These Highlights Do Not Include All The Information Needed To Use Steglatro Safely And Effectively. See Full Prescribing Information For Steglatro.
e6f3e718-bb99-48f1-ab94-b9f0af05fed6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.3 ) 12/2024 Warnings and Precautions ( 5.2 , 5.5 ) 12/2024
Indications and Usage
STEGLATRO ® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Dosage and Administration
Assess renal function before initiating and as clinically indicated. ( 2.1 ) Correct volume depletion before initiating STEGLATRO. ( 2.1 ) Recommended starting dosage is 5 mg orally once daily, taken in the morning, with or without food. ( 2.2 ) Increase dosage to 15 mg orally once daily in those tolerating STEGLATRO and needing additional glycemic control. ( 2.2 ) Use is not recommended in patients with an estimated glomerular filtration rate (eGFR) less than 45 mL/min/1.73 m 2 . ( 2.2 ) Withhold STEGLATRO for at least 4 days, if possible, prior to surgery or procedures associated with prolonged fasting. ( 2.3 )
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue STEGLATRO if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. ( 5.1 ) Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and discontinue if these occur. ( 5.2 ) Volume Depletion: May result in acute kidney injury. Before initiating, assess and correct volume status in patients with renal impairment or low systolic blood pressure, elderly patients, or patients on diuretics. Monitor for signs and symptoms during therapy. ( 5.3 ) Urosepsis and Pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated. ( 5.4 ) Hypoglycemia: Consider a lower dose of insulin or insulin secretagogue to reduce risk of hypoglycemia when used in combination. ( 5.5 ) Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment. ( 5.6 ) Genital Mycotic Infections: Monitor and treat if indicated. ( 5.7 )
Contraindications
STEGLATRO is contraindicated in patients with hypersensitivity to ertugliflozin or any excipient in STEGLATRO. Reactions such as angioedema have occurred [see Adverse Reactions (6.2) ].
Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1) ] Lower Limb Amputation [see Warnings and Precautions (5.2) ] Volume Depletion [see Warnings and Precautions (5.3) ] Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4) ] Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5) ] Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.6) ] Genital Mycotic Infections [see Warnings and Precautions (5.7) ]
Drug Interactions
Table 3: Clinically Significant Drug Interactions with STEGLATRO Insulin or Insulin Secretagogues Clinical Impact: The risk of hypoglycemia is increased when STEGLATRO is used in combination with insulin or an insulin secretagogue. Intervention: A lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with STEGLATRO. Lithium Clinical Impact: Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Intervention: Monitor serum lithium concentration more frequently during STEGLATRO initiation and dosage changes. Positive Urine Glucose Test Clinical Impact: SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. Intervention: Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. Interference with 1,5-anhydroglucitol (1,5-AG) Assay Clinical Impact: Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. Intervention: Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control.
Storage and Handling
STEGLATRO (ertugliflozin) tablets are available as follows: Strength Description How Supplied NDC 5 mg tablets pink, triangular-shaped, biconvex tablets, with “701” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5363-03 unit-of-use bottles of 90 0006-5363-06 15 mg tablets red, triangular-shaped, biconvex tablets, with “702” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5364-03 unit-of-use bottles of 90 0006-5364-06
How Supplied
STEGLATRO (ertugliflozin) tablets are available as follows: Strength Description How Supplied NDC 5 mg tablets pink, triangular-shaped, biconvex tablets, with “701” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5363-03 unit-of-use bottles of 90 0006-5363-06 15 mg tablets red, triangular-shaped, biconvex tablets, with “702” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5364-03 unit-of-use bottles of 90 0006-5364-06
Medication Information
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue STEGLATRO if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. ( 5.1 ) Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and discontinue if these occur. ( 5.2 ) Volume Depletion: May result in acute kidney injury. Before initiating, assess and correct volume status in patients with renal impairment or low systolic blood pressure, elderly patients, or patients on diuretics. Monitor for signs and symptoms during therapy. ( 5.3 ) Urosepsis and Pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated. ( 5.4 ) Hypoglycemia: Consider a lower dose of insulin or insulin secretagogue to reduce risk of hypoglycemia when used in combination. ( 5.5 ) Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment. ( 5.6 ) Genital Mycotic Infections: Monitor and treat if indicated. ( 5.7 )
Indications and Usage
STEGLATRO ® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Dosage and Administration
Assess renal function before initiating and as clinically indicated. ( 2.1 ) Correct volume depletion before initiating STEGLATRO. ( 2.1 ) Recommended starting dosage is 5 mg orally once daily, taken in the morning, with or without food. ( 2.2 ) Increase dosage to 15 mg orally once daily in those tolerating STEGLATRO and needing additional glycemic control. ( 2.2 ) Use is not recommended in patients with an estimated glomerular filtration rate (eGFR) less than 45 mL/min/1.73 m 2 . ( 2.2 ) Withhold STEGLATRO for at least 4 days, if possible, prior to surgery or procedures associated with prolonged fasting. ( 2.3 )
Contraindications
STEGLATRO is contraindicated in patients with hypersensitivity to ertugliflozin or any excipient in STEGLATRO. Reactions such as angioedema have occurred [see Adverse Reactions (6.2) ].
Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1) ] Lower Limb Amputation [see Warnings and Precautions (5.2) ] Volume Depletion [see Warnings and Precautions (5.3) ] Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4) ] Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5) ] Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.6) ] Genital Mycotic Infections [see Warnings and Precautions (5.7) ]
Drug Interactions
Table 3: Clinically Significant Drug Interactions with STEGLATRO Insulin or Insulin Secretagogues Clinical Impact: The risk of hypoglycemia is increased when STEGLATRO is used in combination with insulin or an insulin secretagogue. Intervention: A lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with STEGLATRO. Lithium Clinical Impact: Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Intervention: Monitor serum lithium concentration more frequently during STEGLATRO initiation and dosage changes. Positive Urine Glucose Test Clinical Impact: SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. Intervention: Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. Interference with 1,5-anhydroglucitol (1,5-AG) Assay Clinical Impact: Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. Intervention: Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control.
Storage and Handling
STEGLATRO (ertugliflozin) tablets are available as follows: Strength Description How Supplied NDC 5 mg tablets pink, triangular-shaped, biconvex tablets, with “701” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5363-03 unit-of-use bottles of 90 0006-5363-06 15 mg tablets red, triangular-shaped, biconvex tablets, with “702” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5364-03 unit-of-use bottles of 90 0006-5364-06
How Supplied
STEGLATRO (ertugliflozin) tablets are available as follows: Strength Description How Supplied NDC 5 mg tablets pink, triangular-shaped, biconvex tablets, with “701” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5363-03 unit-of-use bottles of 90 0006-5363-06 15 mg tablets red, triangular-shaped, biconvex tablets, with “702” debossed on one side and plain on the other side unit-of-use bottles of 30 0006-5364-03 unit-of-use bottles of 90 0006-5364-06
Description
Dosage and Administration ( 2.3 ) 12/2024 Warnings and Precautions ( 5.2 , 5.5 ) 12/2024
Section 42229-5
Limitations of Use
- Not recommended for use to improve glycemic control in patients with type 1 diabetes mellitus [see Warnings and Precautions (5.1)].
Section 42231-1
| Medication Guide STEGLATRO® [steh-GLA-troh] (ertugliflozin) tablets, for oral use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 09/2023 | ||
| Read this Medication Guide carefully before you start taking STEGLATRO and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |||
What is the most important information I should know about STEGLATRO? STEGLATRO may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
| Talk to your healthcare provider about what to do if you get symptoms of a yeast infection of the vagina or penis. Your healthcare provider may suggest you use an over-the-counter antifungal medicine. Talk to your healthcare provider right away if you use an over-the-counter antifungal medicine and your symptoms do not go away. | |||
What is STEGLATRO?
|
|||
|
Who should not take STEGLATRO?
|
|||
Before you take STEGLATRO, tell your healthcare provider about all of your medical conditions, including if you:
STEGLATRO may affect the way other medicines work, and other medicines may affect how STEGLATRO works. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I take STEGLATRO?
|
|||
|
What are the possible side effects of STEGLATRO? STEGLATRO may cause serious side effects, including: See " What is the most important information I should know about STEGLATRO? "
|
|||
|
|
|
|
|
|||
|
|
|
|
|
|||
The most common side effects of STEGLATRO include:
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store STEGLATRO?
|
|||
|
General information about the safe and effective use of STEGLATRO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use STEGLATRO for a condition for which it was not prescribed. Do not give STEGLATRO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about STEGLATRO that is written for health professionals. For more information about STEGLATRO, go to www.steglatro.com or call 1-800-622-4477. |
|||
|
What are the ingredients in STEGLATRO? Active ingredient: ertugliflozin. Inactive ingredients: microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, and magnesium stearate. The tablet film coating contains the following inactive ingredients: hypromellose, lactose monohydrate, macrogol, triacetin, titanium dioxide, and iron oxide red. |
|||
| Manufactured for: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA For patent information, go to: www.msd.com/research/patent Copyright © 2017-2023 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved. usmg-mk8835-t-2309r006 |
Section 44425-7
Store at 20°C -25°C (68°F -77°F), excursions permitted between 15°C -30°C (between 59°F -86°F) [see USP Controlled Room Temperature]. Protect from moisture. Store in a dry place.
10 Overdosage
In the event of an overdose with STEGLATRO, contact the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. Employ the usual supportive measures as dictated by the patient's clinical status. Removal of ertugliflozin by hemodialysis has not been studied.
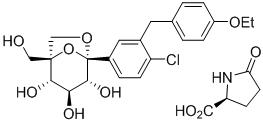
11 Description
STEGLATRO (ertugliflozin) tablets for oral use contain ertugliflozin L-pyroglutamic acid, a SGLT2 inhibitor.
The chemical name of ertugliflozin L-pyroglutamic acid is (1S,2S,3S,4R,5S)-5-(4-chloro-3-(4-ethoxybenzyl)phenyl)-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol, compound with (2S)-5-oxopyrrolidine-2-carboxylic acid. The molecular formula is C27H32ClNO10 and the molecular weight is 566.00.
The chemical structure is:
Ertugliflozin L-pyroglutamic acid is a white to off-white powder that is soluble in ethyl alcohol and acetone, slightly soluble in ethyl acetate and acetonitrile and very slightly soluble in water.
STEGLATRO is supplied as film-coated tablets, containing 6.48 or 19.43 mg of ertugliflozin L-pyroglutamic acid, which is equivalent to 5 and 15 mg of ertugliflozin.
Inactive ingredients are microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, and magnesium stearate.
The film coating contains: hypromellose, lactose monohydrate, macrogol, triacetin, titanium dioxide and iron oxide red.
8.4 Pediatric Use
Safety and effectiveness of STEGLATRO in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
No dosage adjustment of STEGLATRO is recommended based on age. In STEGLATRO clinical trials, a total of 876 (25.7%) patients treated with STEGLATRO were 65 years and older, and 152 (4.5%) patients treated with STEGLATRO were 75 years and older. Patients 65 years and older had a higher incidence of adverse reactions related to volume depletion compared to younger patients; events were reported in 1.1%, 2.2%, and 2.6% of patients treated with comparator, STEGLATRO 5 mg, and STEGLATRO 15 mg, respectively [see Warnings and Precautions (5.3) and Adverse Reactions (6.1)].
In VERTIS CV, a total of 2780 (50.5%) patients treated with STEGLATRO were 65 years and older, and 595 (10.8%) patients treated with STEGLATRO were 75 years and older. Safety and efficacy were generally similar for patients age 65 years and older compared to patients younger than 65.
4 Contraindications
STEGLATRO is contraindicated in patients with hypersensitivity to ertugliflozin or any excipient in STEGLATRO. Reactions such as angioedema have occurred [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Lower Limb Amputation [see Warnings and Precautions (5.2)]
- Volume Depletion [see Warnings and Precautions (5.3)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.6)]
- Genital Mycotic Infections [see Warnings and Precautions (5.7)]
7 Drug Interactions
| Insulin or Insulin Secretagogues | |
|---|---|
| Clinical Impact: | The risk of hypoglycemia is increased when STEGLATRO is used in combination with insulin or an insulin secretagogue. |
| Intervention: | A lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with STEGLATRO. |
| Lithium | |
| Clinical Impact: | Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. |
| Intervention: | Monitor serum lithium concentration more frequently during STEGLATRO initiation and dosage changes. |
| Positive Urine Glucose Test | |
| Clinical Impact: | SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. |
| Intervention: | Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
| Interference with 1,5-anhydroglucitol (1,5-AG) Assay | |
| Clinical Impact: | Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
| Intervention: | Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control. |
5.3 Volume Depletion
STEGLATRO can cause intravascular volume contraction which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine [see Adverse Reactions (6.1)]. There have been postmarketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors, including STEGLATRO. Patients with impaired renal function (eGFR less than 60 mL/min/1.73 m2) [see Use in Specific Populations (8.6)], elderly patients, patients with low systolic blood pressure, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating STEGLATRO in patients with one or more of these characteristics, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating STEGLATRO. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy.
8.6 Renal Impairment
A 26-week placebo-controlled study of 313 patients with Stage 3 Chronic Kidney Disease (eGFR ≥30 to less than 60 mL/min/1.73 m2) treated with STEGLATRO did not demonstrate improvement in glycemic control.
In the VERTIS CV study, there were 1370 patients (25%) with an eGFR ≥90 mL/min/1.73 m2, 2929 patients (53%) with an eGFR of ≥60 to less than 90 mL/min/1.73 m2, 879 patients (16%) with an eGFR of ≥45 to less than 60 mL/min/1.73 m2, and 299 patients (5%) with eGFR of 30 to <45 mL/min/1.73 m2 treated with STEGLATRO. Similar effects on glycemic control at Week 18 were observed in patients treated with STEGLATRO in each eGFR subgroup and also in the overall patient population.
No dosage adjustment is needed in patients with eGFR ≥45 mL/min/1.73 m2.
12.3 Pharmacokinetics
The pharmacokinetics of ertugliflozin are similar in healthy subjects and patients with type 2 diabetes mellitus. The steady state mean plasma AUC and Cmax were 398 ng∙hr/mL and 81.3 ng/mL, respectively, with 5 mg ertugliflozin once-daily treatment, and 1,193 ng∙hr/mL and 268 ng/mL, respectively, with 15 mg ertugliflozin once-daily treatment. Steady-state is reached after 4 to 6 days of once-daily dosing with ertugliflozin. Ertugliflozin does not exhibit time-dependent pharmacokinetics and accumulates in plasma up to 10-40% following multiple dosing.
2.2 Recommended Dosage
- The recommended starting dosage of STEGLATRO is 5 mg orally once daily, taken in the morning, with or without food.
- For additional glycemic control, the dosage may be increased to 15 mg orally once daily in patients tolerating STEGLATRO.
- Use of STEGLATRO is not recommended in patients with an estimated glomerular filtration rate (eGFR) less than 45 mL/min/1.73 m2.
8.7 Hepatic Impairment
No dosage adjustment of STEGLATRO is necessary in patients with mild or moderate hepatic impairment. Ertugliflozin has not been studied in patients with severe hepatic impairment and is not recommended for use in this patient population [see Clinical Pharmacology (12.3)].
1 Indications and Usage
STEGLATRO® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
12.1 Mechanism of Action
SGLT2 is the predominant transporter responsible for reabsorption of glucose from the glomerular filtrate back into the circulation. Ertugliflozin is an inhibitor of SGLT2. By inhibiting SGLT2, ertugliflozin reduces renal reabsorption of filtered glucose and lowers the renal threshold for glucose, and thereby increases urinary glucose excretion.
5.2 Lower Limb Amputation
In a long-term cardiovascular outcomes study [see Clinical Studies (14.2)], in patients with type 2 diabetes mellitus and established cardiovascular disease, the occurrence of non-traumatic lower limb amputations was reported with event rates of 4.7, 5.7, and 6.0 events per 1,000 patient-years in the placebo, STEGLATRO 5 mg, and STEGLATRO 15 mg treatment arms, respectively.
Amputation of the toe and foot were most frequent (81 out of 109 patients with lower limb amputations). Some patients had multiple amputations, some involving both lower limbs.
Lower limb infections, gangrene, and diabetic foot ulcers were the most common precipitating medical events leading to the need for an amputation. Patients with amputations were more likely to be male, have higher A1C (%) at baseline, have a history of peripheral arterial disease, amputation or peripheral revascularization procedure, diabetic foot, and to have been taking diuretics or insulin.
Across seven STEGLATRO clinical trials, non-traumatic lower limb amputations were reported in 1 (0.1%) patient in the comparator group, 3 (0.2%) patients in the STEGLATRO 5 mg group, and 8 (0.5%) patients in the STEGLATRO 15 mg group.
Counsel patients about the importance of routine preventative foot care. Monitor patients receiving STEGLATRO for signs and symptoms of infection (including osteomyelitis), new pain or tenderness, sores or ulcers involving the lower limbs, and discontinue STEGLATRO if these complications occur.
5 Warnings and Precautions
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue STEGLATRO if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. (5.1)
- Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and discontinue if these occur. (5.2)
- Volume Depletion: May result in acute kidney injury. Before initiating, assess and correct volume status in patients with renal impairment or low systolic blood pressure, elderly patients, or patients on diuretics. Monitor for signs and symptoms during therapy. (5.3)
- Urosepsis and Pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated. (5.4)
- Hypoglycemia: Consider a lower dose of insulin or insulin secretagogue to reduce risk of hypoglycemia when used in combination. (5.5)
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment. (5.6)
- Genital Mycotic Infections: Monitor and treat if indicated. (5.7)
2 Dosage and Administration
- Assess renal function before initiating and as clinically indicated. (2.1)
- Correct volume depletion before initiating STEGLATRO. (2.1)
- Recommended starting dosage is 5 mg orally once daily, taken in the morning, with or without food. (2.2)
- Increase dosage to 15 mg orally once daily in those tolerating STEGLATRO and needing additional glycemic control. (2.2)
- Use is not recommended in patients with an estimated glomerular filtration rate (eGFR) less than 45 mL/min/1.73 m2. (2.2)
- Withhold STEGLATRO for at least 4 days, if possible, prior to surgery or procedures associated with prolonged fasting. (2.3)
3 Dosage Forms and Strengths
- Tablets: 5 mg, pink, triangular-shaped debossed with "701" on one side and plain on the other side.
- Tablets: 15 mg, red, triangular-shaped debossed with "702" on one side and plain on the other side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of STEGLATRO. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Infections: necrotizing fasciitis of the perineum (Fournier's Gangrene)
- Skin and Subcutaneous Tissue Disorders: angioedema, rash
8 Use in Specific Populations
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters. (8.1)
- Lactation: Breastfeeding not recommended. (8.2)
- Geriatrics: Higher incidence of adverse reactions related to reduced intravascular volume. (8.5)
- Renal Impairment: Higher incidence of adverse reactions related to reduced intravascular volume and renal function. (8.6)
5.7 Genital Mycotic Infections
STEGLATRO increases the risk of genital mycotic infections. Patients who have a history of genital mycotic infections or who are uncircumcised are more likely to develop genital mycotic infections [see Adverse Reactions (6.1)]. Monitor and treat appropriately.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.4 Urosepsis and Pyelonephritis
There have been postmarketing reports of serious urinary tract infections, including urosepsis and pyelonephritis, requiring hospitalization in patients receiving SGLT2 inhibitors. Treatment with SGLT2 inhibitors increases the risk for urinary tract infections. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6.1)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
STEGLATRO (ertugliflozin) tablets are available as follows:
| Strength | Description | How Supplied | NDC |
|---|---|---|---|
| 5 mg tablets | pink, triangular-shaped, biconvex tablets, with “701” debossed on one side and plain on the other side | unit-of-use bottles of 30 | 0006-5363-03 |
| unit-of-use bottles of 90 | 0006-5363-06 | ||
| 15 mg tablets | red, triangular-shaped, biconvex tablets, with “702” debossed on one side and plain on the other side | unit-of-use bottles of 30 | 0006-5364-03 |
| unit-of-use bottles of 90 | 0006-5364-06 |
2.1 Prior to Initiation of Steglatro
- Assess renal function before initiating STEGLATRO and as clinically indicated [see Warnings and Precautions (5.3)].
- Assess volume status. In patients with volume depletion, correct this condition before initiating STEGLATRO [see Warnings and Precautions (5.3) and Use in Specific Populations (8.5, 8.6)].
2.3 Temporary Interruption for Surgery
Withhold STEGLATRO for at least 4 days, if possible, prior to surgery or procedures associated with prolonged fasting. Resume STEGLATRO when the patient is clinically stable and has resumed oral intake [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2)].
Principal Display Panel 5 Mg Tablet Bottle Label
NDC 0006-5363-03
Steglatro®
(ertugliflozin) tablets
5 mg
Dispense the accompanying Medication Guide
to each patient.
Each tablet contains 6.48 mg ertugliflozin L-pyroglutamic
acid (equivalent to 5 mg ertugliflozin).
Rx only
30 Tablets
Principal Display Panel 15 Mg Tablet Bottle Label
NDC 0006-5364-03
Steglatro®
(ertugliflozin) tablets
15 mg
Dispense the accompanying Medication Guide
to each patient.
Each tablet contains 19.43 mg ertugliflozin L-pyroglutamic
acid (equivalent to 15 mg ertugliflozin).
Rx only
30 Tablets
5.6 Necrotizing Fasciitis of the Perineum (fournier's Gangrene)
Reports of necrotizing fasciitis of the perineum (Fournier's Gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors, including STEGLATRO. Cases have been reported in females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with STEGLATRO presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue STEGLATRO, closely monitor blood glucose levels, and provide appropriate alternative therapy for glycemic control.
14.1 Glycemic Control Trials in Patients With Type 2 Diabetes Mellitus
STEGLATRO has been studied as monotherapy and in combination with metformin HCl, sitagliptin, a sulfonylurea, insulin (with or without metformin HCl), metformin HCl plus sitagliptin, metformin HCl plus a sulfonylurea and compared to a sulfonylurea (glimepiride). STEGLATRO has also been studied in patients with type 2 diabetes mellitus and moderate renal impairment.
In patients with type 2 diabetes mellitus treatment with STEGLATRO reduced hemoglobin A1c (HbA1c) compared to placebo. Reduction in HbA1c was generally similar across subgroups defined by age, sex, race, geographic region, baseline body mass index (BMI), and duration of type 2 diabetes mellitus.
5.5 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues
Insulin and insulin secretagogues (e.g., sulfonylurea) are known to cause hypoglycemia. STEGLATRO may increase the risk of hypoglycemia when used in combination with insulin or an insulin secretagogue [see Adverse Reactions (6.1)]. The risk of hypoglycemia may be lowered by a reduction in the dose of insulin or sulfonylurea (or other concomitantly administered insulin secretagogues). Inform patients using these medications concomitantly of this risk and educate them on the signs and symptoms of hypoglycemia.
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis
In patients with type 1 diabetes mellitus, STEGLATRO significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose transporter 2 (SGLT2) inhibitors compared to patients who received placebo; this risk may be greater with higher doses. STEGLATRO is not indicated for glycemic control in patients with type 1 diabetes mellitus.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes mellitus using SGLT2 inhibitors.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include under-insulinization due to insulin dose reduction or missed insulin doses, acute febrile illness, reduced caloric intake, ketogenic diet, surgery, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected. Urinary glucose excretion persists for 4 days after discontinuing STEGLATRO [see Clinical Pharmacology (12.2)]; however, there have been postmarketing reports of ketoacidosis and/or glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue STEGLATRO, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting STEGLATRO.
Withhold STEGLATRO, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume STEGLATRO when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.3)].
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue STEGLATRO and seek medical attention immediately if signs and symptoms occur.
14.2 Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Established Cardiovascular Disease
The effect of STEGLATRO on cardiovascular risk in adult patients with type 2 diabetes and established atherosclerotic cardiovascular disease was evaluated in the VERTIS CV study (NCT01986881), a multicenter, multi-national, randomized, double-blind, placebo-controlled, event-driven trial. The study compared the risk of experiencing a major adverse cardiovascular event (MACE) between STEGLATRO and placebo when these were added to and used concomitantly with standard of care treatments for diabetes and atherosclerotic cardiovascular disease.
A total of 8,246 patients were randomized to placebo (N=2,747), oral once daily STEGLATRO 5 mg (N=2,752) or oral once daily STEGLATRO 15 mg (N=2,747) and followed for a median of 3 years. Approximately 88% of the study population was White, 6% Asian, and 3% Black or African American. The mean age was 64 years and approximately 70% were male.
All patients in the study had inadequately controlled type 2 diabetes mellitus at baseline (HbA1c greater than or equal to 7%). The mean duration of type 2 diabetes mellitus was 13 years, the mean HbA1c at baseline was 8.2% and the mean eGFR was 76 mL/min/1.73 m2. At baseline, patients were treated with one (32%) or more (67%) antidiabetic medications including biguanides (metformin HCl) (76%), insulin (47%), sulfonylureas (41%) DPP-4 inhibitors (11%) and GLP-1 receptor agonists (3%).
Almost all patients (99%) had established atherosclerotic cardiovascular disease at baseline including: a documented history of coronary artery disease (76%), cerebrovascular disease (23%) or peripheral artery disease (19%). Approximately 24% patients had a history of heart failure (HF). At baseline, the mean systolic blood pressure was 133 mmHg, the mean diastolic blood pressure was 77 mmHg, the mean LDL was 89 mg/dL, and the mean HDL was 44 mg/dL. At baseline, approximately 81% of patients were treated with renin angiotensin system inhibitors, 69% with beta-blockers, 43% with diuretics, 82% with statins, 4% with ezetimibe, and 89% with antiplatelet agents.
The primary endpoint in VERTIS CV was the time to first occurrence of MACE. A major adverse cardiovascular event was defined as occurrence of either a cardiovascular death or a nonfatal myocardial infarction (MI) or a nonfatal stroke. The statistical analysis plan pre-specified that the 5 and 15 mg doses would be combined for the analysis. A Cox proportional hazards model was used to test for non-inferiority against the pre-specified risk margin of 1.3 for the hazard ratio of MACE. Type-1 error was controlled across multiple tests using a hierarchical testing strategy.
The incidence rate of MACE was similar between the STEGLATRO-treated and placebo-treated patients. The estimated hazard ratio of MACE associated with STEGLATRO relative to placebo was 0.97 with 95.6% confidence interval (0.85, 1.11). The upper bound of this confidence interval excluded a risk larger than 1.3 (Table 10). Results for the individual 5 mg and 15 mg doses were consistent with results for the combined dose group.
| Endpoint MACE was evaluated in subjects who took at least one dose of study medication and, for subjects who discontinued study medication prior to the end of the study, censored events that occurred more than 365 days after the last dose of study medication. Other endpoints were evaluated using all randomized subjects and events that occurred any time after the first dose of study medication until the last contact date. The total number of first events was analyzed for each endpoint.
|
Placebo (N=2747) | STEGLATRO (N=5499) | Hazard Ratio vs Placebo (CI) HR and CI are based on Cox proportional hazards regression model, stratified by cohorts. For MACE a 95.6% CI is presented, for other endpoints a 95% CI is presented.
|
||
|---|---|---|---|---|---|
| N (%) | Event Rate (per 100 person-years) | N (%) | Event Rate (per 100 person-years) | ||
| N=Number of patients, CI=Confidence interval, CV=Cardiovascular, MI=Myocardial infarction. | |||||
| MACE (CV death, non-fatal MI, or non-fatal stroke) Composite | 327 (11.9) | 4.0 | 653 (11.9) | 3.9 | 0.97 (0.85, 1.11) |
| Components of Composite Endpoint | |||||
| Non-fatal MI | 148 (5.4) | 1.6 | 310 (5.6) | 1.7 | 1.04 (0.86, 1.27) |
| Non-fatal Stroke | 78 (2.8) | 0.8 | 157 (2.9) | 0.8 | 1.00 (0.76, 1.32) |
| CV death | 184 (6.7) | 1.9 | 341 (6.2) | 1.8 | 0.92 (0.77, 1.11) |
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
- Not recommended for use to improve glycemic control in patients with type 1 diabetes mellitus [see Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
| Medication Guide STEGLATRO® [steh-GLA-troh] (ertugliflozin) tablets, for oral use |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 09/2023 | ||
| Read this Medication Guide carefully before you start taking STEGLATRO and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |||
What is the most important information I should know about STEGLATRO? STEGLATRO may cause serious side effects, including:
|
|||
|
|
||
|
|||
|
|
||
| Talk to your healthcare provider about what to do if you get symptoms of a yeast infection of the vagina or penis. Your healthcare provider may suggest you use an over-the-counter antifungal medicine. Talk to your healthcare provider right away if you use an over-the-counter antifungal medicine and your symptoms do not go away. | |||
What is STEGLATRO?
|
|||
|
Who should not take STEGLATRO?
|
|||
Before you take STEGLATRO, tell your healthcare provider about all of your medical conditions, including if you:
STEGLATRO may affect the way other medicines work, and other medicines may affect how STEGLATRO works. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
How should I take STEGLATRO?
|
|||
|
What are the possible side effects of STEGLATRO? STEGLATRO may cause serious side effects, including: See " What is the most important information I should know about STEGLATRO? "
|
|||
|
|
|
|
|
|||
|
|
|
|
|
|||
The most common side effects of STEGLATRO include:
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store STEGLATRO?
|
|||
|
General information about the safe and effective use of STEGLATRO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use STEGLATRO for a condition for which it was not prescribed. Do not give STEGLATRO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about STEGLATRO that is written for health professionals. For more information about STEGLATRO, go to www.steglatro.com or call 1-800-622-4477. |
|||
|
What are the ingredients in STEGLATRO? Active ingredient: ertugliflozin. Inactive ingredients: microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, and magnesium stearate. The tablet film coating contains the following inactive ingredients: hypromellose, lactose monohydrate, macrogol, triacetin, titanium dioxide, and iron oxide red. |
|||
| Manufactured for: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA For patent information, go to: www.msd.com/research/patent Copyright © 2017-2023 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved. usmg-mk8835-t-2309r006 |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store at 20°C -25°C (68°F -77°F), excursions permitted between 15°C -30°C (between 59°F -86°F) [see USP Controlled Room Temperature]. Protect from moisture. Store in a dry place.
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose with STEGLATRO, contact the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. Employ the usual supportive measures as dictated by the patient's clinical status. Removal of ertugliflozin by hemodialysis has not been studied.
11 Description (11 DESCRIPTION)
STEGLATRO (ertugliflozin) tablets for oral use contain ertugliflozin L-pyroglutamic acid, a SGLT2 inhibitor.
The chemical name of ertugliflozin L-pyroglutamic acid is (1S,2S,3S,4R,5S)-5-(4-chloro-3-(4-ethoxybenzyl)phenyl)-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol, compound with (2S)-5-oxopyrrolidine-2-carboxylic acid. The molecular formula is C27H32ClNO10 and the molecular weight is 566.00.
The chemical structure is:
Ertugliflozin L-pyroglutamic acid is a white to off-white powder that is soluble in ethyl alcohol and acetone, slightly soluble in ethyl acetate and acetonitrile and very slightly soluble in water.
STEGLATRO is supplied as film-coated tablets, containing 6.48 or 19.43 mg of ertugliflozin L-pyroglutamic acid, which is equivalent to 5 and 15 mg of ertugliflozin.
Inactive ingredients are microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, and magnesium stearate.
The film coating contains: hypromellose, lactose monohydrate, macrogol, triacetin, titanium dioxide and iron oxide red.
8.4 Pediatric Use
Safety and effectiveness of STEGLATRO in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
No dosage adjustment of STEGLATRO is recommended based on age. In STEGLATRO clinical trials, a total of 876 (25.7%) patients treated with STEGLATRO were 65 years and older, and 152 (4.5%) patients treated with STEGLATRO were 75 years and older. Patients 65 years and older had a higher incidence of adverse reactions related to volume depletion compared to younger patients; events were reported in 1.1%, 2.2%, and 2.6% of patients treated with comparator, STEGLATRO 5 mg, and STEGLATRO 15 mg, respectively [see Warnings and Precautions (5.3) and Adverse Reactions (6.1)].
In VERTIS CV, a total of 2780 (50.5%) patients treated with STEGLATRO were 65 years and older, and 595 (10.8%) patients treated with STEGLATRO were 75 years and older. Safety and efficacy were generally similar for patients age 65 years and older compared to patients younger than 65.
4 Contraindications (4 CONTRAINDICATIONS)
STEGLATRO is contraindicated in patients with hypersensitivity to ertugliflozin or any excipient in STEGLATRO. Reactions such as angioedema have occurred [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Lower Limb Amputation [see Warnings and Precautions (5.2)]
- Volume Depletion [see Warnings and Precautions (5.3)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.6)]
- Genital Mycotic Infections [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
| Insulin or Insulin Secretagogues | |
|---|---|
| Clinical Impact: | The risk of hypoglycemia is increased when STEGLATRO is used in combination with insulin or an insulin secretagogue. |
| Intervention: | A lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with STEGLATRO. |
| Lithium | |
| Clinical Impact: | Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. |
| Intervention: | Monitor serum lithium concentration more frequently during STEGLATRO initiation and dosage changes. |
| Positive Urine Glucose Test | |
| Clinical Impact: | SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. |
| Intervention: | Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
| Interference with 1,5-anhydroglucitol (1,5-AG) Assay | |
| Clinical Impact: | Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
| Intervention: | Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control. |
5.3 Volume Depletion
STEGLATRO can cause intravascular volume contraction which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine [see Adverse Reactions (6.1)]. There have been postmarketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors, including STEGLATRO. Patients with impaired renal function (eGFR less than 60 mL/min/1.73 m2) [see Use in Specific Populations (8.6)], elderly patients, patients with low systolic blood pressure, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating STEGLATRO in patients with one or more of these characteristics, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating STEGLATRO. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy.
8.6 Renal Impairment
A 26-week placebo-controlled study of 313 patients with Stage 3 Chronic Kidney Disease (eGFR ≥30 to less than 60 mL/min/1.73 m2) treated with STEGLATRO did not demonstrate improvement in glycemic control.
In the VERTIS CV study, there were 1370 patients (25%) with an eGFR ≥90 mL/min/1.73 m2, 2929 patients (53%) with an eGFR of ≥60 to less than 90 mL/min/1.73 m2, 879 patients (16%) with an eGFR of ≥45 to less than 60 mL/min/1.73 m2, and 299 patients (5%) with eGFR of 30 to <45 mL/min/1.73 m2 treated with STEGLATRO. Similar effects on glycemic control at Week 18 were observed in patients treated with STEGLATRO in each eGFR subgroup and also in the overall patient population.
No dosage adjustment is needed in patients with eGFR ≥45 mL/min/1.73 m2.
12.3 Pharmacokinetics
The pharmacokinetics of ertugliflozin are similar in healthy subjects and patients with type 2 diabetes mellitus. The steady state mean plasma AUC and Cmax were 398 ng∙hr/mL and 81.3 ng/mL, respectively, with 5 mg ertugliflozin once-daily treatment, and 1,193 ng∙hr/mL and 268 ng/mL, respectively, with 15 mg ertugliflozin once-daily treatment. Steady-state is reached after 4 to 6 days of once-daily dosing with ertugliflozin. Ertugliflozin does not exhibit time-dependent pharmacokinetics and accumulates in plasma up to 10-40% following multiple dosing.
2.2 Recommended Dosage
- The recommended starting dosage of STEGLATRO is 5 mg orally once daily, taken in the morning, with or without food.
- For additional glycemic control, the dosage may be increased to 15 mg orally once daily in patients tolerating STEGLATRO.
- Use of STEGLATRO is not recommended in patients with an estimated glomerular filtration rate (eGFR) less than 45 mL/min/1.73 m2.
8.7 Hepatic Impairment
No dosage adjustment of STEGLATRO is necessary in patients with mild or moderate hepatic impairment. Ertugliflozin has not been studied in patients with severe hepatic impairment and is not recommended for use in this patient population [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
STEGLATRO® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
12.1 Mechanism of Action
SGLT2 is the predominant transporter responsible for reabsorption of glucose from the glomerular filtrate back into the circulation. Ertugliflozin is an inhibitor of SGLT2. By inhibiting SGLT2, ertugliflozin reduces renal reabsorption of filtered glucose and lowers the renal threshold for glucose, and thereby increases urinary glucose excretion.
5.2 Lower Limb Amputation
In a long-term cardiovascular outcomes study [see Clinical Studies (14.2)], in patients with type 2 diabetes mellitus and established cardiovascular disease, the occurrence of non-traumatic lower limb amputations was reported with event rates of 4.7, 5.7, and 6.0 events per 1,000 patient-years in the placebo, STEGLATRO 5 mg, and STEGLATRO 15 mg treatment arms, respectively.
Amputation of the toe and foot were most frequent (81 out of 109 patients with lower limb amputations). Some patients had multiple amputations, some involving both lower limbs.
Lower limb infections, gangrene, and diabetic foot ulcers were the most common precipitating medical events leading to the need for an amputation. Patients with amputations were more likely to be male, have higher A1C (%) at baseline, have a history of peripheral arterial disease, amputation or peripheral revascularization procedure, diabetic foot, and to have been taking diuretics or insulin.
Across seven STEGLATRO clinical trials, non-traumatic lower limb amputations were reported in 1 (0.1%) patient in the comparator group, 3 (0.2%) patients in the STEGLATRO 5 mg group, and 8 (0.5%) patients in the STEGLATRO 15 mg group.
Counsel patients about the importance of routine preventative foot care. Monitor patients receiving STEGLATRO for signs and symptoms of infection (including osteomyelitis), new pain or tenderness, sores or ulcers involving the lower limbs, and discontinue STEGLATRO if these complications occur.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue STEGLATRO if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. (5.1)
- Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and discontinue if these occur. (5.2)
- Volume Depletion: May result in acute kidney injury. Before initiating, assess and correct volume status in patients with renal impairment or low systolic blood pressure, elderly patients, or patients on diuretics. Monitor for signs and symptoms during therapy. (5.3)
- Urosepsis and Pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated. (5.4)
- Hypoglycemia: Consider a lower dose of insulin or insulin secretagogue to reduce risk of hypoglycemia when used in combination. (5.5)
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment. (5.6)
- Genital Mycotic Infections: Monitor and treat if indicated. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Assess renal function before initiating and as clinically indicated. (2.1)
- Correct volume depletion before initiating STEGLATRO. (2.1)
- Recommended starting dosage is 5 mg orally once daily, taken in the morning, with or without food. (2.2)
- Increase dosage to 15 mg orally once daily in those tolerating STEGLATRO and needing additional glycemic control. (2.2)
- Use is not recommended in patients with an estimated glomerular filtration rate (eGFR) less than 45 mL/min/1.73 m2. (2.2)
- Withhold STEGLATRO for at least 4 days, if possible, prior to surgery or procedures associated with prolonged fasting. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Tablets: 5 mg, pink, triangular-shaped debossed with "701" on one side and plain on the other side.
- Tablets: 15 mg, red, triangular-shaped debossed with "702" on one side and plain on the other side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of STEGLATRO. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Infections: necrotizing fasciitis of the perineum (Fournier's Gangrene)
- Skin and Subcutaneous Tissue Disorders: angioedema, rash
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters. (8.1)
- Lactation: Breastfeeding not recommended. (8.2)
- Geriatrics: Higher incidence of adverse reactions related to reduced intravascular volume. (8.5)
- Renal Impairment: Higher incidence of adverse reactions related to reduced intravascular volume and renal function. (8.6)
5.7 Genital Mycotic Infections
STEGLATRO increases the risk of genital mycotic infections. Patients who have a history of genital mycotic infections or who are uncircumcised are more likely to develop genital mycotic infections [see Adverse Reactions (6.1)]. Monitor and treat appropriately.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.4 Urosepsis and Pyelonephritis
There have been postmarketing reports of serious urinary tract infections, including urosepsis and pyelonephritis, requiring hospitalization in patients receiving SGLT2 inhibitors. Treatment with SGLT2 inhibitors increases the risk for urinary tract infections. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6.1)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
STEGLATRO (ertugliflozin) tablets are available as follows:
| Strength | Description | How Supplied | NDC |
|---|---|---|---|
| 5 mg tablets | pink, triangular-shaped, biconvex tablets, with “701” debossed on one side and plain on the other side | unit-of-use bottles of 30 | 0006-5363-03 |
| unit-of-use bottles of 90 | 0006-5363-06 | ||
| 15 mg tablets | red, triangular-shaped, biconvex tablets, with “702” debossed on one side and plain on the other side | unit-of-use bottles of 30 | 0006-5364-03 |
| unit-of-use bottles of 90 | 0006-5364-06 |
2.1 Prior to Initiation of Steglatro (2.1 Prior to Initiation of STEGLATRO)
- Assess renal function before initiating STEGLATRO and as clinically indicated [see Warnings and Precautions (5.3)].
- Assess volume status. In patients with volume depletion, correct this condition before initiating STEGLATRO [see Warnings and Precautions (5.3) and Use in Specific Populations (8.5, 8.6)].
2.3 Temporary Interruption for Surgery
Withhold STEGLATRO for at least 4 days, if possible, prior to surgery or procedures associated with prolonged fasting. Resume STEGLATRO when the patient is clinically stable and has resumed oral intake [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2)].
Principal Display Panel 5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label)
NDC 0006-5363-03
Steglatro®
(ertugliflozin) tablets
5 mg
Dispense the accompanying Medication Guide
to each patient.
Each tablet contains 6.48 mg ertugliflozin L-pyroglutamic
acid (equivalent to 5 mg ertugliflozin).
Rx only
30 Tablets
Principal Display Panel 15 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 15 mg Tablet Bottle Label)
NDC 0006-5364-03
Steglatro®
(ertugliflozin) tablets
15 mg
Dispense the accompanying Medication Guide
to each patient.
Each tablet contains 19.43 mg ertugliflozin L-pyroglutamic
acid (equivalent to 15 mg ertugliflozin).
Rx only
30 Tablets
5.6 Necrotizing Fasciitis of the Perineum (fournier's Gangrene) (5.6 Necrotizing Fasciitis of the Perineum (Fournier's Gangrene))
Reports of necrotizing fasciitis of the perineum (Fournier's Gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors, including STEGLATRO. Cases have been reported in females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with STEGLATRO presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue STEGLATRO, closely monitor blood glucose levels, and provide appropriate alternative therapy for glycemic control.
14.1 Glycemic Control Trials in Patients With Type 2 Diabetes Mellitus (14.1 Glycemic Control Trials in Patients with Type 2 Diabetes Mellitus)
STEGLATRO has been studied as monotherapy and in combination with metformin HCl, sitagliptin, a sulfonylurea, insulin (with or without metformin HCl), metformin HCl plus sitagliptin, metformin HCl plus a sulfonylurea and compared to a sulfonylurea (glimepiride). STEGLATRO has also been studied in patients with type 2 diabetes mellitus and moderate renal impairment.
In patients with type 2 diabetes mellitus treatment with STEGLATRO reduced hemoglobin A1c (HbA1c) compared to placebo. Reduction in HbA1c was generally similar across subgroups defined by age, sex, race, geographic region, baseline body mass index (BMI), and duration of type 2 diabetes mellitus.
5.5 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues (5.5 Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues)
Insulin and insulin secretagogues (e.g., sulfonylurea) are known to cause hypoglycemia. STEGLATRO may increase the risk of hypoglycemia when used in combination with insulin or an insulin secretagogue [see Adverse Reactions (6.1)]. The risk of hypoglycemia may be lowered by a reduction in the dose of insulin or sulfonylurea (or other concomitantly administered insulin secretagogues). Inform patients using these medications concomitantly of this risk and educate them on the signs and symptoms of hypoglycemia.
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis (5.1 Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis)
In patients with type 1 diabetes mellitus, STEGLATRO significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose transporter 2 (SGLT2) inhibitors compared to patients who received placebo; this risk may be greater with higher doses. STEGLATRO is not indicated for glycemic control in patients with type 1 diabetes mellitus.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes mellitus using SGLT2 inhibitors.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include under-insulinization due to insulin dose reduction or missed insulin doses, acute febrile illness, reduced caloric intake, ketogenic diet, surgery, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected. Urinary glucose excretion persists for 4 days after discontinuing STEGLATRO [see Clinical Pharmacology (12.2)]; however, there have been postmarketing reports of ketoacidosis and/or glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue STEGLATRO, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting STEGLATRO.
Withhold STEGLATRO, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume STEGLATRO when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.3)].
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue STEGLATRO and seek medical attention immediately if signs and symptoms occur.
14.2 Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Established Cardiovascular Disease (14.2 Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus and Established Cardiovascular Disease)
The effect of STEGLATRO on cardiovascular risk in adult patients with type 2 diabetes and established atherosclerotic cardiovascular disease was evaluated in the VERTIS CV study (NCT01986881), a multicenter, multi-national, randomized, double-blind, placebo-controlled, event-driven trial. The study compared the risk of experiencing a major adverse cardiovascular event (MACE) between STEGLATRO and placebo when these were added to and used concomitantly with standard of care treatments for diabetes and atherosclerotic cardiovascular disease.
A total of 8,246 patients were randomized to placebo (N=2,747), oral once daily STEGLATRO 5 mg (N=2,752) or oral once daily STEGLATRO 15 mg (N=2,747) and followed for a median of 3 years. Approximately 88% of the study population was White, 6% Asian, and 3% Black or African American. The mean age was 64 years and approximately 70% were male.
All patients in the study had inadequately controlled type 2 diabetes mellitus at baseline (HbA1c greater than or equal to 7%). The mean duration of type 2 diabetes mellitus was 13 years, the mean HbA1c at baseline was 8.2% and the mean eGFR was 76 mL/min/1.73 m2. At baseline, patients were treated with one (32%) or more (67%) antidiabetic medications including biguanides (metformin HCl) (76%), insulin (47%), sulfonylureas (41%) DPP-4 inhibitors (11%) and GLP-1 receptor agonists (3%).
Almost all patients (99%) had established atherosclerotic cardiovascular disease at baseline including: a documented history of coronary artery disease (76%), cerebrovascular disease (23%) or peripheral artery disease (19%). Approximately 24% patients had a history of heart failure (HF). At baseline, the mean systolic blood pressure was 133 mmHg, the mean diastolic blood pressure was 77 mmHg, the mean LDL was 89 mg/dL, and the mean HDL was 44 mg/dL. At baseline, approximately 81% of patients were treated with renin angiotensin system inhibitors, 69% with beta-blockers, 43% with diuretics, 82% with statins, 4% with ezetimibe, and 89% with antiplatelet agents.
The primary endpoint in VERTIS CV was the time to first occurrence of MACE. A major adverse cardiovascular event was defined as occurrence of either a cardiovascular death or a nonfatal myocardial infarction (MI) or a nonfatal stroke. The statistical analysis plan pre-specified that the 5 and 15 mg doses would be combined for the analysis. A Cox proportional hazards model was used to test for non-inferiority against the pre-specified risk margin of 1.3 for the hazard ratio of MACE. Type-1 error was controlled across multiple tests using a hierarchical testing strategy.
The incidence rate of MACE was similar between the STEGLATRO-treated and placebo-treated patients. The estimated hazard ratio of MACE associated with STEGLATRO relative to placebo was 0.97 with 95.6% confidence interval (0.85, 1.11). The upper bound of this confidence interval excluded a risk larger than 1.3 (Table 10). Results for the individual 5 mg and 15 mg doses were consistent with results for the combined dose group.
| Endpoint MACE was evaluated in subjects who took at least one dose of study medication and, for subjects who discontinued study medication prior to the end of the study, censored events that occurred more than 365 days after the last dose of study medication. Other endpoints were evaluated using all randomized subjects and events that occurred any time after the first dose of study medication until the last contact date. The total number of first events was analyzed for each endpoint.
|
Placebo (N=2747) | STEGLATRO (N=5499) | Hazard Ratio vs Placebo (CI) HR and CI are based on Cox proportional hazards regression model, stratified by cohorts. For MACE a 95.6% CI is presented, for other endpoints a 95% CI is presented.
|
||
|---|---|---|---|---|---|
| N (%) | Event Rate (per 100 person-years) | N (%) | Event Rate (per 100 person-years) | ||
| N=Number of patients, CI=Confidence interval, CV=Cardiovascular, MI=Myocardial infarction. | |||||
| MACE (CV death, non-fatal MI, or non-fatal stroke) Composite | 327 (11.9) | 4.0 | 653 (11.9) | 3.9 | 0.97 (0.85, 1.11) |
| Components of Composite Endpoint | |||||
| Non-fatal MI | 148 (5.4) | 1.6 | 310 (5.6) | 1.7 | 1.04 (0.86, 1.27) |
| Non-fatal Stroke | 78 (2.8) | 0.8 | 157 (2.9) | 0.8 | 1.00 (0.76, 1.32) |
| CV death | 184 (6.7) | 1.9 | 341 (6.2) | 1.8 | 0.92 (0.77, 1.11) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:53.797668 · Updated: 2026-03-14T22:21:24.209091