Obagi Nu-derm®
e5c450af-958e-41e3-9c2f-fda76b3596f5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rx Only For external use only
Indications and Usage
For the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines, and other unwanted areas of melanin hyperpigmentation.
Dosage and Administration
A thin application should be applied once or twice daily or as directed by a physician. If no improvement is seen after three (3) months of treatment, use of this product should be discontinued. Sun exposure should be limited by using a sunscreen agent, a sunblocking agent, or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring.
Contraindications
People with prior history of sensitivity or allergic reaction to this product or any of its ingredients should not use it. The safety of topical hydroquinone use during pregnancy or in children (12 years and under) has not been established.
Adverse Reactions
No systemic adverse reactions have been reported. Occasional hypersensitivity (localized contact dermatitis) may occur, in which case the product should be discontinued and the physician notified immediately.
How Supplied
Obagi Nu-Derm Clear is available as follows: Net wt. 2 oz. (57 g) bottle NDC 62032-101-36 Obagi Nu-Derm Blender is available as follows: Net wt. 2 oz. (57 g) bottle NDC 62032-100-36 Net wt. 1 oz. (28 g) bottle NDC 62032-100-10 Obagi Nu-Derm Sunfader is available as follows: Net wt. 2 oz. (57 g) bottle NDC 62032-116-36
Medication Information
Indications and Usage
For the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines, and other unwanted areas of melanin hyperpigmentation.
Dosage and Administration
A thin application should be applied once or twice daily or as directed by a physician. If no improvement is seen after three (3) months of treatment, use of this product should be discontinued. Sun exposure should be limited by using a sunscreen agent, a sunblocking agent, or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring.
Contraindications
People with prior history of sensitivity or allergic reaction to this product or any of its ingredients should not use it. The safety of topical hydroquinone use during pregnancy or in children (12 years and under) has not been established.
Adverse Reactions
No systemic adverse reactions have been reported. Occasional hypersensitivity (localized contact dermatitis) may occur, in which case the product should be discontinued and the physician notified immediately.
How Supplied
Obagi Nu-Derm Clear is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-101-36
Obagi Nu-Derm Blender is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-100-36
Net wt. 1 oz. (28 g) bottle
NDC 62032-100-10
Obagi Nu-Derm Sunfader is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-116-36
Description
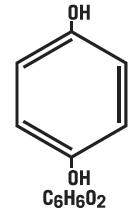
Hydroquinone is 1, 4-benzenediol. The drug is freely soluble in water and in alcohol. Chemically, hydroquinone is designed as p-dihydroxybenzene; the empirical formula is C6H6O2; molecular weight is 110.11 g/mol. The chemical structure is in the diagram below.
Each gram of Obagi Nu-Derm Clear contains:
Active ingredient: Hydroquinone USP, 4% (40 mg/g)
Inactive ingredients: water, cetyl alcohol, glycerin, sodium lauryl sulfate, stearyl alcohol, lactic acid, tocopheryl acetate, ascorbic acid, sodium metabisulfite, disodium EDTA, methylparaben, BHT, propylparaben, saponins, butylparaben
Each gram of Obagi Nu-Derm Blender contains:
Active ingredient: Hydroquinone USP, 4% (40 mg/g)
Inactive ingredients: water, glycerin, cetyl alcohol, PPG-2 myristyl ether propionate, sodium lauryl sulfate, TEA-salicylate, lactic acid, phenyl trimethicone, tocopheryl acetate, sodium metabisulfite, ascorbic acid, methylparaben, disodium EDTA, propylparaben, saponins, BHT
Each gram of Obagi Nu-Derm Sunfader contains:
Active ingredients: Hydroquinone USP, 4% (40 mg/g); Octinoxate USP, 7.5%; Oxybenzone USP, 5.5%
Inactive ingredients: water, cetyl alcohol, glycerin, sodium lauryl sulfate, stearyl alcohol, tocopheryl acetate, ascorbic acid, sodium metabisulfite, disodium EDTA, methylparaben, BHT, saponins, propylparaben, butylparaben
Section 42229-5
Rx Only
For external use only
Section 44425-7
Store at controlled room temperature: 15°C-25°C (59°F-77°F). Keep out of direct sunlight.
Warnings
Hydroquinone is a skin-bleaching agent, which may produce unwanted cosmetic effects if not used as directed. The physician should be familiar with the contents of this insert before prescribing or dispensing this product.
Test for skin sensitivity before using by applying a small amount to an unbroken patch of skin and check within 24 hours. Minor redness is not a contraindication, but where there is itching or vesicle formation or excessive inflammatory response, product should be discontinued and physician consulted. Close patient supervision is recommended.
Warnings: Avoid contact with eyes, nose, mouth, and lips.In case of accidental contact, patient should rinse thoroughly with water and contact a physician. Sunscreen use is an essential aspect of hydroquinone therapy because even minimal sunlight exposure sustains melanocytic activity.
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Precautions
(Also see WARNINGS)
Treatment should be limited to relatively small areas of the body at one time since some patients experience a transient skin reddening and a mild burning sensation, which does not preclude treatment.
Nursing Mothers
It is not known whether topical hydroquinone is absorbed or excreted in human milk. Caution is advised when topical hydroquinone is used by a nursing mother.
Pediatric Usage
Safety and effectiveness in children below the age of 12 years have not been established.
Pregnancy Category C
Animal reproduction studies have not been conducted with topical hydroquinone. It is also not known whether hydroquinone can cause fetal harm when used topically on a pregnant woman or affect reproductive capacity. It is not known to what degree, if any, topical hydroquinone is absorbed systemically. Topical hydroquinone should be used on pregnant women only when clearly indicated.
Clinical Pharmacology
Topical application of hydroquinone produces a reversible depigmentation of the skin by inhibition of the enzymatic oxidation of tyrosine to 3, 4-dihydroxyphenylalanine (DOPA) and suppression of other melanocyte metabolic processes. Exposure to sunlight or ultraviolet light will cause repigmentation of the bleached areas, which may be prevented by the use of sunblocking agents or sunscreen agents contained in Obagi Nu-Derm Sunfader and Obagi Nu-Derm Sun Shield Matte Broad Spectrum SPF 50.
Principal Display Panel 57 G Bottle Label 3
OBAGI
NU-DERM®
NDC 62032-101-36
AM CLEAR
PM
3
SKIN BLEACHING
& CORRECTOR CREAM
HYDROQUINONE USP, 4%
Rx ONLY
Net wt. 2 oz. (57g)
Principal Display Panel 57 G Bottle Label 5
OBAGI
NU-DERM®
NDC 62032-100-36
BLENDER®
PM
5
SKIN LIGHTENER
& BLENDING CREAM
HYDROQUINONE USP, 4%
Rx ONLY
Net wt. 2 oz. (57g)
Principal Display Panel 57 G Bottle Label 6
OBAGI
NU-DERM®
NDC 62032-116-36
AM
6
SUNFADER®
HYDROQUINONE USP, 4%
SKIN LIGHTENER
WITH SUNSCREEN
(SPF 15) PABA-FREE
Rx only
Net wt. 2 oz. (57 g)
Structured Label Content
Section 42229-5 (42229-5)
Rx Only
For external use only
Section 44425-7 (44425-7)
Store at controlled room temperature: 15°C-25°C (59°F-77°F). Keep out of direct sunlight.
Warnings (WARNINGS)
Hydroquinone is a skin-bleaching agent, which may produce unwanted cosmetic effects if not used as directed. The physician should be familiar with the contents of this insert before prescribing or dispensing this product.
Test for skin sensitivity before using by applying a small amount to an unbroken patch of skin and check within 24 hours. Minor redness is not a contraindication, but where there is itching or vesicle formation or excessive inflammatory response, product should be discontinued and physician consulted. Close patient supervision is recommended.
Warnings: Avoid contact with eyes, nose, mouth, and lips.In case of accidental contact, patient should rinse thoroughly with water and contact a physician. Sunscreen use is an essential aspect of hydroquinone therapy because even minimal sunlight exposure sustains melanocytic activity.
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Description (DESCRIPTION)
Hydroquinone is 1, 4-benzenediol. The drug is freely soluble in water and in alcohol. Chemically, hydroquinone is designed as p-dihydroxybenzene; the empirical formula is C6H6O2; molecular weight is 110.11 g/mol. The chemical structure is in the diagram below.
Each gram of Obagi Nu-Derm Clear contains:
Active ingredient: Hydroquinone USP, 4% (40 mg/g)
Inactive ingredients: water, cetyl alcohol, glycerin, sodium lauryl sulfate, stearyl alcohol, lactic acid, tocopheryl acetate, ascorbic acid, sodium metabisulfite, disodium EDTA, methylparaben, BHT, propylparaben, saponins, butylparaben
Each gram of Obagi Nu-Derm Blender contains:
Active ingredient: Hydroquinone USP, 4% (40 mg/g)
Inactive ingredients: water, glycerin, cetyl alcohol, PPG-2 myristyl ether propionate, sodium lauryl sulfate, TEA-salicylate, lactic acid, phenyl trimethicone, tocopheryl acetate, sodium metabisulfite, ascorbic acid, methylparaben, disodium EDTA, propylparaben, saponins, BHT
Each gram of Obagi Nu-Derm Sunfader contains:
Active ingredients: Hydroquinone USP, 4% (40 mg/g); Octinoxate USP, 7.5%; Oxybenzone USP, 5.5%
Inactive ingredients: water, cetyl alcohol, glycerin, sodium lauryl sulfate, stearyl alcohol, tocopheryl acetate, ascorbic acid, sodium metabisulfite, disodium EDTA, methylparaben, BHT, saponins, propylparaben, butylparaben
Precautions (PRECAUTIONS)
(Also see WARNINGS)
Treatment should be limited to relatively small areas of the body at one time since some patients experience a transient skin reddening and a mild burning sensation, which does not preclude treatment.
How Supplied (HOW SUPPLIED)
Obagi Nu-Derm Clear is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-101-36
Obagi Nu-Derm Blender is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-100-36
Net wt. 1 oz. (28 g) bottle
NDC 62032-100-10
Obagi Nu-Derm Sunfader is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-116-36
Nursing Mothers
It is not known whether topical hydroquinone is absorbed or excreted in human milk. Caution is advised when topical hydroquinone is used by a nursing mother.
Pediatric Usage
Safety and effectiveness in children below the age of 12 years have not been established.
Adverse Reactions (ADVERSE REACTIONS)
No systemic adverse reactions have been reported. Occasional hypersensitivity (localized contact dermatitis) may occur, in which case the product should be discontinued and the physician notified immediately.
Contraindications (CONTRAINDICATIONS)
People with prior history of sensitivity or allergic reaction to this product or any of its ingredients should not use it. The safety of topical hydroquinone use during pregnancy or in children (12 years and under) has not been established.
Pregnancy Category C
Animal reproduction studies have not been conducted with topical hydroquinone. It is also not known whether hydroquinone can cause fetal harm when used topically on a pregnant woman or affect reproductive capacity. It is not known to what degree, if any, topical hydroquinone is absorbed systemically. Topical hydroquinone should be used on pregnant women only when clearly indicated.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Topical application of hydroquinone produces a reversible depigmentation of the skin by inhibition of the enzymatic oxidation of tyrosine to 3, 4-dihydroxyphenylalanine (DOPA) and suppression of other melanocyte metabolic processes. Exposure to sunlight or ultraviolet light will cause repigmentation of the bleached areas, which may be prevented by the use of sunblocking agents or sunscreen agents contained in Obagi Nu-Derm Sunfader and Obagi Nu-Derm Sun Shield Matte Broad Spectrum SPF 50.
Indications and Usage (INDICATIONS AND USAGE)
For the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines, and other unwanted areas of melanin hyperpigmentation.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
A thin application should be applied once or twice daily or as directed by a physician. If no improvement is seen after three (3) months of treatment, use of this product should be discontinued. Sun exposure should be limited by using a sunscreen agent, a sunblocking agent, or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring.
Principal Display Panel 57 G Bottle Label 3 (PRINCIPAL DISPLAY PANEL - 57 g Bottle Label - 3)
OBAGI
NU-DERM®
NDC 62032-101-36
AM CLEAR
PM
3
SKIN BLEACHING
& CORRECTOR CREAM
HYDROQUINONE USP, 4%
Rx ONLY
Net wt. 2 oz. (57g)
Principal Display Panel 57 G Bottle Label 5 (PRINCIPAL DISPLAY PANEL - 57 g Bottle Label - 5)
OBAGI
NU-DERM®
NDC 62032-100-36
BLENDER®
PM
5
SKIN LIGHTENER
& BLENDING CREAM
HYDROQUINONE USP, 4%
Rx ONLY
Net wt. 2 oz. (57g)
Principal Display Panel 57 G Bottle Label 6 (PRINCIPAL DISPLAY PANEL - 57 g Bottle Label - 6)
OBAGI
NU-DERM®
NDC 62032-116-36
AM
6
SUNFADER®
HYDROQUINONE USP, 4%
SKIN LIGHTENER
WITH SUNSCREEN
(SPF 15) PABA-FREE
Rx only
Net wt. 2 oz. (57 g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:08.029117 · Updated: 2026-03-14T22:24:05.784289