These Highlights Do Not Include All The Information Needed To Use Caprelsa Safely And Effectively. See Full Prescribing Information For Caprelsa.
e5721cb8-4185-47b9-bbb3-1c587e558a03
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
CAPRELSA can prolong the QT interval. Torsades de pointes and sudden death have occurred in patients receiving CAPRELSA. Do not use CAPRELSA in patients with hypocalcemia, hypokalemia, hypomagnesemia, or long QT syndrome. Correct hypocalcemia, hypokalemia and/or hypomagnesemia prior to CAPRELSA administration. Monitor electrolytes periodically. Avoid drugs known to prolong the QT interval [see Warnings and Precautions (5.1) ] .
Indications and Usage
CAPRELSA is indicated for the treatment of symptomatic or progressive medullary thyroid cancer in patients with unresectable locally advanced or metastatic disease. Use CAPRELSA in patients with indolent, asymptomatic or slowly progressing disease only after careful consideration of the treatment related risks of CAPRELSA.
Dosage and Administration
The recommended dose of CAPRELSA is 300 mg taken orally once daily until disease progression or unacceptable toxicity occurs. CAPRELSA may be taken with or without food. Do not take a missed dose within 12 hours of the next dose. Do not crush CAPRELSA tablets. The tablets can be dispersed in 2 ounces of water by stirring for approximately 10 minutes (will not completely dissolve). Do not use other liquids for dispersion. Swallow immediately after dispersion. Mix any remaining residue with 4 additional ounces of water and swallow. The dispersion can also be administered through nasogastric or gastrostomy tubes.
Warnings and Precautions
Prolonged QT interval, torsades de pointes, and sudden death: Monitor electrocardiograms and levels of serum potassium, calcium, magnesium and TSH. Reduce CAPRELSA dose as appropriate. ( 2.1 , 5.1 ) Severe skin reactions, including toxic epidermal necrolysis and Stevens-Johnson syndrome, some fatal. Discontinue CAPRELSA for severe skin reactions. ( 2.1 , 5.2 ) Interstitial lung disease (ILD), including fatalities: investigate unexplained non-specific respiratory signs and symptoms. Discontinue CAPRELSA for confirmed ILD. ( 2.1 , 5.3 ) Ischemic cerebrovascular events, hemorrhage, heart failure, diarrhea, hypertension, and reversible posterior leukoencephalopathy syndrome: Discontinue or interrupt CAPRELSA. ( 2.1 , 5.4 , 5.5 , 5.6 , 5.7 , 5.9 , 5.10 ) Impaired wound healing: Withhold for at least 1 month prior to elective surgery. Do not administer CAPRELSA for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment with CAPRELSA after resolution of wound healing complications has not been established. ( 5.14 ) Embryo-fetal toxicity: Can cause fetal harm. Advise women of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.15 , 8.1 ) Osteonecrosis, including osteonecrosis of the jaw: Withhold CAPRELSA for 1 month prior to scheduled dental surgery and permanently discontinue if osteonecrosis occurs ( 5.16 , 6.2 ).
Contraindications
Do not use in patients with congenital long QT syndrome [see Boxed Warning ] .
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the label: QT Prolongation and Torsades de Pointes [see Boxed Warning , Warnings and Precautions (5.1) ] Severe Skin Reactions [see Warnings and Precautions (5.2) ] Interstitial Lung Disease [see Warnings and Precautions (5.3) ] Ischemic Cerebrovascular Events [see Warnings and Precautions (5.4) ] Hemorrhage [see Warnings and Precautions (5.5) ] Heart Failure [see Warnings and Precautions (5.6) ] Diarrhea [see Warnings and Precautions (5.7) ] Hypothyroidism [see Warnings and Precautions (5.8) ] Hypertension [see Warnings and Precautions (5.9) ] Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.10) ] Renal Failure [see Warnings and Precautions (5.12) ] Embryo-Fetal Toxicity [see Warnings and Precautions (5.15) ] Osteonecrosis [see Warnings and Precautions (5.16) ]
Drug Interactions
Avoid administration of CAPRELSA with anti-arrhythmic drugs (including but not limited to amiodarone, disopyramide, procainamide, sotalol, dofetilide) and other drugs that may prolong the QT interval (including but not limited to chloroquine, clarithromycin, dolasetron, granisetron, haloperidol, methadone, moxifloxacin, and pimozide) [see Drug Interactions (7.4) and Clinical Pharmacology (12.2) ] .
Storage and Handling
100 mg Tablets available in bottles containing 30 tablets (NDC 58468-7820-3). 300 mg Tablets available in bottles containing 30 tablets (NDC 58468-7840-3).
How Supplied
100 mg Tablets available in bottles containing 30 tablets (NDC 58468-7820-3). 300 mg Tablets available in bottles containing 30 tablets (NDC 58468-7840-3).
Medication Information
Warnings and Precautions
Prolonged QT interval, torsades de pointes, and sudden death: Monitor electrocardiograms and levels of serum potassium, calcium, magnesium and TSH. Reduce CAPRELSA dose as appropriate. ( 2.1 , 5.1 ) Severe skin reactions, including toxic epidermal necrolysis and Stevens-Johnson syndrome, some fatal. Discontinue CAPRELSA for severe skin reactions. ( 2.1 , 5.2 ) Interstitial lung disease (ILD), including fatalities: investigate unexplained non-specific respiratory signs and symptoms. Discontinue CAPRELSA for confirmed ILD. ( 2.1 , 5.3 ) Ischemic cerebrovascular events, hemorrhage, heart failure, diarrhea, hypertension, and reversible posterior leukoencephalopathy syndrome: Discontinue or interrupt CAPRELSA. ( 2.1 , 5.4 , 5.5 , 5.6 , 5.7 , 5.9 , 5.10 ) Impaired wound healing: Withhold for at least 1 month prior to elective surgery. Do not administer CAPRELSA for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment with CAPRELSA after resolution of wound healing complications has not been established. ( 5.14 ) Embryo-fetal toxicity: Can cause fetal harm. Advise women of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.15 , 8.1 ) Osteonecrosis, including osteonecrosis of the jaw: Withhold CAPRELSA for 1 month prior to scheduled dental surgery and permanently discontinue if osteonecrosis occurs ( 5.16 , 6.2 ).
Indications and Usage
CAPRELSA is indicated for the treatment of symptomatic or progressive medullary thyroid cancer in patients with unresectable locally advanced or metastatic disease. Use CAPRELSA in patients with indolent, asymptomatic or slowly progressing disease only after careful consideration of the treatment related risks of CAPRELSA.
Dosage and Administration
The recommended dose of CAPRELSA is 300 mg taken orally once daily until disease progression or unacceptable toxicity occurs. CAPRELSA may be taken with or without food. Do not take a missed dose within 12 hours of the next dose. Do not crush CAPRELSA tablets. The tablets can be dispersed in 2 ounces of water by stirring for approximately 10 minutes (will not completely dissolve). Do not use other liquids for dispersion. Swallow immediately after dispersion. Mix any remaining residue with 4 additional ounces of water and swallow. The dispersion can also be administered through nasogastric or gastrostomy tubes.
Contraindications
Do not use in patients with congenital long QT syndrome [see Boxed Warning ] .
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the label: QT Prolongation and Torsades de Pointes [see Boxed Warning , Warnings and Precautions (5.1) ] Severe Skin Reactions [see Warnings and Precautions (5.2) ] Interstitial Lung Disease [see Warnings and Precautions (5.3) ] Ischemic Cerebrovascular Events [see Warnings and Precautions (5.4) ] Hemorrhage [see Warnings and Precautions (5.5) ] Heart Failure [see Warnings and Precautions (5.6) ] Diarrhea [see Warnings and Precautions (5.7) ] Hypothyroidism [see Warnings and Precautions (5.8) ] Hypertension [see Warnings and Precautions (5.9) ] Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.10) ] Renal Failure [see Warnings and Precautions (5.12) ] Embryo-Fetal Toxicity [see Warnings and Precautions (5.15) ] Osteonecrosis [see Warnings and Precautions (5.16) ]
Drug Interactions
Avoid administration of CAPRELSA with anti-arrhythmic drugs (including but not limited to amiodarone, disopyramide, procainamide, sotalol, dofetilide) and other drugs that may prolong the QT interval (including but not limited to chloroquine, clarithromycin, dolasetron, granisetron, haloperidol, methadone, moxifloxacin, and pimozide) [see Drug Interactions (7.4) and Clinical Pharmacology (12.2) ] .
Storage and Handling
100 mg Tablets available in bottles containing 30 tablets (NDC 58468-7820-3). 300 mg Tablets available in bottles containing 30 tablets (NDC 58468-7840-3).
How Supplied
100 mg Tablets available in bottles containing 30 tablets (NDC 58468-7820-3). 300 mg Tablets available in bottles containing 30 tablets (NDC 58468-7840-3).
Description
CAPRELSA can prolong the QT interval. Torsades de pointes and sudden death have occurred in patients receiving CAPRELSA. Do not use CAPRELSA in patients with hypocalcemia, hypokalemia, hypomagnesemia, or long QT syndrome. Correct hypocalcemia, hypokalemia and/or hypomagnesemia prior to CAPRELSA administration. Monitor electrolytes periodically. Avoid drugs known to prolong the QT interval [see Warnings and Precautions (5.1) ] .
Section 42229-5
For Adverse Reactions
The 300 mg daily dose can be reduced to 200 mg (two 100 mg tablets) and then to 100 mg for Common Terminology Criteria for Adverse Events (CTCAE) Grade 3 or greater toxicities.
Interrupt CAPRELSA for the following:
- Corrected QT interval, Fridericia (QTcF) greater than 500 ms: Resume at a reduced dose when the QTcF returns to less than 450 ms.
- CTCAE Grade 3 or greater toxicity: Resume at a reduced dose when the toxicity resolves or improves to CTCAE Grade 1.
For recurrent toxicities, reduce the dose of CAPRELSA to 100 mg after resolution or improvement to CTCAE Grade 1 severity, if continued treatment is warranted.
Adverse events including QT interval prolongation should be monitored closely as they may not resolve fully until approximately three plasma half-lives of the drug. Monitor appropriately [see Warnings and Precautions (5.1), (5.2), (5.3), (5.4), (5.5), (5.6), (5.7), and (5.9)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 1/2026 | |
|
MEDICATION GUIDE
CAPRELSA® (kap-rel-sah) (vandetanib) tablets |
||
| Read this Medication Guide before you start taking CAPRELSA and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment. | ||
|
What is the most important information I should know about CAPRELSA? CAPRELSA can cause a change in the electrical activity of your heart called QT prolongation, which can cause irregular heartbeats and that may lead to death. You should not take CAPRELSA if you have had a condition called long QT syndrome since birth. Your healthcare provider should perform tests to check the levels of your blood potassium, calcium, magnesium, and thyroid-stimulating hormone (TSH), as well as the electrical activity of your heart with a test called an electrocardiogram (ECG). You should have these tests:
Call your healthcare provider right away if you feel faint, light-headed, or feel your heart beating irregularly during treatment with CAPRELSA. These may be symptoms related to QT prolongation. See "What are the possible side effects of CAPRELSA?" for more information about side effects. |
||
|
What is CAPRELSA?
CAPRELSA is a prescription medicine used to treat medullary thyroid cancer that cannot be removed by surgery or that has spread to other parts of the body. It takes a long time to get rid of CAPRELSA from your body and you may be at risk for side effects related to CAPRELSA after you have stopped your treatment. It is not known if CAPRELSA is safe and effective in children. |
||
|
Who should not take CAPRELSA? Do not take CAPRELSA if you have QT prolongation. |
||
Before you take CAPRELSA, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
Do not take other medicines during treatment with CAPRELSA until you have talked with your healthcare provider or pharmacist. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
||
How should I take CAPRELSA?
|
||
What should I avoid while taking CAPRELSA?
|
||
|
What are the possible side effects of CAPRELSA? CAPRELSA may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
|
||
| The most common side effects of CAPRELSA include: | ||
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of CAPRELSA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store CAPRELSA?
|
||
|
General information about the safe and effective use of CAPRELSA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CAPRELSA for a condition for which it was not prescribed. Do not give CAPRELSA to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about CAPRELSA that is written for health professionals. |
||
|
What are the ingredients in CAPRELSA? Active ingredient: vandetanib Inactive ingredients:
CAPRELSA is a registered trademark of Genzyme Corporation. ©2026 Genzyme Corporation. For more information, go to www.caprelsa.com or call 1-800-633-1610. |
Section 43683-2
| Warnings and Precautions (5.16) | 05/2025 |
5.7 Diarrhea
Diarrhea of Grade 3 or greater severity occurred in 11% of patients receiving CAPRELSA in the randomized MTC study. If diarrhea occurs, carefully monitor serum electrolytes and ECGs to reduce the risk and enable early detection of QT prolongation resulting from dehydration [see Warnings and Precautions (5.1)]. Interrupt CAPRELSA for severe diarrhea. Upon improvement, resume CAPRELSA at a reduced dose [see Dosage and Administration (2.1)].
10 Overdosage
In the event of an overdose, monitor patients closely for QTc prolongation. Adverse events including QT interval prolongation should be monitored closely as they may not resolve fully until approximately three plasma half-lives of the drug.
15 References
- OSHA Hazardous Drugs (OSHA Technical Manual). OSHA.
11 Description
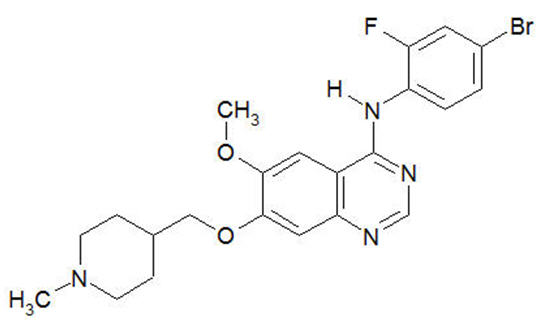
Vandetanib has the chemical name N-(4-bromo-2-fluorophenyl)-6-methoxy-7-[(1-methylpiperidin-4-yl) methoxy]quinazolin-4-amine.
The structural and molecular formulas are:
C22H24BrFN4O2
Vandetanib has a molecular weight of 475.36 g/mol. Vandetanib exhibits pH-dependent solubility, with increased solubility at lower pH. Vandetanib is practically insoluble in water with a value of 0.008 mg/mL at 25°C (77°F).
CAPRELSA tablets for daily oral administration are available in two dosage strengths containing either 100 mg or 300 mg of vandetanib. The tablet cores contain the following inactive ingredients: calcium hydrogen phosphate dihydrate, crospovidone, magnesium stearate, microcrystalline cellulose, and povidone. The tablet film-coat contains the following inactive ingredients: hypromellose 2910, macrogol 300, and titanium dioxide E171.
5.5 Hemorrhage
Serious hemorrhagic events, including fatalities, occurred in patients treated with CAPRELSA. Do not administer CAPRELSA to patients with a recent history of hemoptysis of ≥1/2 teaspoon of red blood. Discontinue CAPRELSA in patients with severe hemorrhage.
5.9 Hypertension
Hypertension, including hypertensive crisis, has occurred in patients treated with CAPRELSA. Monitor all patients for hypertension. Dose reduction or interruption for hypertension may be necessary. If hypertension cannot be controlled, do not resume CAPRELSA [see Dosage and Administration (2.1)].
5.6 Heart Failure
Heart failure, including fatalities, occurred in patients treated with CAPRELSA. Monitor for signs and symptoms of heart failure. Consider discontinuation of CAPRELSA in patients with heart failure. Heart failure may not be reversible upon stopping CAPRELSA.
8.4 Pediatric Use
Safety and efficacy of CAPRELSA in pediatric patients have not been established.
8.5 Geriatric Use
The MTC study of CAPRELSA did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently compared to younger patients.
5.12 Renal Failure
Renal failure occurred in patients treated with CAPRELSA [see Adverse Reactions (6.1)]. Withhold, reduce the dose or permanently discontinue based on severity [see Dosage and Administration (2.1)].
Vandetanib exposure is increased in patients with impaired renal function. Reduce the starting dose to 200 mg in patients with moderate renal impairment and monitor the QT interval closely [see Dosage and Administration (2.1)]. Vandetanib is not recommended for use in patients with severe renal impairment (clearance below 30 mL/min). There is no information available for patients with end-stage renal disease requiring dialysis [see Boxed Warning , Dosage and Administration (2.1) , Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.16 Osteonecrosis
Osteonecrosis, including osteonecrosis of the jaw (ONJ), has occurred during treatment with CAPRELSA. Concomitant exposure to other risk factors, such as bisphosphonates, denosumab, dental disease or invasive dental procedures, may increase the risk of ONJ. Perform an oral examination prior to and periodically during treatment with CAPRELSA. Advise patients regarding good oral hygiene practices. Avoid invasive dental procedures while on CAPRELSA treatment, particularly in patients at higher risk. Withhold CAPRELSA for at least one month prior to scheduled dental surgery or invasive dental procedures. Permanently discontinue CAPRELSA if ONJ develops. [see Adverse Reactions (6.2)].
5.8 Hypothyroidism
In the randomized MTC study in which 90% of the patients enrolled had prior thyroidectomy, increased dosing of thyroid replacement therapy was required in 49% of CAPRELSA-treated patients compared to 17% of placebo-treated patients. Obtain Thyroid-stimulating hormone (TSH) at baseline, at 2 to 4 weeks and 8 to 12 weeks after starting treatment with CAPRELSA, and every 3 months thereafter. If signs or symptoms of hypothyroidism occur, examine thyroid hormone levels and adjust thyroid replacement therapy accordingly.
4 Contraindications
Do not use in patients with congenital long QT syndrome [see Boxed Warning].
6 Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the label:
- QT Prolongation and Torsades de Pointes [see Boxed Warning, Warnings and Precautions (5.1)]
- Severe Skin Reactions [see Warnings and Precautions (5.2)]
- Interstitial Lung Disease [see Warnings and Precautions (5.3)]
- Ischemic Cerebrovascular Events [see Warnings and Precautions (5.4)]
- Hemorrhage [see Warnings and Precautions (5.5)]
- Heart Failure [see Warnings and Precautions (5.6)]
- Diarrhea [see Warnings and Precautions (5.7)]
- Hypothyroidism [see Warnings and Precautions (5.8)]
- Hypertension [see Warnings and Precautions (5.9)]
- Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.10)]
- Renal Failure [see Warnings and Precautions (5.12)]
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.15)]
- Osteonecrosis [see Warnings and Precautions (5.16)]
7 Drug Interactions
8.6 Renal Impairment
Vandetanib exposure is increased in patients with impaired renal function. Reduce the starting dose to 200 mg in patients with moderate (creatinine clearance ≥30 to <50 mL/min) renal impairment [see Dosage and Administration (2.1), Warnings and Precautions (5.12), and Clinical Pharmacology (12.3)].
Vandetanib is not recommended for use in patients with severe renal impairment (clearance below 30 mL/min) [see Warnings and Precautions (5.12)]. Patients with end-stage renal disease requiring dialysis were not studied [see Adverse Reactions (6.1)].
12.3 Pharmacokinetics
A population pharmacokinetic analysis of CAPRELSA was conducted in 231 patients with MTC following oral administration of 300 mg daily doses. The pharmacokinetics of CAPRELSA at the 300 mg dose in MTC patients are characterized by a mean clearance of approximately 13.2 L/h, a mean volume of distribution of approximately 7450 L, and a median plasma half-life of 19 days.
5.11 Drug Interactions
Avoid administration of CAPRELSA with anti-arrhythmic drugs (including but not limited to amiodarone, disopyramide, procainamide, sotalol, dofetilide) and other drugs that may prolong the QT interval (including but not limited to chloroquine, clarithromycin, dolasetron, granisetron, haloperidol, methadone, moxifloxacin, and pimozide) [see Drug Interactions (7.4) and Clinical Pharmacology (12.2)].
8.7 Hepatic Impairment
The pharmacokinetics of CAPRELSA were evaluated after a single dose of 800 mg in subjects with mild (n=8), moderate (n=7), and severe (n=6) hepatic impairment and normal hepatic function (n=5). Subjects with mild (Child-Pugh class A), moderate (Child-Pugh class B), and severe (Child-Pugh class C) hepatic impairment had comparable mean AUC and clearance values to those with normal hepatic function.
There are limited data in patients with liver impairment (serum bilirubin greater than 1.5 times the upper limit of normal). CAPRELSA is not recommended for use in patients with moderate and severe hepatic impairment, as safety and efficacy have not been established [see Dosage and Administration (2.1) and Warnings and Precautions (5.13)].
1 Indications and Usage
CAPRELSA is indicated for the treatment of symptomatic or progressive medullary thyroid cancer in patients with unresectable locally advanced or metastatic disease.
Use CAPRELSA in patients with indolent, asymptomatic or slowly progressing disease only after careful consideration of the treatment related risks of CAPRELSA.
5.13 Hepatic Impairment
CAPRELSA is not recommended for use in patients with moderate and severe hepatic impairment, as safety and efficacy have not been established [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
In vitro studies have shown that vandetanib inhibits the tyrosine kinase activity of the EGFR and VEGFR families, RET, BRK, TIE2, and members of the EPH receptor and Src kinase families. These receptor tyrosine kinases are involved in both normal cellular function and pathologic processes such as oncogenesis, metastasis, tumor angiogenesis, and maintenance of the tumor microenvironment. In addition, the N-desmethyl metabolite of the drug, representing 7 to 17.1% of vandetanib exposure, has similar inhibitory activity to the parent compound for VEGF receptors (KDR and Flt-1) and EGFR.
In vitro, vandetanib inhibited epidermal growth factor (EGF)-stimulated receptor tyrosine kinase phosphorylation in tumor cells and endothelial cells and VEGF-stimulated tyrosine kinase phosphorylation in endothelial cells.
In vivo, vandetanib administration reduced tumor cell-induced angiogenesis, tumor vessel permeability, and inhibited tumor growth and metastasis in mouse models of cancer.
16.1 Storage and Handling
CAPRELSA tablets should be stored at room temperature between 68°F and 77°F (20°C and 25°C); excursions permitted to 59°F–86°F (15°C-30°C) [See USP controlled room temperature].
Procedures for proper handling and disposal of anticancer drugs should be considered. A guideline on this subject has been published.1 Do not crush CAPRELSA tablets.
5.2 Severe Skin Reactions
Severe and sometimes fatal skin reactions, including toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome, have occurred in patients treated with CAPRELSA. Permanently discontinue CAPRELSA for severe skin reactions and refer the patient for urgent medical evaluation. Systemic therapies such as corticosteroids may be required.
Photosensitivity reactions can occur during CAPRELSA treatment and up to 4 months after treatment discontinuation.
5 Warnings and Precautions
- Prolonged QT interval, torsades de pointes, and sudden death: Monitor electrocardiograms and levels of serum potassium, calcium, magnesium and TSH. Reduce CAPRELSA dose as appropriate. (2.1, 5.1)
- Severe skin reactions, including toxic epidermal necrolysis and Stevens-Johnson syndrome, some fatal. Discontinue CAPRELSA for severe skin reactions. (2.1, 5.2)
- Interstitial lung disease (ILD), including fatalities: investigate unexplained non-specific respiratory signs and symptoms. Discontinue CAPRELSA for confirmed ILD. (2.1, 5.3)
- Ischemic cerebrovascular events, hemorrhage, heart failure, diarrhea, hypertension, and reversible posterior leukoencephalopathy syndrome: Discontinue or interrupt CAPRELSA. (2.1, 5.4, 5.5, 5.6, 5.7, 5.9, 5.10)
- Impaired wound healing: Withhold for at least 1 month prior to elective surgery. Do not administer CAPRELSA for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment with CAPRELSA after resolution of wound healing complications has not been established. (5.14)
- Embryo-fetal toxicity: Can cause fetal harm. Advise women of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.15, 8.1)
- Osteonecrosis, including osteonecrosis of the jaw: Withhold CAPRELSA for 1 month prior to scheduled dental surgery and permanently discontinue if osteonecrosis occurs (5.16, 6.2).
5.15 Embryo Fetal Toxicity
Based on its mechanism of action and findings in animals, CAPRELSA can cause fetal harm when administered to a pregnant woman. In rats, vandetanib was embryotoxic, fetotoxic, and induced fetal malformations at exposures equivalent to or lower than those expected at the 300 mg clinical dose and had adverse effects on female fertility, embryofetal development, and postnatal development of pups.
Advise patients of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with CAPRELSA and for 4 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with CAPRELSA and for 4 months after the last dose [see Use in Specific Populations (8.1), (8.3)].
2 Dosage and Administration
The recommended dose of CAPRELSA is 300 mg taken orally once daily until disease progression or unacceptable toxicity occurs.
CAPRELSA may be taken with or without food.
Do not take a missed dose within 12 hours of the next dose.
Do not crush CAPRELSA tablets. The tablets can be dispersed in 2 ounces of water by stirring for approximately 10 minutes (will not completely dissolve). Do not use other liquids for dispersion. Swallow immediately after dispersion. Mix any remaining residue with 4 additional ounces of water and swallow.
The dispersion can also be administered through nasogastric or gastrostomy tubes.
5.14 Impaired Wound Healing
Impaired wound healing can occur in patients who receive drugs that inhibit the VEGF signaling pathway. Impaired wound healing has occurred in patients treated with CAPRELSA.
Withhold CAPRELSA for at least 1 month prior to elective surgery. Do not administer CAPRELSA for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment with CAPRELSA after resolution of wound healing complications has not been established.
3 Dosage Forms and Strengths
CAPRELSA 100 mg tablets are white, round, biconvex, film-coated, and intagliated with 'Z 100' on one side and plain on the reverse side.
CAPRELSA 300 mg tablets are white, oval, biconvex, film-coated, and intagliated with 'Z 300' on one side and plain on the reverse side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of CAPRELSA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Vascular disorders: Arterial (including aortic) aneurysms, dissections, and rupture
Musculoskeletal and connective tissue disorders: Osteonecrosis
General disorders: Impaired wound healing
5.3 Interstitial Lung Disease
Interstitial Lung Disease (ILD) or pneumonitis, including fatalities, has occurred in patients treated with CAPRELSA. Consider a diagnosis of ILD in patients presenting with non-specific respiratory signs and symptoms.
Interrupt CAPRELSA for acute or worsening pulmonary symptoms. Discontinue CAPRELSA if ILD is confirmed.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Patients with unresectable locally advanced or metastatic medullary thyroid cancer were treated with CAPRELSA 300 mg (n=231) or Placebo (n=99). The population exposed to CAPRELSA was 58% male, 94% white, and had a median age of 50 years. The data described below reflect a median exposure to CAPRELSA for 607 days.
The most commonly reported adverse drug reactions which occurred in >20% of CAPRELSA-treated patients and with a between-arm difference of ≥5% included, in order of decreasing frequency: diarrhea/colitis, rash, acneiform dermatitis, hypertension, nausea, headache, upper respiratory tract infection, decreased appetite, and abdominal pain.
Among CAPRELSA-treated patients, dose interruption occurred in 109 (47%) and dose reduction occurred in 83 (36%). Adverse reactions led to study treatment discontinuation in 28 of 231 patients (12%) receiving CAPRELSA and in 3 of 99 patients (3.0%) receiving placebo. Adverse reactions leading to permanent discontinuation in 2 or more (≥0.9%) patients treated with CAPRELSA were: asthenia (1.7%), rash (1.7%), diarrhea (0.9%), fatigue (0.9%), pyrexia (0.9%), elevated creatinine (0.9%), QT prolongation (0.9%), and hypertension (0.9%).
| System Organ Class | CAPRELSA 300 mg | Placebo | ||
|---|---|---|---|---|
| Preferred Term | N=231 | N=99 | ||
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| Gastrointestinal Disorders | ||||
| Diarrhea/Colitis | 57 | 11 | 27 | 2 |
| Nausea | 33 | 1 | 16 | 0 |
| Abdominal Pain Includes abdominal pain, abdominal pain upper, lower abdominal pain, and abdominal discomfort.
|
21 | 3 | 11 | 0 |
| Vomiting | 15 | 1 | 7 | 0 |
| Dyspepsia | 11 | 0 | 4 | 0 |
| Dry Mouth | 9 | 0 | 3 | 0 |
| Skin and Cutaneous Disorders | ||||
| Rash Includes rash, rash (erythematous, generalized, macular, maculopapular, papular, pruritic, and exfoliative), dermatitis, dermatitis bullous, generalized erythema, and eczema.
|
53 | 5 | 12 | 0 |
| Dermatitis Acneiform/Acne | 35 | 1 | 7 | 0 |
| Dry Skin | 15 | 0 | 5 | 0 |
| Photosensitivity Reaction | 13 | 2 | 0 | 0 |
| Pruritus | 11 | 1 | 4 | 0 |
| Nail abnormalities Includes nail disorder, nail bed inflammation, nail bed tenderness, paronychia, nail bed infection, and nail infection.
|
9 | 0 | 0 | 0 |
| Alopecia | 8 | N/A | 0 | N/A |
| Vascular Disorders | ||||
| Hypertension/Hypertensive Crisis/Accelerated Hypertension | 33 | 9 | 5 | 1 |
| Nervous System Disorders | ||||
| Headache | 26 | 1 | 9 | 0 |
| Dysgeusia | 8 | 0 | 3 | 0 |
| General Disorders | ||||
| Fatigue Included in Table 1 due to the increased incidence of severe fatigue in the CAPRELSA group compared to the placebo group.
|
24 | 6 | 23 | 1 |
| Infections | ||||
| Upper Respiratory Tract Infections Includes laryngitis, nasopharyngitis, pharyngitis, sinusitis, upper respiratory tract infection, acute sinusitis, rhinitis, and tracheitis.
|
23 | 0 | 16 | 0 |
| Metabolic and Nutritional Disorders | ||||
| Decreased Appetite | 21 | 4 | 12 | 0 |
| Hypocalcemia | 11 | 2 | 3 | 0 |
| Investigations | ||||
| ECG QT Prolonged 69% had QT prolongation >450 ms and 7% had QT prolongation >500 ms by ECG using Fridericia correction.
|
14 | 8 | 1 | 1 |
| Eye Disorders | ||||
| Corneal Abnormalities Includes corneal edema, corneal opacity, corneal dystrophy, corneal pigmentation, keratopathy, arcus lipoides, corneal deposits, and acquired corneal dystrophy.
|
13 | 0 | 1 | 0 |
| Blurred Vision | 9 | 0 | 1 | 0 |
| Renal Disorders | ||||
| Proteinuria | 10 | 0 | 2 | 0 |
| Psychiatric Disorders | ||||
| Depression | 10 | 2 | 3 | 0 |
| Endocrine Disorders | ||||
| Hypothyroidism | 6 | 0 | 0 | 0 |
| Musculoskeletal Disorders | ||||
| Muscle Spasms | 6 | 0 | 1 | 0 |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
7.3 Effect of Caprelsa On Digoxin
CAPRELSA increased plasma concentrations of digoxin. Use caution and closely monitor for toxicities when administering CAPRELSA with digoxin [see Clinical Pharmacology (12.3)].
5.4 Ischemic Cerebrovascular Events
Ischemic cerebrovascular events, including fatalities, occurred in patients treated with CAPRELSA. In the randomized medullary thyroid cancer (MTC) study, ischemic cerebrovascular events occurred more frequently with CAPRELSA compared to placebo (1.3% compared to 0%). The safety of resumption of CAPRELSA therapy after resolution of an ischemic cerebrovascular event has not been studied. Discontinue CAPRELSA in patients who experience a severe ischemic cerebrovascular event.
16 How Supplied/storage and Handling
100 mg Tablets available in bottles containing 30 tablets (NDC 58468-7820-3).
300 mg Tablets available in bottles containing 30 tablets (NDC 58468-7840-3).
7.4 Drugs That Prolong the Qt Interval
Avoid concomitant use of CAPRELSA with agents that may prolong the QT interval [see Warnings and Precautions (5.11)].
7.1 Effect of Cyp3a4 Inducers On Caprelsa
Rifampicin, a strong CYP3A4 inducer, decreased vandetanib plasma concentrations. Avoid concomitant use of known strong CYP3A4 inducers during CAPRELSA therapy. Avoid concomitant use of St. John's wort because it can decrease vandetanib exposure unpredictably [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology And/or Pharmacology
In an animal model of wound-healing, mice dosed with vandetanib had reduced skin-breaking strength compared with controls. This suggests that CAPRELSA slows but does not prevent wound healing. The appropriate interval between discontinuation of CAPRELSA and subsequent elective surgery required to avoid the risks of impaired wound healing has not been determined.
7.2 Effect of Caprelsa On Oct2 Transporter
CAPRELSA increased plasma concentrations of metformin that is transported by the organic cation transporter type 2 (OCT2). Use caution and closely monitor for toxicities when administering CAPRELSA with drugs that are transported by OCT2 [see Clinical Pharmacology (12.3)].
5.1 Qt Prolongation and Torsades De Pointes
CAPRELSA can prolong the QT interval in a concentration-dependent manner [see Clinical Pharmacology (12.2)]. Torsades de pointes, ventricular tachycardia and sudden deaths have occurred in patients treated with CAPRELSA.
Do not start CAPRELSA treatment in patients whose QTcF interval is greater than 450 ms. Do not administer CAPRELSA to patients who have a history of Torsades de pointes, congenital long QT syndrome, bradyarrhythmias or uncompensated heart failure. CAPRELSA has not been studied in patients with ventricular arrhythmias or recent myocardial infarction. Vandetanib exposure is increased in patients with impaired renal function. Reduce the starting dose to 200 mg in patients with moderate renal impairment and monitor QT interval frequently.
Obtain an ECG and serum potassium, calcium, magnesium and TSH at baseline, 2 to 4 weeks and 8 to 12 weeks after starting treatment with CAPRELSA, and every 3 months thereafter. Monitor electrolytes and ECGs more frequently in patients who experience diarrhea. Following any dose reduction for QT prolongation or any dose interruption greater than 2 weeks, conduct QT assessments as described above. Maintain serum potassium levels of 4 mEq/L or higher (within normal range) and maintain serum magnesium and calcium levels within normal ranges to reduce the risk of QT prolongation.
Avoid using CAPRELSA with drugs known to prolong the QT interval [see Warnings and Precautions (5.11) and Drug Interactions (7.4)]. If such drugs are given to patients already receiving CAPRELSA and no alternative therapy exists, perform ECG monitoring of the QT interval more frequently.
Stop CAPRELSA in patients who develop a QTcF greater than 500 ms until the QTcF returns to less than 450 ms. Dosing of CAPRELSA can then be resumed at a reduced dose [see Dosage and Administration (2.1)].
Principal Display Panel 100 Mg Tablet Bottle Carton
NDC 58468-7820-3
Rx only
Caprelsa®
(vandetanib) tablets
100 mg
Dispense enclosed medication
guide to each patient.
30 Tablets
Principal Display Panel 300 Mg Tablet Bottle Carton
NDC 58468-7840-3
Rx only
Caprelsa®
(vandetanib) tablets
300 mg
Dispense enclosed medication
guide to each patient.
30 Tablets
5.10 Reversible Posterior Leukoencephalopathy Syndrome
Reversible posterior leukoencephalopathy syndrome (RPLS), a syndrome of subcortical vasogenic edema diagnosed by an MRI of the brain, has occurred in patients treated with CAPRELSA. Consider this syndrome in any patient presenting with seizures, headache, visual disturbances, confusion or altered mental function. In clinical studies, three of four patients who developed RPLS while taking CAPRELSA also had hypertension. Discontinue CAPRELSA treatment in patients with RPLS.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Vandetanib was not carcinogenic in a 2-year study in rats when administered by daily oral gavage at doses of up to 10 mg/kg (0.7 times the human Cmax at the 300 mg clinical dose), or in the Tg∙RasH2 mouse when administered by daily oral gavage at doses of up to 30 mg/kg (~5 times the human Cmax at the clinical dose of 300 mg) for 26 weeks. Vandetanib was not mutagenic in vitro in the bacterial reverse mutation (Ames) assay and was not clastogenic in either the in vitro cytogenetic assay using human lymphocytes or in the in vivo rat micronucleus assay.
Based on nonclinical findings, male and female fertility may be impaired by treatment with CAPRELSA. In a fertility study of male rats, vandetanib had no effect on copulation or fertility rate when untreated females were mated with males administered 1, 5, or 20 mg/kg/day of vandetanib (approximately 0.03, 0.22, or 0.40 times, respectively, the human exposure based on area under the curve (AUC) in patients with cancer at the 300 mg clinical dose); however, in the same study there was a slight decrease in the number of live embryos in females mated with males treated at the 20 mg/kg/day dose level and an increase in preimplantation loss in females mated with males administered vandetanib at doses of ≥5 mg/kg/day. In a female fertility study, there was a trend towards increased estrus cycle irregularity, a slight reduction in pregnancy incidence and an increase in implantation loss. In a one-month repeat-dose toxicity study in rats, there was a decrease in the number of corpora lutea in the ovaries of rats administered 75 mg/kg/day vandetanib (approximately 1.8 times the human exposure based on AUC at the 300 mg clinical dose).
14.1 Clinical Trial in Patients With Medullary Thyroid Cancer
A double-blind, placebo-controlled study (Study D4200C00058, NCT00410761) randomized patients with unresectable locally advanced or metastatic medullary thyroid cancer to CAPRELSA 300 mg (n=231) versus placebo (n=100).
The major efficacy outcome measure was progression-free survival (PFS) with CAPRELSA compared to placebo. Other efficacy outcome measures included evaluation of overall survival (OS) and overall objective response rate (ORR). Centralized, independent blinded review of the imaging data was used in the assessment of PFS and ORR. Upon objective disease progression based on the investigator's assessment, patients were discontinued from blinded study treatment and given the option to receive open-label CAPRELSA. Forty-seven percent (109/231) of the patients initially randomized to CAPRELSA opted to receive open-label CAPRELSA after disease progression, and 79% (79/100) of the patients initially randomized to placebo opted to receive open-label CAPRELSA after disease progression.
The result of the PFS analysis, based on the central review RECIST assessment, showed a statistically significant improvement in PFS for patients randomized to CAPRELSA (Hazard Ratio (HR) = 0.35; 95% Confidence Interval (CI) = 0.24-0.53; p<0.0001). Analyses in the subgroups of patients who were symptomatic or had progressed within 6 months prior to their enrollment showed similar PFS results (HR = 0.31 95% CI: 0.19, 0.53 for symptomatic patients; HR = 0.41 95% CI: 0.25, 0.66 for patients who had progressed within 6 months prior to enrollment). Median final OS were similar across both treatment arms.
The overall objective response rate (ORR) for patients randomized to CAPRELSA was 44% compared to 1% for patients randomized to placebo. All objective responses were partial responses.
| Figure 1: Kaplan-Meier Curves for Progression Free Survival in Study D4200C00058 |
| Vandetanib 300 mg (N=231) |
Placebo (N=100) |
|
|---|---|---|
| Progression Free Survival | ||
| Events (%) | 59 (26.0) | 41 (41.0) |
| Median, months | NR Not reached
|
16.4 |
| (95% CI) | (22.6, NE Not estimable ) |
(8.3, 19.7) |
| Hazard Ratio (95% CI) | 0.35 (0.24, 0.53) | |
| p-value | <0.0001 | |
| Overall Survival | ||
| Deaths (%) | 116 (50.2) | 52 (52.0) |
| Median, months | 81.6 | 80.4 |
| (95% CI) | (64.6, 98.5) | (52.5, NE) |
| Hazard Ratio (95% CI) | 0.99 (0.72, 1.38) | |
| p-value | 0.975 |
Warning: Qt Prolongation, Torsades De Pointes, and Sudden Death
CAPRELSA can prolong the QT interval. Torsades de pointes and sudden death have occurred in patients receiving CAPRELSA. Do not use CAPRELSA in patients with hypocalcemia, hypokalemia, hypomagnesemia, or long QT syndrome. Correct hypocalcemia, hypokalemia and/or hypomagnesemia prior to CAPRELSA administration. Monitor electrolytes periodically. Avoid drugs known to prolong the QT interval [see Warnings and Precautions (5.1)].
Structured Label Content
Section 42229-5 (42229-5)
For Adverse Reactions
The 300 mg daily dose can be reduced to 200 mg (two 100 mg tablets) and then to 100 mg for Common Terminology Criteria for Adverse Events (CTCAE) Grade 3 or greater toxicities.
Interrupt CAPRELSA for the following:
- Corrected QT interval, Fridericia (QTcF) greater than 500 ms: Resume at a reduced dose when the QTcF returns to less than 450 ms.
- CTCAE Grade 3 or greater toxicity: Resume at a reduced dose when the toxicity resolves or improves to CTCAE Grade 1.
For recurrent toxicities, reduce the dose of CAPRELSA to 100 mg after resolution or improvement to CTCAE Grade 1 severity, if continued treatment is warranted.
Adverse events including QT interval prolongation should be monitored closely as they may not resolve fully until approximately three plasma half-lives of the drug. Monitor appropriately [see Warnings and Precautions (5.1), (5.2), (5.3), (5.4), (5.5), (5.6), (5.7), and (5.9)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 1/2026 | |
|
MEDICATION GUIDE
CAPRELSA® (kap-rel-sah) (vandetanib) tablets |
||
| Read this Medication Guide before you start taking CAPRELSA and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment. | ||
|
What is the most important information I should know about CAPRELSA? CAPRELSA can cause a change in the electrical activity of your heart called QT prolongation, which can cause irregular heartbeats and that may lead to death. You should not take CAPRELSA if you have had a condition called long QT syndrome since birth. Your healthcare provider should perform tests to check the levels of your blood potassium, calcium, magnesium, and thyroid-stimulating hormone (TSH), as well as the electrical activity of your heart with a test called an electrocardiogram (ECG). You should have these tests:
Call your healthcare provider right away if you feel faint, light-headed, or feel your heart beating irregularly during treatment with CAPRELSA. These may be symptoms related to QT prolongation. See "What are the possible side effects of CAPRELSA?" for more information about side effects. |
||
|
What is CAPRELSA?
CAPRELSA is a prescription medicine used to treat medullary thyroid cancer that cannot be removed by surgery or that has spread to other parts of the body. It takes a long time to get rid of CAPRELSA from your body and you may be at risk for side effects related to CAPRELSA after you have stopped your treatment. It is not known if CAPRELSA is safe and effective in children. |
||
|
Who should not take CAPRELSA? Do not take CAPRELSA if you have QT prolongation. |
||
Before you take CAPRELSA, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
Do not take other medicines during treatment with CAPRELSA until you have talked with your healthcare provider or pharmacist. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
||
How should I take CAPRELSA?
|
||
What should I avoid while taking CAPRELSA?
|
||
|
What are the possible side effects of CAPRELSA? CAPRELSA may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
|
||
| The most common side effects of CAPRELSA include: | ||
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of CAPRELSA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store CAPRELSA?
|
||
|
General information about the safe and effective use of CAPRELSA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CAPRELSA for a condition for which it was not prescribed. Do not give CAPRELSA to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about CAPRELSA that is written for health professionals. |
||
|
What are the ingredients in CAPRELSA? Active ingredient: vandetanib Inactive ingredients:
CAPRELSA is a registered trademark of Genzyme Corporation. ©2026 Genzyme Corporation. For more information, go to www.caprelsa.com or call 1-800-633-1610. |
Section 43683-2 (43683-2)
| Warnings and Precautions (5.16) | 05/2025 |
5.7 Diarrhea
Diarrhea of Grade 3 or greater severity occurred in 11% of patients receiving CAPRELSA in the randomized MTC study. If diarrhea occurs, carefully monitor serum electrolytes and ECGs to reduce the risk and enable early detection of QT prolongation resulting from dehydration [see Warnings and Precautions (5.1)]. Interrupt CAPRELSA for severe diarrhea. Upon improvement, resume CAPRELSA at a reduced dose [see Dosage and Administration (2.1)].
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose, monitor patients closely for QTc prolongation. Adverse events including QT interval prolongation should be monitored closely as they may not resolve fully until approximately three plasma half-lives of the drug.
15 References (15 REFERENCES)
- OSHA Hazardous Drugs (OSHA Technical Manual). OSHA.
11 Description (11 DESCRIPTION)
Vandetanib has the chemical name N-(4-bromo-2-fluorophenyl)-6-methoxy-7-[(1-methylpiperidin-4-yl) methoxy]quinazolin-4-amine.
The structural and molecular formulas are:
C22H24BrFN4O2
Vandetanib has a molecular weight of 475.36 g/mol. Vandetanib exhibits pH-dependent solubility, with increased solubility at lower pH. Vandetanib is practically insoluble in water with a value of 0.008 mg/mL at 25°C (77°F).
CAPRELSA tablets for daily oral administration are available in two dosage strengths containing either 100 mg or 300 mg of vandetanib. The tablet cores contain the following inactive ingredients: calcium hydrogen phosphate dihydrate, crospovidone, magnesium stearate, microcrystalline cellulose, and povidone. The tablet film-coat contains the following inactive ingredients: hypromellose 2910, macrogol 300, and titanium dioxide E171.
5.5 Hemorrhage
Serious hemorrhagic events, including fatalities, occurred in patients treated with CAPRELSA. Do not administer CAPRELSA to patients with a recent history of hemoptysis of ≥1/2 teaspoon of red blood. Discontinue CAPRELSA in patients with severe hemorrhage.
5.9 Hypertension
Hypertension, including hypertensive crisis, has occurred in patients treated with CAPRELSA. Monitor all patients for hypertension. Dose reduction or interruption for hypertension may be necessary. If hypertension cannot be controlled, do not resume CAPRELSA [see Dosage and Administration (2.1)].
5.6 Heart Failure
Heart failure, including fatalities, occurred in patients treated with CAPRELSA. Monitor for signs and symptoms of heart failure. Consider discontinuation of CAPRELSA in patients with heart failure. Heart failure may not be reversible upon stopping CAPRELSA.
8.4 Pediatric Use
Safety and efficacy of CAPRELSA in pediatric patients have not been established.
8.5 Geriatric Use
The MTC study of CAPRELSA did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently compared to younger patients.
5.12 Renal Failure
Renal failure occurred in patients treated with CAPRELSA [see Adverse Reactions (6.1)]. Withhold, reduce the dose or permanently discontinue based on severity [see Dosage and Administration (2.1)].
Vandetanib exposure is increased in patients with impaired renal function. Reduce the starting dose to 200 mg in patients with moderate renal impairment and monitor the QT interval closely [see Dosage and Administration (2.1)]. Vandetanib is not recommended for use in patients with severe renal impairment (clearance below 30 mL/min). There is no information available for patients with end-stage renal disease requiring dialysis [see Boxed Warning , Dosage and Administration (2.1) , Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.16 Osteonecrosis
Osteonecrosis, including osteonecrosis of the jaw (ONJ), has occurred during treatment with CAPRELSA. Concomitant exposure to other risk factors, such as bisphosphonates, denosumab, dental disease or invasive dental procedures, may increase the risk of ONJ. Perform an oral examination prior to and periodically during treatment with CAPRELSA. Advise patients regarding good oral hygiene practices. Avoid invasive dental procedures while on CAPRELSA treatment, particularly in patients at higher risk. Withhold CAPRELSA for at least one month prior to scheduled dental surgery or invasive dental procedures. Permanently discontinue CAPRELSA if ONJ develops. [see Adverse Reactions (6.2)].
5.8 Hypothyroidism
In the randomized MTC study in which 90% of the patients enrolled had prior thyroidectomy, increased dosing of thyroid replacement therapy was required in 49% of CAPRELSA-treated patients compared to 17% of placebo-treated patients. Obtain Thyroid-stimulating hormone (TSH) at baseline, at 2 to 4 weeks and 8 to 12 weeks after starting treatment with CAPRELSA, and every 3 months thereafter. If signs or symptoms of hypothyroidism occur, examine thyroid hormone levels and adjust thyroid replacement therapy accordingly.
4 Contraindications (4 CONTRAINDICATIONS)
Do not use in patients with congenital long QT syndrome [see Boxed Warning].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed elsewhere in the label:
- QT Prolongation and Torsades de Pointes [see Boxed Warning, Warnings and Precautions (5.1)]
- Severe Skin Reactions [see Warnings and Precautions (5.2)]
- Interstitial Lung Disease [see Warnings and Precautions (5.3)]
- Ischemic Cerebrovascular Events [see Warnings and Precautions (5.4)]
- Hemorrhage [see Warnings and Precautions (5.5)]
- Heart Failure [see Warnings and Precautions (5.6)]
- Diarrhea [see Warnings and Precautions (5.7)]
- Hypothyroidism [see Warnings and Precautions (5.8)]
- Hypertension [see Warnings and Precautions (5.9)]
- Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.10)]
- Renal Failure [see Warnings and Precautions (5.12)]
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.15)]
- Osteonecrosis [see Warnings and Precautions (5.16)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.6 Renal Impairment
Vandetanib exposure is increased in patients with impaired renal function. Reduce the starting dose to 200 mg in patients with moderate (creatinine clearance ≥30 to <50 mL/min) renal impairment [see Dosage and Administration (2.1), Warnings and Precautions (5.12), and Clinical Pharmacology (12.3)].
Vandetanib is not recommended for use in patients with severe renal impairment (clearance below 30 mL/min) [see Warnings and Precautions (5.12)]. Patients with end-stage renal disease requiring dialysis were not studied [see Adverse Reactions (6.1)].
12.3 Pharmacokinetics
A population pharmacokinetic analysis of CAPRELSA was conducted in 231 patients with MTC following oral administration of 300 mg daily doses. The pharmacokinetics of CAPRELSA at the 300 mg dose in MTC patients are characterized by a mean clearance of approximately 13.2 L/h, a mean volume of distribution of approximately 7450 L, and a median plasma half-life of 19 days.
5.11 Drug Interactions
Avoid administration of CAPRELSA with anti-arrhythmic drugs (including but not limited to amiodarone, disopyramide, procainamide, sotalol, dofetilide) and other drugs that may prolong the QT interval (including but not limited to chloroquine, clarithromycin, dolasetron, granisetron, haloperidol, methadone, moxifloxacin, and pimozide) [see Drug Interactions (7.4) and Clinical Pharmacology (12.2)].
8.7 Hepatic Impairment
The pharmacokinetics of CAPRELSA were evaluated after a single dose of 800 mg in subjects with mild (n=8), moderate (n=7), and severe (n=6) hepatic impairment and normal hepatic function (n=5). Subjects with mild (Child-Pugh class A), moderate (Child-Pugh class B), and severe (Child-Pugh class C) hepatic impairment had comparable mean AUC and clearance values to those with normal hepatic function.
There are limited data in patients with liver impairment (serum bilirubin greater than 1.5 times the upper limit of normal). CAPRELSA is not recommended for use in patients with moderate and severe hepatic impairment, as safety and efficacy have not been established [see Dosage and Administration (2.1) and Warnings and Precautions (5.13)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
CAPRELSA is indicated for the treatment of symptomatic or progressive medullary thyroid cancer in patients with unresectable locally advanced or metastatic disease.
Use CAPRELSA in patients with indolent, asymptomatic or slowly progressing disease only after careful consideration of the treatment related risks of CAPRELSA.
5.13 Hepatic Impairment
CAPRELSA is not recommended for use in patients with moderate and severe hepatic impairment, as safety and efficacy have not been established [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
In vitro studies have shown that vandetanib inhibits the tyrosine kinase activity of the EGFR and VEGFR families, RET, BRK, TIE2, and members of the EPH receptor and Src kinase families. These receptor tyrosine kinases are involved in both normal cellular function and pathologic processes such as oncogenesis, metastasis, tumor angiogenesis, and maintenance of the tumor microenvironment. In addition, the N-desmethyl metabolite of the drug, representing 7 to 17.1% of vandetanib exposure, has similar inhibitory activity to the parent compound for VEGF receptors (KDR and Flt-1) and EGFR.
In vitro, vandetanib inhibited epidermal growth factor (EGF)-stimulated receptor tyrosine kinase phosphorylation in tumor cells and endothelial cells and VEGF-stimulated tyrosine kinase phosphorylation in endothelial cells.
In vivo, vandetanib administration reduced tumor cell-induced angiogenesis, tumor vessel permeability, and inhibited tumor growth and metastasis in mouse models of cancer.
16.1 Storage and Handling
CAPRELSA tablets should be stored at room temperature between 68°F and 77°F (20°C and 25°C); excursions permitted to 59°F–86°F (15°C-30°C) [See USP controlled room temperature].
Procedures for proper handling and disposal of anticancer drugs should be considered. A guideline on this subject has been published.1 Do not crush CAPRELSA tablets.
5.2 Severe Skin Reactions
Severe and sometimes fatal skin reactions, including toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome, have occurred in patients treated with CAPRELSA. Permanently discontinue CAPRELSA for severe skin reactions and refer the patient for urgent medical evaluation. Systemic therapies such as corticosteroids may be required.
Photosensitivity reactions can occur during CAPRELSA treatment and up to 4 months after treatment discontinuation.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Prolonged QT interval, torsades de pointes, and sudden death: Monitor electrocardiograms and levels of serum potassium, calcium, magnesium and TSH. Reduce CAPRELSA dose as appropriate. (2.1, 5.1)
- Severe skin reactions, including toxic epidermal necrolysis and Stevens-Johnson syndrome, some fatal. Discontinue CAPRELSA for severe skin reactions. (2.1, 5.2)
- Interstitial lung disease (ILD), including fatalities: investigate unexplained non-specific respiratory signs and symptoms. Discontinue CAPRELSA for confirmed ILD. (2.1, 5.3)
- Ischemic cerebrovascular events, hemorrhage, heart failure, diarrhea, hypertension, and reversible posterior leukoencephalopathy syndrome: Discontinue or interrupt CAPRELSA. (2.1, 5.4, 5.5, 5.6, 5.7, 5.9, 5.10)
- Impaired wound healing: Withhold for at least 1 month prior to elective surgery. Do not administer CAPRELSA for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment with CAPRELSA after resolution of wound healing complications has not been established. (5.14)
- Embryo-fetal toxicity: Can cause fetal harm. Advise women of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.15, 8.1)
- Osteonecrosis, including osteonecrosis of the jaw: Withhold CAPRELSA for 1 month prior to scheduled dental surgery and permanently discontinue if osteonecrosis occurs (5.16, 6.2).
5.15 Embryo Fetal Toxicity (5.15 Embryo-Fetal Toxicity)
Based on its mechanism of action and findings in animals, CAPRELSA can cause fetal harm when administered to a pregnant woman. In rats, vandetanib was embryotoxic, fetotoxic, and induced fetal malformations at exposures equivalent to or lower than those expected at the 300 mg clinical dose and had adverse effects on female fertility, embryofetal development, and postnatal development of pups.
Advise patients of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with CAPRELSA and for 4 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with CAPRELSA and for 4 months after the last dose [see Use in Specific Populations (8.1), (8.3)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dose of CAPRELSA is 300 mg taken orally once daily until disease progression or unacceptable toxicity occurs.
CAPRELSA may be taken with or without food.
Do not take a missed dose within 12 hours of the next dose.
Do not crush CAPRELSA tablets. The tablets can be dispersed in 2 ounces of water by stirring for approximately 10 minutes (will not completely dissolve). Do not use other liquids for dispersion. Swallow immediately after dispersion. Mix any remaining residue with 4 additional ounces of water and swallow.
The dispersion can also be administered through nasogastric or gastrostomy tubes.
5.14 Impaired Wound Healing
Impaired wound healing can occur in patients who receive drugs that inhibit the VEGF signaling pathway. Impaired wound healing has occurred in patients treated with CAPRELSA.
Withhold CAPRELSA for at least 1 month prior to elective surgery. Do not administer CAPRELSA for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment with CAPRELSA after resolution of wound healing complications has not been established.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
CAPRELSA 100 mg tablets are white, round, biconvex, film-coated, and intagliated with 'Z 100' on one side and plain on the reverse side.
CAPRELSA 300 mg tablets are white, oval, biconvex, film-coated, and intagliated with 'Z 300' on one side and plain on the reverse side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of CAPRELSA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Vascular disorders: Arterial (including aortic) aneurysms, dissections, and rupture
Musculoskeletal and connective tissue disorders: Osteonecrosis
General disorders: Impaired wound healing
5.3 Interstitial Lung Disease
Interstitial Lung Disease (ILD) or pneumonitis, including fatalities, has occurred in patients treated with CAPRELSA. Consider a diagnosis of ILD in patients presenting with non-specific respiratory signs and symptoms.
Interrupt CAPRELSA for acute or worsening pulmonary symptoms. Discontinue CAPRELSA if ILD is confirmed.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Patients with unresectable locally advanced or metastatic medullary thyroid cancer were treated with CAPRELSA 300 mg (n=231) or Placebo (n=99). The population exposed to CAPRELSA was 58% male, 94% white, and had a median age of 50 years. The data described below reflect a median exposure to CAPRELSA for 607 days.
The most commonly reported adverse drug reactions which occurred in >20% of CAPRELSA-treated patients and with a between-arm difference of ≥5% included, in order of decreasing frequency: diarrhea/colitis, rash, acneiform dermatitis, hypertension, nausea, headache, upper respiratory tract infection, decreased appetite, and abdominal pain.
Among CAPRELSA-treated patients, dose interruption occurred in 109 (47%) and dose reduction occurred in 83 (36%). Adverse reactions led to study treatment discontinuation in 28 of 231 patients (12%) receiving CAPRELSA and in 3 of 99 patients (3.0%) receiving placebo. Adverse reactions leading to permanent discontinuation in 2 or more (≥0.9%) patients treated with CAPRELSA were: asthenia (1.7%), rash (1.7%), diarrhea (0.9%), fatigue (0.9%), pyrexia (0.9%), elevated creatinine (0.9%), QT prolongation (0.9%), and hypertension (0.9%).
| System Organ Class | CAPRELSA 300 mg | Placebo | ||
|---|---|---|---|---|
| Preferred Term | N=231 | N=99 | ||
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| Gastrointestinal Disorders | ||||
| Diarrhea/Colitis | 57 | 11 | 27 | 2 |
| Nausea | 33 | 1 | 16 | 0 |
| Abdominal Pain Includes abdominal pain, abdominal pain upper, lower abdominal pain, and abdominal discomfort.
|
21 | 3 | 11 | 0 |
| Vomiting | 15 | 1 | 7 | 0 |
| Dyspepsia | 11 | 0 | 4 | 0 |
| Dry Mouth | 9 | 0 | 3 | 0 |
| Skin and Cutaneous Disorders | ||||
| Rash Includes rash, rash (erythematous, generalized, macular, maculopapular, papular, pruritic, and exfoliative), dermatitis, dermatitis bullous, generalized erythema, and eczema.
|
53 | 5 | 12 | 0 |
| Dermatitis Acneiform/Acne | 35 | 1 | 7 | 0 |
| Dry Skin | 15 | 0 | 5 | 0 |
| Photosensitivity Reaction | 13 | 2 | 0 | 0 |
| Pruritus | 11 | 1 | 4 | 0 |
| Nail abnormalities Includes nail disorder, nail bed inflammation, nail bed tenderness, paronychia, nail bed infection, and nail infection.
|
9 | 0 | 0 | 0 |
| Alopecia | 8 | N/A | 0 | N/A |
| Vascular Disorders | ||||
| Hypertension/Hypertensive Crisis/Accelerated Hypertension | 33 | 9 | 5 | 1 |
| Nervous System Disorders | ||||
| Headache | 26 | 1 | 9 | 0 |
| Dysgeusia | 8 | 0 | 3 | 0 |
| General Disorders | ||||
| Fatigue Included in Table 1 due to the increased incidence of severe fatigue in the CAPRELSA group compared to the placebo group.
|
24 | 6 | 23 | 1 |
| Infections | ||||
| Upper Respiratory Tract Infections Includes laryngitis, nasopharyngitis, pharyngitis, sinusitis, upper respiratory tract infection, acute sinusitis, rhinitis, and tracheitis.
|
23 | 0 | 16 | 0 |
| Metabolic and Nutritional Disorders | ||||
| Decreased Appetite | 21 | 4 | 12 | 0 |
| Hypocalcemia | 11 | 2 | 3 | 0 |
| Investigations | ||||
| ECG QT Prolonged 69% had QT prolongation >450 ms and 7% had QT prolongation >500 ms by ECG using Fridericia correction.
|
14 | 8 | 1 | 1 |
| Eye Disorders | ||||
| Corneal Abnormalities Includes corneal edema, corneal opacity, corneal dystrophy, corneal pigmentation, keratopathy, arcus lipoides, corneal deposits, and acquired corneal dystrophy.
|
13 | 0 | 1 | 0 |
| Blurred Vision | 9 | 0 | 1 | 0 |
| Renal Disorders | ||||
| Proteinuria | 10 | 0 | 2 | 0 |
| Psychiatric Disorders | ||||
| Depression | 10 | 2 | 3 | 0 |
| Endocrine Disorders | ||||
| Hypothyroidism | 6 | 0 | 0 | 0 |
| Musculoskeletal Disorders | ||||
| Muscle Spasms | 6 | 0 | 1 | 0 |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
7.3 Effect of Caprelsa On Digoxin (7.3 Effect of CAPRELSA on Digoxin)
CAPRELSA increased plasma concentrations of digoxin. Use caution and closely monitor for toxicities when administering CAPRELSA with digoxin [see Clinical Pharmacology (12.3)].
5.4 Ischemic Cerebrovascular Events
Ischemic cerebrovascular events, including fatalities, occurred in patients treated with CAPRELSA. In the randomized medullary thyroid cancer (MTC) study, ischemic cerebrovascular events occurred more frequently with CAPRELSA compared to placebo (1.3% compared to 0%). The safety of resumption of CAPRELSA therapy after resolution of an ischemic cerebrovascular event has not been studied. Discontinue CAPRELSA in patients who experience a severe ischemic cerebrovascular event.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
100 mg Tablets available in bottles containing 30 tablets (NDC 58468-7820-3).
300 mg Tablets available in bottles containing 30 tablets (NDC 58468-7840-3).
7.4 Drugs That Prolong the Qt Interval (7.4 Drugs that Prolong the QT Interval)
Avoid concomitant use of CAPRELSA with agents that may prolong the QT interval [see Warnings and Precautions (5.11)].
7.1 Effect of Cyp3a4 Inducers On Caprelsa (7.1 Effect of CYP3A4 Inducers on CAPRELSA)
Rifampicin, a strong CYP3A4 inducer, decreased vandetanib plasma concentrations. Avoid concomitant use of known strong CYP3A4 inducers during CAPRELSA therapy. Avoid concomitant use of St. John's wort because it can decrease vandetanib exposure unpredictably [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In an animal model of wound-healing, mice dosed with vandetanib had reduced skin-breaking strength compared with controls. This suggests that CAPRELSA slows but does not prevent wound healing. The appropriate interval between discontinuation of CAPRELSA and subsequent elective surgery required to avoid the risks of impaired wound healing has not been determined.
7.2 Effect of Caprelsa On Oct2 Transporter (7.2 Effect of CAPRELSA on OCT2 Transporter)
CAPRELSA increased plasma concentrations of metformin that is transported by the organic cation transporter type 2 (OCT2). Use caution and closely monitor for toxicities when administering CAPRELSA with drugs that are transported by OCT2 [see Clinical Pharmacology (12.3)].
5.1 Qt Prolongation and Torsades De Pointes (5.1 QT Prolongation and Torsades de Pointes)
CAPRELSA can prolong the QT interval in a concentration-dependent manner [see Clinical Pharmacology (12.2)]. Torsades de pointes, ventricular tachycardia and sudden deaths have occurred in patients treated with CAPRELSA.
Do not start CAPRELSA treatment in patients whose QTcF interval is greater than 450 ms. Do not administer CAPRELSA to patients who have a history of Torsades de pointes, congenital long QT syndrome, bradyarrhythmias or uncompensated heart failure. CAPRELSA has not been studied in patients with ventricular arrhythmias or recent myocardial infarction. Vandetanib exposure is increased in patients with impaired renal function. Reduce the starting dose to 200 mg in patients with moderate renal impairment and monitor QT interval frequently.
Obtain an ECG and serum potassium, calcium, magnesium and TSH at baseline, 2 to 4 weeks and 8 to 12 weeks after starting treatment with CAPRELSA, and every 3 months thereafter. Monitor electrolytes and ECGs more frequently in patients who experience diarrhea. Following any dose reduction for QT prolongation or any dose interruption greater than 2 weeks, conduct QT assessments as described above. Maintain serum potassium levels of 4 mEq/L or higher (within normal range) and maintain serum magnesium and calcium levels within normal ranges to reduce the risk of QT prolongation.
Avoid using CAPRELSA with drugs known to prolong the QT interval [see Warnings and Precautions (5.11) and Drug Interactions (7.4)]. If such drugs are given to patients already receiving CAPRELSA and no alternative therapy exists, perform ECG monitoring of the QT interval more frequently.
Stop CAPRELSA in patients who develop a QTcF greater than 500 ms until the QTcF returns to less than 450 ms. Dosing of CAPRELSA can then be resumed at a reduced dose [see Dosage and Administration (2.1)].
Principal Display Panel 100 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Carton)
NDC 58468-7820-3
Rx only
Caprelsa®
(vandetanib) tablets
100 mg
Dispense enclosed medication
guide to each patient.
30 Tablets
Principal Display Panel 300 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 300 mg Tablet Bottle Carton)
NDC 58468-7840-3
Rx only
Caprelsa®
(vandetanib) tablets
300 mg
Dispense enclosed medication
guide to each patient.
30 Tablets
5.10 Reversible Posterior Leukoencephalopathy Syndrome
Reversible posterior leukoencephalopathy syndrome (RPLS), a syndrome of subcortical vasogenic edema diagnosed by an MRI of the brain, has occurred in patients treated with CAPRELSA. Consider this syndrome in any patient presenting with seizures, headache, visual disturbances, confusion or altered mental function. In clinical studies, three of four patients who developed RPLS while taking CAPRELSA also had hypertension. Discontinue CAPRELSA treatment in patients with RPLS.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Vandetanib was not carcinogenic in a 2-year study in rats when administered by daily oral gavage at doses of up to 10 mg/kg (0.7 times the human Cmax at the 300 mg clinical dose), or in the Tg∙RasH2 mouse when administered by daily oral gavage at doses of up to 30 mg/kg (~5 times the human Cmax at the clinical dose of 300 mg) for 26 weeks. Vandetanib was not mutagenic in vitro in the bacterial reverse mutation (Ames) assay and was not clastogenic in either the in vitro cytogenetic assay using human lymphocytes or in the in vivo rat micronucleus assay.
Based on nonclinical findings, male and female fertility may be impaired by treatment with CAPRELSA. In a fertility study of male rats, vandetanib had no effect on copulation or fertility rate when untreated females were mated with males administered 1, 5, or 20 mg/kg/day of vandetanib (approximately 0.03, 0.22, or 0.40 times, respectively, the human exposure based on area under the curve (AUC) in patients with cancer at the 300 mg clinical dose); however, in the same study there was a slight decrease in the number of live embryos in females mated with males treated at the 20 mg/kg/day dose level and an increase in preimplantation loss in females mated with males administered vandetanib at doses of ≥5 mg/kg/day. In a female fertility study, there was a trend towards increased estrus cycle irregularity, a slight reduction in pregnancy incidence and an increase in implantation loss. In a one-month repeat-dose toxicity study in rats, there was a decrease in the number of corpora lutea in the ovaries of rats administered 75 mg/kg/day vandetanib (approximately 1.8 times the human exposure based on AUC at the 300 mg clinical dose).
14.1 Clinical Trial in Patients With Medullary Thyroid Cancer (14.1 Clinical Trial in Patients with Medullary Thyroid Cancer)
A double-blind, placebo-controlled study (Study D4200C00058, NCT00410761) randomized patients with unresectable locally advanced or metastatic medullary thyroid cancer to CAPRELSA 300 mg (n=231) versus placebo (n=100).
The major efficacy outcome measure was progression-free survival (PFS) with CAPRELSA compared to placebo. Other efficacy outcome measures included evaluation of overall survival (OS) and overall objective response rate (ORR). Centralized, independent blinded review of the imaging data was used in the assessment of PFS and ORR. Upon objective disease progression based on the investigator's assessment, patients were discontinued from blinded study treatment and given the option to receive open-label CAPRELSA. Forty-seven percent (109/231) of the patients initially randomized to CAPRELSA opted to receive open-label CAPRELSA after disease progression, and 79% (79/100) of the patients initially randomized to placebo opted to receive open-label CAPRELSA after disease progression.
The result of the PFS analysis, based on the central review RECIST assessment, showed a statistically significant improvement in PFS for patients randomized to CAPRELSA (Hazard Ratio (HR) = 0.35; 95% Confidence Interval (CI) = 0.24-0.53; p<0.0001). Analyses in the subgroups of patients who were symptomatic or had progressed within 6 months prior to their enrollment showed similar PFS results (HR = 0.31 95% CI: 0.19, 0.53 for symptomatic patients; HR = 0.41 95% CI: 0.25, 0.66 for patients who had progressed within 6 months prior to enrollment). Median final OS were similar across both treatment arms.
The overall objective response rate (ORR) for patients randomized to CAPRELSA was 44% compared to 1% for patients randomized to placebo. All objective responses were partial responses.
| Figure 1: Kaplan-Meier Curves for Progression Free Survival in Study D4200C00058 |
| Vandetanib 300 mg (N=231) |
Placebo (N=100) |
|
|---|---|---|
| Progression Free Survival | ||
| Events (%) | 59 (26.0) | 41 (41.0) |
| Median, months | NR Not reached
|
16.4 |
| (95% CI) | (22.6, NE Not estimable ) |
(8.3, 19.7) |
| Hazard Ratio (95% CI) | 0.35 (0.24, 0.53) | |
| p-value | <0.0001 | |
| Overall Survival | ||
| Deaths (%) | 116 (50.2) | 52 (52.0) |
| Median, months | 81.6 | 80.4 |
| (95% CI) | (64.6, 98.5) | (52.5, NE) |
| Hazard Ratio (95% CI) | 0.99 (0.72, 1.38) | |
| p-value | 0.975 |
Warning: Qt Prolongation, Torsades De Pointes, and Sudden Death (WARNING: QT PROLONGATION, TORSADES DE POINTES, AND SUDDEN DEATH)
CAPRELSA can prolong the QT interval. Torsades de pointes and sudden death have occurred in patients receiving CAPRELSA. Do not use CAPRELSA in patients with hypocalcemia, hypokalemia, hypomagnesemia, or long QT syndrome. Correct hypocalcemia, hypokalemia and/or hypomagnesemia prior to CAPRELSA administration. Monitor electrolytes periodically. Avoid drugs known to prolong the QT interval [see Warnings and Precautions (5.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:06.257720 · Updated: 2026-03-14T22:45:29.874304