These Highlights Do Not Include All The Information Needed To Use Savaysa Safely And Effectively. See Full Prescribing Information For Savaysa.
e77d3400-56ad-11e3-949a-0800200c9a66
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: (A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CREATININE CLEARANCE (CRCL) > 95 ML/MIN (B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS (C) SPINAL/EPIDURAL HEMATOMA See full prescribing information for complete boxed warning. (A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CRCL > 95 ML/MIN: SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the ENGAGE AF-TIMI 48 study, nonvalvular atrial fibrillation patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg once daily compared to patients treated with warfarin. In these patients another anticoagulant should be used ( 5.1 ). (B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS: Premature discontinuation of any oral anticoagulant in the absence of adequate alternative anticoagulation increases the risk of ischemic events. If SAVAYSA is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant as described in the transition guidance ( 2.4 , 5.2 , 14 ). (C) SPINAL/EPIDURAL HEMATOMA: Epidural or spinal hematomas may occur in patients treated with SAVAYSA who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures ( 5.4 ).
Indications and Usage
SAVAYSA is a factor Xa inhibitor indicated: To reduce the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF) ( 1.1 ) Limitation of Use for NVAF SAVAYSA should not be used in patients with creatinine clearance (CrCL) > 95 mL/min because of increased risk of ischemic stroke compared to warfarin at the highest dose studied (60 mg) ( 1.1 ) SAVAYSA is indicated for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5 to 10 days of initial therapy with a parenteral anticoagulant ( 1.2 )
Dosage and Administration
Treatment of NVAF: Assess CrCL before initiating therapy ( 2.1 ) The recommended dose is 60 mg once daily in patients with CrCL >50 to ≤ 95 mL/min. Do not use SAVAYSA in patients with CrCL > 95 mL/min ( 2.1 ) Reduce dose to 30 mg once daily in patients with creatinine clearance 15 to 50 mL/min ( 2.1 ) Treatment of DVT and PE: The recommended dose is 60 mg once daily ( 2.2 ) Reduce dose to 30 mg once daily for patients with CrCL 15 to 50 mL/min or body weight less than or equal to 60 kg or who use certain P-gp inhibitors ( 2.2 )
Warnings and Precautions
Bleeding: Serious and potentially fatal bleeding. Promptly evaluate signs and symptoms of blood loss ( 5.3 ) Mechanical Heart Valves or Moderate to Severe Mitral Stenosis: Use is not recommended ( 5.5 ) Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: SAVAYSA use not recommended. ( 5.6 )
Contraindications
SAVAYSA is contraindicated in patients with: Active pathological bleeding [see Warnings and Precautions (5.3) and Adverse Reactions (6.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the prescribing information. Increased Risk of Stroke with Discontinuation of SAVAYSA in Patients with Nonvalvular Atrial Fibrillation [see Warnings and Precautions (5.2) ] Risk of Bleeding [see Warnings and Precautions (5.3) ] Spinal/Epidural Anesthesia or Puncture [see Warnings and Precautions (5.4) ]
Drug Interactions
Anticoagulants, Antiplatelets, Thrombolytics, and Selective Serotonin Reuptake Inhibitors (SSRIs)/Serotonin Norepinephrine Reuptake Inhibitors (SNRIs): Avoid concomitant use due to increased risk of bleeding. ( 7.1 ) Rifampin: Avoid concomitant use ( 7.2 )
Storage and Handling
SAVAYSA (edoxaban) is supplied as round shaped, film-coated, non-scored tablets containing edoxaban tosylate equivalent to 60, 30 or 15 mg of SAVAYSA, packaged in bottles and blisters. NDC 65597-xxx-yy Strength Color Deboss yy xxx Bottle of Blister of 30 90 500 10 × 10 10 blister cards of 10 counts 10 × 5 5 blister cards of 10 counts 15 mg orange DSC L15 201 30 - - - - 30 mg pink DSC L30 202 30 90 50 10 05 60 mg yellow DSC L60 203 30 90 50 10 05
How Supplied
SAVAYSA (edoxaban) is supplied as round shaped, film-coated, non-scored tablets containing edoxaban tosylate equivalent to 60, 30 or 15 mg of SAVAYSA, packaged in bottles and blisters. NDC 65597-xxx-yy Strength Color Deboss yy xxx Bottle of Blister of 30 90 500 10 × 10 10 blister cards of 10 counts 10 × 5 5 blister cards of 10 counts 15 mg orange DSC L15 201 30 - - - - 30 mg pink DSC L30 202 30 90 50 10 05 60 mg yellow DSC L60 203 30 90 50 10 05
Medication Information
Warnings and Precautions
Bleeding: Serious and potentially fatal bleeding. Promptly evaluate signs and symptoms of blood loss ( 5.3 ) Mechanical Heart Valves or Moderate to Severe Mitral Stenosis: Use is not recommended ( 5.5 ) Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: SAVAYSA use not recommended. ( 5.6 )
Indications and Usage
SAVAYSA is a factor Xa inhibitor indicated: To reduce the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF) ( 1.1 ) Limitation of Use for NVAF SAVAYSA should not be used in patients with creatinine clearance (CrCL) > 95 mL/min because of increased risk of ischemic stroke compared to warfarin at the highest dose studied (60 mg) ( 1.1 ) SAVAYSA is indicated for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5 to 10 days of initial therapy with a parenteral anticoagulant ( 1.2 )
Dosage and Administration
Treatment of NVAF: Assess CrCL before initiating therapy ( 2.1 ) The recommended dose is 60 mg once daily in patients with CrCL >50 to ≤ 95 mL/min. Do not use SAVAYSA in patients with CrCL > 95 mL/min ( 2.1 ) Reduce dose to 30 mg once daily in patients with creatinine clearance 15 to 50 mL/min ( 2.1 ) Treatment of DVT and PE: The recommended dose is 60 mg once daily ( 2.2 ) Reduce dose to 30 mg once daily for patients with CrCL 15 to 50 mL/min or body weight less than or equal to 60 kg or who use certain P-gp inhibitors ( 2.2 )
Contraindications
SAVAYSA is contraindicated in patients with: Active pathological bleeding [see Warnings and Precautions (5.3) and Adverse Reactions (6.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the prescribing information. Increased Risk of Stroke with Discontinuation of SAVAYSA in Patients with Nonvalvular Atrial Fibrillation [see Warnings and Precautions (5.2) ] Risk of Bleeding [see Warnings and Precautions (5.3) ] Spinal/Epidural Anesthesia or Puncture [see Warnings and Precautions (5.4) ]
Drug Interactions
Anticoagulants, Antiplatelets, Thrombolytics, and Selective Serotonin Reuptake Inhibitors (SSRIs)/Serotonin Norepinephrine Reuptake Inhibitors (SNRIs): Avoid concomitant use due to increased risk of bleeding. ( 7.1 ) Rifampin: Avoid concomitant use ( 7.2 )
Storage and Handling
SAVAYSA (edoxaban) is supplied as round shaped, film-coated, non-scored tablets containing edoxaban tosylate equivalent to 60, 30 or 15 mg of SAVAYSA, packaged in bottles and blisters. NDC 65597-xxx-yy Strength Color Deboss yy xxx Bottle of Blister of 30 90 500 10 × 10 10 blister cards of 10 counts 10 × 5 5 blister cards of 10 counts 15 mg orange DSC L15 201 30 - - - - 30 mg pink DSC L30 202 30 90 50 10 05 60 mg yellow DSC L60 203 30 90 50 10 05
How Supplied
SAVAYSA (edoxaban) is supplied as round shaped, film-coated, non-scored tablets containing edoxaban tosylate equivalent to 60, 30 or 15 mg of SAVAYSA, packaged in bottles and blisters. NDC 65597-xxx-yy Strength Color Deboss yy xxx Bottle of Blister of 30 90 500 10 × 10 10 blister cards of 10 counts 10 × 5 5 blister cards of 10 counts 15 mg orange DSC L15 201 30 - - - - 30 mg pink DSC L30 202 30 90 50 10 05 60 mg yellow DSC L60 203 30 90 50 10 05
Description
WARNING: (A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CREATININE CLEARANCE (CRCL) > 95 ML/MIN (B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS (C) SPINAL/EPIDURAL HEMATOMA See full prescribing information for complete boxed warning. (A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CRCL > 95 ML/MIN: SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the ENGAGE AF-TIMI 48 study, nonvalvular atrial fibrillation patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg once daily compared to patients treated with warfarin. In these patients another anticoagulant should be used ( 5.1 ). (B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS: Premature discontinuation of any oral anticoagulant in the absence of adequate alternative anticoagulation increases the risk of ischemic events. If SAVAYSA is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant as described in the transition guidance ( 2.4 , 5.2 , 14 ). (C) SPINAL/EPIDURAL HEMATOMA: Epidural or spinal hematomas may occur in patients treated with SAVAYSA who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures ( 5.4 ).
Section 42229-5
A. REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CRCL > 95 ML/MIN
SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the ENGAGE AF-TIMI 48 study, nonvalvular atrial fibrillation patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg once daily compared to patients treated with warfarin. In these patients another anticoagulant should be used [see Dosage and Administration (2.1), Warnings and Precautions (5.1), and Clinical Studies (14.1)].
Section 42231-1
| MEDICATION GUIDE SAVAYSA (sa vaye' sah) (edoxaban) tablets |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 10/2023 |
What is the most important information I should know about SAVAYSA?
|
|
|
What is SAVAYSA?
SAVAYSA is a prescription medicine used to:
|
|
|
Who should not take SAVAYSA?
Do not take SAVAYSA if you currently have certain types of abnormal bleeding. |
|
Before taking SAVAYSA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some of your other medicines may affect the way SAVAYSA works. Certain medicines may increase your risk of bleeding or stroke when taken with SAVAYSA. See "What is the most important information I should know about SAVAYSA?" Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
How should I take SAVAYSA?
|
|
|
What are the possible side effects of SAVAYSA? SAVAYSA can cause serious side effects. See "What is the most important information I should know about SAVAYSA?" The most common side effects in people who take SAVAYSA for nonvalvular atrial fibrillation include bleeding and low red blood cell count (anemia). The most common side effects in people who take SAVAYSA for deep vein thrombosis and pulmonary embolism include bleeding, rash, abnormal liver function tests, and low red blood cell count (anemia). Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store SAVAYSA?
|
|
|
General information about the safe and effective use of SAVAYSA
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use SAVAYSA for a condition for which it was not prescribed. Do not give SAVAYSA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about SAVAYSA that is written for health professionals. |
|
|
What are the ingredients in SAVAYSA?
Active ingredient: edoxaban tosylate monohydrate Inactive ingredients: mannitol, pregelatinized starch, crospovidone, hydroxypropyl cellulose, magnesium stearate, talc, and carnauba wax. The color coatings contain hypromellose, titanium dioxide, talc, polyethylene glycol 8000, iron oxide yellow (60 mg tablets and 15 mg tablets), and iron oxide red (30 mg tablets and 15 mg tablets). Manufactured by: Daiichi Sankyo Co., LTD., Tokyo 103-8426, Japan Distributed by: Daiichi Sankyo, Inc., Basking Ridge, NJ 07920-2311 USA The brands listed above are trademarks of their respective owners. Copyright© 2023, Daiichi Sankyo, Inc. USMG-SAV-C9-1023-r103 For more information, call 1-877-437-7763 or go to https://savaysa.com. |
Section 44425-7
Store at 20-25°C (68-77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature].
Keep out of the reach of children.
Section 51945-4
PRINCIPAL DISPLAY PANEL
NDC 65597-201-30
Savaysa
(edoxaban) tablets
15 mg
30 TABLETS
Rx Only
PRINCIPAL DISPLAY PANEL
NDC 65597-202-30
Savaysa
(edoxaban) tablets
30 mg
30 TABLETS
Rx Only
10 Overdosage
A specific reversal agent for edoxaban is not available. Overdose of SAVAYSA increases the risk of bleeding.
The following are not expected to reverse the anticoagulant effects of edoxaban: protamine sulfate, vitamin K, and tranexamic acid [see Warnings and Precautions (5.3)].
Hemodialysis does not significantly contribute to edoxaban clearance [see Clinical Pharmacology (12.3)].
11 Description
Edoxaban, a factor Xa inhibitor, is supplied as edoxaban tosylate monohydrate. The chemical name is N-(5-Chloropyridin-2-yl)-N'-[(1S,2R,4S)-4-(N,N-dimethylcarbamoyl)-2-(5-methyl-4,5,6,7-tetrahydro[1,3]thiazolo[5,4-c]pyridine-2-carboxamido)cyclohexyl] oxamide mono (4-methylbenzenesulfonate) monohydrate. Edoxaban tosylate monohydrate has the empirical formula C24H30ClN7O4S∙C7H8O3S∙H2O representing a molecular weight of 738.27. The chemical structure of edoxaban tosylate monohydrate is:
It is a white to pale yellowish-white crystalline powder. The solubility of edoxaban tosylate (pKa 6.7) decreases with increasing pH. It is slightly soluble in water, pH 3 to 5 buffer, very slightly soluble at pH 6 to 7; and practically insoluble at pH 8 to 9.
SAVAYSA is available for oral administration as a 60 mg, 30 mg, or 15 mg round shaped, film-coated tablet, debossed with product identification markings. Each 60 mg tablet contains 80.82 mg edoxaban tosylate monohydrate equivalent to 60 mg of edoxaban. Each 30 mg tablet contains 40.41 mg edoxaban tosylate monohydrate equivalent to 30 mg of edoxaban. Each 15 mg tablet contains 20.20 mg edoxaban tosylate monohydrate equivalent to 15 mg of edoxaban. The inactive ingredients are: mannitol, pregelatinized starch, crospovidone, hydroxypropyl cellulose, magnesium stearate, talc, and carnauba wax. The color coatings contain hypromellose, titanium dioxide, talc, polyethylene glycol 8000, iron oxide yellow (60 mg tablets and 15 mg tablets), and iron oxide red (30 mg tablets and 15 mg tablets).
7.2 P Gp Inducers
Avoid the concomitant use of SAVAYSA with rifampin [see Clinical Pharmacology (12.3)].
8.4 Pediatric Use
The safety and effectiveness of SAVAYSA have not been established in pediatric patients with confirmed VTE (PE and/or DVT). Effectiveness was not demonstrated in an adequate and well-controlled study conducted in 145 SAVAYSA-treated pediatric patients, from birth to less than 18 years of age with confirmed VTE (PE and/or DVT), treated for 3 months up to a maximum of 12 months.
8.5 Geriatric Use
Of the total patients in the ENGAGE AF-TIMI 48 study, 5182 (74%) were 65 years and older, while 2838 (41%) were 75 years and older. In Hokusai VTE, 1334 (32%) patients were 65 years and older, while 560 (14%) patients were 75 years and older. In the Hokusai VTE Cancer Study, 539 (52%) patients were 65 years and older and 176 (17%) were 75 years and older. In clinical trials the efficacy and safety of SAVAYSA in elderly (65 years or older) and younger patients were similar [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
4 Contraindications
SAVAYSA is contraindicated in patients with:
- Active pathological bleeding [see Warnings and Precautions (5.3) and Adverse Reactions (6.1)].
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the prescribing information.
- Increased Risk of Stroke with Discontinuation of SAVAYSA in Patients with Nonvalvular Atrial Fibrillation [see Warnings and Precautions (5.2)]
- Risk of Bleeding [see Warnings and Precautions (5.3)]
- Spinal/Epidural Anesthesia or Puncture [see Warnings and Precautions (5.4)]
7 Drug Interactions
5.3 Risk of Bleeding
SAVAYSA increases the risk of bleeding and can cause serious and potentially fatal bleeding. Promptly evaluate any signs or symptoms of blood loss.
Discontinue SAVAYSA in patients with active pathological bleeding.
Concomitant use of drugs affecting hemostasis may increase the risk of bleeding. These include aspirin and other antiplatelet agents, other antithrombotic agents, fibrinolytic therapy, chronic use of nonsteroidal anti-inflammatory drugs (NSAIDs), selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) [see Drug Interactions (7.1)].
8.6 Renal Impairment
Renal clearance accounts for approximately 50% of the total clearance of edoxaban. Consequently, edoxaban blood levels are increased in patients with poor renal function compared to those with higher renal function. Reduce SAVAYSA dose to 30 mg once daily in patients with CrCL 15-50 mL/min. There are limited clinical data with SAVAYSA in patients with CrCL < 15 mL/min; SAVAYSA is therefore not recommended in these patients. Hemodialysis does not significantly contribute to SAVAYSA clearance [see Dosage and Administration (2.1, 2.2) and Clinical Pharmacology (12.3)].
As renal function improves and edoxaban blood levels decrease, the risk for ischemic stroke increases in patients with NVAF [see Indications and Usage (1.1), Dosage and Administration (2.1), and Clinical Studies (14.1)].
12.2 Pharmacodynamics
As a result of FXa inhibition, edoxaban prolongs clotting time tests such as prothrombin time (PT), and activated partial thromboplastin time (aPTT). Changes observed in PT, INR, and aPTT at the expected therapeutic dose, however, are small, subject to a high degree of variability and not useful in monitoring the anticoagulant effect of edoxaban. Following oral administration, peak pharmacodynamic effects are observed within 1-2 hours, which correspond with peak edoxaban concentrations (Cmax).
12.3 Pharmacokinetics
Edoxaban displays approximately dose-proportional pharmacokinetics for doses of 15 to 150 mg and 60 to 120 mg following single and repeat doses, respectively, in healthy subjects.
8.7 Hepatic Impairment
The use of SAVAYSA in patients with moderate or severe hepatic impairment (Child-Pugh B and C) is not recommended as these patients may have intrinsic coagulation abnormalities. No dose reduction is required in patients with mild hepatic impairment (Child-Pugh A) [see Clinical Pharmacology (12.3)].
1 Indications and Usage
SAVAYSA is a factor Xa inhibitor indicated:
To reduce the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF) (1.1)
-
Limitation of Use for NVAF
SAVAYSA should not be used in patients with creatinine clearance (CrCL) > 95 mL/min because of increased risk of ischemic stroke compared to warfarin at the highest dose studied (60 mg) (1.1)
SAVAYSA is indicated for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5 to 10 days of initial therapy with a parenteral anticoagulant (1.2)
12.1 Mechanism of Action
Edoxaban is a selective inhibitor of FXa. It does not require antithrombin III for antithrombotic activity. Edoxaban inhibits free FXa, and prothrombinase activity and inhibits thrombin-induced platelet aggregation. Inhibition of FXa in the coagulation cascade reduces thrombin generation and reduces thrombus formation.
2.6 Administration Options
For patients who are unable to swallow whole tablets, SAVAYSA tablets may be crushed and mixed with 2 to 3 ounces of water and immediately administered by mouth or through a gastric tube. The crushed tablets may also be mixed into applesauce and immediately administered orally [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions
- Bleeding: Serious and potentially fatal bleeding. Promptly evaluate signs and symptoms of blood loss (5.3)
- Mechanical Heart Valves or Moderate to Severe Mitral Stenosis: Use is not recommended (5.5)
- Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: SAVAYSA use not recommended. (5.6)
2 Dosage and Administration
3 Dosage Forms and Strengths
- 60 mg, yellow round shaped, film-coated tablets, debossed with DSC L60 on one side
- 30 mg, pink round shaped, film-coated tablets, debossed with DSC L30 on one side
- 15 mg, orange round shaped, film-coated tablets, debossed with DSC L15 on one side
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of SAVAYSA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: thrombocytopenia
Gastrointestinal disorders: abdominal pain
Immune system disorders: angioedema, hypersensitivity
Nervous system disorders: dizziness, headache
Renal and urinary disorders: anticoagulant-related nephropathy
Skin and subcutaneous tissue disorders: urticaria
8 Use in Specific Populations
2.3 Administration Information
If a dose of SAVAYSA is missed, the dose should be taken as soon as possible on the same day. Dosing should resume the next day according to the normal dosing schedule. The dose should not be doubled to make up for a missed dose.
SAVAYSA can be taken without regard to food [see Clinical Pharmacology (12.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of SAVAYSA was evaluated in the ENGAGE AF-TIMI 48, Hokusai VTE, and Hokusai VTE Cancer studies including 11,530 patients exposed to SAVAYSA 60 mg and 7124 patients exposed to SAVAYSA 30 mg once daily [see Clinical Studies (14)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Advise patients of the following:
2.4 Transition to Or From Savaysa
| From | To | Recommendation |
|---|---|---|
| Warfarin or other Vitamin K Antagonists | SAVAYSA | Discontinue warfarin and start SAVAYSA when the INR is ≤ 2.5 |
| Oral anticoagulants other than warfarin or other Vitamin K Antagonists | SAVAYSA | Discontinue current oral anticoagulant and start SAVAYSA at the time of the next scheduled dose of the other oral anticoagulant |
| Low Molecular Weight Heparin (LMWH) | SAVAYSA | Discontinue LMWH and start SAVAYSA at the time of the next scheduled administration of LMWH |
| Unfractionated heparin | SAVAYSA | Discontinue the infusion and start SAVAYSA 4 hours later |
| From | To | Recommendation |
|---|---|---|
| Abbreviations: INR=International Normalized Ratio | ||
| SAVAYSA | Warfarin | Oral option : For patients taking 60 mg of SAVAYSA, reduce the dose to 30 mg and begin warfarin concomitantly. For patients receiving 30 mg of SAVAYSA, reduce the dose to 15 mg and begin warfarin concomitantly. INR must be measured at least weekly and just prior to the daily dose of SAVAYSA to minimize the influence of SAVAYSA on INR measurements. Once a stable INR ≥ 2.0 is achieved, SAVAYSA should be discontinued and the warfarin continued |
| SAVAYSA | Warfarin | Parenteral option : Discontinue SAVAYSA and administer a parenteral anticoagulant and warfarin at the time of the next scheduled SAVAYSA dose. Once a stable INR ≥ 2.0 is achieved the parenteral anticoagulant should be discontinued and the warfarin continued |
| SAVAYSA | Non-Vitamin-K-Dependent Oral anticoagulants | Discontinue SAVAYSA and start the other oral anticoagulant at the time of the next dose of SAVAYSA |
| SAVAYSA | Parenteral anticoagulants | Discontinue SAVAYSA and start the parenteral anticoagulant at the time of the next dose of SAVAYSA |
2.1 Nonvalvular Atrial Fibrillation
The recommended dose of SAVAYSA is 60 mg taken orally once daily [see Warnings and Precautions (5.1) and Clinical Studies (14.1)]. Assess creatinine clearance, as calculated using the Cockcroft-Gault equation
Reduce SAVAYSA dose to 30 mg once daily in patients with CrCL 15 to 50 mL/min [see Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling
SAVAYSA (edoxaban) is supplied as round shaped, film-coated, non-scored tablets containing edoxaban tosylate equivalent to 60, 30 or 15 mg of SAVAYSA, packaged in bottles and blisters.
| NDC 65597-xxx-yy | ||||||||
|---|---|---|---|---|---|---|---|---|
| Strength | Color | Deboss | yy | |||||
| xxx | Bottle of | Blister of | ||||||
| 30 | 90 | 500 | 10 × 10 10 blister cards of 10 counts
|
10 × 5 5 blister cards of 10 counts
|
||||
| 15 mg | orange | DSC L15 | 201 | 30 | - | - | - | - |
| 30 mg | pink | DSC L30 | 202 | 30 | 90 | 50 | 10 | 05 |
| 60 mg | yellow | DSC L60 | 203 | 30 | 90 | 50 | 10 | 05 |
5.4 Spinal/epidural Anesthesia Or Puncture
When neuraxial anesthesia (spinal/epidural anesthesia) or spinal/epidural puncture is employed, patients treated with antithrombotic agents for prevention of thromboembolic complications are at risk of developing an epidural or spinal hematoma, which can result in long-term or permanent paralysis.
The risk of these events may be increased by the postoperative use of indwelling epidural catheters or the concomitant use of medicinal products affecting hemostasis. Indwelling epidural or intrathecal catheters should not be removed earlier than 12 hours after the last administration of SAVAYSA. The next dose of SAVAYSA should not be administered earlier than 2 hours after the removal of the catheter. The risk may also be increased by traumatic or repeated epidural or spinal puncture.
Monitor patients frequently for signs and symptoms of neurological impairment (e.g., numbness or weakness of the legs, bowel, or bladder dysfunction). If neurological compromise is noted, urgent diagnosis and treatment is necessary. Prior to neuraxial intervention the physician should consider the potential benefit versus the risk in anticoagulated patients or in patients to be anticoagulated for thromboprophylaxis.
8.3 Females and Males of Reproductive Potential
Females of reproductive potential requiring anticoagulation should discuss pregnancy planning with their physician.
The risk of clinically significant uterine bleeding, potentially requiring gynecological surgical interventions, identified with oral anticoagulants including SAVAYSA should be assessed in females of reproductive potential and those with abnormal uterine bleeding.
Principal Display Panel 30 Mg Blister Pack Carton
NDC 65597-202-05
Package contains 5 blister packs
of 10 tablets each.
Savaysa®
(edoxaban) tablets
30 mg
Dispense with medication guide
For Hospital Use Only
Daiichi-Sankyo
Rx only
Principal Display Panel 60 Mg Blister Pack Carton
NDC 65597-203-05
Package contains 5 blister packs
of 10 tablets each.
Savaysa®
(edoxaban) tablets
60 mg
Dispense with medication guide
For Hospital Use Only
Daiichi-Sankyo
Rx only
2.5 Discontinuation for Surgery and Other Interventions
Discontinue SAVAYSA at least 24 hours before invasive or surgical procedures because of the risk of bleeding [see Warnings and Precautions (5.3)].
If surgery cannot be delayed, there is an increased risk of bleeding. This risk of bleeding should be weighed against the urgency of intervention [see Warnings and Precautions (5.3)].
SAVAYSA can be restarted after the surgical or other procedure as soon as adequate hemostasis has been established noting that the time to onset of pharmacodynamic effect is 1-2 hours [see Warnings and Precautions (5.2)]. Administer a parenteral anticoagulant and then switch to oral SAVAYSA, if oral medication cannot be taken during or after surgical intervention.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Edoxaban was not carcinogenic when administered daily to mice and rats by oral gavage for up to 104 weeks. The highest dose tested (500 mg/kg/day) in male and female mice was 3 and 6 times, respectively, the human exposure (AUC) at the human dose of 60 mg/day, and the highest doses tested in male (600/400 mg/kg/day) and female (200 mg/kg/day) rats were 8 and 14 times, respectively, the human exposure at the human dose of 60 mg/day.
Edoxaban and its human-specific metabolite, M-4, were genotoxic in in vitro chromosomal aberration tests but were not genotoxic in the in vitro bacterial reverse mutation (Ames test), in in vitro human lymphocytes micronucleus test, in in vivo rat bone marrow micronucleus test, in in vivo rat liver micronucleus test, and in in vivo unscheduled DNA synthesis tests.
Edoxaban showed no effects on fertility and early embryonic development in rats at doses of up to 1000 mg/kg/day (162 times the human dose of 60 mg/day normalized to body surface area).
1.2 Treatment of Deep Vein Thrombosis and Pulmonary Embolism
SAVAYSA is indicated for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5 to 10 days of initial therapy with a parenteral anticoagulant.
2.2 Treatment of Deep Vein Thrombosis and Pulmonary Embolism
The recommended dose of SAVAYSA is 60 mg taken orally once daily following 5 to 10 days of initial therapy with a parenteral anticoagulant [see Clinical Studies (14.2)].
Reduce SAVAYSA dose to 30 mg once daily in patients with CrCL 15 to 50 mL/min, patients who weigh less than or equal to 60 kg, or patients who are taking certain concomitant P-gp inhibitor medications [see Clinical Studies (14.2)].
7.1 Anticoagulants, Antiplatelets, Thrombolytics, and Ssris/snris
Co-administration of anticoagulants, antiplatelet drugs, thrombolytics and SSRIs or SNRIs may increase the risk of bleeding. Promptly evaluate any signs or symptoms of blood loss if patients are treated concomitantly with anticoagulants, aspirin, other platelet aggregation inhibitors, and/or NSAIDs [see Warnings and Precautions (5.3)].
Long-term concomitant treatment with SAVAYSA and other anticoagulants is not recommended because of increased risk of bleeding [see Warnings and Precautions (5.3)]. Short term co-administration may be needed for patients transitioning to or from SAVAYSA [see Dosage and Administration (2.4)].
In clinical studies with SAVAYSA concomitant use of aspirin (low dose ≤ 100 mg/day) or thienopyridines, and NSAIDs was permitted and resulted in increased rates of Clinically Relevant Bleeding. Carefully monitor for bleeding in patients who require chronic treatment with low dose aspirin and/or NSAIDs [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)].
As with other anticoagulants the possibility may exist that patients are at an increased risk of bleeding in case of concomitant use with SSRIs or SNRIs due to their reported effect on platelets [see Warnings and Precautions (5.3)].
8.8 Low Body Weight Consideration for Patients Treated for Dvt And/or Pe
Based on the clinical experience from the Hokusai VTE study, reduce SAVAYSA dose to 30 mg in patients with body weight less than or equal to 60 kg [see Dosage and Administration (2.2) and Clinical Studies (14.2)].
5.5 Patients With Mechanical Heart Valves Or Moderate to Severe Mitral Stenosis
The safety and efficacy of SAVAYSA has not been studied in patients with mechanical heart valves or moderate to severe mitral stenosis. The use of SAVAYSA is not recommended in these patients [see Clinical Studies (14.1)].
5.1 Reduced Efficacy in Nonvalvular Atrial Fibrillation Patients With Crcl > 95 Ml/min
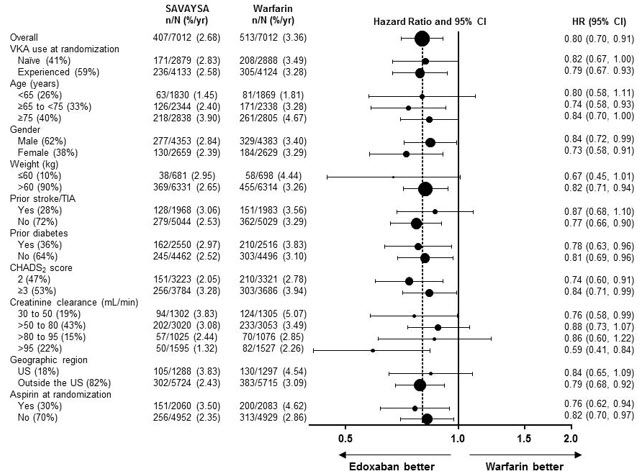
SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the randomized ENGAGE AF-TIMI 48 study, NVAF patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg daily compared to patients treated with warfarin. In these patients another anticoagulant should be used [see Dosage and Administration (2.1) and Clinical Studies (14.1)].
5.6 Increased Risk of Thrombosis in Patients With Triple Positive Antiphospholipid Syndrome
Direct-acting oral anticoagulants (DOACs), including SAVAYSA, are not recommended for use in patients with triple positive antiphospholipid syndrome (APS). For patients with APS (especially those who are triple positive [positive for lupus anticoagulant, anticardiolipin antibodies, and anti-beta 2-glycoprotein I antibodies]), treatment with DOACs has been associated with increased rates of recurrent thrombotic events compared with vitamin K antagonist therapy.
1.1 Reduction in the Risk of Stroke and Systemic Embolism in Nonvalvular Atrial Fibrillation
SAVAYSA is indicated to reduce the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF).
5.2 Increased Risk of Stroke With Discontinuation of Savaysa in Patients With Nonvalvular Atrial Fibrillation
Premature discontinuation of any oral anticoagulant in the absence of adequate alternative anticoagulation increases the risk of ischemic events. If SAVAYSA is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant as described in the transition guidance [see Dosage and Administration (2.4) and Clinical Studies (14.1)].
Warning: (a) Reduced Efficacy in Nonvalvular Atrial Fibrillation Patients With Creatinine Clearance (crcl) > 95 Ml/min (b) Premature Discontinuation of Savaysa Increases the Risk of Ischemic Events (c) Spinal/epidural Hematoma
WARNING: (A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CREATININE CLEARANCE (CRCL) > 95 ML/MIN (B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS (C) SPINAL/EPIDURAL HEMATOMA
See full prescribing information for complete boxed warning.
(A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CRCL > 95 ML/MIN: SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the ENGAGE AF-TIMI 48 study, nonvalvular atrial fibrillation patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg once daily compared to patients treated with warfarin. In these patients another anticoagulant should be used (5.1).
(B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS: Premature discontinuation of any oral anticoagulant in the absence of adequate alternative anticoagulation increases the risk of ischemic events. If SAVAYSA is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant as described in the transition guidance (2.4, 5.2, 14).
(C) SPINAL/EPIDURAL HEMATOMA: Epidural or spinal hematomas may occur in patients treated with SAVAYSA who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures (5.4).
Structured Label Content
Section 42229-5 (42229-5)
A. REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CRCL > 95 ML/MIN
SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the ENGAGE AF-TIMI 48 study, nonvalvular atrial fibrillation patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg once daily compared to patients treated with warfarin. In these patients another anticoagulant should be used [see Dosage and Administration (2.1), Warnings and Precautions (5.1), and Clinical Studies (14.1)].
Section 42231-1 (42231-1)
| MEDICATION GUIDE SAVAYSA (sa vaye' sah) (edoxaban) tablets |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 10/2023 |
What is the most important information I should know about SAVAYSA?
|
|
|
What is SAVAYSA?
SAVAYSA is a prescription medicine used to:
|
|
|
Who should not take SAVAYSA?
Do not take SAVAYSA if you currently have certain types of abnormal bleeding. |
|
Before taking SAVAYSA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some of your other medicines may affect the way SAVAYSA works. Certain medicines may increase your risk of bleeding or stroke when taken with SAVAYSA. See "What is the most important information I should know about SAVAYSA?" Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
How should I take SAVAYSA?
|
|
|
What are the possible side effects of SAVAYSA? SAVAYSA can cause serious side effects. See "What is the most important information I should know about SAVAYSA?" The most common side effects in people who take SAVAYSA for nonvalvular atrial fibrillation include bleeding and low red blood cell count (anemia). The most common side effects in people who take SAVAYSA for deep vein thrombosis and pulmonary embolism include bleeding, rash, abnormal liver function tests, and low red blood cell count (anemia). Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store SAVAYSA?
|
|
|
General information about the safe and effective use of SAVAYSA
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use SAVAYSA for a condition for which it was not prescribed. Do not give SAVAYSA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about SAVAYSA that is written for health professionals. |
|
|
What are the ingredients in SAVAYSA?
Active ingredient: edoxaban tosylate monohydrate Inactive ingredients: mannitol, pregelatinized starch, crospovidone, hydroxypropyl cellulose, magnesium stearate, talc, and carnauba wax. The color coatings contain hypromellose, titanium dioxide, talc, polyethylene glycol 8000, iron oxide yellow (60 mg tablets and 15 mg tablets), and iron oxide red (30 mg tablets and 15 mg tablets). Manufactured by: Daiichi Sankyo Co., LTD., Tokyo 103-8426, Japan Distributed by: Daiichi Sankyo, Inc., Basking Ridge, NJ 07920-2311 USA The brands listed above are trademarks of their respective owners. Copyright© 2023, Daiichi Sankyo, Inc. USMG-SAV-C9-1023-r103 For more information, call 1-877-437-7763 or go to https://savaysa.com. |
Section 44425-7 (44425-7)
Store at 20-25°C (68-77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature].
Keep out of the reach of children.
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL
NDC 65597-201-30
Savaysa
(edoxaban) tablets
15 mg
30 TABLETS
Rx Only
PRINCIPAL DISPLAY PANEL
NDC 65597-202-30
Savaysa
(edoxaban) tablets
30 mg
30 TABLETS
Rx Only
10 Overdosage (10 OVERDOSAGE)
A specific reversal agent for edoxaban is not available. Overdose of SAVAYSA increases the risk of bleeding.
The following are not expected to reverse the anticoagulant effects of edoxaban: protamine sulfate, vitamin K, and tranexamic acid [see Warnings and Precautions (5.3)].
Hemodialysis does not significantly contribute to edoxaban clearance [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Edoxaban, a factor Xa inhibitor, is supplied as edoxaban tosylate monohydrate. The chemical name is N-(5-Chloropyridin-2-yl)-N'-[(1S,2R,4S)-4-(N,N-dimethylcarbamoyl)-2-(5-methyl-4,5,6,7-tetrahydro[1,3]thiazolo[5,4-c]pyridine-2-carboxamido)cyclohexyl] oxamide mono (4-methylbenzenesulfonate) monohydrate. Edoxaban tosylate monohydrate has the empirical formula C24H30ClN7O4S∙C7H8O3S∙H2O representing a molecular weight of 738.27. The chemical structure of edoxaban tosylate monohydrate is:
It is a white to pale yellowish-white crystalline powder. The solubility of edoxaban tosylate (pKa 6.7) decreases with increasing pH. It is slightly soluble in water, pH 3 to 5 buffer, very slightly soluble at pH 6 to 7; and practically insoluble at pH 8 to 9.
SAVAYSA is available for oral administration as a 60 mg, 30 mg, or 15 mg round shaped, film-coated tablet, debossed with product identification markings. Each 60 mg tablet contains 80.82 mg edoxaban tosylate monohydrate equivalent to 60 mg of edoxaban. Each 30 mg tablet contains 40.41 mg edoxaban tosylate monohydrate equivalent to 30 mg of edoxaban. Each 15 mg tablet contains 20.20 mg edoxaban tosylate monohydrate equivalent to 15 mg of edoxaban. The inactive ingredients are: mannitol, pregelatinized starch, crospovidone, hydroxypropyl cellulose, magnesium stearate, talc, and carnauba wax. The color coatings contain hypromellose, titanium dioxide, talc, polyethylene glycol 8000, iron oxide yellow (60 mg tablets and 15 mg tablets), and iron oxide red (30 mg tablets and 15 mg tablets).
7.2 P Gp Inducers (7.2 P-gp Inducers)
Avoid the concomitant use of SAVAYSA with rifampin [see Clinical Pharmacology (12.3)].
8.4 Pediatric Use
The safety and effectiveness of SAVAYSA have not been established in pediatric patients with confirmed VTE (PE and/or DVT). Effectiveness was not demonstrated in an adequate and well-controlled study conducted in 145 SAVAYSA-treated pediatric patients, from birth to less than 18 years of age with confirmed VTE (PE and/or DVT), treated for 3 months up to a maximum of 12 months.
8.5 Geriatric Use
Of the total patients in the ENGAGE AF-TIMI 48 study, 5182 (74%) were 65 years and older, while 2838 (41%) were 75 years and older. In Hokusai VTE, 1334 (32%) patients were 65 years and older, while 560 (14%) patients were 75 years and older. In the Hokusai VTE Cancer Study, 539 (52%) patients were 65 years and older and 176 (17%) were 75 years and older. In clinical trials the efficacy and safety of SAVAYSA in elderly (65 years or older) and younger patients were similar [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
4 Contraindications (4 CONTRAINDICATIONS)
SAVAYSA is contraindicated in patients with:
- Active pathological bleeding [see Warnings and Precautions (5.3) and Adverse Reactions (6.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in greater detail in other sections of the prescribing information.
- Increased Risk of Stroke with Discontinuation of SAVAYSA in Patients with Nonvalvular Atrial Fibrillation [see Warnings and Precautions (5.2)]
- Risk of Bleeding [see Warnings and Precautions (5.3)]
- Spinal/Epidural Anesthesia or Puncture [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.3 Risk of Bleeding
SAVAYSA increases the risk of bleeding and can cause serious and potentially fatal bleeding. Promptly evaluate any signs or symptoms of blood loss.
Discontinue SAVAYSA in patients with active pathological bleeding.
Concomitant use of drugs affecting hemostasis may increase the risk of bleeding. These include aspirin and other antiplatelet agents, other antithrombotic agents, fibrinolytic therapy, chronic use of nonsteroidal anti-inflammatory drugs (NSAIDs), selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) [see Drug Interactions (7.1)].
8.6 Renal Impairment
Renal clearance accounts for approximately 50% of the total clearance of edoxaban. Consequently, edoxaban blood levels are increased in patients with poor renal function compared to those with higher renal function. Reduce SAVAYSA dose to 30 mg once daily in patients with CrCL 15-50 mL/min. There are limited clinical data with SAVAYSA in patients with CrCL < 15 mL/min; SAVAYSA is therefore not recommended in these patients. Hemodialysis does not significantly contribute to SAVAYSA clearance [see Dosage and Administration (2.1, 2.2) and Clinical Pharmacology (12.3)].
As renal function improves and edoxaban blood levels decrease, the risk for ischemic stroke increases in patients with NVAF [see Indications and Usage (1.1), Dosage and Administration (2.1), and Clinical Studies (14.1)].
12.2 Pharmacodynamics
As a result of FXa inhibition, edoxaban prolongs clotting time tests such as prothrombin time (PT), and activated partial thromboplastin time (aPTT). Changes observed in PT, INR, and aPTT at the expected therapeutic dose, however, are small, subject to a high degree of variability and not useful in monitoring the anticoagulant effect of edoxaban. Following oral administration, peak pharmacodynamic effects are observed within 1-2 hours, which correspond with peak edoxaban concentrations (Cmax).
12.3 Pharmacokinetics
Edoxaban displays approximately dose-proportional pharmacokinetics for doses of 15 to 150 mg and 60 to 120 mg following single and repeat doses, respectively, in healthy subjects.
8.7 Hepatic Impairment
The use of SAVAYSA in patients with moderate or severe hepatic impairment (Child-Pugh B and C) is not recommended as these patients may have intrinsic coagulation abnormalities. No dose reduction is required in patients with mild hepatic impairment (Child-Pugh A) [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
SAVAYSA is a factor Xa inhibitor indicated:
To reduce the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF) (1.1)
-
Limitation of Use for NVAF
SAVAYSA should not be used in patients with creatinine clearance (CrCL) > 95 mL/min because of increased risk of ischemic stroke compared to warfarin at the highest dose studied (60 mg) (1.1)
SAVAYSA is indicated for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5 to 10 days of initial therapy with a parenteral anticoagulant (1.2)
12.1 Mechanism of Action
Edoxaban is a selective inhibitor of FXa. It does not require antithrombin III for antithrombotic activity. Edoxaban inhibits free FXa, and prothrombinase activity and inhibits thrombin-induced platelet aggregation. Inhibition of FXa in the coagulation cascade reduces thrombin generation and reduces thrombus formation.
2.6 Administration Options
For patients who are unable to swallow whole tablets, SAVAYSA tablets may be crushed and mixed with 2 to 3 ounces of water and immediately administered by mouth or through a gastric tube. The crushed tablets may also be mixed into applesauce and immediately administered orally [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Bleeding: Serious and potentially fatal bleeding. Promptly evaluate signs and symptoms of blood loss (5.3)
- Mechanical Heart Valves or Moderate to Severe Mitral Stenosis: Use is not recommended (5.5)
- Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: SAVAYSA use not recommended. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 60 mg, yellow round shaped, film-coated tablets, debossed with DSC L60 on one side
- 30 mg, pink round shaped, film-coated tablets, debossed with DSC L30 on one side
- 15 mg, orange round shaped, film-coated tablets, debossed with DSC L15 on one side
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of SAVAYSA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: thrombocytopenia
Gastrointestinal disorders: abdominal pain
Immune system disorders: angioedema, hypersensitivity
Nervous system disorders: dizziness, headache
Renal and urinary disorders: anticoagulant-related nephropathy
Skin and subcutaneous tissue disorders: urticaria
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
2.3 Administration Information
If a dose of SAVAYSA is missed, the dose should be taken as soon as possible on the same day. Dosing should resume the next day according to the normal dosing schedule. The dose should not be doubled to make up for a missed dose.
SAVAYSA can be taken without regard to food [see Clinical Pharmacology (12.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of SAVAYSA was evaluated in the ENGAGE AF-TIMI 48, Hokusai VTE, and Hokusai VTE Cancer studies including 11,530 patients exposed to SAVAYSA 60 mg and 7124 patients exposed to SAVAYSA 30 mg once daily [see Clinical Studies (14)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Advise patients of the following:
2.4 Transition to Or From Savaysa (2.4 Transition to or from SAVAYSA)
| From | To | Recommendation |
|---|---|---|
| Warfarin or other Vitamin K Antagonists | SAVAYSA | Discontinue warfarin and start SAVAYSA when the INR is ≤ 2.5 |
| Oral anticoagulants other than warfarin or other Vitamin K Antagonists | SAVAYSA | Discontinue current oral anticoagulant and start SAVAYSA at the time of the next scheduled dose of the other oral anticoagulant |
| Low Molecular Weight Heparin (LMWH) | SAVAYSA | Discontinue LMWH and start SAVAYSA at the time of the next scheduled administration of LMWH |
| Unfractionated heparin | SAVAYSA | Discontinue the infusion and start SAVAYSA 4 hours later |
| From | To | Recommendation |
|---|---|---|
| Abbreviations: INR=International Normalized Ratio | ||
| SAVAYSA | Warfarin | Oral option : For patients taking 60 mg of SAVAYSA, reduce the dose to 30 mg and begin warfarin concomitantly. For patients receiving 30 mg of SAVAYSA, reduce the dose to 15 mg and begin warfarin concomitantly. INR must be measured at least weekly and just prior to the daily dose of SAVAYSA to minimize the influence of SAVAYSA on INR measurements. Once a stable INR ≥ 2.0 is achieved, SAVAYSA should be discontinued and the warfarin continued |
| SAVAYSA | Warfarin | Parenteral option : Discontinue SAVAYSA and administer a parenteral anticoagulant and warfarin at the time of the next scheduled SAVAYSA dose. Once a stable INR ≥ 2.0 is achieved the parenteral anticoagulant should be discontinued and the warfarin continued |
| SAVAYSA | Non-Vitamin-K-Dependent Oral anticoagulants | Discontinue SAVAYSA and start the other oral anticoagulant at the time of the next dose of SAVAYSA |
| SAVAYSA | Parenteral anticoagulants | Discontinue SAVAYSA and start the parenteral anticoagulant at the time of the next dose of SAVAYSA |
2.1 Nonvalvular Atrial Fibrillation
The recommended dose of SAVAYSA is 60 mg taken orally once daily [see Warnings and Precautions (5.1) and Clinical Studies (14.1)]. Assess creatinine clearance, as calculated using the Cockcroft-Gault equation
Reduce SAVAYSA dose to 30 mg once daily in patients with CrCL 15 to 50 mL/min [see Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
SAVAYSA (edoxaban) is supplied as round shaped, film-coated, non-scored tablets containing edoxaban tosylate equivalent to 60, 30 or 15 mg of SAVAYSA, packaged in bottles and blisters.
| NDC 65597-xxx-yy | ||||||||
|---|---|---|---|---|---|---|---|---|
| Strength | Color | Deboss | yy | |||||
| xxx | Bottle of | Blister of | ||||||
| 30 | 90 | 500 | 10 × 10 10 blister cards of 10 counts
|
10 × 5 5 blister cards of 10 counts
|
||||
| 15 mg | orange | DSC L15 | 201 | 30 | - | - | - | - |
| 30 mg | pink | DSC L30 | 202 | 30 | 90 | 50 | 10 | 05 |
| 60 mg | yellow | DSC L60 | 203 | 30 | 90 | 50 | 10 | 05 |
5.4 Spinal/epidural Anesthesia Or Puncture (5.4 Spinal/Epidural Anesthesia or Puncture)
When neuraxial anesthesia (spinal/epidural anesthesia) or spinal/epidural puncture is employed, patients treated with antithrombotic agents for prevention of thromboembolic complications are at risk of developing an epidural or spinal hematoma, which can result in long-term or permanent paralysis.
The risk of these events may be increased by the postoperative use of indwelling epidural catheters or the concomitant use of medicinal products affecting hemostasis. Indwelling epidural or intrathecal catheters should not be removed earlier than 12 hours after the last administration of SAVAYSA. The next dose of SAVAYSA should not be administered earlier than 2 hours after the removal of the catheter. The risk may also be increased by traumatic or repeated epidural or spinal puncture.
Monitor patients frequently for signs and symptoms of neurological impairment (e.g., numbness or weakness of the legs, bowel, or bladder dysfunction). If neurological compromise is noted, urgent diagnosis and treatment is necessary. Prior to neuraxial intervention the physician should consider the potential benefit versus the risk in anticoagulated patients or in patients to be anticoagulated for thromboprophylaxis.
8.3 Females and Males of Reproductive Potential
Females of reproductive potential requiring anticoagulation should discuss pregnancy planning with their physician.
The risk of clinically significant uterine bleeding, potentially requiring gynecological surgical interventions, identified with oral anticoagulants including SAVAYSA should be assessed in females of reproductive potential and those with abnormal uterine bleeding.
Principal Display Panel 30 Mg Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 30 mg Blister Pack Carton)
NDC 65597-202-05
Package contains 5 blister packs
of 10 tablets each.
Savaysa®
(edoxaban) tablets
30 mg
Dispense with medication guide
For Hospital Use Only
Daiichi-Sankyo
Rx only
Principal Display Panel 60 Mg Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 60 mg Blister Pack Carton)
NDC 65597-203-05
Package contains 5 blister packs
of 10 tablets each.
Savaysa®
(edoxaban) tablets
60 mg
Dispense with medication guide
For Hospital Use Only
Daiichi-Sankyo
Rx only
2.5 Discontinuation for Surgery and Other Interventions
Discontinue SAVAYSA at least 24 hours before invasive or surgical procedures because of the risk of bleeding [see Warnings and Precautions (5.3)].
If surgery cannot be delayed, there is an increased risk of bleeding. This risk of bleeding should be weighed against the urgency of intervention [see Warnings and Precautions (5.3)].
SAVAYSA can be restarted after the surgical or other procedure as soon as adequate hemostasis has been established noting that the time to onset of pharmacodynamic effect is 1-2 hours [see Warnings and Precautions (5.2)]. Administer a parenteral anticoagulant and then switch to oral SAVAYSA, if oral medication cannot be taken during or after surgical intervention.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Edoxaban was not carcinogenic when administered daily to mice and rats by oral gavage for up to 104 weeks. The highest dose tested (500 mg/kg/day) in male and female mice was 3 and 6 times, respectively, the human exposure (AUC) at the human dose of 60 mg/day, and the highest doses tested in male (600/400 mg/kg/day) and female (200 mg/kg/day) rats were 8 and 14 times, respectively, the human exposure at the human dose of 60 mg/day.
Edoxaban and its human-specific metabolite, M-4, were genotoxic in in vitro chromosomal aberration tests but were not genotoxic in the in vitro bacterial reverse mutation (Ames test), in in vitro human lymphocytes micronucleus test, in in vivo rat bone marrow micronucleus test, in in vivo rat liver micronucleus test, and in in vivo unscheduled DNA synthesis tests.
Edoxaban showed no effects on fertility and early embryonic development in rats at doses of up to 1000 mg/kg/day (162 times the human dose of 60 mg/day normalized to body surface area).
1.2 Treatment of Deep Vein Thrombosis and Pulmonary Embolism
SAVAYSA is indicated for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5 to 10 days of initial therapy with a parenteral anticoagulant.
2.2 Treatment of Deep Vein Thrombosis and Pulmonary Embolism
The recommended dose of SAVAYSA is 60 mg taken orally once daily following 5 to 10 days of initial therapy with a parenteral anticoagulant [see Clinical Studies (14.2)].
Reduce SAVAYSA dose to 30 mg once daily in patients with CrCL 15 to 50 mL/min, patients who weigh less than or equal to 60 kg, or patients who are taking certain concomitant P-gp inhibitor medications [see Clinical Studies (14.2)].
7.1 Anticoagulants, Antiplatelets, Thrombolytics, and Ssris/snris (7.1 Anticoagulants, Antiplatelets, Thrombolytics, and SSRIs/SNRIs)
Co-administration of anticoagulants, antiplatelet drugs, thrombolytics and SSRIs or SNRIs may increase the risk of bleeding. Promptly evaluate any signs or symptoms of blood loss if patients are treated concomitantly with anticoagulants, aspirin, other platelet aggregation inhibitors, and/or NSAIDs [see Warnings and Precautions (5.3)].
Long-term concomitant treatment with SAVAYSA and other anticoagulants is not recommended because of increased risk of bleeding [see Warnings and Precautions (5.3)]. Short term co-administration may be needed for patients transitioning to or from SAVAYSA [see Dosage and Administration (2.4)].
In clinical studies with SAVAYSA concomitant use of aspirin (low dose ≤ 100 mg/day) or thienopyridines, and NSAIDs was permitted and resulted in increased rates of Clinically Relevant Bleeding. Carefully monitor for bleeding in patients who require chronic treatment with low dose aspirin and/or NSAIDs [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)].
As with other anticoagulants the possibility may exist that patients are at an increased risk of bleeding in case of concomitant use with SSRIs or SNRIs due to their reported effect on platelets [see Warnings and Precautions (5.3)].
8.8 Low Body Weight Consideration for Patients Treated for Dvt And/or Pe (8.8 Low Body Weight Consideration for Patients Treated for DVT and/or PE)
Based on the clinical experience from the Hokusai VTE study, reduce SAVAYSA dose to 30 mg in patients with body weight less than or equal to 60 kg [see Dosage and Administration (2.2) and Clinical Studies (14.2)].
5.5 Patients With Mechanical Heart Valves Or Moderate to Severe Mitral Stenosis (5.5 Patients with Mechanical Heart Valves or Moderate to Severe Mitral Stenosis)
The safety and efficacy of SAVAYSA has not been studied in patients with mechanical heart valves or moderate to severe mitral stenosis. The use of SAVAYSA is not recommended in these patients [see Clinical Studies (14.1)].
5.1 Reduced Efficacy in Nonvalvular Atrial Fibrillation Patients With Crcl > 95 Ml/min (5.1 Reduced Efficacy in Nonvalvular Atrial Fibrillation Patients with CrCL > 95 mL/min)
SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the randomized ENGAGE AF-TIMI 48 study, NVAF patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg daily compared to patients treated with warfarin. In these patients another anticoagulant should be used [see Dosage and Administration (2.1) and Clinical Studies (14.1)].
5.6 Increased Risk of Thrombosis in Patients With Triple Positive Antiphospholipid Syndrome (5.6 Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome)
Direct-acting oral anticoagulants (DOACs), including SAVAYSA, are not recommended for use in patients with triple positive antiphospholipid syndrome (APS). For patients with APS (especially those who are triple positive [positive for lupus anticoagulant, anticardiolipin antibodies, and anti-beta 2-glycoprotein I antibodies]), treatment with DOACs has been associated with increased rates of recurrent thrombotic events compared with vitamin K antagonist therapy.
1.1 Reduction in the Risk of Stroke and Systemic Embolism in Nonvalvular Atrial Fibrillation
SAVAYSA is indicated to reduce the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF).
5.2 Increased Risk of Stroke With Discontinuation of Savaysa in Patients With Nonvalvular Atrial Fibrillation (5.2 Increased Risk of Stroke with Discontinuation of SAVAYSA in Patients with Nonvalvular Atrial Fibrillation)
Premature discontinuation of any oral anticoagulant in the absence of adequate alternative anticoagulation increases the risk of ischemic events. If SAVAYSA is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant as described in the transition guidance [see Dosage and Administration (2.4) and Clinical Studies (14.1)].
Warning: (a) Reduced Efficacy in Nonvalvular Atrial Fibrillation Patients With Creatinine Clearance (crcl) > 95 Ml/min (b) Premature Discontinuation of Savaysa Increases the Risk of Ischemic Events (c) Spinal/epidural Hematoma (WARNING: (A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CREATININE CLEARANCE (CRCL) > 95 ML/MIN (B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS (C) SPINAL/EPIDURAL HEMATOMA)
WARNING: (A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CREATININE CLEARANCE (CRCL) > 95 ML/MIN (B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS (C) SPINAL/EPIDURAL HEMATOMA
See full prescribing information for complete boxed warning.
(A) REDUCED EFFICACY IN NONVALVULAR ATRIAL FIBRILLATION PATIENTS WITH CRCL > 95 ML/MIN: SAVAYSA should not be used in patients with CrCL > 95 mL/min. In the ENGAGE AF-TIMI 48 study, nonvalvular atrial fibrillation patients with CrCL > 95 mL/min had an increased rate of ischemic stroke with SAVAYSA 60 mg once daily compared to patients treated with warfarin. In these patients another anticoagulant should be used (5.1).
(B) PREMATURE DISCONTINUATION OF SAVAYSA INCREASES THE RISK OF ISCHEMIC EVENTS: Premature discontinuation of any oral anticoagulant in the absence of adequate alternative anticoagulation increases the risk of ischemic events. If SAVAYSA is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant as described in the transition guidance (2.4, 5.2, 14).
(C) SPINAL/EPIDURAL HEMATOMA: Epidural or spinal hematomas may occur in patients treated with SAVAYSA who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures (5.4).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:10.848589 · Updated: 2026-03-14T22:29:13.416831