating Machinery
Advise patients to use caution driving or operating machinery until they know how quitting smoking and/or varenicline may affect them [see Warnings and Precautions (5.4)].
Cardiovascular Events
Patients should be instructed to notify their healthcare providers of symptoms of new or worsening cardiovascular events and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke [see Warnings and Precautions (5.5), Adverse Reactions (6.1)].
Somnambulism
Patients should be instructed to discontinue varenicline tablets and notify their healthcare providers if they experience somnambulism [see Warnings and Precautions (5.6)].
Angioedema
Inform patients that there have been reports of angioedema, with swelling of the face, mouth (lip, gum, tongue) and neck (larynx and pharynx) that can lead to life-threatening respiratory compromise. Instruct patients to discontinue varenicline tablets and immediately seek medical care if they experience these symptoms [see Warnings and Precautions (5.7), Adverse Reactions (6.2)].
Serious Skin Reactions
Inform patients that serious skin reactions, such as Stevens-Johnson Syndrome and erythema multiforme, were reported by some patients taking varenicline tablets. Advise patients to stop taking varenicline tablets at the first sign of rash with mucosal lesions or skin reaction and contact a healthcare provider immediately [see Warnings and Precautions (5.8), Adverse Reactions (6.2)].
Vivid, Unusual, or Strange Dreams
Inform patients that they may experience vivid, unusual or strange dreams during treatment with varenicline tablets.
Pregnancy and Lactation
Patients who are pregnant or breastfeeding or planning to become pregnant should be advised of: the risks of smoking to a pregnant mother and her developing baby, the potential risks of varenicline tablets use during pregnancy and breastfeeding, and the benefits of smoking cessation with and without varenicline tablets. Advise breastfeeding women to monitor the infant for seizures and vomiting [see Use in Specific Populations (8.1 and 8.2)].
This product's label may have been updated. For full prescribing information, please visit www.glenmarkpharma-us.com
Distributed by:
Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
Code No.: HP/Drugs/N-MNB/17/194
Revised: August 2025
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Varenicline tablets are supplied for oral administration in two strengths: a 0.5 mg modified capsule shaped, white to off-white, film coated tablets debossed with 'L' on one side and '11' on other side and a 1 mg modified capsule shaped, light pink to pink color, film coated tablets debossed with 'L' on one side and '12' on other side.
Varenicline tablets are supplied in the following package configurations:
|
Description
|
NDC
|
|
|
Packs |
Starting 4-week card: 0.5 mg x 11 tablets and 1 mg x 42 tablets |
NDC 68462-895-04 |
|
Continuing 4-week card: 1 mg x 56 tablets |
NDC 68462-894-04 |
|
|
Starting Month Box: 0.5 mg x 11 tablets and 1 mg x 42 tablets |
NDC 68462-895-04 |
|
|
Continuing Month Box: 1 mg x 56 tablets |
NDC 68462-894-04 |
|
|
Bottles |
0.5 mg - bottle of 56 |
NDC 68462-893-56 |
|
1 mg - bottle of 56 |
NDC 68462-894-56 |
Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP.
This package is child-resistant
14.5 Gradual Approach to Quitting Smoking
Varenicline tablets was evaluated in a 52-week double-blind placebo-controlled study of 1,510 subjects who were not able or willing to quit smoking within four weeks, but were willing to gradually reduce their smoking over a 12 week period before quitting. Subjects were randomized to either varenicline tablets 1 mg twice daily (N=760) or placebo (N=750) for 24 weeks and followed up post-treatment through week 52. Subjects were instructed to reduce the number of cigarettes smoked by at least 50 percent by the end of the first four weeks of treatment, followed by a further 50 percent reduction from week four to week eight of treatment, with the goal of reaching complete abstinence by 12 weeks. After the initial 12-week reduction phase, subjects continued treatment for another 12 weeks. Subjects treated with varenicline tablets had a significantly higher Continuous Abstinence Rate compared with placebo at weeks 15 through 24 (32% vs. 7%) and weeks 15 through 52 (24% vs. 6%).
14.9 Subjects With Major Depressive Disorder (14.9 Subjects with Major Depressive Disorder)
Varenicline tablets was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 18 to 75 years with major depressive disorder without psychotic features (DSM-IV TR). If on medication, subjects were to be on a stable antidepressant regimen for at least two months. If not on medication, subjects were to have experienced a major depressive episode in the past 2 years, which was successfully treated. Subjects were randomized to varenicline tablets 1 mg twice daily (N=256) or placebo (N=269) for a treatment of 12 weeks and then followed for 40 weeks post-treatment. Subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (36%) compared to subjects treated with placebo (16%) and from week 9 through 52 (20%) compared to subjects treated with placebo (10%).
Table 14. Continuous Abstinence (95% confidence interval), Study in Patients with Major Depressive Disorder (MDD)
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
MDD Study |
36% |
16% |
20% |
10% |
BID = twice daily
5.7 Angioedema and Hypersensitivity Reactions
There have been postmarketing reports of hypersensitivity reactions including angioedema in patients treated with varenicline tablets [see Adverse Reactions (6.2), Patient Counseling Information (17)]. Clinical signs included swelling of the face, mouth (tongue, lips, and gums), extremities, and neck (throat and larynx). There were infrequent reports of life-threatening angioedema requiring emergent medical attention due to respiratory compromise. Instruct patients to discontinue varenicline tablets and immediately seek medical care if they experience these symptoms.
7.1 Use With Other Drugs for Smoking Cessation (7.1 Use with Other Drugs for Smoking Cessation)
Safety and efficacy of varenicline tablets in combination with other smoking cessation therapies have not been studied.
7.2 Effect of Smoking Cessation On Other Drugs (7.2 Effect of Smoking Cessation on Other Drugs)
Physiological changes resulting from smoking cessation, with or without treatment with varenicline tablets, may alter the pharmacokinetics or pharmacodynamics of certain drugs (e.g., theophylline, warfarin, insulin) for which dosage adjustment may be necessary.
14.4 Alternative Instructions for Setting A Quit Date (14.4 Alternative Instructions for Setting a Quit Date)
Varenicline tablets was evaluated in a double-blind, placebo-controlled trial where patients were instructed to select a target quit date between Day 8 and Day 35 of treatment. Subjects were randomized 3:1 to varenicline tablets 1 mg twice daily (N=486) or placebo (N=165) for 12 weeks of treatment and followed for another 12 weeks posttreatment. Patients treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (54%) compared to patients treated with placebo (19%) and from weeks 9 through 24 (35%) compared to subjects treated with placebo (13%).
14.7 Subjects With Chronic Obstructive Pulmonary Disease (14.7 Subjects with Chronic Obstructive Pulmonary Disease)
Varenicline tablets was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged ≥ 35 years with mild-to-moderate COPD with post-bronchodilator FEV1/FVC <70% and FEV1 ≥ 50% of predicted normal value. Subjects were randomized to varenicline tablets 1 mg twice daily (N=223) or placebo (N=237) for a treatment of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (41%) compared to subjects treated with placebo (9%) and from week 9 through 52 (19%) compared to subjects treated with placebo (6%).
Table 10. Continuous Abstinence (95% confidence interval), Studies in Patients with Chronic Obstructive Pulmonary Disease (COPD)
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
COPD Study |
41% |
9% |
19% |
6% |
BID = twice daily
14.10 Postmarketing Neuropsychiatric Safety Outcome Trial
Varenicline tablets was evaluated in a randomized, double-blind, active and placebo-controlled trial that included subjects without a history of psychiatric disorder (non-psychiatric cohort, N=3912) and with a history of psychiatric disorder (psychiatric cohort, N=4003). Subjects aged 18-75 years, smoking 10 or more cigarettes per day were randomized 1:1:1:1 to varenicline tablets 1 mg BID, bupropion SR 150 mg BID, NRT patch 21 mg/day with taper or placebo for a treatment period of 12 weeks; they were then followed for another 12 weeks post-treatment. [See Warnings and Precautions (5.1)]
A composite safety endpoint intended to capture clinically significant neuropsychiatric (NPS) adverse events included the following NPS adverse events: anxiety, depression, feeling abnormal, hostility, agitation, aggression, delusions, hallucinations, homicidal ideation, mania, panic, paranoia, psychosis, irritability, suicidal ideation, suicidal behavior or completed suicide.
As shown in Table 15, the use of varenicline tablets, bupropion, and NRT in the non-psychiatric cohort was not associated with an increased risk of clinically significant NPS adverse events compared with placebo. Similarly, in the non-psychiatric cohort, the use of varenicline tablets was not associated with an increased risk of clinically significant NPS adverse events in the composite safety endpoint compared with bupropion or NRT.
Table 15. Number of Patients with Clinically Significant or Serious NPS Adverse Events by Treatment Group Among Patients without a History of Psychiatric Disorder
|
Varenicline tablets
|
Bupropion
|
NRT
|
Placebo
|
|
|
Clinically Significant NPS |
30 (3.1) |
34 (3.5) |
33 (3.3) |
40 (4.1) |
|
Serious NPS |
1 (0.1) |
5 (0.5) |
1 (0.1) |
4 (0.4) |
|
Psychiatric Hospitalizations |
1 (0.1) |
2 (0.2) |
0 (0.0) |
1 (0.1) |
As shown in Table 16, there were more clinically significant NPS adverse events reported in patients in the psychiatric cohort in each treatment group compared with the non-psychiatric cohort. The incidence of events in the composite endpoint was higher for each of the active treatments compared to placebo: Risk Differences (RDs) (95%CI) vs placebo were 2.7% (-0.05, 5.4) for varenicline tablets, 2.2% (-0.5, 4.9) for bupropion, and 0.4% (-2.2, 3.0) for NRT transdermal nicotine.
Table 16. Number of Patients with Clinically Significant or Serious NPS Adverse Events by Treatment Group Among Patients with a History of Psychiatric Disorder
|
Varenicline tablets
|
Bupropion
|
NRT
|
Placebo
|
|
|
Clinically Significant NPS |
123 (12.2) |
118 (11.8) |
98 (9.8) |
95 (9.5) |
|
Serious NPS |
6 (0.6) |
8 (0.8) |
4 (0.4) |
6 (0.6) |
|
Psychiatric hospitalizations |
5 (0.5) |
8 (0.8) |
4 (0.4) |
2 (0.2) |
There was one completed suicide, which occurred during treatment in a patient treated with placebo in the non-psychiatric cohort. There were no completed suicides reported in the psychiatric cohort.
In both cohorts, subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 and 9 through 24 compared to subjects treated with bupropion, nicotine patch and placebo.
Table 17 Continuous Abstinence (95% confidence interval), Study in Patients with or without a History of Psychiatric Disorder
|
Varenicline tablets
|
Bupropion SR
|
NRT
|
Placebo
|
|
|
Weeks 9 through 12
|
||||
|
Non- |
38% |
26% |
26% |
14% |
|
Psychiatric |
(35%, 41%) |
(23%, 29%) |
(24%, 29%) |
(12%, 16%) |
|
Cohort |
||||
|
Psychiatric |
29% |
19% |
20% |
11% |
|
Cohort |
(26%, 32%) |
(17%, 22%) |
(18%, 23%) |
(10%, 14%) |
|
Weeks 9 through 24
|
||||
|
Non- |
25% |
19% |
18% |
11% |
|
Psychiatric |
(23%, 28%) |
(16%, 21%) |
(16%, 21%) |
(9%, 13%) |
|
Cohort |
||||
|
Psychiatric |
18% |
14% |
13% |
8% |
|
Cohort |
(16%, 21%) |
(12%, 16%) |
(11%, 15%) |
(7%, 10%) |
BID = twice daily
5.1 Neuropsychiatric Adverse Events Including Suicidality (5.1 Neuropsychiatric Adverse Events including Suicidality)
Serious neuropsychiatric adverse events have been reported in patients being treated with varenicline tablets [see Adverse Reactions (6.2)]. These postmarketing reports have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Some patients who stopped smoking may have been experiencing symptoms of nicotine withdrawal, including depressed mood. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these adverse events occurred in patients taking varenicline tablets who continued to smoke.
Neuropsychiatric adverse events occurred in patients without and with pre-existing psychiatric disease; some patients experienced worsening of their psychiatric illnesses. Some neuropsychiatric adverse events, including unusual and sometimes aggressive behavior directed to oneself or others, may have been worsened by concomitant use of alcohol [see Warnings and Precautions (5.3) , Adverse Reactions (6.2)]. Observe patients for the occurrence of neuropsychiatric adverse events. Advise patients and caregivers that the patient should stop taking varenicline tablets and contact a healthcare provider immediately if agitation, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. The healthcare provider should evaluate the severity of the symptoms and the extent to which the patient is benefiting from treatment, and consider options including dose reduction, continued treatment under closer monitoring, or discontinuing treatment. In many postmarketing cases, resolution of symptoms after discontinuation of varenicline tablets was reported. However, the symptoms persisted in some cases; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.
The neuropsychiatric safety of varenicline tablets was evaluated in a randomized, double-blind, active and placebo-controlled study that included patients without a history of psychiatric disorder (non-psychiatric cohort, N=3912) and patients with a history of psychiatric disorder (psychiatric cohort, N=4003). In the non-psychiatric cohort, varenicline tablets was not associated with an increased incidence of clinically significant neuropsychiatric adverse events in a composite endpoint comprising anxiety, depression, feeling abnormal, hostility, agitation, aggression, delusions, hallucinations, homicidal ideation, mania, panic, and irritability. In the psychiatric cohort, there were more events reported in each treatment group compared to the non-psychiatric cohort, and the incidence of events in the composite endpoint was higher for each of the active treatments compared to placebo: Risk Differences (RDs) (95%CI) vs. placebo were 2.7% (-0.05, 5.4) for varenicline tablets, 2.2% (-0.5, 4.9) for bupropion, and 0.4% (-2.2, 3.0) for transdermal nicotine. In the non-psychiatric cohort, neuropsychiatric adverse events of a serious nature were reported in 0.1% of varenicline tablets-treated patients and 0.4% of placebo-treated patients. In the psychiatric cohort, neuropsychiatric events of a serious nature were reported in 0.6% of varenicline tablets-treated patients, with 0.5% involving psychiatric hospitalization. In placebo-treated patients, serious neuropsychiatric events occurred in 0.6%, with 0.2% requiring psychiatric hospitalization [see Clinical Studies (14.10)].
Principal Display Panel 0.5 / 1 Mg Tablet Starting Pack Carton (PRINCIPAL DISPLAY PANEL - 0.5 / 1 mg Tablet Starting Pack Carton)
NDC 68462-895-04
Varenicline Tablets
Dispense the Medication Guide provided separately to each patient.
STARTING MONTH BOX
(Your first 4 weeks)
Contains:
1 Starting Week (0.5 mg* x 11 tablets)
3 Continuing Weeks (1 mg† x 42 tablets)
Use the Starting Week (green) blister card first when beginning to take Varenicline Tablets.
Rx only
PRINCIPAL DISPLAY PANEL - 0.5 / 1 mg Tablet Starting Pack
NDC 68462-895-04
Varenicline Tablets
Dispense the Medication Guide provided separately to each patient.
STARTING PACK
Use the Starting Week (green) blister card first when beginning to take Varenicline Tablets.
Contains:
1 Starting Week (0.5 mg* x 11 tablets)
3 Continuing Weeks (1 mg† x 42 tablets)
Rx only
PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Bottle Label
NDC 68462-893-56
Varenicline Tablets
0.5 mg*
Dispense the Medication Guide provided separately to each patient.
Rx only 56 Tablets
PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label
NDC 68462-894-56
Varenicline Tablets
1 mg*
Dispense the Medication Guide provided separately to each patient.
56 Tablets
Rx only
14.8 Subjects With Cardiovascular Disease and Other Cardiovascular Analyses (14.8 Subjects with Cardiovascular Disease and Other Cardiovascular Analyses)
Varenicline tablets was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 35 to 75 years with stable, documented cardiovascular disease (diagnoses other than, or in addition to, hypertension) that had been diagnosed for more than 2 months. Subjects were randomized to varenicline tablets 1 mg twice daily (N=353) or placebo (N=350) for a treatment period of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with varenicline tablets had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (47%) compared to subjects treated with placebo (14%) and from week 9 through 52 (20%) compared to subjects treated with placebo (7%).
Table 11. Continuous Abstinence (95% confidence interval), Studies in Patients with Cardiovascular Disease (CVD)
|
Weeks 9 through 12
|
Weeks 9 through 52
|
|||
|
Varenicline tablets |
Placebo |
Varenicline tablets |
Placebo |
|
|
CVD Study |
47% |
14% |
20% |
7% |
BID = twice daily
In this study, all-cause and CV mortality was lower in patients treated with varenicline tablets, but certain nonfatal CV events occurred more frequently in patients treated with varenicline tablets than in patients treated with placebo [see Warnings and Precautions (5.5), Adverse Reactions (6.1)]. Table 12 below shows mortality and the incidence of selected nonfatal serious CV events occurring more frequently in the varenicline tablets arm compared to the placebo arm. These events were adjudicated by an independent blinded committee. Nonfatal serious CV events not listed occurred at the same incidence or more commonly in the placebo arm. Patients with more than one CV event of the same type are counted only once per row. Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina.
Table 12. Mortality and Adjudicated Nonfatal Serious Cardiovascular Events in the Placebo-Controlled Varenicline Tablets Trial in Patients with Stable Cardiovascular Disease
|
|
Varenicline tablets
|
Placebo
|
|
Mortality (Cardiovascular and All-cause up to 52 weeks)
|
||
|
Cardiovascular |
1 (0.3) |
2 (0.6) |
|
All-cause |
2 (0.6) |
5 (1.4) |
|
Nonfatal Cardiovascular Events (rate on varenicline tablets > Placebo)
|
||
|
Up to 30 days after treatment
|
||
|
Nonfatal myocardial infarction |
4 (1.1) |
1 (0.3) |
|
Nonfatal Stroke |
2 (0.6) |
0 (0) |
|
Beyond 30 days after treatment and
|
||
|
Nonfatal myocardial infarction |
3 (0.8) |
2 (0.6) |
|
Need for coronary |
7 (2.0) |
2 (0.6) |
|
Hospitalization for angina pectoris |
6 (1.7) |
4 (1.1) |
|
Transient ischemia attack |
1 (0.3) |
0 (0) |
|
New diagnosis of peripheral |
|
|
Following the CVD study, a meta-analysis of 15 clinical trials of ≥12 weeks treatment duration, including 7002 patients (4190 varenicline tablets, 2812 placebo), was conducted to systematically assess the CV safety of varenicline tablets. The study in patients with stable CV disease described above was included in the meta-analysis. There were lower rates of all-cause mortality (varenicline tablets 6 [0.14%]; placebo 7 [0.25%]) and CV mortality (varenicline tablets 2 [0.05%]; placebo 2 [0.07%]) in the varenicline tablets arms compared with the placebo arms in the meta-analysis.
The key CV safety analysis included occurrence and timing of a composite endpoint of Major Adverse Cardiovascular Events (MACE), defined as CV death, nonfatal MI, and nonfatal stroke. These events included in the endpoint were adjudicated by a blinded, independent committee. Overall, a small number of MACE occurred in the trials included in the meta-analysis, as described in Table 13. These events occurred primarily in patients with known CV disease.
Table 13. Number of MACE cases, Hazard Ratio and Rate Difference in a Meta-Analysis of 15 Clinical Trials Comparing Varenicline tablets to Placebo*
|
Varenicline tablets
|
Placebo N=2812
|
|
|
MACE cases, n (%)
|
13 (0.31%) |
6 (0.21%) |
|
Patient-years of exposure |
1316 |
839 |
|
Hazard Ratio (95% CI)
|
||
|
1.95 (0.79, 4.82) |
||
|
Rate Difference per 1,000 patient-years (95% CI)
|
||
|
6.30 (-2.40, 15.10) |
*Includes MACE occurring up to 30 days post-treatment.
The meta-analysis showed that exposure to varenicline tablets resulted in a hazard ratio for MACE of 1.95 (95% confidence interval from 0.79 to 4.82) for patients up to 30 days after treatment; this is equivalent to an estimated increase of 6.3 MACE events per 1,000 patient-years of exposure. The meta-analysis showed higher rates of CV endpoints in patients on varenicline tablets relative to placebo across different time frames and pre-specified sensitivity analyses, including various study groupings and CV outcomes. Although these findings were not statistically significant they were consistent. Because the number of events was small overall, the power for finding a statistically significant difference in a signal of this magnitude is low.
Additionally, a cardiovascular endpoint analysis was added to the postmarketing neuropsychiatric safety outcome study along with a non-treatment extension, [see Warnings and Precautions (5.5), Adverse Reactions (6.1), Clinical Studies (14.10)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:09.285058 · Updated: 2026-03-14T22:42:54.492171