Act Anticavity Fluoride Rinse Mint
e81c83a2-363a-442f-8731-2598a4f4c21e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Anticavity
Medication Information
Purpose
Anticavity
Description
Drug Facts
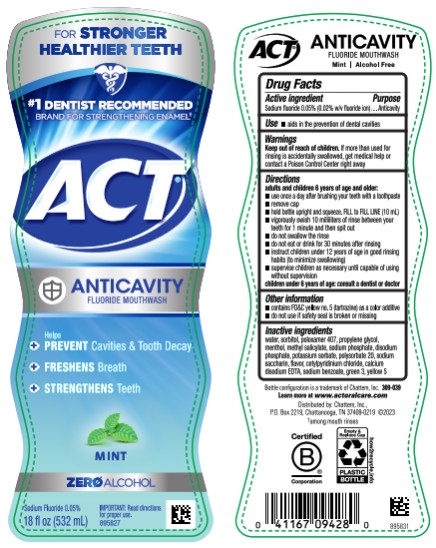
Use
- aids in the prevention of dental cavities
Section 42229-5
Drug Facts
Directions
adults and children 6 years of age and older:
- use once a day after brushing your teeth with a toothpaste
- remove cap
- hold bottle upright and squeeze. Fill to FILL LINE (10 mL)
- vigorously swish 10 milliliters of rinse between your teeth for 1 minute and then spit out
- do not swallow the rinse
- do not eat or drink for 30 minutes after rinsing
- instruct children under 12 years of age in good rinsing habits (to minimize swallowing)
- supervise children as necessary until capable of using without supervision
children under 6 years of age: consult a dentist or doctor
Active Ingredient
Sodium fluoride 0.05% (0.02% w/v fluoride ion)
Other Information
- contains FD&C yellow no. 5 (tartrazine) as a color additive
- do not use if safety seal is broken or missing
Inactive Ingredients
water, sorbitol, poloxamer 407, propylene glycol, menthol, methyl salicylate, sodium phosphate, disodium phosphate, potassium sorbate, polysorbate 20, sodium saccharin, flavor, cetylpyridinium chloride, calcium disodium EDTA, sodium benzoate, green 3, yellow 5
Learn more at www.ACTFLUORIDE.com
Principal Display Panel
ZERO ALCOHOL
#1 DENTIST RECOMMENDED
FLUORIDE BRAND
ACT
1X Daily
ANTICAVITY
FLUORIDE MOUTHWASH
MINT
18
fl
oz (532 mL)
Keep Out of Reach of Children.
If more than used for rinsing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use
- aids in the prevention of dental cavities
Section 42229-5 (42229-5)
Drug Facts
Purpose
Anticavity
Directions
adults and children 6 years of age and older:
- use once a day after brushing your teeth with a toothpaste
- remove cap
- hold bottle upright and squeeze. Fill to FILL LINE (10 mL)
- vigorously swish 10 milliliters of rinse between your teeth for 1 minute and then spit out
- do not swallow the rinse
- do not eat or drink for 30 minutes after rinsing
- instruct children under 12 years of age in good rinsing habits (to minimize swallowing)
- supervise children as necessary until capable of using without supervision
children under 6 years of age: consult a dentist or doctor
Active Ingredient (Active ingredient)
Sodium fluoride 0.05% (0.02% w/v fluoride ion)
Other Information (Other information)
- contains FD&C yellow no. 5 (tartrazine) as a color additive
- do not use if safety seal is broken or missing
Inactive Ingredients (Inactive ingredients)
water, sorbitol, poloxamer 407, propylene glycol, menthol, methyl salicylate, sodium phosphate, disodium phosphate, potassium sorbate, polysorbate 20, sodium saccharin, flavor, cetylpyridinium chloride, calcium disodium EDTA, sodium benzoate, green 3, yellow 5
Learn more at www.ACTFLUORIDE.com
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
ZERO ALCOHOL
#1 DENTIST RECOMMENDED
FLUORIDE BRAND
ACT
1X Daily
ANTICAVITY
FLUORIDE MOUTHWASH
MINT
18
fl
oz (532 mL)
Keep Out of Reach of Children. (Keep out of reach of children.)
If more than used for rinsing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:09.984475 · Updated: 2026-03-14T23:12:19.459812