Gly-sal®
e73bd26c-cdc2-47ab-94e8-3bc49a9b19e8
34390-5
HUMAN OTC DRUG LABEL
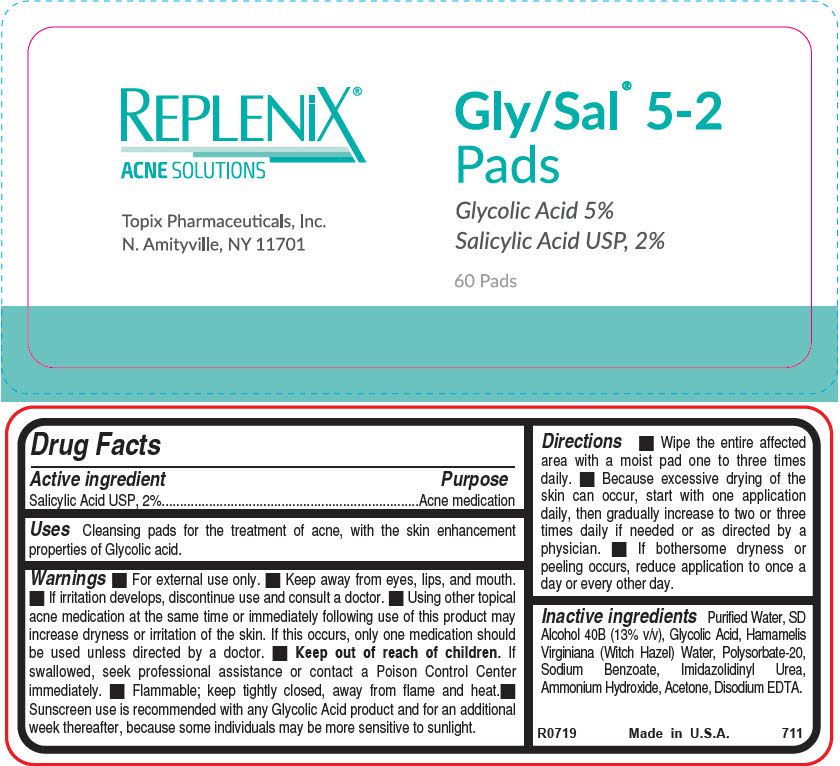
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Acne medication
Medication Information
Purpose
Acne medication
Description
Drug Facts
Uses
Cleansing pads for the treatment of acne, with the skin enhancement properties of Glycolic acid.
Section 42229-5
Drug Facts
Section 50565-1
- Keep out of reach of children. If swallowed, seek professional assistance or contact a Poison Control Center immediately.
Section 50566-9
- If irritation develops, discontinue use and consult a doctor.
Warnings
- For external use only.
- Keep away from eyes, lips, and mouth.
Directions
- Wipe the entire affected area with a moist pad one to three times daily.
- Because excessive drying of the skin can occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a physician.
- If bothersome dryness or peeling occurs, reduce application to once a day or every other day.
Active Ingredient
Salicylic Acid USP, 2%
Inactive Ingredients
Purified Water, SD Alcohol 40B (13% v/v), Glycolic Acid, Hamamelis Virginiana (Witch Hazel) Water, Polysorbate-20, Sodium Benzoate, Imidazolidinyl Urea, Ammonium Hydroxide, Acetone, Disodium EDTA.
Principal Display Panel 60 Pad Jar Label
REPLENiX®
ACNE SOLUTIONS
Topix Pharmaceuticals, Inc.
N. Amityville, NY 11701
Gly/Sal® 5-2
Pads
Glycolic Acid 5%
Salicylic Acid USP, 2%
60 Pads
Structured Label Content
Uses
Cleansing pads for the treatment of acne, with the skin enhancement properties of Glycolic acid.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
- Keep out of reach of children. If swallowed, seek professional assistance or contact a Poison Control Center immediately.
Section 50566-9 (50566-9)
- If irritation develops, discontinue use and consult a doctor.
Purpose
Acne medication
Warnings
- For external use only.
- Keep away from eyes, lips, and mouth.
Directions
- Wipe the entire affected area with a moist pad one to three times daily.
- Because excessive drying of the skin can occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a physician.
- If bothersome dryness or peeling occurs, reduce application to once a day or every other day.
Active Ingredient (Active ingredient)
Salicylic Acid USP, 2%
Inactive Ingredients (Inactive ingredients)
Purified Water, SD Alcohol 40B (13% v/v), Glycolic Acid, Hamamelis Virginiana (Witch Hazel) Water, Polysorbate-20, Sodium Benzoate, Imidazolidinyl Urea, Ammonium Hydroxide, Acetone, Disodium EDTA.
Principal Display Panel 60 Pad Jar Label (PRINCIPAL DISPLAY PANEL - 60 Pad Jar Label)
REPLENiX®
ACNE SOLUTIONS
Topix Pharmaceuticals, Inc.
N. Amityville, NY 11701
Gly/Sal® 5-2
Pads
Glycolic Acid 5%
Salicylic Acid USP, 2%
60 Pads
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:17.380546 · Updated: 2026-03-14T23:01:45.300207