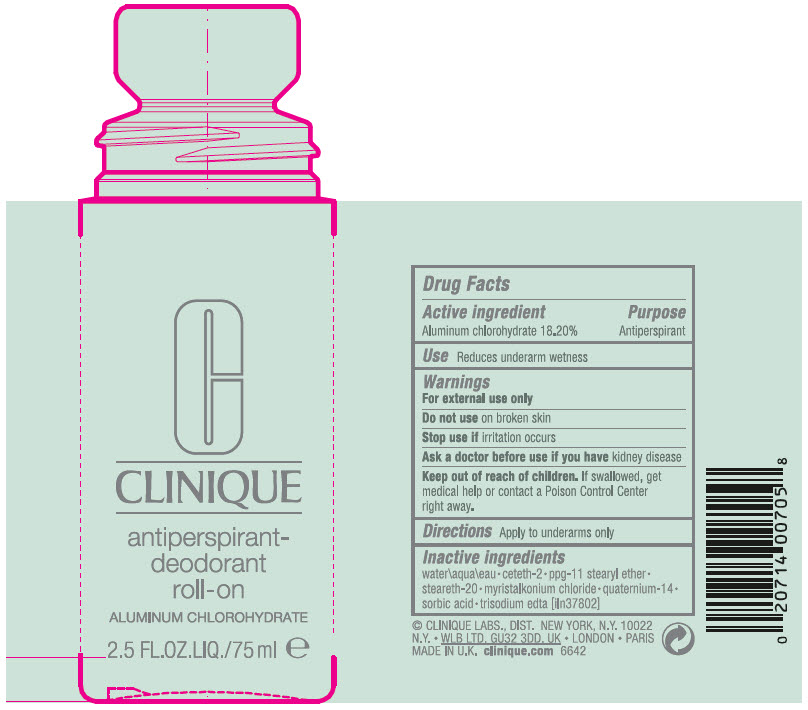
antiperspirant-deodorant roll-on
e496528c-60cf-46ed-9cff-cb4e210a15ab
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antiperspirant
Description
Drug Facts
Medication Information
Warnings
For external use only
Purpose
Antiperspirant
Directions
Apply to underarms only
Inactive Ingredients
water\aqua\eau • ceteth-2 • ppg-11 stearyl ether • steareth-20 • myristalkonium chloride • quaternium-14 • sorbic acid • trisodium edta [iln37802]
Description
Drug Facts
Use
Reduces underarm wetness
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use if irritation occurs
Section 50569-3
Ask a doctor before use if you have kidney disease
Section 50570-1
Do not use on broken skin
Active Ingredient
Aluminum chlorohydrate 18.20%
Principal Display Panel 75 Ml Bottle Label
C
CLINIQUE
antiperspirant-
deodorant
roll-on
ALUMINUM CHLOROHYDRATE
2.5 FL.OZ.LIQ./75 ml e
Structured Label Content
Warnings
For external use only
Purpose
Antiperspirant
Directions
Apply to underarms only
Inactive Ingredients (Inactive ingredients)
water\aqua\eau • ceteth-2 • ppg-11 stearyl ether • steareth-20 • myristalkonium chloride • quaternium-14 • sorbic acid • trisodium edta [iln37802]
Use
Reduces underarm wetness
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use if irritation occurs
Section 50569-3 (50569-3)
Ask a doctor before use if you have kidney disease
Section 50570-1 (50570-1)
Do not use on broken skin
Active Ingredient (Active ingredient)
Aluminum chlorohydrate 18.20%
Principal Display Panel 75 Ml Bottle Label (PRINCIPAL DISPLAY PANEL - 75 ml Bottle Label)
C
CLINIQUE
antiperspirant-
deodorant
roll-on
ALUMINUM CHLOROHYDRATE
2.5 FL.OZ.LIQ./75 ml e
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:01:28.335682 · Updated: 2026-03-14T23:01:25.991637