These Highlights Do Not Include All The Information Needed To Use Tnkase Safely And Effectively. See Full Prescribing Information For Tnkase.
e647640d-c395-4b4b-a0be-1162f9c21d84
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1.1 ) 02/2025 Dosage and Administration ( 2.1 , 2.3 ) 02/2025 Contraindications ( 4 ) 02/2025 Warnings and Precautions ( 5.1 ) 02/2025
Indications and Usage
TNKase is a tissue plasminogen activator (tPA) indicated: for the treatment of acute ischemic stroke (AIS) in adults. ( 1.1 ) to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI) in adults. ( 1.2 )
Dosage and Administration
TNKase is for intravenous administration only, administered as a single bolus over 5 seconds. ( 2.1 , 2.2 ) AIS Initiate treatment as soon as possible and within 3 hours after the onset of stroke symptoms. ( 2.1 ) Individualize dosage based on patient's weight; the maximum recommended dose is 25 mg (5 mL). ( 2.1 ) Acute STEMI Initiate treatment as soon as possible after the onset of STEMI symptoms. ( 2.2 ) Individualize dosage based on patient's weight; the maximum recommended dose is 50 mg (10 mL). ( 2.2 )
Warnings and Precautions
Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. ( 5.1 ) Hypersensitivity: Monitor patients treated with TNKase during and for several hours after administration. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids, epinephrine). ( 5.2 ) Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. ( 5.3 ) Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. ( 5.4 ) Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. ( 5.5 ) Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention in STEMI: In patients with a large ST segment elevation myocardial infarction, choose either thrombolysis or PCI as the primary treatment for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. ( 5.6 )
Contraindications
AIS and STEMI Active internal bleeding ( 4 ) Intracranial or intraspinal surgery or trauma within 2 months ( 4 ) Known bleeding diathesis ( 4 ) Current severe uncontrolled hypertension ( 4 ) Presence of intracranial conditions that may increase the risk of bleeding (e.g., intracranial neoplasm, arteriovenous malformation, or aneurysm) ( 4 ) AIS Active intracranial hemorrhage ( 4 ) Acute STEMI History of intracranial hemorrhage History of ischemic stroke within 3 months ( 4 )
Adverse Reactions
The following clinically significant adverse reactions are discussed in other sections of the label: Bleeding [see Contraindications (4) , Warnings and Precautions (5.1) ] Hypersensitivity [see Warnings and Precautions (5.2) ] Thromboembolism [see Warnings and Precautions (5.3) ] Cholesterol Embolization [see Warnings and Precautions (5.4) ] Arrhythmias [see Warnings and Precautions (5.5) ] Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI [see Warnings and Precautions (5.6) ]
Drug Interactions
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. ( 7.1 )
Storage and Handling
Store lyophilized TNKase at room temperature up to 30°C (86°F) or refrigerated at 2°C to 8°C (36°F to 46°F). Do not use beyond the expiration date stamped on the vial. For storage information for reconstituted TNKase, see Dosage and Administration (2.4) .
How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in single-dose vials under partial vacuum, co-packaged with a single-dose vial of Sterile Water for Injection, USP, for reconstitution, as follows: TNKase Strength Sterile Water for Injection Volume NDC 25 mg 5.2 mL 50242-014-03 50 mg 10 mL 50242-176-01
Medication Information
Warnings and Precautions
Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. ( 5.1 ) Hypersensitivity: Monitor patients treated with TNKase during and for several hours after administration. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids, epinephrine). ( 5.2 ) Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. ( 5.3 ) Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. ( 5.4 ) Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. ( 5.5 ) Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention in STEMI: In patients with a large ST segment elevation myocardial infarction, choose either thrombolysis or PCI as the primary treatment for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. ( 5.6 )
Indications and Usage
TNKase is a tissue plasminogen activator (tPA) indicated: for the treatment of acute ischemic stroke (AIS) in adults. ( 1.1 ) to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI) in adults. ( 1.2 )
Dosage and Administration
TNKase is for intravenous administration only, administered as a single bolus over 5 seconds. ( 2.1 , 2.2 ) AIS Initiate treatment as soon as possible and within 3 hours after the onset of stroke symptoms. ( 2.1 ) Individualize dosage based on patient's weight; the maximum recommended dose is 25 mg (5 mL). ( 2.1 ) Acute STEMI Initiate treatment as soon as possible after the onset of STEMI symptoms. ( 2.2 ) Individualize dosage based on patient's weight; the maximum recommended dose is 50 mg (10 mL). ( 2.2 )
Contraindications
AIS and STEMI Active internal bleeding ( 4 ) Intracranial or intraspinal surgery or trauma within 2 months ( 4 ) Known bleeding diathesis ( 4 ) Current severe uncontrolled hypertension ( 4 ) Presence of intracranial conditions that may increase the risk of bleeding (e.g., intracranial neoplasm, arteriovenous malformation, or aneurysm) ( 4 ) AIS Active intracranial hemorrhage ( 4 ) Acute STEMI History of intracranial hemorrhage History of ischemic stroke within 3 months ( 4 )
Adverse Reactions
The following clinically significant adverse reactions are discussed in other sections of the label: Bleeding [see Contraindications (4) , Warnings and Precautions (5.1) ] Hypersensitivity [see Warnings and Precautions (5.2) ] Thromboembolism [see Warnings and Precautions (5.3) ] Cholesterol Embolization [see Warnings and Precautions (5.4) ] Arrhythmias [see Warnings and Precautions (5.5) ] Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI [see Warnings and Precautions (5.6) ]
Drug Interactions
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. ( 7.1 )
Storage and Handling
Store lyophilized TNKase at room temperature up to 30°C (86°F) or refrigerated at 2°C to 8°C (36°F to 46°F). Do not use beyond the expiration date stamped on the vial. For storage information for reconstituted TNKase, see Dosage and Administration (2.4) .
How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in single-dose vials under partial vacuum, co-packaged with a single-dose vial of Sterile Water for Injection, USP, for reconstitution, as follows: TNKase Strength Sterile Water for Injection Volume NDC 25 mg 5.2 mL 50242-014-03 50 mg 10 mL 50242-176-01
Description
Indications and Usage ( 1.1 ) 02/2025 Dosage and Administration ( 2.1 , 2.3 ) 02/2025 Contraindications ( 4 ) 02/2025 Warnings and Precautions ( 5.1 ) 02/2025
Section 42229-5
AIS and Acute STEMI
TNKase is contraindicated in any patients with:
- Active internal bleeding
- Intracranial or intraspinal surgery or trauma within 2 months
- Known bleeding diathesis
- Current severe uncontrolled hypertension
- Presence of intracranial conditions that may increase the risk of bleeding (e.g., intracranial neoplasm, arteriovenous malformation, or aneurysm)
Section 43683-2
5.1 Bleeding
TNKase can cause significant, sometimes fatal, internal or external bleeding, especially at arterial and venous puncture sites. Concomitant use of other drugs that impair hemostasis increases the risk of bleeding. Avoid intramuscular injections and trauma to the patient while on TNKase. Perform arterial and venous punctures carefully and only as required. To minimize bleeding from noncompressible sites, avoid internal jugular and subclavian venous punctures. If an arterial puncture is necessary during TNKase administration, use an upper extremity vessel that is accessible to manual compression, apply pressure for at least 30 minutes, and monitor the puncture site closely.
Should serious bleeding that is not controlled by local pressure occur, discontinue any concomitant heparin or antiplatelet agents immediately and treat appropriately. Because of the higher risk of intracranial hemorrhage in patients treated for acute ischemic stroke, limit treatment to facilities that can provide timely access to appropriate evaluation and management of intracranial hemorrhage.
Aspirin and heparin have been administered concomitantly with and following administration of TNKase in the management of acute myocardial infarction, but the concomitant administration of heparin and aspirin with and following administration of TNKase for the treatment of acute ischemic stroke during the first 24 hours after symptom onset has not been investigated. Because heparin, aspirin, or TNKase may cause bleeding complications, carefully monitor for bleeding, especially at arterial puncture sites. Hemorrhage can occur 1 or more days after administration of TNKase, while patients are still receiving anticoagulant therapy.
If serious bleeding occurs, treat appropriately. In the following conditions, the risks of bleeding with TNKase therapy for all approved indications are increased and should be weighed against the anticipated benefits:
- Recent major surgery or procedure (e.g., coronary artery bypass graft, obstetrical delivery, organ biopsy, previous puncture of noncompressible vessels)
- Cerebrovascular disease
- Recent intracranial hemorrhage (if not contraindicated)
- Recent gastrointestinal or genitourinary bleeding
- Recent trauma
- Hypertension: systolic BP above 175 mm Hg or diastolic BP above 110 mm Hg
- Acute pericarditis
- Subacute bacterial endocarditis
- Hemostatic defects including those secondary to severe hepatic or renal disease
- Significant hepatic dysfunction
- Pregnancy
- Diabetic hemorrhagic retinopathy, or other hemorrhagic ophthalmic conditions
- Septic thrombophlebitis or occluded AV cannula at seriously infected site
- Advanced age [see Use in Specific Populations (8.5)]
- Patients currently receiving anticoagulants (e.g., warfarin sodium)
Any other condition in which bleeding constitutes a significant hazard or would be particularly difficult to manage because of its location.
11 Description
Tenecteplase is a tissue plasminogen activator (tPA) produced by recombinant DNA technology using a mammalian cell line (Chinese Hamster Ovary cells). Tenecteplase is a 527-amino acid glycoprotein developed by introducing the following modifications to the complementary DNA (cDNA) for natural human tPA: a substitution of threonine 103 with asparagine, and a substitution of asparagine 117 with glutamine, both within the kringle 1 domain, and a tetra-alanine substitution at amino acids 296–299 in the protease domain. It has a molecular weight of 58,742 daltons. Biological potency is determined by an in vitro clot lysis assay and is expressed in tenecteplase specific units. The specific activity of tenecteplase has been defined as 200 units/mg.
TNKase (tenecteplase) for injection is a sterile, white to pale yellow, lyophilized powder for intravenous bolus administration after reconstitution with Sterile Water for Injection, USP.
Each 25 mg single-dose vial of TNKase nominally contains 25 mg of tenecteplase, arginine (261 mg), phosphoric acid (approximately 80 mg), and polysorbate 20 (2.0 mg). Following reconstitution with the supplied 5.2 mL single-dose vial of Sterile Water for Injection, USP, the final concentration is 5 mg/mL with a pH of approximately 7.3.
Each 50 mg single-dose vial of TNKase nominally contains 50 mg of tenecteplase, arginine (522 mg), phosphoric acid (approximately 160 mg), and polysorbate 20 (4.0 mg). Following reconstitution with the supplied 10 mL single-dose vial of Sterile Water for Injection, USP, the final concentration is 5 mg/mL with a pH of approximately 7.3.
2.3 Preparation
Follow the steps below to prepare TNKase for administration:
- Only use the supplied Sterile Water for Injection diluent vial for reconstitution as shown below.
| TNKase Vial Strength | Sterile Water for Injection Vial Volume |
|---|---|
| 25 mg | 5.2 mL |
| 50 mg | 10 mL |
- Using a sterile syringe, aseptically withdraw the Sterile Water for Injection from the diluent vial and reconstitute the TNKase vial by directing the stream into the lyophilized powder to obtain a final concentration of 5 mg/mL. Slight foaming upon reconstitution is not unusual; any large bubbles will dissipate if the product is allowed to stand undisturbed for several minutes.
- Gently swirl until contents are completely dissolved. DO NOT SHAKE. The reconstituted preparation results in a colorless to pale yellow transparent solution.
- Determine the appropriate dose of TNKase [see Dosage and Administration (2.1, 2.2)] and withdraw the required volume (in milliliters) from the reconstituted vial into the syringe. Discard any unused solution.
5.5 Arrhythmias
Coronary thrombolysis may result in arrhythmias associated with reperfusion. These arrhythmias (such as sinus bradycardia, accelerated idioventricular rhythm, ventricular premature depolarizations, ventricular tachycardia) may be managed with standard anti-arrhythmic measures. It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered.
16.1 How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in single-dose vials under partial vacuum, co-packaged with a single-dose vial of Sterile Water for Injection, USP, for reconstitution, as follows:
| TNKase Strength | Sterile Water for Injection Volume | NDC |
|---|---|---|
| 25 mg | 5.2 mL | 50242-014-03 |
| 50 mg | 10 mL | 50242-176-01 |
8.4 Pediatric Use
The safety and effectiveness of TNKase in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of TNKase-treated patients within 0-3 hours in Trial 1 for acute ischemic stroke (AIS), 426 (72%) were 65 years of age and older, and 290 (49%) were 75 years of age and older [see Clinical Studies (14.1)]. No overall differences in safety were observed between patients over 65 years old with AIS and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
In the ASSENT-2 study, 41% (3500/8458) of STEMI patients who were treated with TNKase were aged 65 years or older. In this population, rates of 30-day mortality, stroke, intracranial hemorrhage and major bleeds requiring blood transfusion or leading to hemodynamic complications were higher than in those aged less than 65 years.
2.4 Administration
Follow the steps below for administration of TNKase:
- Visually inspect the reconstituted product in the syringe for particulate matter and discoloration prior to administration.
- Precipitation may occur when TNKase is administered in an intravenous line containing dextrose. Flush dextrose-containing lines with 0.9% Sodium Chloride Injection solution prior to and following single bolus administration of TNKase.
- Using sterile technique, connect the syringe directly to the intravenous port.
- Administer reconstituted TNKase as a single intravenous bolus over 5 seconds.
- Because TNKase contains no antibacterial preservatives, reconstitute immediately before use. If the reconstituted TNKase is not used immediately, refrigerate the TNKase vial at 2°C to 8°C (36°F to 46°F) and use within 8 hours.
- Dispose of the syringe per established procedures.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of tenecteplase or of other tenecteplase products.
In a study of subjects with STEMI, four of 625 (0.64%) STEMI patients tested for antibody formation to TNKase had a positive antibody titer at 30 days in studies with TNKase.
4 Contraindications
-
AIS and STEMI
- Active internal bleeding (4)
- Intracranial or intraspinal surgery or trauma within 2 months (4)
- Known bleeding diathesis (4)
- Current severe uncontrolled hypertension (4)
- Presence of intracranial conditions that may increase the risk of bleeding (e.g., intracranial neoplasm, arteriovenous malformation, or aneurysm) (4)
-
AIS
- Active intracranial hemorrhage (4)
-
Acute STEMI
- History of intracranial hemorrhage
- History of ischemic stroke within 3 months (4)
5.3 Thromboembolism
The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus, such as patients with mitral stenosis or atrial fibrillation.
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in other sections of the label:
- Bleeding [see Contraindications (4), Warnings and Precautions (5.1)]
- Hypersensitivity [see Warnings and Precautions (5.2)]
- Thromboembolism [see Warnings and Precautions (5.3)]
- Cholesterol Embolization [see Warnings and Precautions (5.4)]
- Arrhythmias [see Warnings and Precautions (5.5)]
- Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI [see Warnings and Precautions (5.6)]
7 Drug Interactions
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. (7.1)
5.2 Hypersensitivity
Hypersensitivity, including urticarial / anaphylactic reactions, have been reported after administration of TNKase (e.g., anaphylaxis, angioedema, laryngeal edema, rash, and urticaria). Monitor patients treated with TNKase during and for several hours after administration. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids, epinephrine).
12.2 Pharmacodynamics
Following administration of 30, 40, or 50 mg of TNKase in patients with STEMI, there are decreases in circulating fibrinogen (4%–15%) and plasminogen (11%–24%).
1 Indications and Usage
12.1 Mechanism of Action
Tenecteplase is a modified form of human tissue plasminogen activator (tPA) that binds to fibrin and converts plasminogen to plasmin. In the presence of fibrin, in vitro studies demonstrate that tenecteplase-mediated conversion of plasminogen to plasmin is increased relative to its conversion in the absence of fibrin. This fibrin specificity decreases systemic activation of plasminogen and the resulting degradation of circulating fibrinogen as compared to a molecule lacking this property. The clinical significance of fibrin-specificity on safety (e.g., bleeding) or efficacy has not been established.
1.1 Acute Ischemic Stroke
TNKase is indicated for the treatment of acute ischemic stroke (AIS) in adults.
16.2 Storage and Handling
Store lyophilized TNKase at room temperature up to 30°C (86°F) or refrigerated at 2°C to 8°C (36°F to 46°F). Do not use beyond the expiration date stamped on the vial.
For storage information for reconstituted TNKase, see Dosage and Administration (2.4) .
14.1 Acute Ischemic Stroke
The Alteplase compared to Tenecteplase (AcT) trial (NCT03889249, Trial 1) was a registry-linked, multicenter, parallel group, open-label, randomized trial with blinded outcome assessment that investigated the non-inferiority of TNKase (tenecteplase) compared to Activase (alteplase) in treating patients with acute ischemic stroke (AIS) that presented with a disabling neurologic deficit. Endovascular thrombectomy was allowed if patients were eligible based on standard of care.
A total of 1147 patients were treated within 3 hours of symptom onset and were included in the intent to treat (ITT) population. In the ITT population, the mean age was 72 years, 53% of patients were male, 24% were White, 5% were Asian, and 70% did not have a race reported or their race was unknown. Among the ITT population, the median baseline National Institute of Health Stroke Scale (NIHSS) score was 10, 27% of patients had a large vessel occlusion on baseline CT angiography, and the median stroke symptom onset to thrombolysis time was 108 minutes. Demographics and other baseline characteristics were generally balanced between the treatment groups.
Patients were randomized (1:1) to receive a single IV bolus of TNKase or a one-hour infusion of 0.9 mg/kg Activase (10% administered as an IV bolus over 1 minute and the remaining 90% given as an infusion over 1 hour; maximum dose 90 mg). TNKase was dosed using actual or estimated weight in a body weight-tiered fashion at the recommended dosage [see Dosage and Administration (2.1)]. Administration of antiplatelet agents or other antithrombotic products were prohibited within the first 24 hours after administration of TNKase or Activase.
In Trial 1, efficacy was assessed as a pre-defined favorable clinical outcome based on the proportion of patients that were treated within 3 hours of symptom onset who achieved a modified Rankin Scale (mRS) score of 0–1 at 90–120 days, as assessed by the Rankin Focused Assessment. The mRS is a 7-point scale which captures disability after stroke; higher scores indicate an increased amount of disability. A score of 0 represents no symptoms or disability, where a score of 6 represents death. A score of 0 or 1 represents a favorable clinical outcome. The results comparing TNKase with Activase in patients treated within 3 hours of symptom onset are presented in Table 4. Study results demonstrated no significant differences between treatment groups. Although the trial enrolled patients who were treated within 0-4.5 hours of stroke symptom onset, the subset of patients treated within 0-3 hours was used to assess efficacy of TNKase because the comparator, Activase, is approved for use within 0-3 hours of stroke symptom onset.
| Endpoint | TNKase N = 592 |
Activase N = 555 |
|---|---|---|
| CI: Confidence Interval | ||
| mRS: Modified Rankin Score | ||
| Proportion of Patients with mRS 0-1 at 90-120 days | 36.6% | 35.9% |
| Unadjusted Risk Difference % (95% CI) | 0.7% (-4.9%, 6.3%) |
A similar treatment effect in achieving a mRS score 0–1 was observed in exploratory subgroups defined by age, gender, and baseline NIHSS score.
5 Warnings and Precautions
- Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. (5.1)
- Hypersensitivity: Monitor patients treated with TNKase during and for several hours after administration. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids, epinephrine). (5.2)
- Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. (5.3)
- Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. (5.4)
- Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. (5.5)
- Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention in STEMI: In patients with a large ST segment elevation myocardial infarction, choose either thrombolysis or PCI as the primary treatment for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. (5.6)
2 Dosage and Administration
3 Dosage Forms and Strengths
For injection: 25 mg or 50 mg as a white to pale yellow lyophilized powder in a single-dose vial for reconstitution with co-packaged Sterile Water for Injection, USP (diluent).
5.4 Cholesterol Embolization
Cholesterol embolism has been reported in patients treated with thrombolytic agents. Investigate cause of any new embolic event and treat appropriately.
2.5 Chemical Incompatibilities
TNKase is incompatible with dextrose containing solutions. When used together, precipitation may occur. Flush dextrose containing lines with 0.9% Sodium Chloride Injection solution before using TNKase.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most frequent adverse reaction associated with TNKase in all approved indications is bleeding.
7.1 Drug/laboratory Test Interactions
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. Tenecteplase is an enzyme that, when present in blood in pharmacologic concentrations, remains active under in vitro conditions. This can lead to degradation of fibrinogen in blood samples removed for analysis.
1.2 Acute St Elevation Myocardial Infarction
TNKase is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI) in adults.
Principal Display Panel Kit Carton 25 Mg
NDC 50242-014-03
TNKase®
(tenecteplase)
For Injection
25 mg per vial
For Intravenous Use
after Reconstitution
Single-Dose Vial
Discard Unused Portion
Carton contents:
One 25 mg vial TNKase
One 5.2 mL vial Sterile
Water for Injection, USP
Rx only
Genentech
11006671
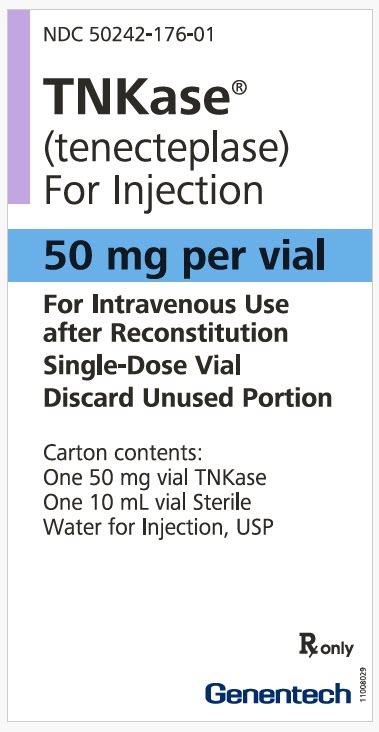
Principal Display Panel Kit Carton 50 Mg
NDC 50242-176-01
TNKase®
(tenecteplase)
For Injection
50 mg per vial
For Intravenous Use
after Reconstitution
Single-Dose Vial
Discard Unused Portion
Carton contents:
One 50 mg vial TNKase
One 10 mL vial Sterile
Water for Injection, USP
Rx only
Genentech
11008029
2.1 Recommended Dosage for Acute Ischemic Stroke
Initiate treatment as soon as possible and within 3 hours after the onset of stroke symptoms.
TNKase is for intravenous (IV) administration only, administered as a single bolus over 5 seconds. Individualize dosage based on the patient's weight (see Table 1). The maximum recommended dose is 25 mg (5 mL).
|
Patient Weight (kg) |
TNKase (mg) |
Volume TNKase to be administered (mL) |
|---|---|---|
| less than 60 kg | 15 mg | 3 mL |
| 60 kg to less than 70 kg | 17.5 mg | 3.5 mL |
| 70 kg to less than 80 kg | 20 mg | 4 mL |
| 80 kg to less than 90 kg | 22.5 mg | 4.5 mL |
| 90 kg or more | 25 mg | 5 mL |
During and following TNKase administration for the treatment of acute ischemic stroke, frequently monitor and control blood pressure.
In patients without recent use of oral anticoagulants or heparin, TNKase treatment can be initiated prior to the availability of coagulation study results. If the pretreatment International Normalized Ratio (INR) is greater than 1.7 or the activated partial thromboplastin time (aPTT) is elevated, closely monitor patients [see Contraindications (4)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals have not been performed to evaluate the carcinogenic potential, mutagenicity, or the effect on fertility.
2.2 Recommended Dosage for Acute St Elevation Myocardial Infarction
Initiate treatment as soon as possible after the onset of STEMI symptoms.
TNKase is for intravenous (IV) administration only, administered as a single bolus over 5 seconds. Individualize dosage based on the patient's weight (see Table 2). The maximum recommended dose is 50 mg (10 mL).
| Patient Weight (kg) |
TNKase (mg) |
Volume TNKase to be administered (mL) |
|---|---|---|
| less than 60 kg | 30 mg | 6 mL |
| 60 kg to less than 70 kg | 35 mg | 7 mL |
| 70 kg to less than 80 kg | 40 mg | 8 mL |
| 80 kg to less than 90 kg | 45 mg | 9 mL |
| 90 kg or more | 50 mg | 10 mL |
5.6 Increased Risk of Heart Failure and Recurrent Ischemia When Used With Planned Percutaneous Coronary Intervention (pci) in Stemi
In a trial of patients with STEMI, there were worse outcomes in the individual components of the primary endpoint between TNKase plus PCI versus PCI alone (mortality 6.7% vs. 4.9%, respectively; cardiogenic shock 6.3% vs. 4.8%, respectively; and CHF 12% vs. 9.2%, respectively). In addition, there were worse outcomes in recurrent MI (6.1% vs. 3.7%, respectively; p = 0.03) and repeat target vessel revascularization (6.6% vs. 3.4%, respectively; p = 0.0045) in patients receiving TNKase plus PCI versus PCI alone [see Clinical Studies (14.2)]. In patients with large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate; however, the optimal use of adjunctive antithrombotic and antiplatelet therapies in this setting is unknown.
Structured Label Content
Section 42229-5 (42229-5)
AIS and Acute STEMI
TNKase is contraindicated in any patients with:
- Active internal bleeding
- Intracranial or intraspinal surgery or trauma within 2 months
- Known bleeding diathesis
- Current severe uncontrolled hypertension
- Presence of intracranial conditions that may increase the risk of bleeding (e.g., intracranial neoplasm, arteriovenous malformation, or aneurysm)
Section 43683-2 (43683-2)
5.1 Bleeding
TNKase can cause significant, sometimes fatal, internal or external bleeding, especially at arterial and venous puncture sites. Concomitant use of other drugs that impair hemostasis increases the risk of bleeding. Avoid intramuscular injections and trauma to the patient while on TNKase. Perform arterial and venous punctures carefully and only as required. To minimize bleeding from noncompressible sites, avoid internal jugular and subclavian venous punctures. If an arterial puncture is necessary during TNKase administration, use an upper extremity vessel that is accessible to manual compression, apply pressure for at least 30 minutes, and monitor the puncture site closely.
Should serious bleeding that is not controlled by local pressure occur, discontinue any concomitant heparin or antiplatelet agents immediately and treat appropriately. Because of the higher risk of intracranial hemorrhage in patients treated for acute ischemic stroke, limit treatment to facilities that can provide timely access to appropriate evaluation and management of intracranial hemorrhage.
Aspirin and heparin have been administered concomitantly with and following administration of TNKase in the management of acute myocardial infarction, but the concomitant administration of heparin and aspirin with and following administration of TNKase for the treatment of acute ischemic stroke during the first 24 hours after symptom onset has not been investigated. Because heparin, aspirin, or TNKase may cause bleeding complications, carefully monitor for bleeding, especially at arterial puncture sites. Hemorrhage can occur 1 or more days after administration of TNKase, while patients are still receiving anticoagulant therapy.
If serious bleeding occurs, treat appropriately. In the following conditions, the risks of bleeding with TNKase therapy for all approved indications are increased and should be weighed against the anticipated benefits:
- Recent major surgery or procedure (e.g., coronary artery bypass graft, obstetrical delivery, organ biopsy, previous puncture of noncompressible vessels)
- Cerebrovascular disease
- Recent intracranial hemorrhage (if not contraindicated)
- Recent gastrointestinal or genitourinary bleeding
- Recent trauma
- Hypertension: systolic BP above 175 mm Hg or diastolic BP above 110 mm Hg
- Acute pericarditis
- Subacute bacterial endocarditis
- Hemostatic defects including those secondary to severe hepatic or renal disease
- Significant hepatic dysfunction
- Pregnancy
- Diabetic hemorrhagic retinopathy, or other hemorrhagic ophthalmic conditions
- Septic thrombophlebitis or occluded AV cannula at seriously infected site
- Advanced age [see Use in Specific Populations (8.5)]
- Patients currently receiving anticoagulants (e.g., warfarin sodium)
Any other condition in which bleeding constitutes a significant hazard or would be particularly difficult to manage because of its location.
11 Description (11 DESCRIPTION)
Tenecteplase is a tissue plasminogen activator (tPA) produced by recombinant DNA technology using a mammalian cell line (Chinese Hamster Ovary cells). Tenecteplase is a 527-amino acid glycoprotein developed by introducing the following modifications to the complementary DNA (cDNA) for natural human tPA: a substitution of threonine 103 with asparagine, and a substitution of asparagine 117 with glutamine, both within the kringle 1 domain, and a tetra-alanine substitution at amino acids 296–299 in the protease domain. It has a molecular weight of 58,742 daltons. Biological potency is determined by an in vitro clot lysis assay and is expressed in tenecteplase specific units. The specific activity of tenecteplase has been defined as 200 units/mg.
TNKase (tenecteplase) for injection is a sterile, white to pale yellow, lyophilized powder for intravenous bolus administration after reconstitution with Sterile Water for Injection, USP.
Each 25 mg single-dose vial of TNKase nominally contains 25 mg of tenecteplase, arginine (261 mg), phosphoric acid (approximately 80 mg), and polysorbate 20 (2.0 mg). Following reconstitution with the supplied 5.2 mL single-dose vial of Sterile Water for Injection, USP, the final concentration is 5 mg/mL with a pH of approximately 7.3.
Each 50 mg single-dose vial of TNKase nominally contains 50 mg of tenecteplase, arginine (522 mg), phosphoric acid (approximately 160 mg), and polysorbate 20 (4.0 mg). Following reconstitution with the supplied 10 mL single-dose vial of Sterile Water for Injection, USP, the final concentration is 5 mg/mL with a pH of approximately 7.3.
2.3 Preparation
Follow the steps below to prepare TNKase for administration:
- Only use the supplied Sterile Water for Injection diluent vial for reconstitution as shown below.
| TNKase Vial Strength | Sterile Water for Injection Vial Volume |
|---|---|
| 25 mg | 5.2 mL |
| 50 mg | 10 mL |
- Using a sterile syringe, aseptically withdraw the Sterile Water for Injection from the diluent vial and reconstitute the TNKase vial by directing the stream into the lyophilized powder to obtain a final concentration of 5 mg/mL. Slight foaming upon reconstitution is not unusual; any large bubbles will dissipate if the product is allowed to stand undisturbed for several minutes.
- Gently swirl until contents are completely dissolved. DO NOT SHAKE. The reconstituted preparation results in a colorless to pale yellow transparent solution.
- Determine the appropriate dose of TNKase [see Dosage and Administration (2.1, 2.2)] and withdraw the required volume (in milliliters) from the reconstituted vial into the syringe. Discard any unused solution.
5.5 Arrhythmias
Coronary thrombolysis may result in arrhythmias associated with reperfusion. These arrhythmias (such as sinus bradycardia, accelerated idioventricular rhythm, ventricular premature depolarizations, ventricular tachycardia) may be managed with standard anti-arrhythmic measures. It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered.
16.1 How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in single-dose vials under partial vacuum, co-packaged with a single-dose vial of Sterile Water for Injection, USP, for reconstitution, as follows:
| TNKase Strength | Sterile Water for Injection Volume | NDC |
|---|---|---|
| 25 mg | 5.2 mL | 50242-014-03 |
| 50 mg | 10 mL | 50242-176-01 |
8.4 Pediatric Use
The safety and effectiveness of TNKase in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of TNKase-treated patients within 0-3 hours in Trial 1 for acute ischemic stroke (AIS), 426 (72%) were 65 years of age and older, and 290 (49%) were 75 years of age and older [see Clinical Studies (14.1)]. No overall differences in safety were observed between patients over 65 years old with AIS and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
In the ASSENT-2 study, 41% (3500/8458) of STEMI patients who were treated with TNKase were aged 65 years or older. In this population, rates of 30-day mortality, stroke, intracranial hemorrhage and major bleeds requiring blood transfusion or leading to hemodynamic complications were higher than in those aged less than 65 years.
2.4 Administration
Follow the steps below for administration of TNKase:
- Visually inspect the reconstituted product in the syringe for particulate matter and discoloration prior to administration.
- Precipitation may occur when TNKase is administered in an intravenous line containing dextrose. Flush dextrose-containing lines with 0.9% Sodium Chloride Injection solution prior to and following single bolus administration of TNKase.
- Using sterile technique, connect the syringe directly to the intravenous port.
- Administer reconstituted TNKase as a single intravenous bolus over 5 seconds.
- Because TNKase contains no antibacterial preservatives, reconstitute immediately before use. If the reconstituted TNKase is not used immediately, refrigerate the TNKase vial at 2°C to 8°C (36°F to 46°F) and use within 8 hours.
- Dispose of the syringe per established procedures.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of tenecteplase or of other tenecteplase products.
In a study of subjects with STEMI, four of 625 (0.64%) STEMI patients tested for antibody formation to TNKase had a positive antibody titer at 30 days in studies with TNKase.
4 Contraindications (4 CONTRAINDICATIONS)
-
AIS and STEMI
- Active internal bleeding (4)
- Intracranial or intraspinal surgery or trauma within 2 months (4)
- Known bleeding diathesis (4)
- Current severe uncontrolled hypertension (4)
- Presence of intracranial conditions that may increase the risk of bleeding (e.g., intracranial neoplasm, arteriovenous malformation, or aneurysm) (4)
-
AIS
- Active intracranial hemorrhage (4)
-
Acute STEMI
- History of intracranial hemorrhage
- History of ischemic stroke within 3 months (4)
5.3 Thromboembolism
The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus, such as patients with mitral stenosis or atrial fibrillation.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in other sections of the label:
- Bleeding [see Contraindications (4), Warnings and Precautions (5.1)]
- Hypersensitivity [see Warnings and Precautions (5.2)]
- Thromboembolism [see Warnings and Precautions (5.3)]
- Cholesterol Embolization [see Warnings and Precautions (5.4)]
- Arrhythmias [see Warnings and Precautions (5.5)]
- Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. (7.1)
5.2 Hypersensitivity
Hypersensitivity, including urticarial / anaphylactic reactions, have been reported after administration of TNKase (e.g., anaphylaxis, angioedema, laryngeal edema, rash, and urticaria). Monitor patients treated with TNKase during and for several hours after administration. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids, epinephrine).
12.2 Pharmacodynamics
Following administration of 30, 40, or 50 mg of TNKase in patients with STEMI, there are decreases in circulating fibrinogen (4%–15%) and plasminogen (11%–24%).
1 Indications and Usage (1 INDICATIONS AND USAGE)
12.1 Mechanism of Action
Tenecteplase is a modified form of human tissue plasminogen activator (tPA) that binds to fibrin and converts plasminogen to plasmin. In the presence of fibrin, in vitro studies demonstrate that tenecteplase-mediated conversion of plasminogen to plasmin is increased relative to its conversion in the absence of fibrin. This fibrin specificity decreases systemic activation of plasminogen and the resulting degradation of circulating fibrinogen as compared to a molecule lacking this property. The clinical significance of fibrin-specificity on safety (e.g., bleeding) or efficacy has not been established.
1.1 Acute Ischemic Stroke
TNKase is indicated for the treatment of acute ischemic stroke (AIS) in adults.
16.2 Storage and Handling
Store lyophilized TNKase at room temperature up to 30°C (86°F) or refrigerated at 2°C to 8°C (36°F to 46°F). Do not use beyond the expiration date stamped on the vial.
For storage information for reconstituted TNKase, see Dosage and Administration (2.4) .
14.1 Acute Ischemic Stroke
The Alteplase compared to Tenecteplase (AcT) trial (NCT03889249, Trial 1) was a registry-linked, multicenter, parallel group, open-label, randomized trial with blinded outcome assessment that investigated the non-inferiority of TNKase (tenecteplase) compared to Activase (alteplase) in treating patients with acute ischemic stroke (AIS) that presented with a disabling neurologic deficit. Endovascular thrombectomy was allowed if patients were eligible based on standard of care.
A total of 1147 patients were treated within 3 hours of symptom onset and were included in the intent to treat (ITT) population. In the ITT population, the mean age was 72 years, 53% of patients were male, 24% were White, 5% were Asian, and 70% did not have a race reported or their race was unknown. Among the ITT population, the median baseline National Institute of Health Stroke Scale (NIHSS) score was 10, 27% of patients had a large vessel occlusion on baseline CT angiography, and the median stroke symptom onset to thrombolysis time was 108 minutes. Demographics and other baseline characteristics were generally balanced between the treatment groups.
Patients were randomized (1:1) to receive a single IV bolus of TNKase or a one-hour infusion of 0.9 mg/kg Activase (10% administered as an IV bolus over 1 minute and the remaining 90% given as an infusion over 1 hour; maximum dose 90 mg). TNKase was dosed using actual or estimated weight in a body weight-tiered fashion at the recommended dosage [see Dosage and Administration (2.1)]. Administration of antiplatelet agents or other antithrombotic products were prohibited within the first 24 hours after administration of TNKase or Activase.
In Trial 1, efficacy was assessed as a pre-defined favorable clinical outcome based on the proportion of patients that were treated within 3 hours of symptom onset who achieved a modified Rankin Scale (mRS) score of 0–1 at 90–120 days, as assessed by the Rankin Focused Assessment. The mRS is a 7-point scale which captures disability after stroke; higher scores indicate an increased amount of disability. A score of 0 represents no symptoms or disability, where a score of 6 represents death. A score of 0 or 1 represents a favorable clinical outcome. The results comparing TNKase with Activase in patients treated within 3 hours of symptom onset are presented in Table 4. Study results demonstrated no significant differences between treatment groups. Although the trial enrolled patients who were treated within 0-4.5 hours of stroke symptom onset, the subset of patients treated within 0-3 hours was used to assess efficacy of TNKase because the comparator, Activase, is approved for use within 0-3 hours of stroke symptom onset.
| Endpoint | TNKase N = 592 |
Activase N = 555 |
|---|---|---|
| CI: Confidence Interval | ||
| mRS: Modified Rankin Score | ||
| Proportion of Patients with mRS 0-1 at 90-120 days | 36.6% | 35.9% |
| Unadjusted Risk Difference % (95% CI) | 0.7% (-4.9%, 6.3%) |
A similar treatment effect in achieving a mRS score 0–1 was observed in exploratory subgroups defined by age, gender, and baseline NIHSS score.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. (5.1)
- Hypersensitivity: Monitor patients treated with TNKase during and for several hours after administration. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids, epinephrine). (5.2)
- Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. (5.3)
- Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. (5.4)
- Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. (5.5)
- Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention in STEMI: In patients with a large ST segment elevation myocardial infarction, choose either thrombolysis or PCI as the primary treatment for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 25 mg or 50 mg as a white to pale yellow lyophilized powder in a single-dose vial for reconstitution with co-packaged Sterile Water for Injection, USP (diluent).
5.4 Cholesterol Embolization
Cholesterol embolism has been reported in patients treated with thrombolytic agents. Investigate cause of any new embolic event and treat appropriately.
2.5 Chemical Incompatibilities
TNKase is incompatible with dextrose containing solutions. When used together, precipitation may occur. Flush dextrose containing lines with 0.9% Sodium Chloride Injection solution before using TNKase.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most frequent adverse reaction associated with TNKase in all approved indications is bleeding.
7.1 Drug/laboratory Test Interactions (7.1 Drug/Laboratory Test Interactions)
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. Tenecteplase is an enzyme that, when present in blood in pharmacologic concentrations, remains active under in vitro conditions. This can lead to degradation of fibrinogen in blood samples removed for analysis.
1.2 Acute St Elevation Myocardial Infarction (1.2 Acute ST Elevation Myocardial Infarction)
TNKase is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI) in adults.
Principal Display Panel Kit Carton 25 Mg (PRINCIPAL DISPLAY PANEL - Kit Carton - 25 mg)
NDC 50242-014-03
TNKase®
(tenecteplase)
For Injection
25 mg per vial
For Intravenous Use
after Reconstitution
Single-Dose Vial
Discard Unused Portion
Carton contents:
One 25 mg vial TNKase
One 5.2 mL vial Sterile
Water for Injection, USP
Rx only
Genentech
11006671
Principal Display Panel Kit Carton 50 Mg (PRINCIPAL DISPLAY PANEL - Kit Carton - 50 mg)
NDC 50242-176-01
TNKase®
(tenecteplase)
For Injection
50 mg per vial
For Intravenous Use
after Reconstitution
Single-Dose Vial
Discard Unused Portion
Carton contents:
One 50 mg vial TNKase
One 10 mL vial Sterile
Water for Injection, USP
Rx only
Genentech
11008029
2.1 Recommended Dosage for Acute Ischemic Stroke
Initiate treatment as soon as possible and within 3 hours after the onset of stroke symptoms.
TNKase is for intravenous (IV) administration only, administered as a single bolus over 5 seconds. Individualize dosage based on the patient's weight (see Table 1). The maximum recommended dose is 25 mg (5 mL).
|
Patient Weight (kg) |
TNKase (mg) |
Volume TNKase to be administered (mL) |
|---|---|---|
| less than 60 kg | 15 mg | 3 mL |
| 60 kg to less than 70 kg | 17.5 mg | 3.5 mL |
| 70 kg to less than 80 kg | 20 mg | 4 mL |
| 80 kg to less than 90 kg | 22.5 mg | 4.5 mL |
| 90 kg or more | 25 mg | 5 mL |
During and following TNKase administration for the treatment of acute ischemic stroke, frequently monitor and control blood pressure.
In patients without recent use of oral anticoagulants or heparin, TNKase treatment can be initiated prior to the availability of coagulation study results. If the pretreatment International Normalized Ratio (INR) is greater than 1.7 or the activated partial thromboplastin time (aPTT) is elevated, closely monitor patients [see Contraindications (4)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals have not been performed to evaluate the carcinogenic potential, mutagenicity, or the effect on fertility.
2.2 Recommended Dosage for Acute St Elevation Myocardial Infarction (2.2 Recommended Dosage for Acute ST Elevation Myocardial Infarction)
Initiate treatment as soon as possible after the onset of STEMI symptoms.
TNKase is for intravenous (IV) administration only, administered as a single bolus over 5 seconds. Individualize dosage based on the patient's weight (see Table 2). The maximum recommended dose is 50 mg (10 mL).
| Patient Weight (kg) |
TNKase (mg) |
Volume TNKase to be administered (mL) |
|---|---|---|
| less than 60 kg | 30 mg | 6 mL |
| 60 kg to less than 70 kg | 35 mg | 7 mL |
| 70 kg to less than 80 kg | 40 mg | 8 mL |
| 80 kg to less than 90 kg | 45 mg | 9 mL |
| 90 kg or more | 50 mg | 10 mL |
5.6 Increased Risk of Heart Failure and Recurrent Ischemia When Used With Planned Percutaneous Coronary Intervention (pci) in Stemi (5.6 Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI)
In a trial of patients with STEMI, there were worse outcomes in the individual components of the primary endpoint between TNKase plus PCI versus PCI alone (mortality 6.7% vs. 4.9%, respectively; cardiogenic shock 6.3% vs. 4.8%, respectively; and CHF 12% vs. 9.2%, respectively). In addition, there were worse outcomes in recurrent MI (6.1% vs. 3.7%, respectively; p = 0.03) and repeat target vessel revascularization (6.6% vs. 3.4%, respectively; p = 0.0045) in patients receiving TNKase plus PCI versus PCI alone [see Clinical Studies (14.2)]. In patients with large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate; however, the optimal use of adjunctive antithrombotic and antiplatelet therapies in this setting is unknown.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:33.931230 · Updated: 2026-03-14T22:40:50.579212