Quinapril Tablets, Usp
e6d36e1e-8e35-4d06-83dc-45e0456365fd
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: FETAL TOXICITY When pregnancy is detected, discontinue quinapril as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Medication Information
Recent Major Changes
WARNING: FETAL TOXICITY
- When pregnancy is detected, discontinue quinapril as soon as possible.
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Description
WARNING: FETAL TOXICITY When pregnancy is detected, discontinue quinapril as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Section 42229-5
Quinapril hydrochloride, USP is the hydrochloride salt of quinapril, the ethyl ester of a non-sulfhydryl, angiotensin-converting enzyme (ACE) inhibitor, quinaprilat.
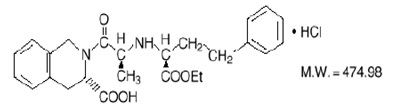
Quinapril hydrochloride, USP is chemically described as [3S-[2[R*(R*)], 3R*]]-2-[2-[1-(ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]-1,2,3,4-tetrahydro-3-isoquinolinecarboxylic acid, monohydrochloride. Its empirical formula is C25H30N2O5 •HCl and its structural formula is:
Quinapril hydrochloride, USP is a white to off-white amorphous powder that is freely soluble in aqueous solvents.
Quinapril tablets, USP contain quinapril hydrochloride equivalent to 5 mg, 10 mg, 20 mg, or 40 mg of quinapril for oral administration. Each tablet also contains lactose monohydrate, magnesium carbonate, magnesium stearate, crospovidone, povidone and opadry brown (hypromellose, titanium dioxide, iron oxide and macrogol).
Package Label.principal Display Panel
NDC 69097-839-05 Rx Only
Quinapril Tablets, USP
5 mg*
90 Tablets
Cipla
NDC 69097-841-05 Rx Only
Quinapril Tablets, USP
10 mg*
90 Tablets
Cipla
NDC 69097-842-05 Rx Only
Quinapril Tablets, USP
20 mg*
90 Tablets
Cipla
NDC 69097-843-05 Rx Only
Quinapril Tablets, USP
40 mg*
90 Tablets
Cipla
Structured Label Content
Recent Major Changes (34066-1)
WARNING: FETAL TOXICITY
- When pregnancy is detected, discontinue quinapril as soon as possible.
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See Warnings: Fetal Toxicity
Section 42229-5 (42229-5)
Quinapril hydrochloride, USP is the hydrochloride salt of quinapril, the ethyl ester of a non-sulfhydryl, angiotensin-converting enzyme (ACE) inhibitor, quinaprilat.
Quinapril hydrochloride, USP is chemically described as [3S-[2[R*(R*)], 3R*]]-2-[2-[1-(ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]-1,2,3,4-tetrahydro-3-isoquinolinecarboxylic acid, monohydrochloride. Its empirical formula is C25H30N2O5 •HCl and its structural formula is:
Quinapril hydrochloride, USP is a white to off-white amorphous powder that is freely soluble in aqueous solvents.
Quinapril tablets, USP contain quinapril hydrochloride equivalent to 5 mg, 10 mg, 20 mg, or 40 mg of quinapril for oral administration. Each tablet also contains lactose monohydrate, magnesium carbonate, magnesium stearate, crospovidone, povidone and opadry brown (hypromellose, titanium dioxide, iron oxide and macrogol).
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 69097-839-05 Rx Only
Quinapril Tablets, USP
5 mg*
90 Tablets
Cipla
NDC 69097-841-05 Rx Only
Quinapril Tablets, USP
10 mg*
90 Tablets
Cipla
NDC 69097-842-05 Rx Only
Quinapril Tablets, USP
20 mg*
90 Tablets
Cipla
NDC 69097-843-05 Rx Only
Quinapril Tablets, USP
40 mg*
90 Tablets
Cipla
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:44.123132 · Updated: 2026-03-14T21:51:39.333033