Quality Choice medicated Pads
e8422db7-5e7e-4c9c-9142-1f042a0f22de
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
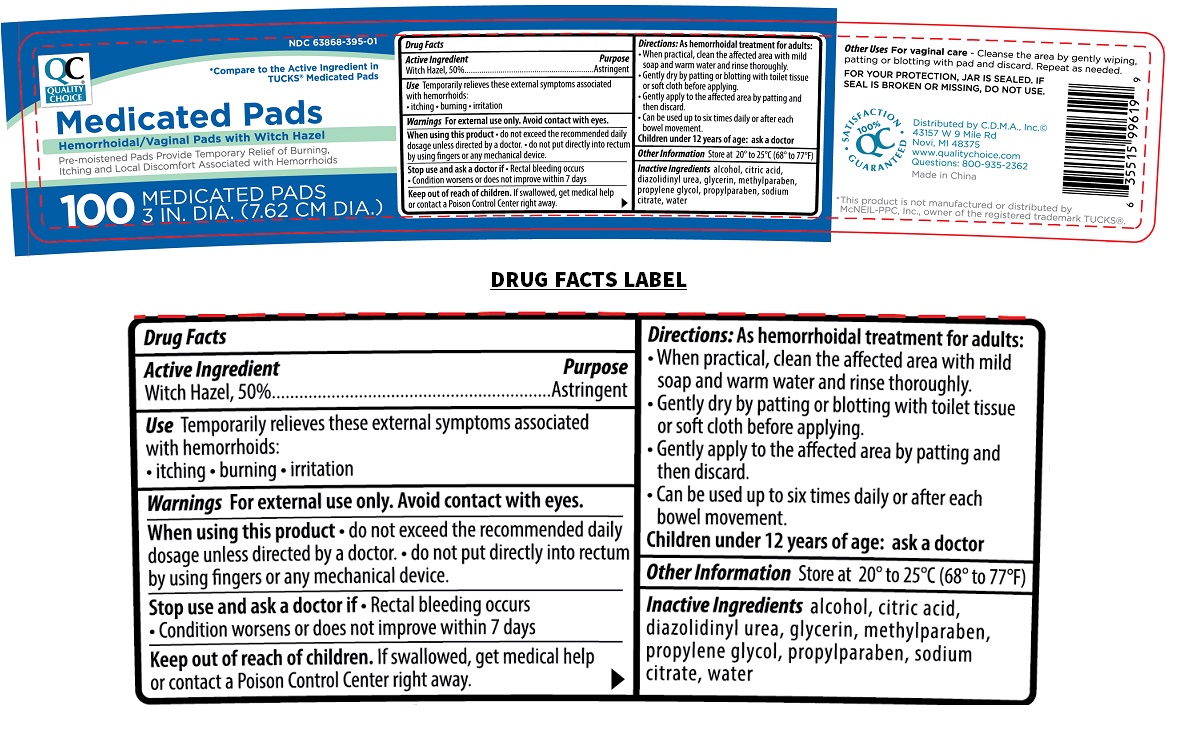
Witch Hazel, 50%
Purpose
Astringent
Medication Information
Purpose
Astringent
Description
Witch Hazel, 50%
Use
Temporarily relieves these external symptoms associated with hemorrhoids:
• itching • burning • irritation
Section 42229-5
* Compare to the Active Ingredient in TUCKS® Medicated pads
Hemorrhoidal/Vaginal pads with Witch Hazel
Pre-moistened Pads Provide Temporary Relief of Burning, Itching and Local Discomfort Associated with Hemorrhoids
Other Uses For vaginal care - Cleanse the area by gently wiping, patting or blotting with pad and discard. Repeat as needed.
FOR YOUR PROTECTION, JAR IS SEALED. IF SEAL IS BROKEN OR MISSING, DO NOT USE.
• SATISFACTION • GUARANTEED 100% QC
Distributed by C.D.M.A., Inc.©
43157 W 9 Mile Rd
Novi, MI 48375
www.qualitychoice.com
Questions: 800-935-2362
Made in China
*This product is not manufactured or distributed by McNEIL-PPC, Inc., owner of the registered trademark TUCKS®.
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only. Avoid contact with eyes.
When using this product • do not exceed the recommended daily dosage unless directed by a doctor. • do not put directly into rectum by using fingers or any mechanical device.
Stop use and ask a doctor if • Rectal bleeding occurs • Condition worsens or does not improve within 7 days
Packaging
Drug Facts
Directions:
As hemorrhoidal treatment for adults:
• When practical, clean the affected area with mild soap and warm water and rinse thoroughly.
• Gently dry by patting or blotting with toilet tissue or soft cloth before applying.
• Gently apply to the affected area by patting and then discard.
• Can be used up to six times daily or after each bowel movement.
Children under 12 years of age: ask a doctor
Active Ingredient
Witch Hazel, 50%
Other Information
Store at 20° to 25°C (68° to 77°F)
Inactive Ingredients
alcohol, citric acid, diazolidinyl urea, glycerin, methylparaben, propylene glycol, propylparaben, sodium citrate, water
Structured Label Content
Use
Temporarily relieves these external symptoms associated with hemorrhoids:
• itching • burning • irritation
Section 42229-5 (42229-5)
* Compare to the Active Ingredient in TUCKS® Medicated pads
Hemorrhoidal/Vaginal pads with Witch Hazel
Pre-moistened Pads Provide Temporary Relief of Burning, Itching and Local Discomfort Associated with Hemorrhoids
Other Uses For vaginal care - Cleanse the area by gently wiping, patting or blotting with pad and discard. Repeat as needed.
FOR YOUR PROTECTION, JAR IS SEALED. IF SEAL IS BROKEN OR MISSING, DO NOT USE.
• SATISFACTION • GUARANTEED 100% QC
Distributed by C.D.M.A., Inc.©
43157 W 9 Mile Rd
Novi, MI 48375
www.qualitychoice.com
Questions: 800-935-2362
Made in China
*This product is not manufactured or distributed by McNEIL-PPC, Inc., owner of the registered trademark TUCKS®.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Astringent
Warnings
For external use only. Avoid contact with eyes.
When using this product • do not exceed the recommended daily dosage unless directed by a doctor. • do not put directly into rectum by using fingers or any mechanical device.
Stop use and ask a doctor if • Rectal bleeding occurs • Condition worsens or does not improve within 7 days
Packaging
Drug Facts
Directions:
As hemorrhoidal treatment for adults:
• When practical, clean the affected area with mild soap and warm water and rinse thoroughly.
• Gently dry by patting or blotting with toilet tissue or soft cloth before applying.
• Gently apply to the affected area by patting and then discard.
• Can be used up to six times daily or after each bowel movement.
Children under 12 years of age: ask a doctor
Active Ingredient
Witch Hazel, 50%
Other Information
Store at 20° to 25°C (68° to 77°F)
Inactive Ingredients
alcohol, citric acid, diazolidinyl urea, glycerin, methylparaben, propylene glycol, propylparaben, sodium citrate, water
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:38.865219 · Updated: 2026-03-14T22:59:43.649746