Methylphenidate Hydrochloride Extended-release Capsules (la)
e5db854a-1c77-4faf-9e60-daf9c9b6cfa0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Methylphenidate hydrochloride extended-release capsules (LA) is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD), in pediatric patients 6 to 12 years of age [see Clinical Studies (14) ] .
Dosage and Administration
Administer orally once daily in the morning ( 2.2 ). Capsules may be swallowed whole, or opened and the entire contents sprinkled on applesauce ( 2.2 ). Should not be crushed, chewed, or divided ( 2.2 ). Patients new to methylphenidate: Start at 20 mg daily, titrating the dose weekly in 10-mg increments. Doses above 60 mg daily are not recommended ( 2.3 ). For patients currently using methylphenidate hydrochloride tablets or methylphenidate hydrochloride extended-release tablets: Dosage is based on current dose regimen ( 2.4 ). If switching from other methylphenidate products, discontinue treatment and titrate with methylphenidate hydrochloride extended-release capsules (LA) ( 2.4 ).
Contraindications
Hypersensitivity to methylphenidate or other components of methylphenidate hydrochloride extended-release capsules (LA). Hypersensitivity reactions, such as angioedema and anaphylactic reactions, have been reported in patients treated with methylphenidate [see Adverse Reactions (6.1) ]. Concomitant treatment with monoamine oxidase inhibitors (MAOIs), or within 14 days following discontinuation of treatment with an MAOI, because of the risk of hypertensive crises [ s ee Drug Interactions (7.1) ].
Warnings and Precautions
Serious Cardiovascular Events : Sudden death has been reported in association with CNS-stimulant treatment at usual doses in pediatric patients with structural cardiac abnormalities or other serious heart problems. In adults, sudden death, stroke, and myocardial infarction have been reported. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm arrhythmias, or coronary artery disease ( 5.2 ). Blood Pressure and Heart Rate Increases : Monitor blood pressure and pulse. Consider the benefits and risk in patients for whom an increase in blood pressure or heart rate would be problematic ( 5.3 ). Psychiatric Adverse Reactions : Use of stimulants may cause psychotic or manic symptoms in patients with no prior history or exacerbation of symptoms in patients with preexisting psychiatric illness. Evaluate for preexisting psychotic or bipolar disorder prior to methylphenidate hydrochloride extended-release capsules (LA) use ( 5.4 ). Priapism : Cases of painful and prolonged penile erections and priapism have been reported with methylphenidate products. Immediate medical attention should be sought if signs or symptoms of prolonged penile erections or priapism are observed ( 5.5 ). Peripheral Vasculopathy, including Raynaud's Phenomenon : Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants ( 5.6 ). Long-Term Suppression of Growth : Monitor height and weight at appropriate intervals in pediatric patients ( 5.7 ).
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Abuse and Dependence [see Boxed Warning , Warnings and Precautions (5.1) , Drug Abuse and Dependence (9.2 , 9.3) ] Known hypersensitivity to methylphenidate or other ingredients of methylphenidate hydrochloride extended-release capsules (LA) [see Contraindications (4) ] Hypertensive crisis when used concomitantly with Monoamine Oxidase Inhibitors [see Contraindications (4) , Drug Interactions (7.1) ] Serious Cardiovascular Reactions [see Warnings and Precautions (5.2) ] Blood Pressure and Heart Rate Increases [see Warnings and Precautions (5.3) ] Psychiatric Adverse Reactions [see Warnings and Precautions (5.4) ] Priapism [see Warnings and Precautions (5.5) ] Peripheral Vasculopathy, Including Raynaud's Phenomenon [see Warnings and Precautions (5.6) ] Long-Term Suppression of Growth [see Warnings and Precautions (5.7) ]
Drug Interactions
Antihypertensive drugs : Monitor blood pressure and heart. Adjust dosage of antihypertensive drug as needed ( 7.1 ). Halogenated Anesthetics: Avoid use of methylphenidate hydrochloride extended-release capsules (LA) on the day of surgery if halogenated anesthetics will be used ( 7.1 ).
How Supplied
10 mg white/light green capsules (imprinted mayne 609 on both the body and the cap) Bottles of 100 NDC 51862-609-01 20 mg white/white capsules (imprinted mayne 610 on both the body and cap Bottles of 100 NDC 51862-610-01 30 mg white/light blue capsules (imprinted mayne 611 on both the body and cap Bottles of 100 NDC 51862-611-01 40 mg white/dark blue capsules (imprinted mayne 612 on both the body and cap) Bottles of 100 NDC 51862-612-01 60 mg light yellow/dark yellow capsules (imprinted mayne 614 on both the body and cap) Bottles of 30 NDC 51862-614-30
Storage and Handling
10 mg white/light green capsules (imprinted mayne 609 on both the body and the cap) Bottles of 100 NDC 51862-609-01 20 mg white/white capsules (imprinted mayne 610 on both the body and cap Bottles of 100 NDC 51862-610-01 30 mg white/light blue capsules (imprinted mayne 611 on both the body and cap Bottles of 100 NDC 51862-611-01 40 mg white/dark blue capsules (imprinted mayne 612 on both the body and cap) Bottles of 100 NDC 51862-612-01 60 mg light yellow/dark yellow capsules (imprinted mayne 614 on both the body and cap) Bottles of 30 NDC 51862-614-30
Description
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Warning and Precautions (5.1) , Drug Abuse and Dependence (9.2 , 9.3) ] .
Medication Information
Warnings and Precautions
Serious Cardiovascular Events : Sudden death has been reported in association with CNS-stimulant treatment at usual doses in pediatric patients with structural cardiac abnormalities or other serious heart problems. In adults, sudden death, stroke, and myocardial infarction have been reported. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm arrhythmias, or coronary artery disease ( 5.2 ). Blood Pressure and Heart Rate Increases : Monitor blood pressure and pulse. Consider the benefits and risk in patients for whom an increase in blood pressure or heart rate would be problematic ( 5.3 ). Psychiatric Adverse Reactions : Use of stimulants may cause psychotic or manic symptoms in patients with no prior history or exacerbation of symptoms in patients with preexisting psychiatric illness. Evaluate for preexisting psychotic or bipolar disorder prior to methylphenidate hydrochloride extended-release capsules (LA) use ( 5.4 ). Priapism : Cases of painful and prolonged penile erections and priapism have been reported with methylphenidate products. Immediate medical attention should be sought if signs or symptoms of prolonged penile erections or priapism are observed ( 5.5 ). Peripheral Vasculopathy, including Raynaud's Phenomenon : Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants ( 5.6 ). Long-Term Suppression of Growth : Monitor height and weight at appropriate intervals in pediatric patients ( 5.7 ).
Indications and Usage
Methylphenidate hydrochloride extended-release capsules (LA) is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD), in pediatric patients 6 to 12 years of age [see Clinical Studies (14) ] .
Dosage and Administration
Administer orally once daily in the morning ( 2.2 ). Capsules may be swallowed whole, or opened and the entire contents sprinkled on applesauce ( 2.2 ). Should not be crushed, chewed, or divided ( 2.2 ). Patients new to methylphenidate: Start at 20 mg daily, titrating the dose weekly in 10-mg increments. Doses above 60 mg daily are not recommended ( 2.3 ). For patients currently using methylphenidate hydrochloride tablets or methylphenidate hydrochloride extended-release tablets: Dosage is based on current dose regimen ( 2.4 ). If switching from other methylphenidate products, discontinue treatment and titrate with methylphenidate hydrochloride extended-release capsules (LA) ( 2.4 ).
Contraindications
Hypersensitivity to methylphenidate or other components of methylphenidate hydrochloride extended-release capsules (LA). Hypersensitivity reactions, such as angioedema and anaphylactic reactions, have been reported in patients treated with methylphenidate [see Adverse Reactions (6.1) ]. Concomitant treatment with monoamine oxidase inhibitors (MAOIs), or within 14 days following discontinuation of treatment with an MAOI, because of the risk of hypertensive crises [ s ee Drug Interactions (7.1) ].
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Abuse and Dependence [see Boxed Warning , Warnings and Precautions (5.1) , Drug Abuse and Dependence (9.2 , 9.3) ] Known hypersensitivity to methylphenidate or other ingredients of methylphenidate hydrochloride extended-release capsules (LA) [see Contraindications (4) ] Hypertensive crisis when used concomitantly with Monoamine Oxidase Inhibitors [see Contraindications (4) , Drug Interactions (7.1) ] Serious Cardiovascular Reactions [see Warnings and Precautions (5.2) ] Blood Pressure and Heart Rate Increases [see Warnings and Precautions (5.3) ] Psychiatric Adverse Reactions [see Warnings and Precautions (5.4) ] Priapism [see Warnings and Precautions (5.5) ] Peripheral Vasculopathy, Including Raynaud's Phenomenon [see Warnings and Precautions (5.6) ] Long-Term Suppression of Growth [see Warnings and Precautions (5.7) ]
Drug Interactions
Antihypertensive drugs : Monitor blood pressure and heart. Adjust dosage of antihypertensive drug as needed ( 7.1 ). Halogenated Anesthetics: Avoid use of methylphenidate hydrochloride extended-release capsules (LA) on the day of surgery if halogenated anesthetics will be used ( 7.1 ).
Storage and Handling
10 mg white/light green capsules (imprinted mayne 609 on both the body and the cap) Bottles of 100 NDC 51862-609-01 20 mg white/white capsules (imprinted mayne 610 on both the body and cap Bottles of 100 NDC 51862-610-01 30 mg white/light blue capsules (imprinted mayne 611 on both the body and cap Bottles of 100 NDC 51862-611-01 40 mg white/dark blue capsules (imprinted mayne 612 on both the body and cap) Bottles of 100 NDC 51862-612-01 60 mg light yellow/dark yellow capsules (imprinted mayne 614 on both the body and cap) Bottles of 30 NDC 51862-614-30
How Supplied
10 mg white/light green capsules (imprinted mayne 609 on both the body and the cap) Bottles of 100 NDC 51862-609-01 20 mg white/white capsules (imprinted mayne 610 on both the body and cap Bottles of 100 NDC 51862-610-01 30 mg white/light blue capsules (imprinted mayne 611 on both the body and cap Bottles of 100 NDC 51862-611-01 40 mg white/dark blue capsules (imprinted mayne 612 on both the body and cap) Bottles of 100 NDC 51862-612-01 60 mg light yellow/dark yellow capsules (imprinted mayne 614 on both the body and cap) Bottles of 30 NDC 51862-614-30
Description
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Warning and Precautions (5.1) , Drug Abuse and Dependence (9.2 , 9.3) ] .
Section 42229-5
Exacerbation of Preexisting Psychosis
CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a preexisting psychotic disorder.
Section 42231-1
| MEDICATION GUIDE
Methylphenidate Hydrochloride (METH-il-FEN-i-date HYE-droe-KLOR-ide) Extended-Release Capsules (LA) CII |
||||
|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 11/2022 | |||
|
What is the most important information I should know about methylphenidate hydrochloride extended-release capsules (LA)?
Methylphenidate hydrochloride extended-release capsules (LA) is a federal controlled substance (CII) because it can be abused or lead to dependence. Keep methylphenidate hydrochloride extended-release capsules (LA) in a safe place to prevent misuse and abuse. Selling or giving away methylphenidate hydrochloride extended-release capsules (LA) may harm others, and is against the law. Tell your doctor if you or your child have ever abused or been dependent on alcohol, prescription medicines or street drugs. The following have been reported with use of methylphenidate hydrochloride and other stimulant medicines. 1. Heart-related problems:
Your doctor should check you or your child carefully for heart problems before starting methylphenidate hydrochloride extended-release capsules (LA). Your doctor should check your or your child's blood pressure and heart rate regularly during treatment with methylphenidate hydrochloride extended-release capsules (LA). Call your doctor right away if you or your child has any signs of heart problems, such as chest pain, shortness of breath, or fainting while taking methylphenidate hydrochloride extended-release capsules (LA). 2. Mental (psychiatric) problems: All Patients
Call your doctor right away if you or your child have any new or worsening mental symptoms or problems while taking methylphenidate hydrochloride extended-release capsules (LA), especially seeing or hearing things that are not real, believing things that are not real, or are suspicious. |
||||
|
What are methylphenidate hydrochloride extended-release capsules (LA)?
Methylphenidate hydrochloride extended-release capsules (LA) are a central nervous system (CNS) stimulant prescription medicine. It is used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD).Methylphenidate hydrochloride extended-release capsules (LA) may help increase attention and decrease impulsiveness and hyperactivity in patients with ADHD. Methylphenidate hydrochloride extended-release capsules (LA) should be used as a part of a total treatment program for ADHD that may include counseling or other therapies. It is not known if methylphenidate hydrochloride extended-release capsules (LA) is safe and effective in children under 6 years of age. |
||||
|
Who should not take methylphenidate hydrochloride extended-release capsules (LA)?
Methylphenidate hydrochloride extended-release capsules (LA) should not be taken if you or your child:
Your doctor will decide whether methylphenidate hydrochloride extended-release capsules (LA) can be taken with other medicines. Especially tell your doctor if you or your child takes:
|
||||
How should methylphenidate hydrochloride extended-release capsules (LA) be taken?
|
||||
What are possible side effects of methylphenidate hydrochloride extended-release capsules (LA)?
|
||||
|
|
|
|
|
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
How should I store methylphenidate hydrochloride extended-release capsules (LA)?
|
||||
|
General information about the safe and effective use methylphenidate hydrochloride extended-release capsules (LA).
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or doctor for information about methylphenidate hydrochloride extended-release capsules (LA)that is written for healthcare professionals. Do not use methylphenidate extended release-capsules (LA) for a condition for which it was not prescribed. Do not give methylphenidate hydrochloride extended-release capsules (LA) to other people, even if they have the same symptoms. It may harm them and it is against the law. |
||||
|
What are the ingredients in methylphenidate hydrochloride extended-release capsules (LA)?
Active Ingredient: methylphenidate HCl Inactive Ingredients:sugar spheres (which contain sucrose and starch (corn)), hypromellose, cellulose acetate butyrate, hypromellose acetate succinate, acetyltributyl citrate, acetone, talc, and purified water. Opaque gelatin capsules contain: titanium dioxide and gelatin. The 10mg capsule contains FD&C Green#3, FD&C #40, and FD&C Yellow #6. The 30 and 40 mg capsules contain D&C Red #28 and FD&C Blue #1. The capsules are imprinted with black ink which contains black iron oxide, shellac and potassium hydroxide. The 60 mg capsules contain iron oxide yellow and sodium lauryl sulfate. The capsules are imprinted with black ink which contains black iron oxide, shellac and potassium hydroxide. The 60 mg black imprinting ink also contains ammonium hydroxide and propylene glycol. Distributed by: Mayne Pharma Raleigh, NC 27609 AB7678 For more information, call 1-844-825-8500. |
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F and 86°F). [See USP controlled room temperature].
Dispense in tight container (USP).
9.2 Abuse
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), have a high potential for abuse. Abuse is characterized by impaired control over drug use despite harm, and craving.
Signs and symptoms of CNS stimulant abuse include increased heart rate, respiratory rate, blood pressure, and/or sweating, dilated pupils, hyperactivity, restlessness, insomnia, decreased appetite, loss of coordination, tremors, flushed skin, vomiting, and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed. Abusers of CNS stimulants may chew, snort, inject, or use other unapproved routes of administration which may result in overdose and death [see Overdosage (10)] .
To reduce the abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), assess the risk of abuse prior to prescribing. After prescribing, keep careful prescription records, educate patients and their families about abuse and on proper storage and disposal of CNS stimulants [see How Supplied/Storage and Handling (16)] , monitor for signs of abuse while on therapy, and reevaluate the need for methylphenidate hydrochloride tablets and methylphenidate hydrochloride extended-release tablets use.
5.5 Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate products in both pediatric and adult patients. Priapism was not reported with drug initiation but developed after some time on the drug, often subsequent to an increase in dose. Priapism has also appeared during a period of drug withdrawal (drug holidays or during discontinuation). Patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
11 Description
Methylphenidate hydrochloride extended-release capsules (LA) contains methylphenidate hydrochloride USP, a CNS stimulant.
Methylphenidate hydrochloride extended-release capsules (LA) is an extended-release formulation of methylphenidate for oral administration with a bi-modal release profile. Each bead-filled methylphenidate hydrochloride extended-release capsule (LA) contains half the dose as immediate-release beads and half as enteric-coated beads, thus providing an immediate release of methylphenidate and a second delayed release of methylphenidate.
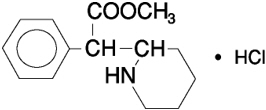
The active substance in methylphenidate hydrochloride extended-release capsules (LA) is methyl α-phenyl-2-piperidineacetate hydrochloride, and its structural formula is
Methylphenidate hydrochloride USP is a white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone. Its molecular weight is 269.77 g/mol.
Inactive ingredients: Sugar spheres (which contain sucrose and starch (corn)), hypromellose, cellulose acetate butyrate, hypromellose acetate succinate, acetyltributyl citrate, acetone, talc, and purified water. Opaque gelatin capsules contain: titanium dioxide and gelatin. The 10mg capsule contains FD&C Green#3, FD&C#40, and FD&C Yellow #6. The 30 and 40 mg capsules contain D&C Red #28 and FD&C Blue #1. The 60 mg capsules contain iron oxide yellow and sodium lauryl sulfate. The capsules are imprinted with black ink which contains black iron oxide, shellac and potassium hydroxide. The 60 mg black imprinting ink also contains ammonium hydroxide and propylene glycol.
8.4 Pediatric Use
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules (LA) for the treatment of ADHD have been established in pediatric patients 6 to 12 years.
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules (LA) in pediatric patients less than 6 years have not been established. The long-term efficacy of methylphenidate hydrochloride extended-release capsules (LA) in pediatric patients has not been established.
8.5 Geriatric Use
Methylphenidate hydrochloride extended-release capsules (LA) has not been studied in the geriatric population.
4 Contraindications
- Hypersensitivity to methylphenidate or other components of methylphenidate hydrochloride extended-release capsules (LA). Hypersensitivity reactions, such as angioedema and anaphylactic reactions, have been reported in patients treated with methylphenidate [see Adverse Reactions (6.1)].
- Concomitant treatment with monoamine oxidase inhibitors (MAOIs), or within 14 days following discontinuation of treatment with an MAOI, because of the risk of hypertensive crises [s ee Drug Interactions (7.1)].
6 Adverse Reactions
The following are discussed in more detail in other sections of the labeling:
- Abuse and Dependence [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)]
- Known hypersensitivity to methylphenidate or other ingredients of methylphenidate hydrochloride extended-release capsules (LA) [see Contraindications (4)]
- Hypertensive crisis when used concomitantly with Monoamine Oxidase Inhibitors [see Contraindications (4), Drug Interactions (7.1)]
- Serious Cardiovascular Reactions [see Warnings and Precautions (5.2)]
- Blood Pressure and Heart Rate Increases [see Warnings and Precautions (5.3)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.5)]
- Peripheral Vasculopathy, Including Raynaud's Phenomenon [see Warnings and Precautions (5.6)]
- Long-Term Suppression of Growth [see Warnings and Precautions (5.7)]
7 Drug Interactions
12.2 Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d-and l-threoenantiomers. The d-threoenantiomer is more pharmacologically active than the l-threoenantiomer. Methylphenidate blocks the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
Methylphenidate hydrochloride extended-release capsules (LA) produces a bi-modal plasma concentration-time profile (i.e., 2 distinct peaks approximately 4 hours apart) when administered orally to children diagnosed with ADHD and healthy adults.
No accumulation of methylphenidate is expected following multiple once daily oral dosing with methylphenidate hydrochloride extended-release capsules (LA), however, there is a slight upward trend in the methylphenidate area under the curve and peak plasma concentrations (C max1and C max2) after oral administration of methylphenidate hydrochloride extended-release capsules (LA) 20 mg and 40 mg to adults.
1 Indications and Usage
Methylphenidate hydrochloride extended-release capsules (LA) is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD), in pediatric patients 6 to 12 years of age [see Clinical Studies (14)] .
12.1 Mechanism of Action
Methylphenidate hydrochloride is a CNS stimulant. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
Methylphenidate hydrochloride extended-release capsules (LA) contains methylphenidate hydrochloride, a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to initiating treatment with central nervous system (CNS) stimulants, including methylphenidate hydrochloride extended-release capsules (LA), assess for the presence of cardiac disease (i.e., perform a careful history, including family history of sudden death or ventricular arrhythmia, and physical examination) [see Warnings and Precautions (5.2)] .
Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy. Maintain careful prescription records, educate patients about abuse, monitor for signs of abuse and overdose, and periodically reevaluate the need for methylphenidate hydrochloride extended-release capsules (LA) use [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)].
5 Warnings and Precautions
- Serious Cardiovascular Events: Sudden death has been reported in association with CNS-stimulant treatment at usual doses in pediatric patients with structural cardiac abnormalities or other serious heart problems. In adults, sudden death, stroke, and myocardial infarction have been reported. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm arrhythmias, or coronary artery disease ( 5.2).
- Blood Pressure and Heart Rate Increases: Monitor blood pressure and pulse. Consider the benefits and risk in patients for whom an increase in blood pressure or heart rate would be problematic ( 5.3).
- Psychiatric Adverse Reactions: Use of stimulants may cause psychotic or manic symptoms in patients with no prior history or exacerbation of symptoms in patients with preexisting psychiatric illness. Evaluate for preexisting psychotic or bipolar disorder prior to methylphenidate hydrochloride extended-release capsules (LA) use ( 5.4).
- Priapism: Cases of painful and prolonged penile erections and priapism have been reported with methylphenidate products. Immediate medical attention should be sought if signs or symptoms of prolonged penile erections or priapism are observed ( 5.5).
- Peripheral Vasculopathy, including Raynaud's Phenomenon: Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants ( 5.6).
- Long-Term Suppression of Growth: Monitor height and weight at appropriate intervals in pediatric patients ( 5.7).
2 Dosage and Administration
- Administer orally once daily in the morning ( 2.2).
- Capsules may be swallowed whole, or opened and the entire contents sprinkled on applesauce ( 2.2).
- Should not be crushed, chewed, or divided ( 2.2).
- Patients new to methylphenidate: Start at 20 mg daily, titrating the dose weekly in 10-mg increments. Doses above 60 mg daily are not recommended ( 2.3).
- For patients currently using methylphenidate hydrochloride tablets or methylphenidate hydrochloride extended-release tablets: Dosage is based on current dose regimen ( 2.4).
- If switching from other methylphenidate products, discontinue treatment and titrate with methylphenidate hydrochloride extended-release capsules (LA) ( 2.4).
3 Dosage Forms and Strengths
- 10 mg extended-release capsules white/light green (imprinted mayne 609 on both the body and cap)
- 20 mg extended-release capsules white/white (imprinted mayne 610 on both the body and cap)
- 30 mg extended-release capsules white/light blue, (imprinted mayne 611 on both body and cap)
- 40 mg extended-release capsules white/dark blue, (imprinted mayne 612 on both body and cap)
- 60 mg extended-release capsules light yellow/dark yellow (imprinted mayne 614 on both body and cap)
6.2 Postmarketing Experience
The following adverse reactions have been identified during the post approval use of methylphenidate products. Because these reactions were reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure.
14.1 Children and Adolescents
Methylphenidate hydrochloride extended-release capsules (LA) was evaluated in a randomized, double-blind, placebo-controlled, parallel group clinical study in which 134 children, ages 6 to 12, with DSM-IV diagnoses of ADHD received a single morning dose of methylphenidate hydrochloride extended-release capsules (LA)in the range of 10 to 40 mg/day, or placebo, for up to 2 weeks. The doses used were the optimal doses established in a previous individual dose titration phase. In that titration phase, 53 of 164 patients (32%) started on a daily dose of 10 mg and 111 of 164 patients (68%) started on a daily dose of 20 mg or higher. The patient's regular schoolteacher completed the Conners ADHD/DSM-IV Scale for Teachers (CADS-T) at baseline and the end of each week. The CADS-T assesses symptoms of hyperactivity and inattention. The change from baseline of the (CADS-T) scores during the last week of treatment was analyzed as the primary efficacy parameter. Patients treated with methylphenidate hydrochloride extended-release capsules (LA) showed a statistically significant improvement in symptom scores from baseline [Mean (final score - baseline) = -10.7 points)] over patients who received placebo [Mean (final score - baseline) = +2.8 points ]. The lower the final score on the CADS-T scale from baseline, the less severe the disease is. This demonstrates that a single morning dose of methylphenidate hydrochloride extended-release capsules (LA) exerts a treatment effect in ADHD.
| Figure 2: CADS-T Total Subscale - Mean Change From Baseline* |
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The clinical program for methylphenidate hydrochloride extended-release capsules (LA) consisted of 6 studies: 2 controlled clinical studies conducted in children with ADHD aged 6 to 12 years and 4 clinical pharmacology studies conducted in healthy adult volunteers. These studies included a total of 256 subjects; 195 children with ADHD and 61 healthy adult volunteers. The subjects received methylphenidate hydrochloride extended-release capsules (LA) in doses of 10 to 40 mg per day. Safety of methylphenidate hydrochloride extended-release capsules (LA) was assessed by evaluating frequency and nature of adverse events, routine laboratory tests, vital signs, and body weight. A placebo-controlled, double-blind, parallel-group study was conducted to evaluate the efficacy and safety of methylphenidate hydrochloride extended-release capsules (LA) in children with ADHD aged 6 to 12 years. All subjects received methylphenidate hydrochloride extended-release capsules (LA) for up to 4 weeks, and had their dose optimally adjusted, prior to entering the double-blind phase of the trial. In the 2-week double-blind treatment phase of this study, patients received either placebo or methylphenidate hydrochloride extended-release capsules (LA) at their individually-titrated dose (range, 10 to 40 mg).
Adverse reactions with an incidence greater than 5% during the initial 4-week single-blind methylphenidate hydrochloride extended-release capsules (LA) titration period of this study were headache, insomnia, upper abdominal pain, appetite decreased, and anorexia.
Adverse reactions with an incidence greater than 2% among methylphenidate hydrochloride extended-release capsules (LA) -treated subjects, during the 2-week double-blind phase of the clinical study, are shown in Table 2:
| Preferred Term | Methylphenidate Hydrochloride Extended-Release Capsules (LA)
N = 65 N (%) |
Placebo
N = 71 N (%) |
|---|---|---|
| Anorexia | 2 (3.1) | 0 (0.0) |
| Insomnia | 2 (3.1) | 0 (0.0) |
Warning: Abuse and Dependence
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Warning and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)] .
2.2 General Dosing Information
The recommended starting dose for methylphenidate hydrochloride extended-release capsules (LA) is 20 mg once daily. Increase dosage gradually, in increments of 10 mg weekly. Daily dosage above 60 mg is not recommended. When a lower initial dose is appropriate, patients may begin treatment with 10 mg.
Administer methylphenidate hydrochloride extended-release capsules (LA) orally once daily in the morning. Methylphenidate hydrochloride extended-release capsules (LA) may be swallowed as whole capsules or may be administered by sprinkling the capsule contents on a small amount of applesauce (see specific instructions below). Methylphenidate hydrochloride extended-release capsules (LA) and/or their contents should not be crushed, chewed, or divided.
The capsules may be carefully opened and the beads sprinkled over a spoonful of applesauce. The applesauce should not be warm because it could affect the modified release properties of this formulation. The mixture of drug and applesauce should be consumed immediately in its entirety. The drug and applesauce mixture should not be stored for future use.
Pharmacological treatment of ADHD may be needed for extended periods. Periodically reevaluate the long-term use of methylphenidate hydrochloride tablets and methylphenidate hydrochloride extended-release tablets, and adjust dosage as needed.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide)
5.7 Long Term Suppression of Growth
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated patients over 36 months (to the ages of 10-13 years), suggests that consistently medicated pediatric patients (i.e., treatment for 7 days per week throughout the year) have a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this period of development.
Closely monitor growth (weight and height) in pediatric patients treated with CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA). Patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
16 How Supplied/storage and Handling
10 mg white/light green capsules (imprinted mayne 609 on both the body and the cap)
| Bottles of 100 | NDC 51862-609-01 |
20 mg white/white capsules (imprinted mayne 610 on both the body and cap
| Bottles of 100 | NDC 51862-610-01 |
30 mg white/light blue capsules (imprinted mayne 611 on both the body and cap
| Bottles of 100 | NDC 51862-611-01 |
40 mg white/dark blue capsules (imprinted mayne 612 on both the body and cap)
| Bottles of 100 | NDC 51862-612-01 |
60 mg light yellow/dark yellow capsules (imprinted mayne 614 on both the body and cap)
| Bottles of 30 | NDC 51862-614-30 |
5.2 Serious Cardiovascular Reactions
Sudden death, stroke, and myocardial infarction have been reported in adults with CNS-stimulant treatment at recommended doses. Sudden death has been reported in pediatric patients with structural cardiac abnormalities and other serious heart problems taking CNS stimulants at recommended doses for ADHD. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, and other serious heart problems. Further evaluate patients who develop exertional chest pain, unexplained syncope, or arrhythmias during methylphenidate hydrochloride extended-release capsules (LA) treatment.
2.5 Dose Reduction and Discontinuation
If paradoxical worsening of symptoms or other adverse reactions occur, reduce the dosage, or, if necessary, discontinue methylphenidate hydrochloride extended-release capsules (LA). If improvement is not observed after appropriate dosage adjustment over a one-month period, the drug should be discontinued.
5.1 Potential for Abuse and Dependence
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Boxed Warning, Drug Abuse and Dependence (9.2, 9.3)].
5.3 Blood Pressure and Heart Rate Increases
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 beats per minute). Individuals may have larger increases. Monitor all patients for hypertension and tachycardia.
2.4 Switching From Other Methylphenidate Products
If switching from other methylphenidate products, discontinue that treatment, and titrate with methylphenidate hydrochloride extended-release capsules (LA) using the titration schedule.
Do not substitute for other methylphenidate products on a milligram-per-milligram basis, because different methylphenidate base compositions and differing pharmacokinetic profiles [see Description (11), Clinical Pharmacology (12.3)] .
Clinical judgment should be used when selecting the starting dose. Daily dosage above 60 mg is not recommended.
Principal Display Panel 10 Mg Capsule Bottle Label
NDC 51862-609-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
10 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 20 Mg Capsule Bottle Label
NDC 51862-610-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
20 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 30 Mg Capsule Bottle Label
NDC 51862-611-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
30 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 40 Mg Capsule Bottle Label
NDC 51862-612-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
40 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 60 Mg Capsule Bottle Label
NDC 51862-614-30
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
60 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
30 Capsules
maynepharma
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon
CNS Stimulants, including methylphenidate hydrochloride extended-release capsules (LA), used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.
7.1 Clinically Important Drug Interactions With Methylphenidate Hydrochloride Extended Release Capsules (la)
Table 3 presents clinically important drug interactions with methylphenidate hydrochloride extended-release capsules (LA)
| Monoamine Oxidase Inhibitors (MAOI) | |
| Clinical Impact | Concomitant use of MAOIs and CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure [see Contraindications (4)] . |
| Intervention | Concomitant use of methylphenidate hydrochloride extended-release capsules (LA) with MAOIs or within 14 days after discontinuing MAOI treatment is contraindicated. |
| Examples | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Antihypertensive Drugs | |
| Clinical Impact | Methylphenidate hydrochloride extended-release capsules (LA) may decrease the effectiveness of drugs used to treat hypertension [see Warnings and Precautions (5.3)] . |
| Intervention | Monitor blood pressure and adjust the dosage of the antihypertensive drug as needed. |
| Examples | Potassium-sparing and thiazide diuretics, calcium channel blockers, angiotensin-converting-enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), beta blockers, centrally acting alpha-2 receptor agonists. |
| Halogenated Anesthetics | |
| Clinical Impact | Concomitant use of halogenated anesthetics and methylphenidate hydrochloride extended-release capsules (LA) may increase the risk of sudden blood pressure and heart rate increase during surgery. |
| Intervention | Avoid use of methylphenidate hydrochloride extended-release capsules (LA) in patients being treated with anesthetics on the day of surgery. |
| Examples | halothane, isoflurane, enflurane, desflurane, sevoflurane |
| Risperidone | |
| Clinical Impact | Combined use of methylphenidate with risperidone when there is a change, whether an increase or decrease, in dosage of either or both medications, may increase the risk of extrapyramidal symptoms (EPS) |
| Intervention | Monitor for signs of EPS |
2.3 Patients Currently Using Methylphenidate Hydrochloride Tablets Or Methylphenidate Hydrochloride Extended Release Tablets
The recommended dose of methylphenidate hydrochloride extended-release capsules (LA) for patients currently taking methylphenidate hydrochloride tablets twice daily or methylphenidate hydrochloride extended-release tablets (SR) is provided below.
| Previous Methylphenidate Hydrochloride Tablets or Methylphenidate Hydrochloride Extended-Release Tablets Dose | Recommended Methylphenidate Hydrochloride Extended-Release Capsules (LA) Dose |
|---|---|
| 5 mg methylphenidate hydrochloride tablets twice daily | 10 mg once daily |
| 10 mg methylphenidate hydrochloride tablets twice daily or 20 mg methylphenidate hydrochloride extended-release tablets | 20 mg once daily |
| 15 mg methylphenidate hydrochloride tablets twice daily | 30 mg once daily |
| 20 mg methylphenidate hydrochloride tablets twice daily or 40 mg of methylphenidate hydrochloride extended-release tablets | 40 mg once daily |
| 30 mg methylphenidate hydrochloride tablets twice daily or 60 mg of methylphenidate hydrochloride extended-release tablets | 60 mg once daily |
Structured Label Content
Section 42229-5 (42229-5)
Exacerbation of Preexisting Psychosis
CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a preexisting psychotic disorder.
Section 42231-1 (42231-1)
| MEDICATION GUIDE
Methylphenidate Hydrochloride (METH-il-FEN-i-date HYE-droe-KLOR-ide) Extended-Release Capsules (LA) CII |
||||
|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 11/2022 | |||
|
What is the most important information I should know about methylphenidate hydrochloride extended-release capsules (LA)?
Methylphenidate hydrochloride extended-release capsules (LA) is a federal controlled substance (CII) because it can be abused or lead to dependence. Keep methylphenidate hydrochloride extended-release capsules (LA) in a safe place to prevent misuse and abuse. Selling or giving away methylphenidate hydrochloride extended-release capsules (LA) may harm others, and is against the law. Tell your doctor if you or your child have ever abused or been dependent on alcohol, prescription medicines or street drugs. The following have been reported with use of methylphenidate hydrochloride and other stimulant medicines. 1. Heart-related problems:
Your doctor should check you or your child carefully for heart problems before starting methylphenidate hydrochloride extended-release capsules (LA). Your doctor should check your or your child's blood pressure and heart rate regularly during treatment with methylphenidate hydrochloride extended-release capsules (LA). Call your doctor right away if you or your child has any signs of heart problems, such as chest pain, shortness of breath, or fainting while taking methylphenidate hydrochloride extended-release capsules (LA). 2. Mental (psychiatric) problems: All Patients
Call your doctor right away if you or your child have any new or worsening mental symptoms or problems while taking methylphenidate hydrochloride extended-release capsules (LA), especially seeing or hearing things that are not real, believing things that are not real, or are suspicious. |
||||
|
What are methylphenidate hydrochloride extended-release capsules (LA)?
Methylphenidate hydrochloride extended-release capsules (LA) are a central nervous system (CNS) stimulant prescription medicine. It is used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD).Methylphenidate hydrochloride extended-release capsules (LA) may help increase attention and decrease impulsiveness and hyperactivity in patients with ADHD. Methylphenidate hydrochloride extended-release capsules (LA) should be used as a part of a total treatment program for ADHD that may include counseling or other therapies. It is not known if methylphenidate hydrochloride extended-release capsules (LA) is safe and effective in children under 6 years of age. |
||||
|
Who should not take methylphenidate hydrochloride extended-release capsules (LA)?
Methylphenidate hydrochloride extended-release capsules (LA) should not be taken if you or your child:
Your doctor will decide whether methylphenidate hydrochloride extended-release capsules (LA) can be taken with other medicines. Especially tell your doctor if you or your child takes:
|
||||
How should methylphenidate hydrochloride extended-release capsules (LA) be taken?
|
||||
What are possible side effects of methylphenidate hydrochloride extended-release capsules (LA)?
|
||||
|
|
|
|
|
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
How should I store methylphenidate hydrochloride extended-release capsules (LA)?
|
||||
|
General information about the safe and effective use methylphenidate hydrochloride extended-release capsules (LA).
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or doctor for information about methylphenidate hydrochloride extended-release capsules (LA)that is written for healthcare professionals. Do not use methylphenidate extended release-capsules (LA) for a condition for which it was not prescribed. Do not give methylphenidate hydrochloride extended-release capsules (LA) to other people, even if they have the same symptoms. It may harm them and it is against the law. |
||||
|
What are the ingredients in methylphenidate hydrochloride extended-release capsules (LA)?
Active Ingredient: methylphenidate HCl Inactive Ingredients:sugar spheres (which contain sucrose and starch (corn)), hypromellose, cellulose acetate butyrate, hypromellose acetate succinate, acetyltributyl citrate, acetone, talc, and purified water. Opaque gelatin capsules contain: titanium dioxide and gelatin. The 10mg capsule contains FD&C Green#3, FD&C #40, and FD&C Yellow #6. The 30 and 40 mg capsules contain D&C Red #28 and FD&C Blue #1. The capsules are imprinted with black ink which contains black iron oxide, shellac and potassium hydroxide. The 60 mg capsules contain iron oxide yellow and sodium lauryl sulfate. The capsules are imprinted with black ink which contains black iron oxide, shellac and potassium hydroxide. The 60 mg black imprinting ink also contains ammonium hydroxide and propylene glycol. Distributed by: Mayne Pharma Raleigh, NC 27609 AB7678 For more information, call 1-844-825-8500. |
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F and 86°F). [See USP controlled room temperature].
Dispense in tight container (USP).
9.2 Abuse
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), have a high potential for abuse. Abuse is characterized by impaired control over drug use despite harm, and craving.
Signs and symptoms of CNS stimulant abuse include increased heart rate, respiratory rate, blood pressure, and/or sweating, dilated pupils, hyperactivity, restlessness, insomnia, decreased appetite, loss of coordination, tremors, flushed skin, vomiting, and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed. Abusers of CNS stimulants may chew, snort, inject, or use other unapproved routes of administration which may result in overdose and death [see Overdosage (10)] .
To reduce the abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), assess the risk of abuse prior to prescribing. After prescribing, keep careful prescription records, educate patients and their families about abuse and on proper storage and disposal of CNS stimulants [see How Supplied/Storage and Handling (16)] , monitor for signs of abuse while on therapy, and reevaluate the need for methylphenidate hydrochloride tablets and methylphenidate hydrochloride extended-release tablets use.
5.5 Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate products in both pediatric and adult patients. Priapism was not reported with drug initiation but developed after some time on the drug, often subsequent to an increase in dose. Priapism has also appeared during a period of drug withdrawal (drug holidays or during discontinuation). Patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
11 Description (11 DESCRIPTION)
Methylphenidate hydrochloride extended-release capsules (LA) contains methylphenidate hydrochloride USP, a CNS stimulant.
Methylphenidate hydrochloride extended-release capsules (LA) is an extended-release formulation of methylphenidate for oral administration with a bi-modal release profile. Each bead-filled methylphenidate hydrochloride extended-release capsule (LA) contains half the dose as immediate-release beads and half as enteric-coated beads, thus providing an immediate release of methylphenidate and a second delayed release of methylphenidate.
The active substance in methylphenidate hydrochloride extended-release capsules (LA) is methyl α-phenyl-2-piperidineacetate hydrochloride, and its structural formula is
Methylphenidate hydrochloride USP is a white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone. Its molecular weight is 269.77 g/mol.
Inactive ingredients: Sugar spheres (which contain sucrose and starch (corn)), hypromellose, cellulose acetate butyrate, hypromellose acetate succinate, acetyltributyl citrate, acetone, talc, and purified water. Opaque gelatin capsules contain: titanium dioxide and gelatin. The 10mg capsule contains FD&C Green#3, FD&C#40, and FD&C Yellow #6. The 30 and 40 mg capsules contain D&C Red #28 and FD&C Blue #1. The 60 mg capsules contain iron oxide yellow and sodium lauryl sulfate. The capsules are imprinted with black ink which contains black iron oxide, shellac and potassium hydroxide. The 60 mg black imprinting ink also contains ammonium hydroxide and propylene glycol.
8.4 Pediatric Use
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules (LA) for the treatment of ADHD have been established in pediatric patients 6 to 12 years.
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules (LA) in pediatric patients less than 6 years have not been established. The long-term efficacy of methylphenidate hydrochloride extended-release capsules (LA) in pediatric patients has not been established.
8.5 Geriatric Use
Methylphenidate hydrochloride extended-release capsules (LA) has not been studied in the geriatric population.
4 Contraindications (4 CONTRAINDICATIONS)
- Hypersensitivity to methylphenidate or other components of methylphenidate hydrochloride extended-release capsules (LA). Hypersensitivity reactions, such as angioedema and anaphylactic reactions, have been reported in patients treated with methylphenidate [see Adverse Reactions (6.1)].
- Concomitant treatment with monoamine oxidase inhibitors (MAOIs), or within 14 days following discontinuation of treatment with an MAOI, because of the risk of hypertensive crises [s ee Drug Interactions (7.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are discussed in more detail in other sections of the labeling:
- Abuse and Dependence [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)]
- Known hypersensitivity to methylphenidate or other ingredients of methylphenidate hydrochloride extended-release capsules (LA) [see Contraindications (4)]
- Hypertensive crisis when used concomitantly with Monoamine Oxidase Inhibitors [see Contraindications (4), Drug Interactions (7.1)]
- Serious Cardiovascular Reactions [see Warnings and Precautions (5.2)]
- Blood Pressure and Heart Rate Increases [see Warnings and Precautions (5.3)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.5)]
- Peripheral Vasculopathy, Including Raynaud's Phenomenon [see Warnings and Precautions (5.6)]
- Long-Term Suppression of Growth [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d-and l-threoenantiomers. The d-threoenantiomer is more pharmacologically active than the l-threoenantiomer. Methylphenidate blocks the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
12.3 Pharmacokinetics
Methylphenidate hydrochloride extended-release capsules (LA) produces a bi-modal plasma concentration-time profile (i.e., 2 distinct peaks approximately 4 hours apart) when administered orally to children diagnosed with ADHD and healthy adults.
No accumulation of methylphenidate is expected following multiple once daily oral dosing with methylphenidate hydrochloride extended-release capsules (LA), however, there is a slight upward trend in the methylphenidate area under the curve and peak plasma concentrations (C max1and C max2) after oral administration of methylphenidate hydrochloride extended-release capsules (LA) 20 mg and 40 mg to adults.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Methylphenidate hydrochloride extended-release capsules (LA) is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD), in pediatric patients 6 to 12 years of age [see Clinical Studies (14)] .
12.1 Mechanism of Action
Methylphenidate hydrochloride is a CNS stimulant. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
Methylphenidate hydrochloride extended-release capsules (LA) contains methylphenidate hydrochloride, a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to initiating treatment with central nervous system (CNS) stimulants, including methylphenidate hydrochloride extended-release capsules (LA), assess for the presence of cardiac disease (i.e., perform a careful history, including family history of sudden death or ventricular arrhythmia, and physical examination) [see Warnings and Precautions (5.2)] .
Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy. Maintain careful prescription records, educate patients about abuse, monitor for signs of abuse and overdose, and periodically reevaluate the need for methylphenidate hydrochloride extended-release capsules (LA) use [see Boxed Warning, Warnings and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serious Cardiovascular Events: Sudden death has been reported in association with CNS-stimulant treatment at usual doses in pediatric patients with structural cardiac abnormalities or other serious heart problems. In adults, sudden death, stroke, and myocardial infarction have been reported. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm arrhythmias, or coronary artery disease ( 5.2).
- Blood Pressure and Heart Rate Increases: Monitor blood pressure and pulse. Consider the benefits and risk in patients for whom an increase in blood pressure or heart rate would be problematic ( 5.3).
- Psychiatric Adverse Reactions: Use of stimulants may cause psychotic or manic symptoms in patients with no prior history or exacerbation of symptoms in patients with preexisting psychiatric illness. Evaluate for preexisting psychotic or bipolar disorder prior to methylphenidate hydrochloride extended-release capsules (LA) use ( 5.4).
- Priapism: Cases of painful and prolonged penile erections and priapism have been reported with methylphenidate products. Immediate medical attention should be sought if signs or symptoms of prolonged penile erections or priapism are observed ( 5.5).
- Peripheral Vasculopathy, including Raynaud's Phenomenon: Stimulants used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with ADHD stimulants ( 5.6).
- Long-Term Suppression of Growth: Monitor height and weight at appropriate intervals in pediatric patients ( 5.7).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer orally once daily in the morning ( 2.2).
- Capsules may be swallowed whole, or opened and the entire contents sprinkled on applesauce ( 2.2).
- Should not be crushed, chewed, or divided ( 2.2).
- Patients new to methylphenidate: Start at 20 mg daily, titrating the dose weekly in 10-mg increments. Doses above 60 mg daily are not recommended ( 2.3).
- For patients currently using methylphenidate hydrochloride tablets or methylphenidate hydrochloride extended-release tablets: Dosage is based on current dose regimen ( 2.4).
- If switching from other methylphenidate products, discontinue treatment and titrate with methylphenidate hydrochloride extended-release capsules (LA) ( 2.4).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 10 mg extended-release capsules white/light green (imprinted mayne 609 on both the body and cap)
- 20 mg extended-release capsules white/white (imprinted mayne 610 on both the body and cap)
- 30 mg extended-release capsules white/light blue, (imprinted mayne 611 on both body and cap)
- 40 mg extended-release capsules white/dark blue, (imprinted mayne 612 on both body and cap)
- 60 mg extended-release capsules light yellow/dark yellow (imprinted mayne 614 on both body and cap)
6.2 Postmarketing Experience
The following adverse reactions have been identified during the post approval use of methylphenidate products. Because these reactions were reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure.
14.1 Children and Adolescents
Methylphenidate hydrochloride extended-release capsules (LA) was evaluated in a randomized, double-blind, placebo-controlled, parallel group clinical study in which 134 children, ages 6 to 12, with DSM-IV diagnoses of ADHD received a single morning dose of methylphenidate hydrochloride extended-release capsules (LA)in the range of 10 to 40 mg/day, or placebo, for up to 2 weeks. The doses used were the optimal doses established in a previous individual dose titration phase. In that titration phase, 53 of 164 patients (32%) started on a daily dose of 10 mg and 111 of 164 patients (68%) started on a daily dose of 20 mg or higher. The patient's regular schoolteacher completed the Conners ADHD/DSM-IV Scale for Teachers (CADS-T) at baseline and the end of each week. The CADS-T assesses symptoms of hyperactivity and inattention. The change from baseline of the (CADS-T) scores during the last week of treatment was analyzed as the primary efficacy parameter. Patients treated with methylphenidate hydrochloride extended-release capsules (LA) showed a statistically significant improvement in symptom scores from baseline [Mean (final score - baseline) = -10.7 points)] over patients who received placebo [Mean (final score - baseline) = +2.8 points ]. The lower the final score on the CADS-T scale from baseline, the less severe the disease is. This demonstrates that a single morning dose of methylphenidate hydrochloride extended-release capsules (LA) exerts a treatment effect in ADHD.
| Figure 2: CADS-T Total Subscale - Mean Change From Baseline* |
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The clinical program for methylphenidate hydrochloride extended-release capsules (LA) consisted of 6 studies: 2 controlled clinical studies conducted in children with ADHD aged 6 to 12 years and 4 clinical pharmacology studies conducted in healthy adult volunteers. These studies included a total of 256 subjects; 195 children with ADHD and 61 healthy adult volunteers. The subjects received methylphenidate hydrochloride extended-release capsules (LA) in doses of 10 to 40 mg per day. Safety of methylphenidate hydrochloride extended-release capsules (LA) was assessed by evaluating frequency and nature of adverse events, routine laboratory tests, vital signs, and body weight. A placebo-controlled, double-blind, parallel-group study was conducted to evaluate the efficacy and safety of methylphenidate hydrochloride extended-release capsules (LA) in children with ADHD aged 6 to 12 years. All subjects received methylphenidate hydrochloride extended-release capsules (LA) for up to 4 weeks, and had their dose optimally adjusted, prior to entering the double-blind phase of the trial. In the 2-week double-blind treatment phase of this study, patients received either placebo or methylphenidate hydrochloride extended-release capsules (LA) at their individually-titrated dose (range, 10 to 40 mg).
Adverse reactions with an incidence greater than 5% during the initial 4-week single-blind methylphenidate hydrochloride extended-release capsules (LA) titration period of this study were headache, insomnia, upper abdominal pain, appetite decreased, and anorexia.
Adverse reactions with an incidence greater than 2% among methylphenidate hydrochloride extended-release capsules (LA) -treated subjects, during the 2-week double-blind phase of the clinical study, are shown in Table 2:
| Preferred Term | Methylphenidate Hydrochloride Extended-Release Capsules (LA)
N = 65 N (%) |
Placebo
N = 71 N (%) |
|---|---|---|
| Anorexia | 2 (3.1) | 0 (0.0) |
| Insomnia | 2 (3.1) | 0 (0.0) |
Warning: Abuse and Dependence (WARNING: ABUSE AND DEPENDENCE)
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Warning and Precautions (5.1), Drug Abuse and Dependence (9.2, 9.3)] .
2.2 General Dosing Information
The recommended starting dose for methylphenidate hydrochloride extended-release capsules (LA) is 20 mg once daily. Increase dosage gradually, in increments of 10 mg weekly. Daily dosage above 60 mg is not recommended. When a lower initial dose is appropriate, patients may begin treatment with 10 mg.
Administer methylphenidate hydrochloride extended-release capsules (LA) orally once daily in the morning. Methylphenidate hydrochloride extended-release capsules (LA) may be swallowed as whole capsules or may be administered by sprinkling the capsule contents on a small amount of applesauce (see specific instructions below). Methylphenidate hydrochloride extended-release capsules (LA) and/or their contents should not be crushed, chewed, or divided.
The capsules may be carefully opened and the beads sprinkled over a spoonful of applesauce. The applesauce should not be warm because it could affect the modified release properties of this formulation. The mixture of drug and applesauce should be consumed immediately in its entirety. The drug and applesauce mixture should not be stored for future use.
Pharmacological treatment of ADHD may be needed for extended periods. Periodically reevaluate the long-term use of methylphenidate hydrochloride tablets and methylphenidate hydrochloride extended-release tablets, and adjust dosage as needed.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide)
5.7 Long Term Suppression of Growth (5.7 Long-Term Suppression of Growth)
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated patients over 36 months (to the ages of 10-13 years), suggests that consistently medicated pediatric patients (i.e., treatment for 7 days per week throughout the year) have a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this period of development.
Closely monitor growth (weight and height) in pediatric patients treated with CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA). Patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
10 mg white/light green capsules (imprinted mayne 609 on both the body and the cap)
| Bottles of 100 | NDC 51862-609-01 |
20 mg white/white capsules (imprinted mayne 610 on both the body and cap
| Bottles of 100 | NDC 51862-610-01 |
30 mg white/light blue capsules (imprinted mayne 611 on both the body and cap
| Bottles of 100 | NDC 51862-611-01 |
40 mg white/dark blue capsules (imprinted mayne 612 on both the body and cap)
| Bottles of 100 | NDC 51862-612-01 |
60 mg light yellow/dark yellow capsules (imprinted mayne 614 on both the body and cap)
| Bottles of 30 | NDC 51862-614-30 |
5.2 Serious Cardiovascular Reactions
Sudden death, stroke, and myocardial infarction have been reported in adults with CNS-stimulant treatment at recommended doses. Sudden death has been reported in pediatric patients with structural cardiac abnormalities and other serious heart problems taking CNS stimulants at recommended doses for ADHD. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, and other serious heart problems. Further evaluate patients who develop exertional chest pain, unexplained syncope, or arrhythmias during methylphenidate hydrochloride extended-release capsules (LA) treatment.
2.5 Dose Reduction and Discontinuation
If paradoxical worsening of symptoms or other adverse reactions occur, reduce the dosage, or, if necessary, discontinue methylphenidate hydrochloride extended-release capsules (LA). If improvement is not observed after appropriate dosage adjustment over a one-month period, the drug should be discontinued.
5.1 Potential for Abuse and Dependence
CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), other methylphenidate-containing products, and amphetamines, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Boxed Warning, Drug Abuse and Dependence (9.2, 9.3)].
5.3 Blood Pressure and Heart Rate Increases
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 beats per minute). Individuals may have larger increases. Monitor all patients for hypertension and tachycardia.
2.4 Switching From Other Methylphenidate Products
If switching from other methylphenidate products, discontinue that treatment, and titrate with methylphenidate hydrochloride extended-release capsules (LA) using the titration schedule.
Do not substitute for other methylphenidate products on a milligram-per-milligram basis, because different methylphenidate base compositions and differing pharmacokinetic profiles [see Description (11), Clinical Pharmacology (12.3)] .
Clinical judgment should be used when selecting the starting dose. Daily dosage above 60 mg is not recommended.
Principal Display Panel 10 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Label)
NDC 51862-609-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
10 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 20 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Label)
NDC 51862-610-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
20 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 30 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Label)
NDC 51862-611-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
30 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 40 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 40 mg Capsule Bottle Label)
NDC 51862-612-01
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
40 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
100 Capsules
maynepharma
Principal Display Panel 60 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 60 mg Capsule Bottle Label)
NDC 51862-614-30
Once Daily
Methylphenidate
Hydrochloride
CII
Extended-Release Capsules (LA)
60 mg
PHARMACIST: Please dispense with
Medication Guide provided separately.
Rx Only
30 Capsules
maynepharma
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon
CNS Stimulants, including methylphenidate hydrochloride extended-release capsules (LA), used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.
7.1 Clinically Important Drug Interactions With Methylphenidate Hydrochloride Extended Release Capsules (la) (7.1 Clinically Important Drug Interactions With Methylphenidate Hydrochloride Extended-Release Capsules (LA))
Table 3 presents clinically important drug interactions with methylphenidate hydrochloride extended-release capsules (LA)
| Monoamine Oxidase Inhibitors (MAOI) | |
| Clinical Impact | Concomitant use of MAOIs and CNS stimulants, including methylphenidate hydrochloride extended-release capsules (LA), can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure [see Contraindications (4)] . |
| Intervention | Concomitant use of methylphenidate hydrochloride extended-release capsules (LA) with MAOIs or within 14 days after discontinuing MAOI treatment is contraindicated. |
| Examples | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Antihypertensive Drugs | |
| Clinical Impact | Methylphenidate hydrochloride extended-release capsules (LA) may decrease the effectiveness of drugs used to treat hypertension [see Warnings and Precautions (5.3)] . |
| Intervention | Monitor blood pressure and adjust the dosage of the antihypertensive drug as needed. |
| Examples | Potassium-sparing and thiazide diuretics, calcium channel blockers, angiotensin-converting-enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), beta blockers, centrally acting alpha-2 receptor agonists. |
| Halogenated Anesthetics | |
| Clinical Impact | Concomitant use of halogenated anesthetics and methylphenidate hydrochloride extended-release capsules (LA) may increase the risk of sudden blood pressure and heart rate increase during surgery. |
| Intervention | Avoid use of methylphenidate hydrochloride extended-release capsules (LA) in patients being treated with anesthetics on the day of surgery. |
| Examples | halothane, isoflurane, enflurane, desflurane, sevoflurane |
| Risperidone | |
| Clinical Impact | Combined use of methylphenidate with risperidone when there is a change, whether an increase or decrease, in dosage of either or both medications, may increase the risk of extrapyramidal symptoms (EPS) |
| Intervention | Monitor for signs of EPS |
2.3 Patients Currently Using Methylphenidate Hydrochloride Tablets Or Methylphenidate Hydrochloride Extended Release Tablets (2.3 Patients Currently Using Methylphenidate Hydrochloride Tablets or Methylphenidate Hydrochloride Extended-Release Tablets)
The recommended dose of methylphenidate hydrochloride extended-release capsules (LA) for patients currently taking methylphenidate hydrochloride tablets twice daily or methylphenidate hydrochloride extended-release tablets (SR) is provided below.
| Previous Methylphenidate Hydrochloride Tablets or Methylphenidate Hydrochloride Extended-Release Tablets Dose | Recommended Methylphenidate Hydrochloride Extended-Release Capsules (LA) Dose |
|---|---|
| 5 mg methylphenidate hydrochloride tablets twice daily | 10 mg once daily |
| 10 mg methylphenidate hydrochloride tablets twice daily or 20 mg methylphenidate hydrochloride extended-release tablets | 20 mg once daily |
| 15 mg methylphenidate hydrochloride tablets twice daily | 30 mg once daily |
| 20 mg methylphenidate hydrochloride tablets twice daily or 40 mg of methylphenidate hydrochloride extended-release tablets | 40 mg once daily |
| 30 mg methylphenidate hydrochloride tablets twice daily or 60 mg of methylphenidate hydrochloride extended-release tablets | 60 mg once daily |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:17.529424 · Updated: 2026-03-14T22:49:18.583726