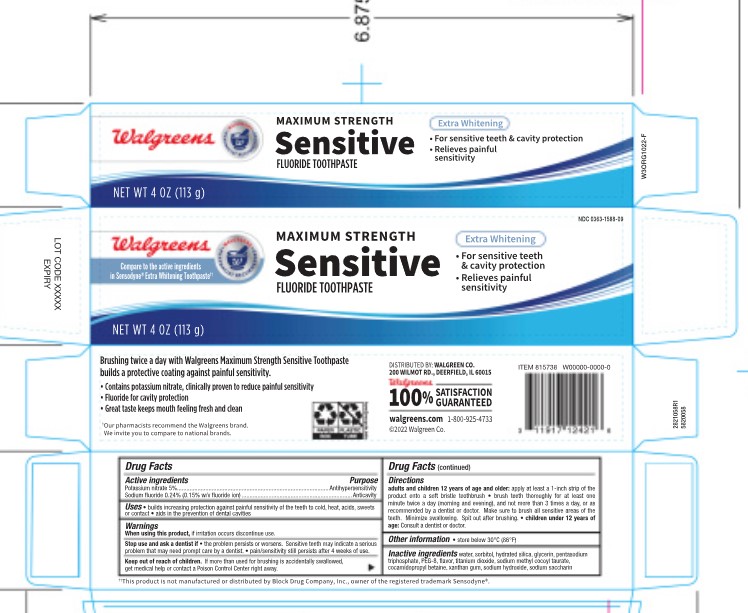
5820058 Walgreens Sensitive Extra Whitening
e807b8a0-6d58-a15f-e053-2995a90a2ea1
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Potassium Nitrate 5% Antihypersensitivity Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Medication Information
Warnings and Precautions
When using this product, if irritation occurs discontinue use. Stop use and ask a dentist if the problem persists or worsens. Sensitive teeth may indicate a serious problem that may need prompt care by a dentist. Pain/sensitivity still persists after 4 weeks of use.
Indications and Usage
Adults and children 12 years of age and older: apply at least a 1-inch strip of the product onto a soft bristle toothbrush. Brush teeth thoroughly for at least one minute twice a day (morning and evening), and not more than 3 time a day, or as recommended by a dentist or doctor. Make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing. Children under 12 years of age: consult a dentist or doctor.
Dosage and Administration
Builds increasing protection against painful sensitivity of the teeth to cold, heat, acids, sweets or contact. Aids in the prevention of dental cavities.
Description
Potassium Nitrate 5% Antihypersensitivity Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Section 50565-1
Kep out of reach of children. If more tahn used for brushing is accidentally swallowed, get medical help or contact a poisoin control center right away.
Section 51727-6
water, sorbitol, hydrated silica, glycerin, petasodium triphosphate, PEG-8, flavor, titanium dioxide, sodium methyl cocoyl taurate, cocamidopropyl betaine, xanthan gum, sodium hydroxide, sodium saccharin
Section 51945-4
Section 55105-1
Store below 30C (86F).
Section 55106-9
Potassium Nitrate 5% Antihypersensitivity
Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Structured Label Content
Indications and Usage (34067-9)
Adults and children 12 years of age and older: apply at least a 1-inch strip of the product onto a soft bristle toothbrush. Brush teeth thoroughly for at least one minute twice a day (morning and evening), and not more than 3 time a day, or as recommended by a dentist or doctor. Make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing. Children under 12 years of age: consult a dentist or doctor.
Dosage and Administration (34068-7)
Builds increasing protection against painful sensitivity of the teeth to cold, heat, acids, sweets or contact. Aids in the prevention of dental cavities.
Warnings and Precautions (34071-1)
When using this product, if irritation occurs discontinue use. Stop use and ask a dentist if the problem persists or worsens. Sensitive teeth may indicate a serious problem that may need prompt care by a dentist. Pain/sensitivity still persists after 4 weeks of use.
Section 50565-1 (50565-1)
Kep out of reach of children. If more tahn used for brushing is accidentally swallowed, get medical help or contact a poisoin control center right away.
Section 51727-6 (51727-6)
water, sorbitol, hydrated silica, glycerin, petasodium triphosphate, PEG-8, flavor, titanium dioxide, sodium methyl cocoyl taurate, cocamidopropyl betaine, xanthan gum, sodium hydroxide, sodium saccharin
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Store below 30C (86F).
Section 55106-9 (55106-9)
Potassium Nitrate 5% Antihypersensitivity
Sodium Fluoride 0.24% (0.15% w/v fluoride ion)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:34.719968 · Updated: 2026-03-14T23:11:10.262850