Private Label Itch Relief Gel
e42f484d-a641-3489-e053-2a95a90a4254
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Do not use more often than directed. Adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily. Children under 2 years of age: ask a doctor.
Medication Information
Warnings and Precautions
For external use only.
Indications and Usage
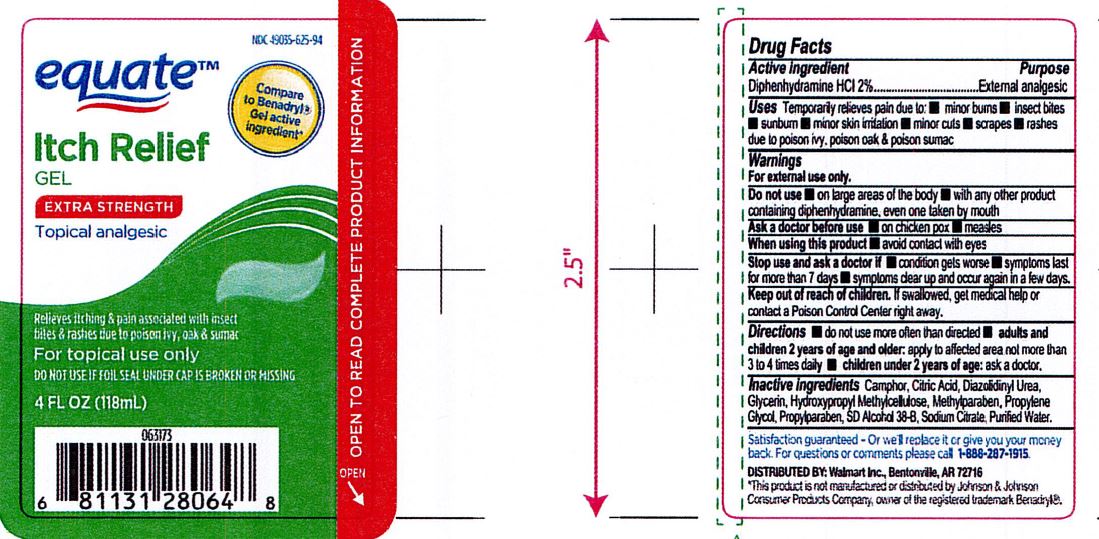
Temporarily relieves pain due to: Minor burns, insect bites, sunburn, minor skin irritation, minor cuts, scrapes, rashes due to poison ivy, poison oak and poison sumac.
Dosage and Administration
Do not use more often than directed.
Adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily.
Children under 2 years of age: ask a doctor.
Description
Do not use more often than directed. Adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily. Children under 2 years of age: ask a doctor.
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
Camphor, Citric Acid, Diazolidnyl Urea, Glycerin, Hydroxypropyl Methylcellulose, Methylparaben, Propylene Glycol, Propylparaben, SD Alcohol 38-B, Sodium Citrate, Purified Water.
Section 55105-1
External Analgesic
Section 55106-9
Diphenhydramine HCl 2%
Equate Label
[
Structured Label Content
Indications and Usage (34067-9)
Temporarily relieves pain due to: Minor burns, insect bites, sunburn, minor skin irritation, minor cuts, scrapes, rashes due to poison ivy, poison oak and poison sumac.
Dosage and Administration (34068-7)
Do not use more often than directed.
Adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily.
Children under 2 years of age: ask a doctor.
Warnings and Precautions (34071-1)
For external use only.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Camphor, Citric Acid, Diazolidnyl Urea, Glycerin, Hydroxypropyl Methylcellulose, Methylparaben, Propylene Glycol, Propylparaben, SD Alcohol 38-B, Sodium Citrate, Purified Water.
Section 55105-1 (55105-1)
External Analgesic
Section 55106-9 (55106-9)
Diphenhydramine HCl 2%
Equate Label
[
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:35.336144 · Updated: 2026-03-14T23:02:13.859151