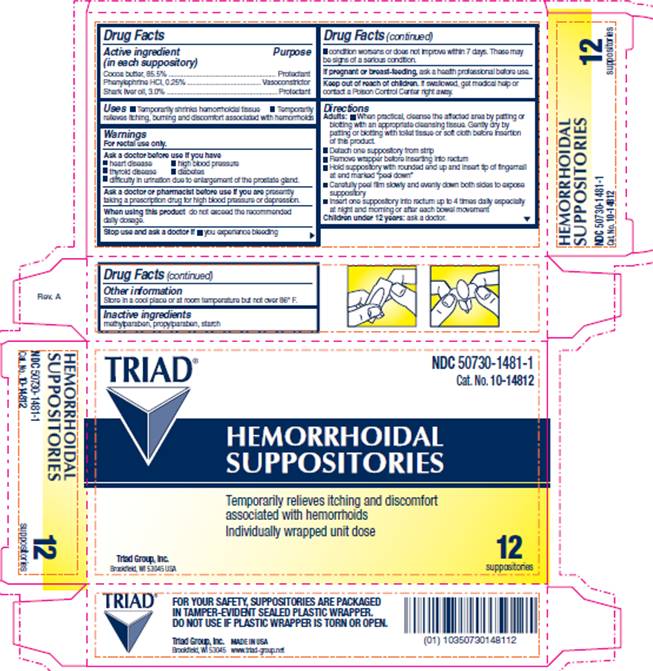
Drug Facts
e56eb547-9a1f-4f37-a0f6-95b3c1f27e65
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cocoa butter, 85.5% Phenylephrine HCI, 0.25% Shark liver oil, 3.0%
Purpose
protectant vasoconstrictor protectant
Medication Information
Warnings and Precautions
Purpose
protectant
vasoconstrictor
protectant
Description
Cocoa butter, 85.5% Phenylephrine HCI, 0.25% Shark liver oil, 3.0%
Uses
- temporarily shrinks hemorrhoidal tissue
- temporarily relieves itching burning and discomfort associated with hemorrhoids
Section 42229-5
Section 50570-1
Section 53413-1
Storage
Warnings
For rectal use only.
Directions
- When practical, cleanse the affected area by patting or blotting with an appropriate cleansing tissue. Gently dry by patting or blotting with toilet tissue or soft cloth before insertion of this product.
- Detach one suppository from strip
- Remove wrapper before inserting into rectum
- Hold suppository with rounded end up and insert tip of fingernail at end marked "peel down"
- Carefully peel film slowly and evenly down both sides to expose suppository
- Insert one suppository into rectum up to 4 times daily especially at night and morning or after each bowel movement
Active Ingredients
Cocoa butter, 85.5%
Phenylephrine HCI, 0.25%
Shark liver oil, 3.0%
Package Information
Cat. No. 10-14812
TRIAD®
HEMORRHOIDAL
SUPPOSITORIES
Temporarily relieves itching and discomfort associated with hemorrhoids
Individually wrapped unit dose
12 suppositories
Triad Group, Inc.
Brookfield, WI 53045 USA
FOR YOUR SAFETY, SUPPOSITORIES ARE PACKAGED IN TAMPER-EVIDENT SEALED PLASTIC WRAPPER.
DO NOT USE IF PLASTIC WRAPPER IS TORN OR OPEN.
Inactive Ingredients
When Using This Product
do not exceed the recommended daily dosage.
If Pregnant Or Breastfeeding
ask a health professional before use.
Stop Use and Ask A Doctor If
- you experience bleeding
- condition worsens or does not improve within 7 days.
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have
- heart disease
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
- high blood pressure
- diabetes
Ask A Doctor Or Pharmacist Before Use If
you are presently taking a prescription drug for high blood pressure or depression.
Structured Label Content
Uses (USES)
- temporarily shrinks hemorrhoidal tissue
- temporarily relieves itching burning and discomfort associated with hemorrhoids
Warnings and Precautions (34071-1)
Section 42229-5 (42229-5)
Section 50570-1 (50570-1)
Section 53413-1 (53413-1)
Purpose (PURPOSE)
protectant
vasoconstrictor
protectant
Storage (STORAGE)
Warnings (WARNINGS)
For rectal use only.
Directions (DIRECTIONS)
- When practical, cleanse the affected area by patting or blotting with an appropriate cleansing tissue. Gently dry by patting or blotting with toilet tissue or soft cloth before insertion of this product.
- Detach one suppository from strip
- Remove wrapper before inserting into rectum
- Hold suppository with rounded end up and insert tip of fingernail at end marked "peel down"
- Carefully peel film slowly and evenly down both sides to expose suppository
- Insert one suppository into rectum up to 4 times daily especially at night and morning or after each bowel movement
Active Ingredients (ACTIVE INGREDIENTS)
Cocoa butter, 85.5%
Phenylephrine HCI, 0.25%
Shark liver oil, 3.0%
Package Information (PACKAGE INFORMATION)
Cat. No. 10-14812
TRIAD®
HEMORRHOIDAL
SUPPOSITORIES
Temporarily relieves itching and discomfort associated with hemorrhoids
Individually wrapped unit dose
12 suppositories
Triad Group, Inc.
Brookfield, WI 53045 USA
FOR YOUR SAFETY, SUPPOSITORIES ARE PACKAGED IN TAMPER-EVIDENT SEALED PLASTIC WRAPPER.
DO NOT USE IF PLASTIC WRAPPER IS TORN OR OPEN.
Inactive Ingredients (INACTIVE INGREDIENTS)
When Using This Product (When using this product)
do not exceed the recommended daily dosage.
If Pregnant Or Breastfeeding (If pregnant or breastfeeding)
ask a health professional before use.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you experience bleeding
- condition worsens or does not improve within 7 days.
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- heart disease
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
- high blood pressure
- diabetes
Ask A Doctor Or Pharmacist Before Use If (Ask a doctor or pharmacist before use if)
you are presently taking a prescription drug for high blood pressure or depression.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:54:58.275970 · Updated: 2026-03-14T22:52:13.708968