e69d7f9e-881e-4502-a239-03308387bbac
34390-5
HUMAN OTC DRUG LABEL
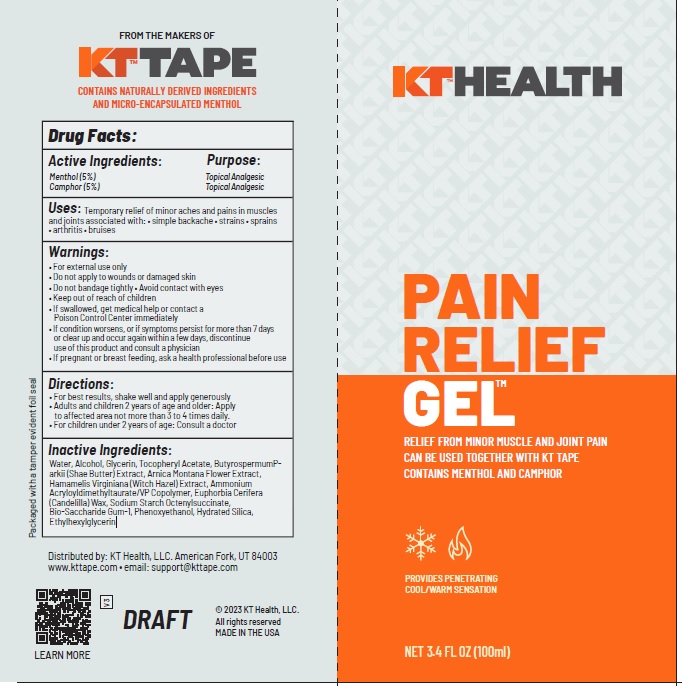
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients Purpose Camphor (5%) Topical Analgesic Menthol (5%) Topical Analgesic
Medication Information
Description
Active Ingredients Purpose Camphor (5%) Topical Analgesic Menthol (5%) Topical Analgesic
Uses
Temporary relief of minor aches and pains in muscles and joints associated with: • simple backache • strains • sprains
• arthritis • bruises
Section 51727-6
Water, Alcohol, Glycerin, Tocopheryl Acetate, ButyrospermumParkii (Shae Butter) Extract, Arnica Montana Flower Extract, Hamamelis Virginiana (Witch Hazel) Extract, Ammonium Acryloyldimethyltaurate/VP Copolymer, Euphorbia Cerifera (Candelilla) Wax, Sodium Starch Octenylsuccinate,
Bio-Saccharide Gum-1, Phenoxyethanol, Hydrated Silica, Ethylhexylglycerin
Section 55105-1
Section 55106-9
Active Ingredients Purpose
Camphor (5%) Topical Analgesic
Menthol (5%) Topical Analgesic
Warnings
• For external use only
• Do not apply to wounds or damaged skin
• Do not bandage tightly • Avoid contact with eyes
• Keep out of reach of children
• If swallowed, get medical help or contact a Poison Control Center immediately
• If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a physician
• If pregnant or breast feeding, ask a health professional before use
Directions
• For best results, shake well and apply generously
• Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
• For children under 2 years of age: Consult a doctor
Product Label
Structured Label Content
Uses
Temporary relief of minor aches and pains in muscles and joints associated with: • simple backache • strains • sprains
• arthritis • bruises
Section 51727-6 (51727-6)
Water, Alcohol, Glycerin, Tocopheryl Acetate, ButyrospermumParkii (Shae Butter) Extract, Arnica Montana Flower Extract, Hamamelis Virginiana (Witch Hazel) Extract, Ammonium Acryloyldimethyltaurate/VP Copolymer, Euphorbia Cerifera (Candelilla) Wax, Sodium Starch Octenylsuccinate,
Bio-Saccharide Gum-1, Phenoxyethanol, Hydrated Silica, Ethylhexylglycerin
Section 55105-1 (55105-1)
Section 55106-9 (55106-9)
Active Ingredients Purpose
Camphor (5%) Topical Analgesic
Menthol (5%) Topical Analgesic
Warnings
• For external use only
• Do not apply to wounds or damaged skin
• Do not bandage tightly • Avoid contact with eyes
• Keep out of reach of children
• If swallowed, get medical help or contact a Poison Control Center immediately
• If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a physician
• If pregnant or breast feeding, ask a health professional before use
Directions
• For best results, shake well and apply generously
• Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
• For children under 2 years of age: Consult a doctor
Product Label (Product label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:27.519597 · Updated: 2026-03-14T23:05:12.885917