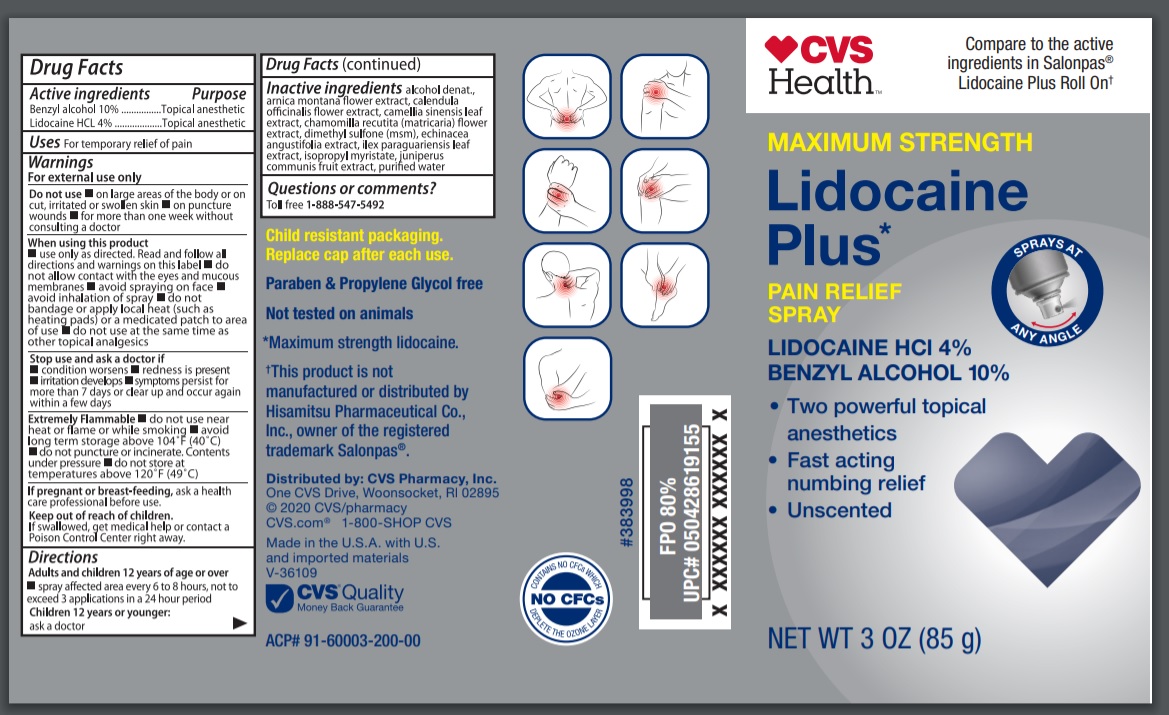
Cvs Health Lidocaine Plus
e6aff579-affb-41bb-8e36-53ea484cb0c5
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzyl alcohol 10% Lidocaine HCl 4%
Purpose
Topical anesthetic
Medication Information
Purpose
Topical anesthetic
Description
Benzyl alcohol 10% Lidocaine HCl 4%
Uses
For temporary relief of pain
Warnings
For external use only
Directions
Adults and children 12 years of age and over:
- spray to affected area not more than 3 to 4 times daily
Children under 12 years of age:
consult a doctor
Do Not Use
- on large areas of the body or cut or wounds or damaged skin
- on puncture wounds
- for more than one week without consulting a doctor
Flammable:
Keep away from fire or flame
-Do not use near heat or flame or while smoking.
-avoid long term storage above 40 degree Celcius.
-do not puncture or incinerate. COntents under pressure
-do not store at temperatures above 49 degree Celcius
Other Information
- Avoid storing product in direct sunlight
- Protect product from excessive moisture
- Store with lid closed tighly
Active Ingredients
Benzyl alcohol 10%
Lidocaine HCl 4%
Inactive Ingredients
alcohol denatured, arnica montana flower extract, calendula officinalis flower extract, camellia sinensis leaf extract, chamomilla recutita (matricaria) flower extract , dimethyl sulfone (msm), Echinacea angustifolia extract, ilex paraguariensis leaf extract, isopropyl myristate, juniperus communis fruit extract, purified water
Questions Or Comments?
Toll free 1-888-547-5492
Principal Display Panel
When Using This Product
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- avoid spraying on face
- avoid inhalation of spray
- do not bandage or apply local heat such as heating pads or a medicated patch to area of use
- do not use at the same time as other topical analgesics
Stop Use and Ask A Doctor If
- skin reactions occur, such as rash, itching, redness, irritaion, pain, swelling and blistering
- conditions worsen
- symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
For temporary relief of pain
Purpose
Topical anesthetic
Warnings
For external use only
Directions
Adults and children 12 years of age and over:
- spray to affected area not more than 3 to 4 times daily
Children under 12 years of age:
consult a doctor
Do Not Use (Do not use)
- on large areas of the body or cut or wounds or damaged skin
- on puncture wounds
- for more than one week without consulting a doctor
Flammable:
Keep away from fire or flame
-Do not use near heat or flame or while smoking.
-avoid long term storage above 40 degree Celcius.
-do not puncture or incinerate. COntents under pressure
-do not store at temperatures above 49 degree Celcius
Other Information (Other information)
- Avoid storing product in direct sunlight
- Protect product from excessive moisture
- Store with lid closed tighly
Active Ingredients (Active ingredients)
Benzyl alcohol 10%
Lidocaine HCl 4%
Inactive Ingredients (Inactive ingredients)
alcohol denatured, arnica montana flower extract, calendula officinalis flower extract, camellia sinensis leaf extract, chamomilla recutita (matricaria) flower extract , dimethyl sulfone (msm), Echinacea angustifolia extract, ilex paraguariensis leaf extract, isopropyl myristate, juniperus communis fruit extract, purified water
Questions Or Comments? (Questions or comments?)
Toll free 1-888-547-5492
Principal Display Panel
When Using This Product (When using this product)
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- avoid spraying on face
- avoid inhalation of spray
- do not bandage or apply local heat such as heating pads or a medicated patch to area of use
- do not use at the same time as other topical analgesics
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- skin reactions occur, such as rash, itching, redness, irritaion, pain, swelling and blistering
- conditions worsen
- symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:23.676985 · Updated: 2026-03-14T23:01:55.379030