Sunlenca
e5652804-29c4-40d7-aeb2-0142ed2a7b5b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
SUNLENCA, in combination with other antiretroviral(s), is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in heavily treatment-experienced adults with multidrug resistant HIV-1 whose current antiretroviral regimen is failing due to resistance, intolerance, or safety considerations.
Dosage and Administration
Recommended dosage – Initiation with one of two options followed by once every 6 months maintenance injection dosing. Tablets may be taken without regard to food. ( 2.2 ) Initiation Option 1 Day 1 927 mg by subcutaneous injection (2 × 1.5 mL injections) 600 mg orally (2 × 300 mg tablets) Day 2 600 mg orally (2 × 300 mg tablets) Initiation Option 2 Day 1 600 mg orally (2 × 300 mg tablets) Day 2 600 mg orally (2 × 300 mg tablets) Day 8 300 mg orally (1 × 300 mg tablet) Day 15 927 mg by subcutaneous injection (2 × 1.5 mL injections) Maintenance 927 mg by subcutaneous injection (2 × 1.5 mL injections) every 6 months (26 weeks) from the date of the last injection +/-2 weeks. Planned missed injections: If scheduled injection is to be missed by more than 2 weeks, SUNLENCA tablets may be used for oral bridging for up to 6 months until injections resume. Recommended dosage is 300 mg orally once every 7 days. ( 2.3 ) Unplanned missed injections: If more than 28 weeks since last injection and tablets have not been taken for oral bridging, restart initiation from Day 1 (using Option 1 or Option 2) if clinically appropriate. ( 2.3 ) SUNLENCA injection is for subcutaneous administration only. Two 1.5 mL injections are required for complete dose. ( 2.4 )
Contraindications
Concomitant administration of SUNLENCA with strong CYP3A inducers is contraindicated due to decreased lenacapavir plasma concentrations, which may result in the loss of therapeutic effect and development of resistance to SUNLENCA [see Drug Interactions (7.1) ] .
Warnings and Precautions
Immune reconstitution syndrome: May necessitate further evaluation and treatment. ( 5.1 ) Residual concentrations of lenacapavir may remain in systemic circulation for up to 12 months or longer. Counsel patients regarding the dosing schedule; non-adherence could lead to loss of virologic response and development of resistance. ( 5.2 ) May increase exposure and risk of adverse reactions to drugs primarily metabolized by CYP3A initiated within 9 months after the last subcutaneous dose of SUNLENCA. ( 5.2 ) If discontinued, initiate an alternative, fully suppressive antiretroviral regimen where possible no later than 28 weeks after the final injection of SUNLENCA. If virologic failure occurs, switch to an alternative regimen if possible. ( 5.2 ) Injection site reactions may occur, and nodules and indurations may be persistent. Improper administration (intradermal injection) has been associated with serious injection site reactions. ( 5.3 )
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Immune Reconstitution Syndrome [see Warnings and Precautions (5.1) ] Injection Site Reactions [see Warnings and Precautions (5.3) ].
Drug Interactions
Table 5 provides a listing of clinically significant drug interactions with recommended prevention or management strategies, but is not all inclusive. The drug interactions described are based on studies conducted with SUNLENCA or are drug interactions that may occur with SUNLENCA [see Contraindications (4) and Clinical Pharmacology (12.3) ] . Table 5 Drug Interactions with SUNLENCA Concomitant Drug Class: Drug Name Effect on Concentration ↑ = Increase, ↓ = Decrease. Clinical Comment Antiarrhythmics: digoxin ↑ digoxin Use with caution and monitor digoxin therapeutic concentration. Anticoagulants: Direct Oral Anticoagulants (DOACs) rivaroxaban dabigatran edoxaban ↑ DOAC Refer to the DOAC prescribing information for concomitant administration with moderate CYP3A inhibitors and/or P-gp inhibitors. Anticonvulsants: carbamazepine oxcarbazepine phenobarbital phenytoin ↓ lenacapavir Concomitant administration of carbamazepine, oxcarbazepine, phenobarbital, or phenytoin may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with carbamazepine or phenytoin is contraindicated. Concomitant administration of SUNLENCA with oxcarbazepine or phenobarbital is not recommended. Consider use of alternative anticonvulsants. Antiretroviral Agents: atazanavir/cobicistat Drug-drug interaction study was conducted. atazanavir/ritonavir ↑ lenacapavir (atazanavir/cobicistat, atazanavir/ritonavir) Concomitant administration of efavirenz, nevirapine, or tipranavir/ritonavir may result in loss of therapeutic effect and development of resistance. efavirenz nevirapine tipranavir/ritonavir ↓ lenacapavir (efavirenz, nevirapine, tipranavir/ritonavir) Concomitant administration with atazanavir/cobicistat, atazanavir/ritonavir, efavirenz, nevirapine, or tipranavir/ritonavir is not recommended. Antimycobacterials: rifabutin rifampin rifapentine ↓ lenacapavir Concomitant administration of rifabutin, rifampin and rifapentine may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with rifampin is contraindicated [see Contraindications (4) ] . Concomitant administration of SUNLENCA with rifabutin or rifapentine is not recommended. Corticosteroids (systemic): cortisone/hydrocortisone dexamethasone ↑ corticosteroids (systemic) Concomitant administration with systemic corticosteroids whose exposures are significantly increased by CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Initiate with the lowest starting dose and titrate carefully while monitoring for safety. ↓ lenacapavir (dexamethasone) Concomitant administration of systemic dexamethasone may result in loss of therapeutic effect of lenacapavir and development of resistance. Alternative corticosteroids to dexamethasone should be considered, particularly for long-term use. Ergot derivatives: dihydroergotamine ergotamine methylergonovine ↑ dihydroergotamine ↑ ergotamine ↑ methylergonovine Concomitant administration of SUNLENCA with dihydroergotamine, ergotamine or methylergonovine is not recommended. Herbal Products: St. John's wort The induction potency of St. John's wort may vary widely based on preparation. (Hypericum perforatum) ↓ lenacapavir Concomitant administration of St. John's wort may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with St. John's wort is contraindicated. HMG-CoA Reductase Inhibitors: lovastatin simvastatin ↑ lovastatin ↑ simvastatin Initiate lovastatin and simvastatin with the lowest starting dose and titrate carefully while monitoring for safety (e.g., myopathy). Narcotic analgesics metabolized by CYP3A: e.g., fentanyl, oxycodone ↑ fentanyl ↑ oxycodone Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. tramadol ↑ tramadol A decrease in dose may be needed for tramadol with concomitant use. Narcotic analgesic for treatment of opioid dependence: buprenorphine, methadone buprenorphine: effects unknown methadone: effects unknown Initiation of buprenorphine or methadone in patients taking SUNLENCA: Carefully titrate the dose of buprenorphine or methadone to the desired effect; use the lowest feasible initial or maintenance dose. Initiation of SUNLENCA in patients taking buprenorphine or methadone: A dose adjustment for buprenorphine or methadone may be needed. Monitor clinical signs and symptoms. Opioid Antagonist: naloxegol ↑ naloxegol Avoid use with SUNLENCA; if unavoidable, decrease the dosage of naloxegol and monitor for adverse reactions. Phosphodiesterase-5 (PDE-5) Inhibitors: sildenafil tadalafil vardenafil ↑ PDE-5 inhibitors Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Concomitant administration of SUNLENCA with tadalafil for the treatment of PAH is not recommended. Use of PDE-5 inhibitors for erectile dysfunction (ED): Refer to the prescribing information of PDE-5 inhibitors for dose recommendations. Sedatives/Hypnotics: midazolam (oral) triazolam ↑ midazolam (oral) ↑ triazolam Use with caution when midazolam or triazolam is concomitantly administered with SUNLENCA
Description
Dosage and Administration ( 2.1 , 2.3 , 2.4 ) 11/2024 Warnings and Precautions ( 5.3 ) 11/2024
Medication Information
Warnings and Precautions
Immune reconstitution syndrome: May necessitate further evaluation and treatment. ( 5.1 ) Residual concentrations of lenacapavir may remain in systemic circulation for up to 12 months or longer. Counsel patients regarding the dosing schedule; non-adherence could lead to loss of virologic response and development of resistance. ( 5.2 ) May increase exposure and risk of adverse reactions to drugs primarily metabolized by CYP3A initiated within 9 months after the last subcutaneous dose of SUNLENCA. ( 5.2 ) If discontinued, initiate an alternative, fully suppressive antiretroviral regimen where possible no later than 28 weeks after the final injection of SUNLENCA. If virologic failure occurs, switch to an alternative regimen if possible. ( 5.2 ) Injection site reactions may occur, and nodules and indurations may be persistent. Improper administration (intradermal injection) has been associated with serious injection site reactions. ( 5.3 )
Indications and Usage
SUNLENCA, in combination with other antiretroviral(s), is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in heavily treatment-experienced adults with multidrug resistant HIV-1 whose current antiretroviral regimen is failing due to resistance, intolerance, or safety considerations.
Dosage and Administration
Recommended dosage – Initiation with one of two options followed by once every 6 months maintenance injection dosing. Tablets may be taken without regard to food. ( 2.2 ) Initiation Option 1 Day 1 927 mg by subcutaneous injection (2 × 1.5 mL injections) 600 mg orally (2 × 300 mg tablets) Day 2 600 mg orally (2 × 300 mg tablets) Initiation Option 2 Day 1 600 mg orally (2 × 300 mg tablets) Day 2 600 mg orally (2 × 300 mg tablets) Day 8 300 mg orally (1 × 300 mg tablet) Day 15 927 mg by subcutaneous injection (2 × 1.5 mL injections) Maintenance 927 mg by subcutaneous injection (2 × 1.5 mL injections) every 6 months (26 weeks) from the date of the last injection +/-2 weeks. Planned missed injections: If scheduled injection is to be missed by more than 2 weeks, SUNLENCA tablets may be used for oral bridging for up to 6 months until injections resume. Recommended dosage is 300 mg orally once every 7 days. ( 2.3 ) Unplanned missed injections: If more than 28 weeks since last injection and tablets have not been taken for oral bridging, restart initiation from Day 1 (using Option 1 or Option 2) if clinically appropriate. ( 2.3 ) SUNLENCA injection is for subcutaneous administration only. Two 1.5 mL injections are required for complete dose. ( 2.4 )
Contraindications
Concomitant administration of SUNLENCA with strong CYP3A inducers is contraindicated due to decreased lenacapavir plasma concentrations, which may result in the loss of therapeutic effect and development of resistance to SUNLENCA [see Drug Interactions (7.1) ] .
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Immune Reconstitution Syndrome [see Warnings and Precautions (5.1) ] Injection Site Reactions [see Warnings and Precautions (5.3) ].
Drug Interactions
Table 5 provides a listing of clinically significant drug interactions with recommended prevention or management strategies, but is not all inclusive. The drug interactions described are based on studies conducted with SUNLENCA or are drug interactions that may occur with SUNLENCA [see Contraindications (4) and Clinical Pharmacology (12.3) ] . Table 5 Drug Interactions with SUNLENCA Concomitant Drug Class: Drug Name Effect on Concentration ↑ = Increase, ↓ = Decrease. Clinical Comment Antiarrhythmics: digoxin ↑ digoxin Use with caution and monitor digoxin therapeutic concentration. Anticoagulants: Direct Oral Anticoagulants (DOACs) rivaroxaban dabigatran edoxaban ↑ DOAC Refer to the DOAC prescribing information for concomitant administration with moderate CYP3A inhibitors and/or P-gp inhibitors. Anticonvulsants: carbamazepine oxcarbazepine phenobarbital phenytoin ↓ lenacapavir Concomitant administration of carbamazepine, oxcarbazepine, phenobarbital, or phenytoin may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with carbamazepine or phenytoin is contraindicated. Concomitant administration of SUNLENCA with oxcarbazepine or phenobarbital is not recommended. Consider use of alternative anticonvulsants. Antiretroviral Agents: atazanavir/cobicistat Drug-drug interaction study was conducted. atazanavir/ritonavir ↑ lenacapavir (atazanavir/cobicistat, atazanavir/ritonavir) Concomitant administration of efavirenz, nevirapine, or tipranavir/ritonavir may result in loss of therapeutic effect and development of resistance. efavirenz nevirapine tipranavir/ritonavir ↓ lenacapavir (efavirenz, nevirapine, tipranavir/ritonavir) Concomitant administration with atazanavir/cobicistat, atazanavir/ritonavir, efavirenz, nevirapine, or tipranavir/ritonavir is not recommended. Antimycobacterials: rifabutin rifampin rifapentine ↓ lenacapavir Concomitant administration of rifabutin, rifampin and rifapentine may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with rifampin is contraindicated [see Contraindications (4) ] . Concomitant administration of SUNLENCA with rifabutin or rifapentine is not recommended. Corticosteroids (systemic): cortisone/hydrocortisone dexamethasone ↑ corticosteroids (systemic) Concomitant administration with systemic corticosteroids whose exposures are significantly increased by CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Initiate with the lowest starting dose and titrate carefully while monitoring for safety. ↓ lenacapavir (dexamethasone) Concomitant administration of systemic dexamethasone may result in loss of therapeutic effect of lenacapavir and development of resistance. Alternative corticosteroids to dexamethasone should be considered, particularly for long-term use. Ergot derivatives: dihydroergotamine ergotamine methylergonovine ↑ dihydroergotamine ↑ ergotamine ↑ methylergonovine Concomitant administration of SUNLENCA with dihydroergotamine, ergotamine or methylergonovine is not recommended. Herbal Products: St. John's wort The induction potency of St. John's wort may vary widely based on preparation. (Hypericum perforatum) ↓ lenacapavir Concomitant administration of St. John's wort may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with St. John's wort is contraindicated. HMG-CoA Reductase Inhibitors: lovastatin simvastatin ↑ lovastatin ↑ simvastatin Initiate lovastatin and simvastatin with the lowest starting dose and titrate carefully while monitoring for safety (e.g., myopathy). Narcotic analgesics metabolized by CYP3A: e.g., fentanyl, oxycodone ↑ fentanyl ↑ oxycodone Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. tramadol ↑ tramadol A decrease in dose may be needed for tramadol with concomitant use. Narcotic analgesic for treatment of opioid dependence: buprenorphine, methadone buprenorphine: effects unknown methadone: effects unknown Initiation of buprenorphine or methadone in patients taking SUNLENCA: Carefully titrate the dose of buprenorphine or methadone to the desired effect; use the lowest feasible initial or maintenance dose. Initiation of SUNLENCA in patients taking buprenorphine or methadone: A dose adjustment for buprenorphine or methadone may be needed. Monitor clinical signs and symptoms. Opioid Antagonist: naloxegol ↑ naloxegol Avoid use with SUNLENCA; if unavoidable, decrease the dosage of naloxegol and monitor for adverse reactions. Phosphodiesterase-5 (PDE-5) Inhibitors: sildenafil tadalafil vardenafil ↑ PDE-5 inhibitors Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Concomitant administration of SUNLENCA with tadalafil for the treatment of PAH is not recommended. Use of PDE-5 inhibitors for erectile dysfunction (ED): Refer to the prescribing information of PDE-5 inhibitors for dose recommendations. Sedatives/Hypnotics: midazolam (oral) triazolam ↑ midazolam (oral) ↑ triazolam Use with caution when midazolam or triazolam is concomitantly administered with SUNLENCA
Description
Dosage and Administration ( 2.1 , 2.3 , 2.4 ) 11/2024 Warnings and Precautions ( 5.3 ) 11/2024
Section 42229-5
Planned Missed Injections
During the maintenance period, if a patient plans to miss a scheduled 6-month injection visit by more than 2 weeks, SUNLENCA tablets may be taken for up to 6 months until injections resume. Refer to Table 3 below for the recommended dosage after planned missed injections.
| Time since Last Injection | Recommendation |
|---|---|
| 26 to 28 weeks | Maintenance oral dosage of 300 mg taken once every 7 days for up to 6 months. Resume the maintenance injection dosage within 7 days after the last oral dose. |
Section 42230-3
| PATIENT INFORMATION | ||
|---|---|---|
| SUNLENCA® (sun-LEN-kuh) (lenacapavir) tablets |
SUNLENCA® (sun-LEN-kuh) (lenacapavir) injection |
|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2024 | |
| What is SUNLENCA? SUNLENCA is a prescription medicine that is used with other human immunodeficiency virus-1 (HIV-1) medicines to treat HIV-1 infection in adults:
It is not known if SUNLENCA is safe and effective in children. |
||
| Do not receive or take SUNLENCA if you also take certain other medicines called strong CYP3A inducers. Ask your healthcare provider if you are not sure. | ||
Before receiving or taking SUNLENCA, tell your healthcare provider about all your medical conditions, including if you:
|
||
How should I receive and take SUNLENCA?
|
||
| What are the possible side effects of SUNLENCA? SUNLENCA may cause serious side effects, including:
These are not all of the possible side effects of SUNLENCA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store SUNLENCA tablets?
|
||
| General information about the safe and effective use of SUNLENCA. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SUNLENCA for a condition for which it was not prescribed. Do not give SUNLENCA to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about SUNLENCA that is written for health professionals. |
||
| What are the ingredients in SUNLENCA? Active ingredient: lenacapavir Inactive ingredients: SUNLENCA tablets: copovidone, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, and poloxamer 407. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. SUNLENCA injection: polyethylene glycol 300 and water for injection. Manufactured and distributed by: Gilead Sciences, Inc. Foster City, CA 94404 SUNLENCA is a trademark of Gilead Sciences, Inc., or its related companies. All other trademarks referenced herein are the property of their respective owners. © 2024 Gilead Sciences, Inc. All rights reserved. 215973-GS-004/IFU-001/IFU-WD-000 For more information, call 1-800-445-3235 or go to www.SUNLENCA.com. |
Section 43683-2
Section 44425-7
Store bottle and blister packs at 20 °C – 25 °C (68 °F – 77 °F), excursions permitted to 15 °C – 30 °C (59 °F – 86 °F) (see USP Controlled Room Temperature).
Dispense and store only in original bottle or blister pack.
10 Overdosage
No data are available on overdose of SUNLENCA in patients. If overdose occurs, monitor the patient for evidence of toxicity. Treatment of overdose with SUNLENCA consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the patient. As lenacapavir is highly bound to plasma proteins, it is unlikely to be significantly removed by dialysis.
11 Description
SUNLENCA tablets and SUNLENCA injection contain lenacapavir sodium, a capsid inhibitor.
The chemical name of lenacapavir sodium is: Sodium (4-chloro-7-(2-((S)-1-(2-((3bS,4aR)-5,5-difluoro-3-(trifluoromethyl)-3b,4,4a,5-tetrahydro-1H-cyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl)acetamido)-2-(3,5-difluorophenyl)ethyl)-6-(3-methyl-3-(methylsulfonyl)but-1-yn-1-yl)pyridin-3-yl)-1-(2,2,2-trifluoroethyl)-1H-indazol-3-yl)(methylsulfonyl)amide.
Lenacapavir sodium has a molecular formula of C39H31ClF10N7NaO5S2, a molecular weight of 990.3, and the following structural formula:
Lenacapavir sodium is a light yellow to yellow solid and is practically insoluble in water.
8.4 Pediatric Use
The safety and effectiveness of SUNLENCA have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of SUNLENCA did not include sufficient numbers of participants aged 65 and over to determine whether they respond differently from younger patients.
14 Clinical Studies
The efficacy and safety of SUNLENCA in heavily treatment-experienced participants with multidrug resistant HIV-1 is based on 52-week data from CAPELLA, a randomized, placebo-controlled, double-blind, multicenter trial (NCT 04150068).
CAPELLA was conducted in 72 heavily treatment-experienced participants with multiclass resistant HIV-1. Participants were required to have a viral load ≥ 400 copies/mL, documented resistance to at least two antiretroviral medications from each of at least 3 of the 4 classes of antiretroviral medications (NRTI, NNRTI, PI and INSTI), and ≤ 2 fully active antiretroviral medications from the 4 classes of antiretroviral medications remaining at baseline due to resistance, intolerability, drug access, contraindication, or other safety concerns.
The trial was composed of two cohorts. Participants were enrolled into the randomized cohort (cohort 1, N=36) if they had a < 0.5 log10 HIV-1 RNA decline compared to the screening visit. Participants were enrolled into the non-randomized cohort (cohort 2, N=36) if they had a ≥ 0.5 log10 HIV-1 RNA decline compared to the screening visit or after cohort 1 reached its planned sample size.
In the 14-day functional monotherapy period, participants in cohort 1 were randomized in a 2:1 ratio in a blinded fashion to receive either SUNLENCA or placebo, while continuing their failing regimen. This period was to establish the virologic activity of SUNLENCA. After the functional monotherapy period, participants who had received SUNLENCA continued on SUNLENCA along with an optimized background regimen (OBR); participants who had received placebo during this period initiated SUNLENCA along with an OBR.
Participants in cohort 1 had a mean age of 52 years (range: 24 to 71), 72% were male, 46% were White, 46% were Black, and 9% were Asian. 29% percent of participants identified as Hispanic/Latino. The mean baseline plasma HIV-1 RNA was 4.3 log10 copies/mL (range: 2.3 to 5.4). 19% of participants had baseline viral loads greater than 100,000 copies/mL. The mean baseline CD4+ cell count was 161 cells/mm3 (range: 6 to 827). 75% of participants had CD4+ cell counts below 200 cells/mm3. The mean number of years since participants first started HIV treatment was 24 years (range: 7 to 33); the mean number of antiretroviral agents in failing regimens at baseline was 4 (range: 1 to 7). The percentage of participants in the randomized cohort with known resistance to at least 2 agents from the NRTI, NNRTI, PI and INSTI classes was 97%, 94%, 78% and 75%, respectively. In cohort 1, 53% of participants had no fully active agents, 31% had 1 fully active agent, and 17% had 2 or more fully active agents within their initial failing regimen, including 6% of participants were who were receiving fostemsavir, which was an investigational agent at the start of the CAPELLA trial.
Participants in cohort 2 initiated SUNLENCA and an OBR on Day 1.
Participants in cohort 2 had a mean age of 48 years (range: 23 to 78), 78% were male, 36% were White, 31% were Black, 33% were Asian, and 14% of participants identified as Hispanic/Latino. The mean baseline plasma HIV-1 RNA was 4.1 log10 copies/mL (range: 1.3 to 5.7). 19% of participants had baseline viral loads greater than 100,000 copies/mL. The mean baseline CD4+ cell count was 258 cells/mm3 (range: 3 to 1296). 53% of participants had CD4+ cell counts below 200 cells/mm3. The mean number of years since participants first started HIV treatment was 19 years (range: 3 to 35); the mean number of antiretroviral agents in failing regimens at baseline was 4 (range: 2 to 7). The percentage of participants in the non-randomized cohort with known resistance to at least 2 agents from the NRTI, NNRTI, PI and INSTI classes was 100%, 100%, 83% and 64%, respectively. In cohort 2, 31% of participants had no fully active agents, 42% had 1 fully active agent, and 28% had 2 or more fully active agents within their initial failing regimen, including 6% of participants who were receiving fostemsavir, which was an investigational agent at the start of the CAPELLA trial.
The primary efficacy endpoint was the proportion of participants in cohort 1 achieving ≥ 0.5 log10 copies/mL reduction from baseline in HIV-1 RNA at the end of the functional monotherapy period. The results of the primary endpoint analysis are shown in Table 10.
| SUNLENCA (N=24) |
Placebo (N=12) |
|
|---|---|---|
| Proportion of Participants Achieving a ≥ 0.5 log10 Decrease in Viral Load | 87.5% | 16.7% |
| Treatment Difference (95% CI) | 70.8% (34.9% to 90.0%) p < 0.0001
|
The results at Weeks 26 and 52 are provided in Table 11 and Table 12.
| SUNLENCA plus OBR (N=36) |
||
|---|---|---|
| Week 26 | Week 52 | |
| OBR = optimized background regimen | ||
| HIV-1 RNA < 50 copies/mL | 81% | 83% |
| HIV-1 RNA ≥ 50 copies/mL Includes participants who had ≥ 50 copies/mL in the Week 26 or 52 window; participant who discontinued early due to lack or loss of efficacy; participants who discontinued for reasons other than an adverse event (AE), death or lack or loss of efficacy and at the time of discontinuation had a viral value of ≥ 50 copies/mL.
|
19% | 14% |
| No virologic data in Week 26 or 52 Window | 0 | 3% |
| Discontinued Study Drug Due to AE or Death Includes participants who discontinued due to AE or death at any time point from Day 1 through the time window if this resulted in no virologic data on treatment during the specified window.
|
0 | 0 |
| Discontinued Study Drug Due to Other Reasons Includes participants who discontinued for reasons other than an AE, death or lack or loss of efficacy, e.g., withdrew consent, loss to follow-up, etc. and Last Available HIV-1 RNA < 50 copies/mL |
0 | 3% |
| Missing Data During Window but on Study Drug | 0 | 0 |
| SUNLENCA plus OBR (N=36) |
||
|---|---|---|
| Week 26 | Week 52 | |
| ARV = antiretroviral; DRV=darunavir; DTG=dolutegravir; INSTI = integrase strand-transfer inhibitor; OBR = optimized background regimen; | ||
| Age (Years) | ||
| < 50 | 100% (9/9) | 89% (8/9) |
| ≥ 50 | 74% (20/27) | 81% (22/27) |
| Gender | ||
| Male | 77% (20/26) | 77% (20/26) |
| Female | 90% (9/10) | 100% (10/10) |
| Race | ||
| Black | 81% (13/16) | 75% (12/16) |
| Non-Black | 84% (16/19) | 89% (17/19) |
| Baseline plasma viral load (copies/mL) | ||
| ≤ 100,000 | 86% (25/29) | 86% (25/29) |
| > 100,000 | 57% (4/7) | 71% (5/7) |
| Baseline CD4+ (cells/mm3) | ||
| < 200 | 78% (21/27) | 78% (21/27) |
| ≥ 200 | 89% (8/9) | 100% (9/9) |
| Baseline INSTI resistance profile | ||
| With INSTI resistance | 85% (23/27) | 81% (22/27) |
| Without INSTI resistance | 63% (5/8) | 88% (7/8) |
| Number of fully active ARV agents in the OBR | ||
| 0 | 67% (4/6) | 67% (4/6) |
| 1 | 86% (12/14) | 79% (11/14) |
| ≥ 2 | 81% (13/16) | 94% (15/16) |
| Use of DTG and/or DRV in the OBR | ||
| With DTG and DRV | 83% (10/12) | 83% (10/12) |
| With DTG, without DRV | 83% (5/6) | 83% (5/6) |
| Without DTG, with DRV | 78% (7/9) | 89% (8/9) |
| Without DTG or DRV | 78% (7/9) | 78% (7/9) |
In cohort 1, at Weeks 26 and 52, the mean change from baseline in CD4+ cell count was 81 cells/mm3 (range: -101 to 522) and 82 cells/mm3 (range: -194 to 467), respectively.
In cohort 2, at Weeks 26 and 52, 81% (29/36) and 72% (26/36) of participants achieved HIV-1 RNA < 50 copies/mL, respectively, and the mean change from baseline in CD4+ cell count was 97 cells/mm3 (range: -103 to 459) and 113 cells/mm3 (range: -124 to 405), respectively.
4 Contraindications
Concomitant administration of SUNLENCA with strong CYP3A inducers is contraindicated due to decreased lenacapavir plasma concentrations, which may result in the loss of therapeutic effect and development of resistance to SUNLENCA [see Drug Interactions (7.1)].
6 Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling:
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.1)]
- Injection Site Reactions [see Warnings and Precautions (5.3)].
7 Drug Interactions
8.6 Renal Impairment
No dosage adjustment of SUNLENCA is recommended in patients with mild, moderate or severe renal impairment (estimated creatinine clearance greater than or equal to 15 mL per minute). SUNLENCA has not been studied in patients with ESRD (estimated creatinine clearance less than 15 mL per minute) [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetic (PK) properties of lenacapavir are provided in Table 6 and Table 7. The estimated lenacapavir exposures are comparable between the two recommended dosing regimens.
| Oral | Subcutaneous | ||
|---|---|---|---|
| Absorption | |||
| % Absolute bioavailability | 6 to 10 | 100 Values reflect absolute bioavailability following subcutaneous administration of the 927 mg dose.
|
|
| Tmax Values reflect administration of lenacapavir with or without food.
|
4 hours | 77 to 84 days Due to slow release from the site of subcutaneous administration, the absorption profile of subcutaneously administered lenacapavir is complex.
|
|
| Effect of Food | |||
| Effect of low-fat meal (relative to fasting) Values refer to geometric mean ratio [low-fat meal/fasting] in PK parameters and (90% confidence interval). Low fat meal is approximately 400 kcal, 25% fat.
|
AUCinf ratio | 98.6 (58.2,167.2) | - |
| Cmax ratio | 115.8 (55.4, 242.1) | - | |
| Effect of high-fat meal (relative to fasting) Values refer to geometric mean ratio [high-fat meal/fasting] in PK parameters and (90% confidence interval). High fat meal is approximately 1000 kcal, 50% fat.
|
AUCinf ratio | 115.2 (72.0, 184.5) | - |
| Cmax ratio | 145.2 (77.9, 270.5) | - | |
| Distribution | |||
| Apparent volume of distribution (Vd/F, L) | 19240 | 9500 to 11700 | |
| % bound to human plasma proteins | >98.5 | ||
| Blood-to-plasma ratio | 0.5 to 0.7 Values reflect the blood-to-plasma ratio of lenacapavir following a single dose intravenous administration of [14C] lenacapavir through 336 hours postdose.
|
||
| Elimination | |||
| t1/2 | 10 to 12 days | 8 to 12 weeks | |
| Clearance (mean apparent clearance, L/h) | 55 | 4.2 | |
| % of dose of unchanged drug in plasma Dosing in mass balance studies: single dose intravenous administration of [14C] lenacapair to participants without HIV-1.
|
69 | ||
| Metabolism | |||
| Metabolic pathway(s) | CYP3A (minor) UGT1A1 (minor) |
||
| Excretion | |||
| Major routes of elimination | Excretion of unchanged drug into feces Metabolized via oxidation, N-dealkylation, hydrogenation, amide hydrolysis, glucuronidation, hexose conjugation, pentose conjugation, and glutathione conjugation; primarily via CYP3A and UGT1A1 and no single circulating metabolite accounted for >10% of plasma drug-related exposure.
|
||
| % of dose excreted in urine | <1 | ||
| % of dose excreted in feces (% unchanged) | 76 (33) |
| Parameter Mean (%CV) | Recommended Dosing Regimen, Option 1 Predicted exposures utilizing population PK analysis.
|
Recommended Dosing Regimen, Option 2 | |
|---|---|---|---|
| Day 1: 600 mg (oral) + 927 mg (SC) Day 2: 600 mg (oral) |
Days 1 and 2: 600 mg (oral), Day 8: 300 mg (oral), Day 15: 927 mg (SC) |
||
| Day 1 to end of Month 6 | Days 1 to 15 | Day 15 to end of Month 6 | |
| CV = coefficient of variation; NA = not applicable; SC = subcutaneous | |||
| Cmax
(ng/mL) |
101.4 (53.1) | 88.0 (72.4) | 86.5 (51.7) |
| AUCtau
(h∙ng/mL) |
242266 (46.0) | 19496 (72.6) | 239163 (47.2) |
| Ctrough
(ng/mL) |
32.5 (57.2) | 46.9 (72.3) | 32.5 (57.5) |
Lenacapavir exposures after subcutaneous administration were similar between heavily treatment experienced participants with HIV-1 and participants without HIV-1 based on population pharmacokinetics analysis. Lenacapavir exposures (AUCtau, Cmax and Ctrough) after oral administration were 28% to 43% higher in participants with HIV-1 who were heavily treatment experienced, compared to participants without HIV-1 based on population PK analysis. These differences were not considered clinically relevant.
2.2 Recommended Dosage
SUNLENCA can be initiated using one of the two recommended dosage regimens in Table 1 and Table 2 below. Maintenance dosing is administered by subcutaneous injection every 6 months regardless of the initiation regimen. Healthcare providers should determine the appropriate initiation regimen for the patient. SUNLENCA oral tablets may be taken with or without food [see Clinical Pharmacology (12.3)].
| Treatment Time | |
|---|---|
| Dosage of SUNLENCA: Initiation | |
| Day 1 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Dosage of SUNLENCA: Maintenance | |
| Every 6 months (26 weeks) From the date of the last injection. +/-2 weeks |
927 mg by subcutaneous injection (2 × 1.5 mL injections) |
| Treatment Time | |
|---|---|
| Dosage of SUNLENCA: Initiation | |
| Day 1 | 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Day 8 | 300 mg orally (1 × 300 mg tablet) |
| Day 15 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) |
| Dosage of SUNLENCA: Maintenance | |
| Every 6 months (26 weeks) From the date of the last injection. +/-2 weeks |
927 mg by subcutaneous injection (2 × 1.5 mL injections) |
8.7 Hepatic Impairment
No dosage adjustment of SUNLENCA is recommended in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. SUNLENCA has not been studied in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)].
1 Indications and Usage
SUNLENCA, in combination with other antiretroviral(s), is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in heavily treatment-experienced adults with multidrug resistant HIV-1 whose current antiretroviral regimen is failing due to resistance, intolerance, or safety considerations.
12.1 Mechanism of Action
SUNLENCA is an HIV-1 antiretroviral agent [see Microbiology (12.4)].
5 Warnings and Precautions
- Immune reconstitution syndrome: May necessitate further evaluation and treatment. (5.1)
- Residual concentrations of lenacapavir may remain in systemic circulation for up to 12 months or longer. Counsel patients regarding the dosing schedule; non-adherence could lead to loss of virologic response and development of resistance. (5.2)
- May increase exposure and risk of adverse reactions to drugs primarily metabolized by CYP3A initiated within 9 months after the last subcutaneous dose of SUNLENCA. (5.2)
- If discontinued, initiate an alternative, fully suppressive antiretroviral regimen where possible no later than 28 weeks after the final injection of SUNLENCA. If virologic failure occurs, switch to an alternative regimen if possible. (5.2)
- Injection site reactions may occur, and nodules and indurations may be persistent. Improper administration (intradermal injection) has been associated with serious injection site reactions. (5.3)
2 Dosage and Administration
- Recommended dosage – Initiation with one of two options followed by once every 6 months maintenance injection dosing. Tablets may be taken without regard to food. (2.2)
| Initiation Option 1 | |
| Day 1 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Initiation Option 2 | |
| Day 1 | 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Day 8 | 300 mg orally (1 × 300 mg tablet) |
| Day 15 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) |
| Maintenance | |
| 927 mg by subcutaneous injection (2 × 1.5 mL injections) every 6 months (26 weeks) from the date of the last injection +/-2 weeks. |
- Planned missed injections: If scheduled injection is to be missed by more than 2 weeks, SUNLENCA tablets may be used for oral bridging for up to 6 months until injections resume. Recommended dosage is 300 mg orally once every 7 days. (2.3)
- Unplanned missed injections: If more than 28 weeks since last injection and tablets have not been taken for oral bridging, restart initiation from Day 1 (using Option 1 or Option 2) if clinically appropriate. (2.3)
- SUNLENCA injection is for subcutaneous administration only. Two 1.5 mL injections are required for complete dose. (2.4)
3 Dosage Forms and Strengths
Tablets: 300 mg
Injection: 463.5 mg/1.5 mL (309 mg/mL) in single-dose vials. (3)
5.3 Injection Site Reactions
Administration of SUNLENCA may result in local injection site reactions (ISRs). If clinically significant ISRs occur, evaluate and institute appropriate therapy and follow-up.
Manifestations of ISRs may include swelling, pain, erythema, nodule, induration, pruritus, extravasation or mass. Nodules and indurations at the injection site may take longer to resolve than other ISRs. In clinical studies, after a median follow-up of 553 days, 30% of nodules and 13% of indurations (in 10% and 1% of participants, respectively) associated with the first injections of SUNLENCA had not fully resolved. Measurements and qualitative assessments of ISRs were not routinely reported. Where described, the majority of the injection site nodules and indurations were palpable but not visible, and had a maximum size of approximately 1 to 4 cm [see Adverse Reactions (6.1)].
The mechanism driving the persistence of injection site nodules and indurations in some patients is not fully understood, but based on available data, they may be related to the presence of the subcutaneous drug depot. In some patients who had a skin biopsy performed of an injection site nodule or induration, dermatopathology revealed foreign body inflammation or granulomatous response.
Improper administration (intradermal injection) has been associated with serious injection site reactions, including necrosis and ulcer [see Adverse Reactions (6)]. Ensure SUNLENCA is only administered subcutaneously in the abdomen [see Dosage and Administration (2.4)].
6.2 Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the following adverse reactions have been identified during postmarketing use. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The primary safety assessment of SUNLENCA was based on data from heavily treatment-experienced adult participants with HIV who received SUNLENCA in a Phase 2/3 trial (CAPELLA; N=72) through Week 52 (median duration on study of 71 weeks) [see Clinical Studies (14)], as well as supportive data in treatment-naïve adult participants with HIV who received SUNLENCA in a Phase 2 trial (CALIBRATE; N=157) through Week 54 (median duration of exposure of 66 weeks).
The most common adverse reactions (all Grades) reported in at least 3% of participants in CAPELLA were nausea and injection site reactions. The proportion of partcipants in CAPELLA who discontinued treatment with SUNLENCA due to adverse events, regardless of severity, was 1% (Grade 1 injection site nodule in 1 participant). Table 3 displays the frequency of adverse reactions (all Grades) greater than or equal to 3% in the SUNLENCA group.
| Adverse Reactions | SUNLENCA + Background Regimen (N=72) |
|---|---|
| Injection Site Reactions | 65% |
| Nausea | 4% |
The majority (96%) of all adverse reactions associated with SUNLENCA were mild or moderate in severity.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Adherence to Treatment Regimen
Prior to starting SUNLENCA, healthcare providers should carefully select patients who agree to the required every 6 month injection dosing schedule and counsel patients about the importance of adherence to scheduled SUNLENCA dosing visits and concomitant oral antiretroviral therapy to help maintain viral suppression and reduce the risk of viral rebound and potential development of resistance with missed doses [see Warnings and Precautions (5.2), Microbiology (12.4)].
5.1 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections [such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia (PCP), or tuberculosis], which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
Principal Display Panel Kit Carton
Rx only
NDC 61958-3002-1
Sunlenca®
(lenacapavir) injection
463.5 mg/1.5 mL (309 mg/mL)
For Subcutaneous Injection
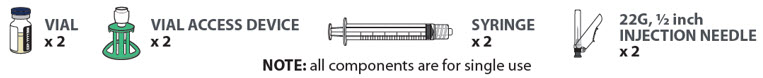
Contents
- 2 x 1.5 mL lenacapavir single-dose vials
- 2 vial access devices
- 2 syringes
- 2 injection needles (22 gauge, 1/2 inch)
- Prescribing Information
- Instructions for Use
- Patient Information
Both 463.5 mg/1.5 mL (2 single-dose vials) must be
administered to receive the 927 mg dose.
For Healthcare Professional administration only.
GILEAD
7.1 Effect of Other Drugs On Sunlenca
Lenacapavir is a substrate of P-gp, UGT1A1, and CYP3A.
7.2 Effect of Sunlenca On Other Drugs
Lenacapavir is a moderate inhibitor of CYP3A. Due to the long half-life of lenacapavir following subcutaneous administration, SUNLENCA may increase the exposure of drugs primarily metabolized by CYP3A [see Clinical Pharmacology (12.3)] initiated within 9 months after the last subcutaneous dose of SUNLENCA, which may increase the potential risk of adverse reactions. See the prescribing information of the sensitive CYP3A substrate for dosing recommendations with moderate inhibitors of CYP3A.
Principal Display Panel Kit Carton 3005
Rx only
NDC 61958-3005-1
Sunlenca®
(lenacapavir) injection
463.5 mg/1.5 mL (309 mg/mL)
For Subcutaneous Injection
Contents
- 2 x 1.5 mL lenacapavir single-dose vials
- 2 withdrawal needles (18 gauge, 1½ inch)
- 2 syringes
- 2 injection needles (22 gauge, ½ inch)
- Prescribing Information
- Instructions for Use
- Patient Information
Both 463.5 mg/1.5 mL (2 single-dose vials) must be
administered to receive the 927 mg dose.
For Healthcare Professional administration only.
GILEAD
Principal Display Panel 4 Tablet Bottle Label
NDC 61958-3001-3
4 tablets
Sunlenca®
(lenacapavir) tablets
300 mg per tablet
Talk to your healthcare provider
before taking Sunlenca tablets.
Your healthcare provider will tell you
when to take Sunlenca tablets.
2.4 Preparation and Administration of Subcutaneous Injection
SUNLENCA injection is only for subcutaneous administration into the abdomen by a healthcare provider. Do NOT administer intradermally due to risk of serious injection site reactions [see Warnings and Precautions (5.3)].
Use aseptic technique. Visually inspect the solution in the vials and prepared syringe for particulate matter and discoloration prior to administration. SUNLENCA injection is a yellow solution. Do not use SUNLENCA injection if the solution is discolored or if it contains particulate matter. Once the solution is withdrawn from the vials, the subcutaneous injections should be administered as soon as possible [see How Supplied/Storage and Handling (16)].
There are two available injection kits, which differ only in how SUNLENCA injection is prepared (the components and associated method for withdrawal of the solution from the vials) [see How Supplied/Storage and Handling (16)]. Refer to the figures below for the relevant injection kit.
The injection kit components are for single use only. Two 1.5 mL injections are required for a complete dose.
Principal Display Panel 5 Tablet Blister Pack Pouch Carton
NDC 61958-3001-2
Sunlenca®
(lenacapavir) tablets
300 mg per tablet
1 pouch containing 5 tablets
Each tablet contains: 300 mg of lenacapavir
(present as 306.8 mg lenacapavir sodium).
Store at 20 °C - 25 °C (68 °F - 77 °F).
Dispense only in original container.
Recommended Dosage: See prescribing information.
KEEP OUT OF THE REACH OF CHILDREN
Talk to your healthcare provider before taking
Sunlenca tablets.
Your healthcare provider will tell you when to take
Sunlenca tablets.
GILEAD
Manufactured for:
Gilead Sciences, Inc.
Foster City, CA 94404
Made in Canada
© 2022 Gilead Sciences, Inc.
Principal Display Panel 300 Mg Tablet Blister Pack Pouch Carton
NDC 61958-3001-1
Sunlenca®
(lenacapavir) tablets
300 mg per tablet
1 pouch containing 4 tablets
Each tablet contains: 300 mg of lenacapavir
(present as 306.8 mg lenacapavir sodium).
Store at 20 °C - 25 °C (68 °F - 77 °F).
Dispense only in original container.
Recommended Dosage: See prescribing information.
KEEP OUT OF THE REACH OF CHILDREN
GILEAD
Manufactured for:
Gilead Sciences, Inc.
Foster City, CA 94404
Made in Canada
© 2022 Gilead Sciences, Inc.
7.3 Established and Other Potentially Significant Drug Interactions
Table 5 provides a listing of clinically significant drug interactions with recommended prevention or management strategies, but is not all inclusive. The drug interactions described are based on studies conducted with SUNLENCA or are drug interactions that may occur with SUNLENCA [see Contraindications (4) and Clinical Pharmacology (12.3)].
| Concomitant Drug Class: Drug Name |
Effect on Concentration ↑ = Increase, ↓ = Decrease.
|
Clinical Comment |
|---|---|---|
|
Antiarrhythmics:
digoxin |
↑ digoxin | Use with caution and monitor digoxin therapeutic concentration. |
|
Anticoagulants:
Direct Oral Anticoagulants (DOACs) rivaroxaban dabigatran edoxaban |
↑ DOAC | Refer to the DOAC prescribing information for concomitant administration with moderate CYP3A inhibitors and/or P-gp inhibitors. |
|
Anticonvulsants:
carbamazepine oxcarbazepine phenobarbital phenytoin |
↓ lenacapavir | Concomitant administration of carbamazepine, oxcarbazepine, phenobarbital, or phenytoin may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with carbamazepine or phenytoin is contraindicated. Concomitant administration of SUNLENCA with oxcarbazepine or phenobarbital is not recommended. Consider use of alternative anticonvulsants. |
|
Antiretroviral Agents:
atazanavir/cobicistat Drug-drug interaction study was conducted.
atazanavir/ritonavir |
↑ lenacapavir (atazanavir/cobicistat, atazanavir/ritonavir) | Concomitant administration of efavirenz, nevirapine, or tipranavir/ritonavir may result in loss of therapeutic effect and development of resistance. |
| efavirenz
nevirapine tipranavir/ritonavir |
↓ lenacapavir (efavirenz, nevirapine, tipranavir/ritonavir) | Concomitant administration with atazanavir/cobicistat, atazanavir/ritonavir, efavirenz, nevirapine, or tipranavir/ritonavir is not recommended. |
|
Antimycobacterials:
rifabutin rifampin rifapentine |
↓ lenacapavir | Concomitant administration of rifabutin, rifampin and rifapentine may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with rifampin is contraindicated [see Contraindications (4)]. Concomitant administration of SUNLENCA with rifabutin or rifapentine is not recommended. |
|
Corticosteroids (systemic):
cortisone/hydrocortisone dexamethasone |
↑ corticosteroids (systemic) | Concomitant administration with systemic corticosteroids whose exposures are significantly increased by CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Initiate with the lowest starting dose and titrate carefully while monitoring for safety. |
| ↓ lenacapavir (dexamethasone) | Concomitant administration of systemic dexamethasone may result in loss of therapeutic effect of lenacapavir and development of resistance. Alternative corticosteroids to dexamethasone should be considered, particularly for long-term use. | |
|
Ergot derivatives:
dihydroergotamine ergotamine methylergonovine |
↑ dihydroergotamine ↑ ergotamine ↑ methylergonovine |
Concomitant administration of SUNLENCA with dihydroergotamine, ergotamine or methylergonovine is not recommended. |
|
Herbal Products:
St. John's wort The induction potency of St. John's wort may vary widely based on preparation.
(Hypericum perforatum) |
↓ lenacapavir | Concomitant administration of St. John's wort may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with St. John's wort is contraindicated. |
|
HMG-CoA Reductase Inhibitors:
lovastatin simvastatin |
↑ lovastatin ↑ simvastatin |
Initiate lovastatin and simvastatin with the lowest starting dose and titrate carefully while monitoring for safety (e.g., myopathy). |
|
Narcotic analgesics metabolized by CYP3A:
e.g., fentanyl, oxycodone |
↑ fentanyl ↑ oxycodone |
Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. |
| tramadol | ↑ tramadol | A decrease in dose may be needed for tramadol with concomitant use. |
|
Narcotic analgesic for treatment of opioid dependence:
buprenorphine, methadone |
buprenorphine: effects unknown methadone: effects unknown |
Initiation of buprenorphine or methadone in patients taking SUNLENCA: Carefully titrate the dose of buprenorphine or methadone to the desired effect; use the lowest feasible initial or maintenance dose. Initiation of SUNLENCA in patients taking buprenorphine or methadone: A dose adjustment for buprenorphine or methadone may be needed. Monitor clinical signs and symptoms. |
|
Opioid Antagonist:
naloxegol |
↑ naloxegol | Avoid use with SUNLENCA; if unavoidable, decrease the dosage of naloxegol and monitor for adverse reactions. |
|
Phosphodiesterase-5 (PDE-5) Inhibitors:
sildenafil tadalafil vardenafil |
↑ PDE-5 inhibitors | Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Concomitant administration of SUNLENCA with tadalafil for the treatment of PAH is not recommended. Use of PDE-5 inhibitors for erectile dysfunction (ED): Refer to the prescribing information of PDE-5 inhibitors for dose recommendations. |
|
Sedatives/Hypnotics:
midazolam (oral) triazolam |
↑ midazolam (oral) ↑ triazolam |
Use with caution when midazolam or triazolam is concomitantly administered with SUNLENCA |
7.4 Drugs Without Clinically Significant Interactions With Sunlenca
Based on drug interaction studies conducted with SUNLENCA, no clinically significant drug interactions have been observed with: darunavir/cobicistat, cobicistat, famotidine, pitavastatin, rosuvastatin, tenofovir alafenamide, and voriconazole.
5.2 Long Acting Properties and Potential Associated Risks With Sunlenca
Residual concentrations of lenacapavir may remain in the systemic circulation of patients for prolonged periods (up to 12 months or longer after the last subcutaneous dose). It is important to counsel patients that maintenance dosing by injection is required every 6 months, because missed doses or non-adherence to injections could lead to loss of virologic response and development of resistance [see Dosage and Administration (2.1)].
Lenacapavir, a moderate CYP3A inhibitor, may increase the exposure to, and therefore potential risk of adverse reactions from, drugs primarily metabolized by CYP3A initiated within 9 months after the last subcutaneous dose of SUNLENCA [see Drug Interactions and Clinical Pharmacology (7.2, 12.3)].
If SUNLENCA is discontinued, to minimize the potential risk of developing viral resistance, it is essential to initiate an alternative, fully suppressive antiretroviral regimen where possible no later than 28 weeks after the final injection of SUNLENCA. If virologic failure occurs during treatment, switch the patient to an alternative regimen if possible [see Dosage and Administration (2.1)].
Structured Label Content
Section 42229-5 (42229-5)
Planned Missed Injections
During the maintenance period, if a patient plans to miss a scheduled 6-month injection visit by more than 2 weeks, SUNLENCA tablets may be taken for up to 6 months until injections resume. Refer to Table 3 below for the recommended dosage after planned missed injections.
| Time since Last Injection | Recommendation |
|---|---|
| 26 to 28 weeks | Maintenance oral dosage of 300 mg taken once every 7 days for up to 6 months. Resume the maintenance injection dosage within 7 days after the last oral dose. |
Section 42230-3 (42230-3)
| PATIENT INFORMATION | ||
|---|---|---|
| SUNLENCA® (sun-LEN-kuh) (lenacapavir) tablets |
SUNLENCA® (sun-LEN-kuh) (lenacapavir) injection |
|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2024 | |
| What is SUNLENCA? SUNLENCA is a prescription medicine that is used with other human immunodeficiency virus-1 (HIV-1) medicines to treat HIV-1 infection in adults:
It is not known if SUNLENCA is safe and effective in children. |
||
| Do not receive or take SUNLENCA if you also take certain other medicines called strong CYP3A inducers. Ask your healthcare provider if you are not sure. | ||
Before receiving or taking SUNLENCA, tell your healthcare provider about all your medical conditions, including if you:
|
||
How should I receive and take SUNLENCA?
|
||
| What are the possible side effects of SUNLENCA? SUNLENCA may cause serious side effects, including:
These are not all of the possible side effects of SUNLENCA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store SUNLENCA tablets?
|
||
| General information about the safe and effective use of SUNLENCA. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SUNLENCA for a condition for which it was not prescribed. Do not give SUNLENCA to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about SUNLENCA that is written for health professionals. |
||
| What are the ingredients in SUNLENCA? Active ingredient: lenacapavir Inactive ingredients: SUNLENCA tablets: copovidone, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, and poloxamer 407. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. SUNLENCA injection: polyethylene glycol 300 and water for injection. Manufactured and distributed by: Gilead Sciences, Inc. Foster City, CA 94404 SUNLENCA is a trademark of Gilead Sciences, Inc., or its related companies. All other trademarks referenced herein are the property of their respective owners. © 2024 Gilead Sciences, Inc. All rights reserved. 215973-GS-004/IFU-001/IFU-WD-000 For more information, call 1-800-445-3235 or go to www.SUNLENCA.com. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store bottle and blister packs at 20 °C – 25 °C (68 °F – 77 °F), excursions permitted to 15 °C – 30 °C (59 °F – 86 °F) (see USP Controlled Room Temperature).
Dispense and store only in original bottle or blister pack.
10 Overdosage (10 OVERDOSAGE)
No data are available on overdose of SUNLENCA in patients. If overdose occurs, monitor the patient for evidence of toxicity. Treatment of overdose with SUNLENCA consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the patient. As lenacapavir is highly bound to plasma proteins, it is unlikely to be significantly removed by dialysis.
11 Description (11 DESCRIPTION)
SUNLENCA tablets and SUNLENCA injection contain lenacapavir sodium, a capsid inhibitor.
The chemical name of lenacapavir sodium is: Sodium (4-chloro-7-(2-((S)-1-(2-((3bS,4aR)-5,5-difluoro-3-(trifluoromethyl)-3b,4,4a,5-tetrahydro-1H-cyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl)acetamido)-2-(3,5-difluorophenyl)ethyl)-6-(3-methyl-3-(methylsulfonyl)but-1-yn-1-yl)pyridin-3-yl)-1-(2,2,2-trifluoroethyl)-1H-indazol-3-yl)(methylsulfonyl)amide.
Lenacapavir sodium has a molecular formula of C39H31ClF10N7NaO5S2, a molecular weight of 990.3, and the following structural formula:
Lenacapavir sodium is a light yellow to yellow solid and is practically insoluble in water.
8.4 Pediatric Use
The safety and effectiveness of SUNLENCA have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of SUNLENCA did not include sufficient numbers of participants aged 65 and over to determine whether they respond differently from younger patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy and safety of SUNLENCA in heavily treatment-experienced participants with multidrug resistant HIV-1 is based on 52-week data from CAPELLA, a randomized, placebo-controlled, double-blind, multicenter trial (NCT 04150068).
CAPELLA was conducted in 72 heavily treatment-experienced participants with multiclass resistant HIV-1. Participants were required to have a viral load ≥ 400 copies/mL, documented resistance to at least two antiretroviral medications from each of at least 3 of the 4 classes of antiretroviral medications (NRTI, NNRTI, PI and INSTI), and ≤ 2 fully active antiretroviral medications from the 4 classes of antiretroviral medications remaining at baseline due to resistance, intolerability, drug access, contraindication, or other safety concerns.
The trial was composed of two cohorts. Participants were enrolled into the randomized cohort (cohort 1, N=36) if they had a < 0.5 log10 HIV-1 RNA decline compared to the screening visit. Participants were enrolled into the non-randomized cohort (cohort 2, N=36) if they had a ≥ 0.5 log10 HIV-1 RNA decline compared to the screening visit or after cohort 1 reached its planned sample size.
In the 14-day functional monotherapy period, participants in cohort 1 were randomized in a 2:1 ratio in a blinded fashion to receive either SUNLENCA or placebo, while continuing their failing regimen. This period was to establish the virologic activity of SUNLENCA. After the functional monotherapy period, participants who had received SUNLENCA continued on SUNLENCA along with an optimized background regimen (OBR); participants who had received placebo during this period initiated SUNLENCA along with an OBR.
Participants in cohort 1 had a mean age of 52 years (range: 24 to 71), 72% were male, 46% were White, 46% were Black, and 9% were Asian. 29% percent of participants identified as Hispanic/Latino. The mean baseline plasma HIV-1 RNA was 4.3 log10 copies/mL (range: 2.3 to 5.4). 19% of participants had baseline viral loads greater than 100,000 copies/mL. The mean baseline CD4+ cell count was 161 cells/mm3 (range: 6 to 827). 75% of participants had CD4+ cell counts below 200 cells/mm3. The mean number of years since participants first started HIV treatment was 24 years (range: 7 to 33); the mean number of antiretroviral agents in failing regimens at baseline was 4 (range: 1 to 7). The percentage of participants in the randomized cohort with known resistance to at least 2 agents from the NRTI, NNRTI, PI and INSTI classes was 97%, 94%, 78% and 75%, respectively. In cohort 1, 53% of participants had no fully active agents, 31% had 1 fully active agent, and 17% had 2 or more fully active agents within their initial failing regimen, including 6% of participants were who were receiving fostemsavir, which was an investigational agent at the start of the CAPELLA trial.
Participants in cohort 2 initiated SUNLENCA and an OBR on Day 1.
Participants in cohort 2 had a mean age of 48 years (range: 23 to 78), 78% were male, 36% were White, 31% were Black, 33% were Asian, and 14% of participants identified as Hispanic/Latino. The mean baseline plasma HIV-1 RNA was 4.1 log10 copies/mL (range: 1.3 to 5.7). 19% of participants had baseline viral loads greater than 100,000 copies/mL. The mean baseline CD4+ cell count was 258 cells/mm3 (range: 3 to 1296). 53% of participants had CD4+ cell counts below 200 cells/mm3. The mean number of years since participants first started HIV treatment was 19 years (range: 3 to 35); the mean number of antiretroviral agents in failing regimens at baseline was 4 (range: 2 to 7). The percentage of participants in the non-randomized cohort with known resistance to at least 2 agents from the NRTI, NNRTI, PI and INSTI classes was 100%, 100%, 83% and 64%, respectively. In cohort 2, 31% of participants had no fully active agents, 42% had 1 fully active agent, and 28% had 2 or more fully active agents within their initial failing regimen, including 6% of participants who were receiving fostemsavir, which was an investigational agent at the start of the CAPELLA trial.
The primary efficacy endpoint was the proportion of participants in cohort 1 achieving ≥ 0.5 log10 copies/mL reduction from baseline in HIV-1 RNA at the end of the functional monotherapy period. The results of the primary endpoint analysis are shown in Table 10.
| SUNLENCA (N=24) |
Placebo (N=12) |
|
|---|---|---|
| Proportion of Participants Achieving a ≥ 0.5 log10 Decrease in Viral Load | 87.5% | 16.7% |
| Treatment Difference (95% CI) | 70.8% (34.9% to 90.0%) p < 0.0001
|
The results at Weeks 26 and 52 are provided in Table 11 and Table 12.
| SUNLENCA plus OBR (N=36) |
||
|---|---|---|
| Week 26 | Week 52 | |
| OBR = optimized background regimen | ||
| HIV-1 RNA < 50 copies/mL | 81% | 83% |
| HIV-1 RNA ≥ 50 copies/mL Includes participants who had ≥ 50 copies/mL in the Week 26 or 52 window; participant who discontinued early due to lack or loss of efficacy; participants who discontinued for reasons other than an adverse event (AE), death or lack or loss of efficacy and at the time of discontinuation had a viral value of ≥ 50 copies/mL.
|
19% | 14% |
| No virologic data in Week 26 or 52 Window | 0 | 3% |
| Discontinued Study Drug Due to AE or Death Includes participants who discontinued due to AE or death at any time point from Day 1 through the time window if this resulted in no virologic data on treatment during the specified window.
|
0 | 0 |
| Discontinued Study Drug Due to Other Reasons Includes participants who discontinued for reasons other than an AE, death or lack or loss of efficacy, e.g., withdrew consent, loss to follow-up, etc. and Last Available HIV-1 RNA < 50 copies/mL |
0 | 3% |
| Missing Data During Window but on Study Drug | 0 | 0 |
| SUNLENCA plus OBR (N=36) |
||
|---|---|---|
| Week 26 | Week 52 | |
| ARV = antiretroviral; DRV=darunavir; DTG=dolutegravir; INSTI = integrase strand-transfer inhibitor; OBR = optimized background regimen; | ||
| Age (Years) | ||
| < 50 | 100% (9/9) | 89% (8/9) |
| ≥ 50 | 74% (20/27) | 81% (22/27) |
| Gender | ||
| Male | 77% (20/26) | 77% (20/26) |
| Female | 90% (9/10) | 100% (10/10) |
| Race | ||
| Black | 81% (13/16) | 75% (12/16) |
| Non-Black | 84% (16/19) | 89% (17/19) |
| Baseline plasma viral load (copies/mL) | ||
| ≤ 100,000 | 86% (25/29) | 86% (25/29) |
| > 100,000 | 57% (4/7) | 71% (5/7) |
| Baseline CD4+ (cells/mm3) | ||
| < 200 | 78% (21/27) | 78% (21/27) |
| ≥ 200 | 89% (8/9) | 100% (9/9) |
| Baseline INSTI resistance profile | ||
| With INSTI resistance | 85% (23/27) | 81% (22/27) |
| Without INSTI resistance | 63% (5/8) | 88% (7/8) |
| Number of fully active ARV agents in the OBR | ||
| 0 | 67% (4/6) | 67% (4/6) |
| 1 | 86% (12/14) | 79% (11/14) |
| ≥ 2 | 81% (13/16) | 94% (15/16) |
| Use of DTG and/or DRV in the OBR | ||
| With DTG and DRV | 83% (10/12) | 83% (10/12) |
| With DTG, without DRV | 83% (5/6) | 83% (5/6) |
| Without DTG, with DRV | 78% (7/9) | 89% (8/9) |
| Without DTG or DRV | 78% (7/9) | 78% (7/9) |
In cohort 1, at Weeks 26 and 52, the mean change from baseline in CD4+ cell count was 81 cells/mm3 (range: -101 to 522) and 82 cells/mm3 (range: -194 to 467), respectively.
In cohort 2, at Weeks 26 and 52, 81% (29/36) and 72% (26/36) of participants achieved HIV-1 RNA < 50 copies/mL, respectively, and the mean change from baseline in CD4+ cell count was 97 cells/mm3 (range: -103 to 459) and 113 cells/mm3 (range: -124 to 405), respectively.
4 Contraindications (4 CONTRAINDICATIONS)
Concomitant administration of SUNLENCA with strong CYP3A inducers is contraindicated due to decreased lenacapavir plasma concentrations, which may result in the loss of therapeutic effect and development of resistance to SUNLENCA [see Drug Interactions (7.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in other sections of the labeling:
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.1)]
- Injection Site Reactions [see Warnings and Precautions (5.3)].
7 Drug Interactions (7 DRUG INTERACTIONS)
8.6 Renal Impairment
No dosage adjustment of SUNLENCA is recommended in patients with mild, moderate or severe renal impairment (estimated creatinine clearance greater than or equal to 15 mL per minute). SUNLENCA has not been studied in patients with ESRD (estimated creatinine clearance less than 15 mL per minute) [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetic (PK) properties of lenacapavir are provided in Table 6 and Table 7. The estimated lenacapavir exposures are comparable between the two recommended dosing regimens.
| Oral | Subcutaneous | ||
|---|---|---|---|
| Absorption | |||
| % Absolute bioavailability | 6 to 10 | 100 Values reflect absolute bioavailability following subcutaneous administration of the 927 mg dose.
|
|
| Tmax Values reflect administration of lenacapavir with or without food.
|
4 hours | 77 to 84 days Due to slow release from the site of subcutaneous administration, the absorption profile of subcutaneously administered lenacapavir is complex.
|
|
| Effect of Food | |||
| Effect of low-fat meal (relative to fasting) Values refer to geometric mean ratio [low-fat meal/fasting] in PK parameters and (90% confidence interval). Low fat meal is approximately 400 kcal, 25% fat.
|
AUCinf ratio | 98.6 (58.2,167.2) | - |
| Cmax ratio | 115.8 (55.4, 242.1) | - | |
| Effect of high-fat meal (relative to fasting) Values refer to geometric mean ratio [high-fat meal/fasting] in PK parameters and (90% confidence interval). High fat meal is approximately 1000 kcal, 50% fat.
|
AUCinf ratio | 115.2 (72.0, 184.5) | - |
| Cmax ratio | 145.2 (77.9, 270.5) | - | |
| Distribution | |||
| Apparent volume of distribution (Vd/F, L) | 19240 | 9500 to 11700 | |
| % bound to human plasma proteins | >98.5 | ||
| Blood-to-plasma ratio | 0.5 to 0.7 Values reflect the blood-to-plasma ratio of lenacapavir following a single dose intravenous administration of [14C] lenacapavir through 336 hours postdose.
|
||
| Elimination | |||
| t1/2 | 10 to 12 days | 8 to 12 weeks | |
| Clearance (mean apparent clearance, L/h) | 55 | 4.2 | |
| % of dose of unchanged drug in plasma Dosing in mass balance studies: single dose intravenous administration of [14C] lenacapair to participants without HIV-1.
|
69 | ||
| Metabolism | |||
| Metabolic pathway(s) | CYP3A (minor) UGT1A1 (minor) |
||
| Excretion | |||
| Major routes of elimination | Excretion of unchanged drug into feces Metabolized via oxidation, N-dealkylation, hydrogenation, amide hydrolysis, glucuronidation, hexose conjugation, pentose conjugation, and glutathione conjugation; primarily via CYP3A and UGT1A1 and no single circulating metabolite accounted for >10% of plasma drug-related exposure.
|
||
| % of dose excreted in urine | <1 | ||
| % of dose excreted in feces (% unchanged) | 76 (33) |
| Parameter Mean (%CV) | Recommended Dosing Regimen, Option 1 Predicted exposures utilizing population PK analysis.
|
Recommended Dosing Regimen, Option 2 | |
|---|---|---|---|
| Day 1: 600 mg (oral) + 927 mg (SC) Day 2: 600 mg (oral) |
Days 1 and 2: 600 mg (oral), Day 8: 300 mg (oral), Day 15: 927 mg (SC) |
||
| Day 1 to end of Month 6 | Days 1 to 15 | Day 15 to end of Month 6 | |
| CV = coefficient of variation; NA = not applicable; SC = subcutaneous | |||
| Cmax
(ng/mL) |
101.4 (53.1) | 88.0 (72.4) | 86.5 (51.7) |
| AUCtau
(h∙ng/mL) |
242266 (46.0) | 19496 (72.6) | 239163 (47.2) |
| Ctrough
(ng/mL) |
32.5 (57.2) | 46.9 (72.3) | 32.5 (57.5) |
Lenacapavir exposures after subcutaneous administration were similar between heavily treatment experienced participants with HIV-1 and participants without HIV-1 based on population pharmacokinetics analysis. Lenacapavir exposures (AUCtau, Cmax and Ctrough) after oral administration were 28% to 43% higher in participants with HIV-1 who were heavily treatment experienced, compared to participants without HIV-1 based on population PK analysis. These differences were not considered clinically relevant.
2.2 Recommended Dosage
SUNLENCA can be initiated using one of the two recommended dosage regimens in Table 1 and Table 2 below. Maintenance dosing is administered by subcutaneous injection every 6 months regardless of the initiation regimen. Healthcare providers should determine the appropriate initiation regimen for the patient. SUNLENCA oral tablets may be taken with or without food [see Clinical Pharmacology (12.3)].
| Treatment Time | |
|---|---|
| Dosage of SUNLENCA: Initiation | |
| Day 1 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Dosage of SUNLENCA: Maintenance | |
| Every 6 months (26 weeks) From the date of the last injection. +/-2 weeks |
927 mg by subcutaneous injection (2 × 1.5 mL injections) |
| Treatment Time | |
|---|---|
| Dosage of SUNLENCA: Initiation | |
| Day 1 | 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Day 8 | 300 mg orally (1 × 300 mg tablet) |
| Day 15 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) |
| Dosage of SUNLENCA: Maintenance | |
| Every 6 months (26 weeks) From the date of the last injection. +/-2 weeks |
927 mg by subcutaneous injection (2 × 1.5 mL injections) |
8.7 Hepatic Impairment
No dosage adjustment of SUNLENCA is recommended in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. SUNLENCA has not been studied in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
SUNLENCA, in combination with other antiretroviral(s), is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in heavily treatment-experienced adults with multidrug resistant HIV-1 whose current antiretroviral regimen is failing due to resistance, intolerance, or safety considerations.
12.1 Mechanism of Action
SUNLENCA is an HIV-1 antiretroviral agent [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Immune reconstitution syndrome: May necessitate further evaluation and treatment. (5.1)
- Residual concentrations of lenacapavir may remain in systemic circulation for up to 12 months or longer. Counsel patients regarding the dosing schedule; non-adherence could lead to loss of virologic response and development of resistance. (5.2)
- May increase exposure and risk of adverse reactions to drugs primarily metabolized by CYP3A initiated within 9 months after the last subcutaneous dose of SUNLENCA. (5.2)
- If discontinued, initiate an alternative, fully suppressive antiretroviral regimen where possible no later than 28 weeks after the final injection of SUNLENCA. If virologic failure occurs, switch to an alternative regimen if possible. (5.2)
- Injection site reactions may occur, and nodules and indurations may be persistent. Improper administration (intradermal injection) has been associated with serious injection site reactions. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dosage – Initiation with one of two options followed by once every 6 months maintenance injection dosing. Tablets may be taken without regard to food. (2.2)
| Initiation Option 1 | |
| Day 1 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Initiation Option 2 | |
| Day 1 | 600 mg orally (2 × 300 mg tablets) |
| Day 2 | 600 mg orally (2 × 300 mg tablets) |
| Day 8 | 300 mg orally (1 × 300 mg tablet) |
| Day 15 | 927 mg by subcutaneous injection (2 × 1.5 mL injections) |
| Maintenance | |
| 927 mg by subcutaneous injection (2 × 1.5 mL injections) every 6 months (26 weeks) from the date of the last injection +/-2 weeks. |
- Planned missed injections: If scheduled injection is to be missed by more than 2 weeks, SUNLENCA tablets may be used for oral bridging for up to 6 months until injections resume. Recommended dosage is 300 mg orally once every 7 days. (2.3)
- Unplanned missed injections: If more than 28 weeks since last injection and tablets have not been taken for oral bridging, restart initiation from Day 1 (using Option 1 or Option 2) if clinically appropriate. (2.3)
- SUNLENCA injection is for subcutaneous administration only. Two 1.5 mL injections are required for complete dose. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 300 mg
Injection: 463.5 mg/1.5 mL (309 mg/mL) in single-dose vials. (3)
5.3 Injection Site Reactions
Administration of SUNLENCA may result in local injection site reactions (ISRs). If clinically significant ISRs occur, evaluate and institute appropriate therapy and follow-up.
Manifestations of ISRs may include swelling, pain, erythema, nodule, induration, pruritus, extravasation or mass. Nodules and indurations at the injection site may take longer to resolve than other ISRs. In clinical studies, after a median follow-up of 553 days, 30% of nodules and 13% of indurations (in 10% and 1% of participants, respectively) associated with the first injections of SUNLENCA had not fully resolved. Measurements and qualitative assessments of ISRs were not routinely reported. Where described, the majority of the injection site nodules and indurations were palpable but not visible, and had a maximum size of approximately 1 to 4 cm [see Adverse Reactions (6.1)].
The mechanism driving the persistence of injection site nodules and indurations in some patients is not fully understood, but based on available data, they may be related to the presence of the subcutaneous drug depot. In some patients who had a skin biopsy performed of an injection site nodule or induration, dermatopathology revealed foreign body inflammation or granulomatous response.
Improper administration (intradermal injection) has been associated with serious injection site reactions, including necrosis and ulcer [see Adverse Reactions (6)]. Ensure SUNLENCA is only administered subcutaneously in the abdomen [see Dosage and Administration (2.4)].
6.2 Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the following adverse reactions have been identified during postmarketing use. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The primary safety assessment of SUNLENCA was based on data from heavily treatment-experienced adult participants with HIV who received SUNLENCA in a Phase 2/3 trial (CAPELLA; N=72) through Week 52 (median duration on study of 71 weeks) [see Clinical Studies (14)], as well as supportive data in treatment-naïve adult participants with HIV who received SUNLENCA in a Phase 2 trial (CALIBRATE; N=157) through Week 54 (median duration of exposure of 66 weeks).
The most common adverse reactions (all Grades) reported in at least 3% of participants in CAPELLA were nausea and injection site reactions. The proportion of partcipants in CAPELLA who discontinued treatment with SUNLENCA due to adverse events, regardless of severity, was 1% (Grade 1 injection site nodule in 1 participant). Table 3 displays the frequency of adverse reactions (all Grades) greater than or equal to 3% in the SUNLENCA group.
| Adverse Reactions | SUNLENCA + Background Regimen (N=72) |
|---|---|
| Injection Site Reactions | 65% |
| Nausea | 4% |
The majority (96%) of all adverse reactions associated with SUNLENCA were mild or moderate in severity.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Adherence to Treatment Regimen
Prior to starting SUNLENCA, healthcare providers should carefully select patients who agree to the required every 6 month injection dosing schedule and counsel patients about the importance of adherence to scheduled SUNLENCA dosing visits and concomitant oral antiretroviral therapy to help maintain viral suppression and reduce the risk of viral rebound and potential development of resistance with missed doses [see Warnings and Precautions (5.2), Microbiology (12.4)].
5.1 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections [such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia (PCP), or tuberculosis], which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
Principal Display Panel Kit Carton (PRINCIPAL DISPLAY PANEL - Kit Carton)
Rx only
NDC 61958-3002-1
Sunlenca®
(lenacapavir) injection
463.5 mg/1.5 mL (309 mg/mL)
For Subcutaneous Injection
Contents
- 2 x 1.5 mL lenacapavir single-dose vials
- 2 vial access devices
- 2 syringes
- 2 injection needles (22 gauge, 1/2 inch)
- Prescribing Information
- Instructions for Use
- Patient Information
Both 463.5 mg/1.5 mL (2 single-dose vials) must be
administered to receive the 927 mg dose.
For Healthcare Professional administration only.
GILEAD
7.1 Effect of Other Drugs On Sunlenca (7.1 Effect of Other Drugs on SUNLENCA)
Lenacapavir is a substrate of P-gp, UGT1A1, and CYP3A.
7.2 Effect of Sunlenca On Other Drugs (7.2 Effect of SUNLENCA on Other Drugs)
Lenacapavir is a moderate inhibitor of CYP3A. Due to the long half-life of lenacapavir following subcutaneous administration, SUNLENCA may increase the exposure of drugs primarily metabolized by CYP3A [see Clinical Pharmacology (12.3)] initiated within 9 months after the last subcutaneous dose of SUNLENCA, which may increase the potential risk of adverse reactions. See the prescribing information of the sensitive CYP3A substrate for dosing recommendations with moderate inhibitors of CYP3A.
Principal Display Panel Kit Carton 3005 (PRINCIPAL DISPLAY PANEL - Kit Carton - 3005)
Rx only
NDC 61958-3005-1
Sunlenca®
(lenacapavir) injection
463.5 mg/1.5 mL (309 mg/mL)
For Subcutaneous Injection
Contents
- 2 x 1.5 mL lenacapavir single-dose vials
- 2 withdrawal needles (18 gauge, 1½ inch)
- 2 syringes
- 2 injection needles (22 gauge, ½ inch)
- Prescribing Information
- Instructions for Use
- Patient Information
Both 463.5 mg/1.5 mL (2 single-dose vials) must be
administered to receive the 927 mg dose.
For Healthcare Professional administration only.
GILEAD
Principal Display Panel 4 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 4 Tablet Bottle Label)
NDC 61958-3001-3
4 tablets
Sunlenca®
(lenacapavir) tablets
300 mg per tablet
Talk to your healthcare provider
before taking Sunlenca tablets.
Your healthcare provider will tell you
when to take Sunlenca tablets.
2.4 Preparation and Administration of Subcutaneous Injection
SUNLENCA injection is only for subcutaneous administration into the abdomen by a healthcare provider. Do NOT administer intradermally due to risk of serious injection site reactions [see Warnings and Precautions (5.3)].
Use aseptic technique. Visually inspect the solution in the vials and prepared syringe for particulate matter and discoloration prior to administration. SUNLENCA injection is a yellow solution. Do not use SUNLENCA injection if the solution is discolored or if it contains particulate matter. Once the solution is withdrawn from the vials, the subcutaneous injections should be administered as soon as possible [see How Supplied/Storage and Handling (16)].
There are two available injection kits, which differ only in how SUNLENCA injection is prepared (the components and associated method for withdrawal of the solution from the vials) [see How Supplied/Storage and Handling (16)]. Refer to the figures below for the relevant injection kit.
The injection kit components are for single use only. Two 1.5 mL injections are required for a complete dose.
Principal Display Panel 5 Tablet Blister Pack Pouch Carton (PRINCIPAL DISPLAY PANEL - 5 Tablet Blister Pack Pouch Carton)
NDC 61958-3001-2
Sunlenca®
(lenacapavir) tablets
300 mg per tablet
1 pouch containing 5 tablets
Each tablet contains: 300 mg of lenacapavir
(present as 306.8 mg lenacapavir sodium).
Store at 20 °C - 25 °C (68 °F - 77 °F).
Dispense only in original container.
Recommended Dosage: See prescribing information.
KEEP OUT OF THE REACH OF CHILDREN
Talk to your healthcare provider before taking
Sunlenca tablets.
Your healthcare provider will tell you when to take
Sunlenca tablets.
GILEAD
Manufactured for:
Gilead Sciences, Inc.
Foster City, CA 94404
Made in Canada
© 2022 Gilead Sciences, Inc.
Principal Display Panel 300 Mg Tablet Blister Pack Pouch Carton (PRINCIPAL DISPLAY PANEL - 300 mg Tablet Blister Pack Pouch Carton)
NDC 61958-3001-1
Sunlenca®
(lenacapavir) tablets
300 mg per tablet
1 pouch containing 4 tablets
Each tablet contains: 300 mg of lenacapavir
(present as 306.8 mg lenacapavir sodium).
Store at 20 °C - 25 °C (68 °F - 77 °F).
Dispense only in original container.
Recommended Dosage: See prescribing information.
KEEP OUT OF THE REACH OF CHILDREN
GILEAD
Manufactured for:
Gilead Sciences, Inc.
Foster City, CA 94404
Made in Canada
© 2022 Gilead Sciences, Inc.
7.3 Established and Other Potentially Significant Drug Interactions
Table 5 provides a listing of clinically significant drug interactions with recommended prevention or management strategies, but is not all inclusive. The drug interactions described are based on studies conducted with SUNLENCA or are drug interactions that may occur with SUNLENCA [see Contraindications (4) and Clinical Pharmacology (12.3)].
| Concomitant Drug Class: Drug Name |
Effect on Concentration ↑ = Increase, ↓ = Decrease.
|
Clinical Comment |
|---|---|---|
|
Antiarrhythmics:
digoxin |
↑ digoxin | Use with caution and monitor digoxin therapeutic concentration. |
|
Anticoagulants:
Direct Oral Anticoagulants (DOACs) rivaroxaban dabigatran edoxaban |
↑ DOAC | Refer to the DOAC prescribing information for concomitant administration with moderate CYP3A inhibitors and/or P-gp inhibitors. |
|
Anticonvulsants:
carbamazepine oxcarbazepine phenobarbital phenytoin |
↓ lenacapavir | Concomitant administration of carbamazepine, oxcarbazepine, phenobarbital, or phenytoin may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with carbamazepine or phenytoin is contraindicated. Concomitant administration of SUNLENCA with oxcarbazepine or phenobarbital is not recommended. Consider use of alternative anticonvulsants. |
|
Antiretroviral Agents:
atazanavir/cobicistat Drug-drug interaction study was conducted.
atazanavir/ritonavir |
↑ lenacapavir (atazanavir/cobicistat, atazanavir/ritonavir) | Concomitant administration of efavirenz, nevirapine, or tipranavir/ritonavir may result in loss of therapeutic effect and development of resistance. |
| efavirenz
nevirapine tipranavir/ritonavir |
↓ lenacapavir (efavirenz, nevirapine, tipranavir/ritonavir) | Concomitant administration with atazanavir/cobicistat, atazanavir/ritonavir, efavirenz, nevirapine, or tipranavir/ritonavir is not recommended. |
|
Antimycobacterials:
rifabutin rifampin rifapentine |
↓ lenacapavir | Concomitant administration of rifabutin, rifampin and rifapentine may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with rifampin is contraindicated [see Contraindications (4)]. Concomitant administration of SUNLENCA with rifabutin or rifapentine is not recommended. |
|
Corticosteroids (systemic):
cortisone/hydrocortisone dexamethasone |
↑ corticosteroids (systemic) | Concomitant administration with systemic corticosteroids whose exposures are significantly increased by CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Initiate with the lowest starting dose and titrate carefully while monitoring for safety. |
| ↓ lenacapavir (dexamethasone) | Concomitant administration of systemic dexamethasone may result in loss of therapeutic effect of lenacapavir and development of resistance. Alternative corticosteroids to dexamethasone should be considered, particularly for long-term use. | |
|
Ergot derivatives:
dihydroergotamine ergotamine methylergonovine |
↑ dihydroergotamine ↑ ergotamine ↑ methylergonovine |
Concomitant administration of SUNLENCA with dihydroergotamine, ergotamine or methylergonovine is not recommended. |
|
Herbal Products:
St. John's wort The induction potency of St. John's wort may vary widely based on preparation.
(Hypericum perforatum) |
↓ lenacapavir | Concomitant administration of St. John's wort may result in loss of therapeutic effect and development of resistance. Concomitant administration of SUNLENCA with St. John's wort is contraindicated. |
|
HMG-CoA Reductase Inhibitors:
lovastatin simvastatin |
↑ lovastatin ↑ simvastatin |
Initiate lovastatin and simvastatin with the lowest starting dose and titrate carefully while monitoring for safety (e.g., myopathy). |
|
Narcotic analgesics metabolized by CYP3A:
e.g., fentanyl, oxycodone |
↑ fentanyl ↑ oxycodone |
Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. |
| tramadol | ↑ tramadol | A decrease in dose may be needed for tramadol with concomitant use. |
|
Narcotic analgesic for treatment of opioid dependence:
buprenorphine, methadone |
buprenorphine: effects unknown methadone: effects unknown |
Initiation of buprenorphine or methadone in patients taking SUNLENCA: Carefully titrate the dose of buprenorphine or methadone to the desired effect; use the lowest feasible initial or maintenance dose. Initiation of SUNLENCA in patients taking buprenorphine or methadone: A dose adjustment for buprenorphine or methadone may be needed. Monitor clinical signs and symptoms. |
|
Opioid Antagonist:
naloxegol |
↑ naloxegol | Avoid use with SUNLENCA; if unavoidable, decrease the dosage of naloxegol and monitor for adverse reactions. |
|
Phosphodiesterase-5 (PDE-5) Inhibitors:
sildenafil tadalafil vardenafil |
↑ PDE-5 inhibitors | Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Concomitant administration of SUNLENCA with tadalafil for the treatment of PAH is not recommended. Use of PDE-5 inhibitors for erectile dysfunction (ED): Refer to the prescribing information of PDE-5 inhibitors for dose recommendations. |
|
Sedatives/Hypnotics:
midazolam (oral) triazolam |
↑ midazolam (oral) ↑ triazolam |
Use with caution when midazolam or triazolam is concomitantly administered with SUNLENCA |
7.4 Drugs Without Clinically Significant Interactions With Sunlenca (7.4 Drugs without Clinically Significant Interactions with SUNLENCA)
Based on drug interaction studies conducted with SUNLENCA, no clinically significant drug interactions have been observed with: darunavir/cobicistat, cobicistat, famotidine, pitavastatin, rosuvastatin, tenofovir alafenamide, and voriconazole.
5.2 Long Acting Properties and Potential Associated Risks With Sunlenca (5.2 Long-Acting Properties and Potential Associated Risks with SUNLENCA)
Residual concentrations of lenacapavir may remain in the systemic circulation of patients for prolonged periods (up to 12 months or longer after the last subcutaneous dose). It is important to counsel patients that maintenance dosing by injection is required every 6 months, because missed doses or non-adherence to injections could lead to loss of virologic response and development of resistance [see Dosage and Administration (2.1)].
Lenacapavir, a moderate CYP3A inhibitor, may increase the exposure to, and therefore potential risk of adverse reactions from, drugs primarily metabolized by CYP3A initiated within 9 months after the last subcutaneous dose of SUNLENCA [see Drug Interactions and Clinical Pharmacology (7.2, 12.3)].
If SUNLENCA is discontinued, to minimize the potential risk of developing viral resistance, it is essential to initiate an alternative, fully suppressive antiretroviral regimen where possible no later than 28 weeks after the final injection of SUNLENCA. If virologic failure occurs during treatment, switch the patient to an alternative regimen if possible [see Dosage and Administration (2.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:32.752426 · Updated: 2026-03-14T22:50:12.261930