These Highlights Do Not Include All The Information Needed To Use Cycloset Safely And Effectively. See Full Prescribing Information For Cycloset.
e42ba916-16b9-4d1c-8d09-9d2fbb1b6c20
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.3 ) 04/2020 Warnings and Precautions ( 5.7 ) 08/2020
Indications and Usage
CYCLOSET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Dosage and Administration
Taken within two hours after waking in the morning with food ( 2.1 ) Initial dose is one tablet (0.8 mg) daily increased weekly by one tablet until maximal tolerated daily dose of 1.6 to 4.8 mg is achieved. ( 2.2 ) Limit dose to 1.6 mg daily during concomitant use of a moderate CYP3A4 inhibitor. Avoid concomitant use with strong CYP3A4 inhibitors. ( 2.3 )
Warnings and Precautions
Hypotension: Can cause orthostatic hypotension and syncope, particularly upon initiation or dose escalation. Use caution in patients taking antihypertensive medications. Assess orthostatic vital signs prior to initiation of CYCLOSET and periodically thereafter. Advise patients during early treatment to avoid situations that could lead to injury if syncope was to occur. ( 5.1 , 6.1 ) Psychosis: May exacerbate psychotic disorders or reduce the effectiveness of drugs that treat psychosis. Use in patients with severe psychotic disorders is not recommended. ( 5.2 ) Impulse control/compulsive behaviors: Ask patients or their caregivers about new or increased gambling urges, sexual urges, uncontrolled spending, or other urges while being treated with CYCLOSET. Consider dose reduction or stopping CYCLOSET if a patient develops such urges. Use of CYCLOSET in patients with impulse control/compulsive behaviors is not recommended. ( 5.3 , 6.2 ). Somnolence: May cause somnolence. Advise patients not to operate heavy machinery if symptoms of somnolence occur. ( 5.4 ) Interaction with dopamine antagonists: Concomitant use with dopamine antagonists such as neuroleptic agents may diminish the effectiveness of both drugs. Concomitant use is not recommended. ( 5.5 , 7 ) Other dopamine receptor agonists: Effectiveness and safety are unknown in patients already taking dopamine receptor agonists for other indications. Concomitant use is not recommended. ( 5.6 ) Risks in Postpartum Patients: Serious and life-threatening adverse reactions have been reported. ( 5.7 , 6.2 )
Contraindications
CYCLOSET is contraindicated in: Patients with known hypersensitivity to bromocriptine, ergot-related drugs, or any of the excipients in CYCLOSET. Patients with syncopal migraine. Bromocriptine increases the likelihood of a hypotensive episode among patients with syncopal migraine. Loss of consciousness during a migraine may reflect dopamine receptor hypersensitivity. CYCLOSET is a dopamine receptor agonist and may, therefore, potentiate the risk for syncope in these patients. Postpartum patients. Serious and life-threatening adverse reactions have been reported with bromocriptine use in this population [see Warnings and Precautions (5.7) , Adverse Reactions (6.2) ] . Lactating patients. CYCLOSET contains bromocriptine which inhibits lactation [see Use in Specific Populations (8.2) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypotension [see Warnings and Precautions (5.1) ] Psychotic Disorders [see Warnings and Precautions (5.2) ] Somnolence [see Warnings and Precautions (5.4) ] Risks in Postpartum Women [see Warnings and Precautions (5.7) ]
Drug Interactions
The active ingredient in CYCLOSET (bromocriptine mesylate) is highly bound to serum proteins. Therefore, CYCLOSET may increase the unbound fraction of other concomitantly used highly protein-bound therapies (e.g., salicylates, sulfonamides, chloramphenicol and probenecid), which may alter their effectiveness and risk for side effects. CYCLOSET is a dopamine receptor agonist. Concomitant use of dopamine receptor antagonists, such as neuroleptics (e.g., phenothiazines, butyrophenones, thioxanthenes), or metoclopramide may diminish the effectiveness of CYCLOSET, and CYCLOSET may diminish the effectiveness of these other therapies. The concurrent use of CYCLOSET with these agents has not been studied in clinical trials and is not recommended [see Warnings and Precautions (5.5) ]. CYCLOSET in combination with ergot-related drugs may cause an increase in the occurrence of ergot-related side effects, such as nausea, vomiting, and fatigue, and may also reduce the effectiveness of these ergot therapies when used to treat migraine. The concurrent use of these ergot agents within 6 hours of CYCLOSET dosing is not recommended. CYCLOSET is extensively metabolized by the liver via CYP3A4. Therefore, potent inhibitors or inducers of CYP3A4 may increase or reduce the circulating levels of CYCLOSET, respectively. Use caution when co-administering drugs that are inhibitors or inducers of CYP3A4. CYCLOSET dose should not exceed 1.6 mg once daily during concomitant use of a moderate CYP3A4 inhibitor (e.g., erythromycin). Concomitant use of strong CYP3A4 inhibitors (e.g., azole antimycotics, HIV protease inhibitors) with CYCLOSET should be avoided. Ensure adequate washout of the strong CYP3A4 inhibitor drug before initiating CYCLOSET treatment [see Clinical Pharmacology (12.3) ] . There are postmarketing reports of hypertension and tachycardia when bromocriptine was co-administered with sympathomimetic drugs (e.g., phenylpropanolamine and isometheptene) in postpartum women. There are limited clinical trial data supporting the safety of co-administering sympathomimetic drugs and CYCLOSET for more than 10 days. Therefore, concomitant use of these agents with CYCLOSET for more than 10 days duration is not recommended. Also, there are limited clinical trial data supporting the safety of selective 5-hydroxytryptamine 1B (5-HT 1B ) agonists (e.g., sumatriptan) used concurrently with CYCLOSET, and the concomitant use of these agents with CYCLOSET should be avoided.
Storage and Handling
CYCLOSET 0.8 mg tablets are WHITE and round with "C" on one side and "9" on the other. The tablets are supplied as follows: NDC 73515-123-30 unit-of-use bottles of 200 NDC 73515-123-21 unit-of-use bottles of 21 (samples only).
How Supplied
CYCLOSET 0.8 mg tablets are WHITE and round with "C" on one side and "9" on the other. The tablets are supplied as follows: NDC 73515-123-30 unit-of-use bottles of 200 NDC 73515-123-21 unit-of-use bottles of 21 (samples only).
Medication Information
Warnings and Precautions
Hypotension: Can cause orthostatic hypotension and syncope, particularly upon initiation or dose escalation. Use caution in patients taking antihypertensive medications. Assess orthostatic vital signs prior to initiation of CYCLOSET and periodically thereafter. Advise patients during early treatment to avoid situations that could lead to injury if syncope was to occur. ( 5.1 , 6.1 ) Psychosis: May exacerbate psychotic disorders or reduce the effectiveness of drugs that treat psychosis. Use in patients with severe psychotic disorders is not recommended. ( 5.2 ) Impulse control/compulsive behaviors: Ask patients or their caregivers about new or increased gambling urges, sexual urges, uncontrolled spending, or other urges while being treated with CYCLOSET. Consider dose reduction or stopping CYCLOSET if a patient develops such urges. Use of CYCLOSET in patients with impulse control/compulsive behaviors is not recommended. ( 5.3 , 6.2 ). Somnolence: May cause somnolence. Advise patients not to operate heavy machinery if symptoms of somnolence occur. ( 5.4 ) Interaction with dopamine antagonists: Concomitant use with dopamine antagonists such as neuroleptic agents may diminish the effectiveness of both drugs. Concomitant use is not recommended. ( 5.5 , 7 ) Other dopamine receptor agonists: Effectiveness and safety are unknown in patients already taking dopamine receptor agonists for other indications. Concomitant use is not recommended. ( 5.6 ) Risks in Postpartum Patients: Serious and life-threatening adverse reactions have been reported. ( 5.7 , 6.2 )
Indications and Usage
CYCLOSET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Dosage and Administration
Taken within two hours after waking in the morning with food ( 2.1 ) Initial dose is one tablet (0.8 mg) daily increased weekly by one tablet until maximal tolerated daily dose of 1.6 to 4.8 mg is achieved. ( 2.2 ) Limit dose to 1.6 mg daily during concomitant use of a moderate CYP3A4 inhibitor. Avoid concomitant use with strong CYP3A4 inhibitors. ( 2.3 )
Contraindications
CYCLOSET is contraindicated in: Patients with known hypersensitivity to bromocriptine, ergot-related drugs, or any of the excipients in CYCLOSET. Patients with syncopal migraine. Bromocriptine increases the likelihood of a hypotensive episode among patients with syncopal migraine. Loss of consciousness during a migraine may reflect dopamine receptor hypersensitivity. CYCLOSET is a dopamine receptor agonist and may, therefore, potentiate the risk for syncope in these patients. Postpartum patients. Serious and life-threatening adverse reactions have been reported with bromocriptine use in this population [see Warnings and Precautions (5.7) , Adverse Reactions (6.2) ] . Lactating patients. CYCLOSET contains bromocriptine which inhibits lactation [see Use in Specific Populations (8.2) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypotension [see Warnings and Precautions (5.1) ] Psychotic Disorders [see Warnings and Precautions (5.2) ] Somnolence [see Warnings and Precautions (5.4) ] Risks in Postpartum Women [see Warnings and Precautions (5.7) ]
Drug Interactions
The active ingredient in CYCLOSET (bromocriptine mesylate) is highly bound to serum proteins. Therefore, CYCLOSET may increase the unbound fraction of other concomitantly used highly protein-bound therapies (e.g., salicylates, sulfonamides, chloramphenicol and probenecid), which may alter their effectiveness and risk for side effects. CYCLOSET is a dopamine receptor agonist. Concomitant use of dopamine receptor antagonists, such as neuroleptics (e.g., phenothiazines, butyrophenones, thioxanthenes), or metoclopramide may diminish the effectiveness of CYCLOSET, and CYCLOSET may diminish the effectiveness of these other therapies. The concurrent use of CYCLOSET with these agents has not been studied in clinical trials and is not recommended [see Warnings and Precautions (5.5) ]. CYCLOSET in combination with ergot-related drugs may cause an increase in the occurrence of ergot-related side effects, such as nausea, vomiting, and fatigue, and may also reduce the effectiveness of these ergot therapies when used to treat migraine. The concurrent use of these ergot agents within 6 hours of CYCLOSET dosing is not recommended. CYCLOSET is extensively metabolized by the liver via CYP3A4. Therefore, potent inhibitors or inducers of CYP3A4 may increase or reduce the circulating levels of CYCLOSET, respectively. Use caution when co-administering drugs that are inhibitors or inducers of CYP3A4. CYCLOSET dose should not exceed 1.6 mg once daily during concomitant use of a moderate CYP3A4 inhibitor (e.g., erythromycin). Concomitant use of strong CYP3A4 inhibitors (e.g., azole antimycotics, HIV protease inhibitors) with CYCLOSET should be avoided. Ensure adequate washout of the strong CYP3A4 inhibitor drug before initiating CYCLOSET treatment [see Clinical Pharmacology (12.3) ] . There are postmarketing reports of hypertension and tachycardia when bromocriptine was co-administered with sympathomimetic drugs (e.g., phenylpropanolamine and isometheptene) in postpartum women. There are limited clinical trial data supporting the safety of co-administering sympathomimetic drugs and CYCLOSET for more than 10 days. Therefore, concomitant use of these agents with CYCLOSET for more than 10 days duration is not recommended. Also, there are limited clinical trial data supporting the safety of selective 5-hydroxytryptamine 1B (5-HT 1B ) agonists (e.g., sumatriptan) used concurrently with CYCLOSET, and the concomitant use of these agents with CYCLOSET should be avoided.
Storage and Handling
CYCLOSET 0.8 mg tablets are WHITE and round with "C" on one side and "9" on the other. The tablets are supplied as follows: NDC 73515-123-30 unit-of-use bottles of 200 NDC 73515-123-21 unit-of-use bottles of 21 (samples only).
How Supplied
CYCLOSET 0.8 mg tablets are WHITE and round with "C" on one side and "9" on the other. The tablets are supplied as follows: NDC 73515-123-30 unit-of-use bottles of 200 NDC 73515-123-21 unit-of-use bottles of 21 (samples only).
Description
Warnings and Precautions ( 5.3 ) 04/2020 Warnings and Precautions ( 5.7 ) 08/2020
Section 42229-5
Limitations of Use
- CYCLOSET should not be used to treat type 1 diabetes or diabetic ketoacidosis.
- Limited efficacy data in combination with thiazolidinediones.
- Efficacy has not been confirmed in combination with insulin.
Section 42230-3
| Patient Information CYCLOSET® (Sikloset) (bromocriptine mesylate tablets) for oral use |
||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 08/2020 | |||
| Read this Patient Information that comes with CYCLOSET before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. | ||||
| What is CYCLOSET? | ||||
|
||||
| Who should not take CYCLOSET? | ||||
| Do not take CYCLOSET if you: | ||||
|
||||
| Talk to your healthcare provider before taking CYCLOSET if you have any of these conditions. | ||||
| What should I tell my healthcare provider before taking CYCLOSET? | ||||
| Before taking CYCLOSET, tell your healthcare provider about all of your medical conditions, including if you: | ||||
|
||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | ||||
| Especially tell your healthcare provider if you take: | ||||
|
||||
| Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure. | ||||
| CYCLOSET may affect the way other medicines work, and other medicines may affect how CYCLOSET works. | ||||
| Know the medicines you take. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine. | ||||
| How should I take CYCLOSET? | ||||
|
||||
| What are the possible side effects of CYCLOSET? | ||||
| CYCLOSET may cause serious side effects, including: | ||||
|
||||
| The most common side effects of CYCLOSET include: | ||||
|
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. | ||||
| These are not all the possible side effects of CYCLOSET. For more information, ask your healthcare provider or pharmacist. | ||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
| How should I store CYCLOSET? | ||||
|
||||
| Keep CYCLOSET and all medicines out of the reach of children. | ||||
| General information about the safe and effective use of CYCLOSET. | ||||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use CYCLOSET for a condition for which it was not prescribed. Do not give CYCLOSET to other people, even if they have the same symptoms that you have. It may harm them. | ||||
| This leaflet summarizes the most important information about CYCLOSET. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about CYCLOSET that is written for health professionals. | ||||
| What are the ingredients in CYCLOSET? | ||||
| Active ingredient: bromocriptine mesylate | ||||
| Inactive ingredients: lactose, corn starch, magnesium stearate, colloidal silicon dioxide, and citric acid | ||||
|
Manufactured for:
VeroScience, LLC Tiverton, RI 02878 |
||||
|
Distributed by:
Avvisto Therapeutics, LLC Tiverton, RI 02878 USA |
||||
| For information for healthcare professionals, call 1-888-621-1215. | ||||
| CYCLOSET is a registered trademark of: VeroScience, LLC Tiverton, RI 02878 |
||||
| For more information, go to www.CYCLOSET.com or call 1-888-621-1215. |
Section 44425-7
Storage
Store and dispense: At 20-25°C (68-77°F) in a tight, light-resistant container. See USP Controlled Room Temperature.
10 Overdosage
With another formulation of bromocriptine mesylate, the most commonly reported signs and symptoms associated with acute overdose were nausea, vomiting, constipation, diaphoresis, dizziness, pallor, severe hypotension, malaise, confusion, lethargy, drowsiness, delusions, hallucinations, and repetitive yawning. The lethal dose has not been established.
Treatment of overdose consists of removal of the drug by emesis (if conscious), gastric lavage, activated charcoal, or saline catharsis. Careful supervision and recording of fluid intake and output is essential. Hypotension should be treated by placing the patient in the Trendelenburg position and administering intravenous fluids. If satisfactory relief of hypotension cannot be achieved by using the above measures to their fullest extent, vasopressors should be considered.
2.2 Titration
CYCLOSET should be initiated at one tablet (0.8 mg) and increased by one tablet per week until a maximum daily dose of 6 tablets (4.8 mg) or until the maximal tolerated number of tablets between 2 and 6 per day is reached.
11 Description
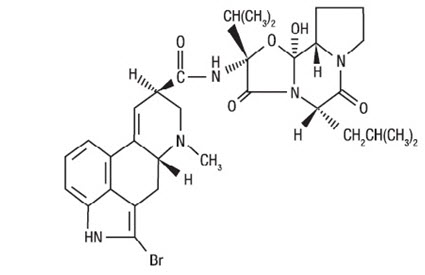
CYCLOSET Tablets contain micronized bromocriptine mesylate, an ergot derivative. Bromocriptine mesylate is chemically designated [Ergotaman-3',6',18-trione, 2-bromo-12'-hydroxy-2'-(1-methylethyl)-5'-(2-methylpropyl)-, monomethanesulfonate (salt), (5'α)-]. CYCLOSET is a single enantiomer with absolute configuration 5R, 8R, 2'R, 5'S, 11'S, 12'S.
The structural formula of bromocriptine is shown below:
Bromocriptine mesylate in CYCLOSET is a white or slightly colored micronized crystalline powder with a molecular formula of C32H40BrN5O5∙CH4SO3 and a molecular weight of 750.72. CYCLOSET Tablets contain bromocriptine mesylate USP in an amount equivalent to 0.8 mg. of bromocriptine. Each tablet contains the following inactive ingredients: lactose, corn starch, magnesium stearate, colloidal silicon dioxide, and citric acid.
5.4 Somnolence
CYCLOSET may cause somnolence. In a 52-week, randomized clinical trial, 4.3% of CYCLOSET-treated patients and 1.3% of placebo-treated patients reported somnolence. None of these events were reported as serious, and the majority of patients reported resolution of somnolence over time. Inform patients of the risk of somnolence, particularly when initiating therapy with CYCLOSET. Patients experiencing somnolence should refrain from driving or operating heavy machinery.
5.1 Hypotension
Hypotension, including orthostatic hypotension, can occur, particularly upon initiation of CYCLOSET therapy and with dose escalation. In a 52-week, randomized clinical trial of 3070 patients, hypotension was reported in 2.2% of patients randomized to CYCLOSET compared to 0.8% of patients randomized to placebo. Among CYCLOSET-treated patients reporting symptomatic hypotension, 98% were on at least one blood pressure medication compared to 73% on such medication in the total study population. In this trial, six CYCLOSET-treated patients (0.3%) reported orthostatic hypotension compared to 2 (0.2%) placebo-treated patients. All six patients were taking antihypertensive medications. Hypotension can result in syncope. In this trial, syncope due to any cause was reported in 1.6% of CYCLOSET-treated patients and 0.7% of placebo-treated patients [see Adverse Reactions (6.1)]. As a precaution, assessment of orthostatic vital signs is recommended prior to initiation of CYCLOSET and periodically thereafter. Advise patients during early treatment with CYCLOSET to make slow postural changes and to avoid situations that could lead to serious injury if syncope was to occur. Use caution in patients taking antihypertensive medications.
14.1 Monotherapy
A total of 159 adults with type 2 diabetes, overweight (body mass index ≥26.0 kg/m2 for males and ≥28.0 kg/m2 for females), and inadequate glycemic control (HbA1c 7.5-11%) participated in a 24-week, placebo-controlled, monotherapy trial that evaluated the efficacy and safety of CYCLOSET as an adjunct to diet and exercise. Mean body weight at baseline was 93 kg in the CYCLOSET group and 96 kg in the placebo group. Mean HbA1c at baseline was 9.0% in the CYCLOSET group and 8.8% in the placebo group. Mean duration of diabetes at baseline was 5 years in the CYCLOSET group and 4 years in the placebo group. Of the 80 patients in the CYCLOSET group, 69% (N = 55) achieved the maximum daily dose of 4.8 mg. CYCLOSET improved HbA1c and fasting plasma glucose compared to placebo (Table 2). Mean change from baseline in body weight was +0.2 kg in the CYCLOSET group (N = 78) and +0.5 kg in the placebo group (N = 77).
| CYCLOSET N = 80 (1.6 - 4.8 mg) |
Placebo N = 79 |
|
|---|---|---|
| P-value calculated by ANOVA | ||
| HbA1c (%) | N = 74 | N = 74 |
| Baseline (mean) | 9.0 | 8.8 |
| Change from baseline (adj. mean) | -0.1 | 0.3 |
| Difference from placebo (adj. mean) | -0.4 p = 0.05
|
|
| Fasting Plasma Glucose (mg/dL) | N = 76 | N = 75 |
| Baseline (mean) | 215 | 205 |
| Change from baseline (adj. mean) | 0 | 23 |
| Difference from placebo (adj. mean) | -23 p = 0.005
|
8.4 Pediatric Use
The safety and effectiveness of CYCLOSET in pediatric patients have not been established.
8.5 Geriatric Use
In the two clinical trials of CYCLOSET add-on to sulfonylurea therapy and in the monotherapy trial, a total of 54 patients randomized to CYCLOSET were ≥65 years old. In the 52-week safety trial, 601 of the 2,054 CYCLOSET-treated patients (29%) were ≥65 years old. No overall differences in safety or effectiveness were observed between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out [See Clinical Studies (14)].
14 Clinical Studies
A total of 3,723 patients with type 2 diabetes were randomized across 4 double-blind, placebo-controlled clinical trials conducted to evaluate the safety and glycemic efficacy of CYCLOSET. In the pooled 24-week monotherapy trial and the two 24-week add-on to sulfonylurea trials (N = 653), the mean age of the CYCLOSET-treated patients (N = 324) was 55 years, 71% were male and 73% Caucasian. In the 52-week safety trial (N = 3,070), the mean age for the entire study population was 60 years and 43% of patients were female, 68% were Caucasian, 17% were Black, 13% were Hispanic, and 1% were Asian.
In all 4 clinical trials, patients assigned to treatment with CYCLOSET received an initial dose of 0.8 mg, which was increased by 0.8 mg each week for 6 weeks (4.8 mg/day final dose) if no intolerance occurred or until the maximum tolerated dose ≥1.6 mg/day was reached. In patients with type 2 diabetes, treatment with CYCLOSET produced clinically significant improvements in HbA1c and postprandial glucose (PPG).
4 Contraindications
CYCLOSET is contraindicated in:
- Patients with known hypersensitivity to bromocriptine, ergot-related drugs, or any of the excipients in CYCLOSET.
- Patients with syncopal migraine. Bromocriptine increases the likelihood of a hypotensive episode among patients with syncopal migraine. Loss of consciousness during a migraine may reflect dopamine receptor hypersensitivity. CYCLOSET is a dopamine receptor agonist and may, therefore, potentiate the risk for syncope in these patients.
- Postpartum patients. Serious and life-threatening adverse reactions have been reported with bromocriptine use in this population [see Warnings and Precautions (5.7), Adverse Reactions (6.2)].
- Lactating patients. CYCLOSET contains bromocriptine which inhibits lactation [see Use in Specific Populations (8.2)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypotension [see Warnings and Precautions (5.1)]
- Psychotic Disorders [see Warnings and Precautions (5.2)]
- Somnolence [see Warnings and Precautions (5.4)]
- Risks in Postpartum Women [see Warnings and Precautions (5.7)]
7 Drug Interactions
- The active ingredient in CYCLOSET (bromocriptine mesylate) is highly bound to serum proteins. Therefore, CYCLOSET may increase the unbound fraction of other concomitantly used highly protein-bound therapies (e.g., salicylates, sulfonamides, chloramphenicol and probenecid), which may alter their effectiveness and risk for side effects.
- CYCLOSET is a dopamine receptor agonist. Concomitant use of dopamine receptor antagonists, such as neuroleptics (e.g., phenothiazines, butyrophenones, thioxanthenes), or metoclopramide may diminish the effectiveness of CYCLOSET, and CYCLOSET may diminish the effectiveness of these other therapies. The concurrent use of CYCLOSET with these agents has not been studied in clinical trials and is not recommended [see Warnings and Precautions (5.5)].
- CYCLOSET in combination with ergot-related drugs may cause an increase in the occurrence of ergot-related side effects, such as nausea, vomiting, and fatigue, and may also reduce the effectiveness of these ergot therapies when used to treat migraine. The concurrent use of these ergot agents within 6 hours of CYCLOSET dosing is not recommended.
- CYCLOSET is extensively metabolized by the liver via CYP3A4. Therefore, potent inhibitors or inducers of CYP3A4 may increase or reduce the circulating levels of CYCLOSET, respectively. Use caution when co-administering drugs that are inhibitors or inducers of CYP3A4. CYCLOSET dose should not exceed 1.6 mg once daily during concomitant use of a moderate CYP3A4 inhibitor (e.g., erythromycin). Concomitant use of strong CYP3A4 inhibitors (e.g., azole antimycotics, HIV protease inhibitors) with CYCLOSET should be avoided. Ensure adequate washout of the strong CYP3A4 inhibitor drug before initiating CYCLOSET treatment [see Clinical Pharmacology (12.3)].
- There are postmarketing reports of hypertension and tachycardia when bromocriptine was co-administered with sympathomimetic drugs (e.g., phenylpropanolamine and isometheptene) in postpartum women. There are limited clinical trial data supporting the safety of co-administering sympathomimetic drugs and CYCLOSET for more than 10 days. Therefore, concomitant use of these agents with CYCLOSET for more than 10 days duration is not recommended. Also, there are limited clinical trial data supporting the safety of selective 5-hydroxytryptamine1B (5-HT1B) agonists (e.g., sumatriptan) used concurrently with CYCLOSET, and the concomitant use of these agents with CYCLOSET should be avoided.
2.1 Recommended Dosing
The recommended dose of CYCLOSET is 1.6 mg to 4.8 mg administered once daily within two hours after waking in the morning. CYCLOSET should be taken with food to potentially reduce gastrointestinal side effects such as nausea. If the morning dose is missed, instruct patients to take their usual dose the following morning. Doses of CYCLOSET should not be doubled the following morning.
1 Indications and Usage
CYCLOSET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
5.2 Psychotic Disorders
In patients with severe psychotic disorders, treatment with a dopamine receptor agonist such as CYCLOSET may exacerbate the disorder or may diminish the effectiveness of drugs used to treat the disorder. Therefore, the use of CYCLOSET in patients with severe psychotic disorders is not recommended.
12.1 Mechanism of Action
CYCLOSET contains bromocriptine mesylate, a sympatholytic, dopamine D2 receptor agonist. In patients with type 2 diabetes, timed morning administration of CYCLOSET is associated with increased insulin sensitivity and glucose disposal and reduced fasting and postprandial hyperglycemia throughout the meals of the day without raising plasma insulin levels.
5 Warnings and Precautions
- Hypotension: Can cause orthostatic hypotension and syncope, particularly upon initiation or dose escalation. Use caution in patients taking antihypertensive medications. Assess orthostatic vital signs prior to initiation of CYCLOSET and periodically thereafter. Advise patients during early treatment to avoid situations that could lead to injury if syncope was to occur. (5.1, 6.1)
- Psychosis: May exacerbate psychotic disorders or reduce the effectiveness of drugs that treat psychosis. Use in patients with severe psychotic disorders is not recommended. (5.2)
- Impulse control/compulsive behaviors: Ask patients or their caregivers about new or increased gambling urges, sexual urges, uncontrolled spending, or other urges while being treated with CYCLOSET. Consider dose reduction or stopping CYCLOSET if a patient develops such urges. Use of CYCLOSET in patients with impulse control/compulsive behaviors is not recommended. (5.3, 6.2).
- Somnolence: May cause somnolence. Advise patients not to operate heavy machinery if symptoms of somnolence occur. (5.4)
- Interaction with dopamine antagonists: Concomitant use with dopamine antagonists such as neuroleptic agents may diminish the effectiveness of both drugs. Concomitant use is not recommended. (5.5, 7)
- Other dopamine receptor agonists: Effectiveness and safety are unknown in patients already taking dopamine receptor agonists for other indications. Concomitant use is not recommended. (5.6)
- Risks in Postpartum Patients: Serious and life-threatening adverse reactions have been reported. (5.7, 6.2)
2 Dosage and Administration
- Taken within two hours after waking in the morning with food (2.1)
- Initial dose is one tablet (0.8 mg) daily increased weekly by one tablet until maximal tolerated daily dose of 1.6 to 4.8 mg is achieved. (2.2)
- Limit dose to 1.6 mg daily during concomitant use of a moderate CYP3A4 inhibitor. Avoid concomitant use with strong CYP3A4 inhibitors. (2.3)
3 Dosage Forms and Strengths
0.8 mg tablets are white and round, imprinted with "C" on one side and "9" on the other.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of other formulations of bromocriptine mesylate for indications for which CYCLOSET is not approved (e.g., hyperprolactinemia, acromegaly, and Parkinson's disease), generally at doses higher than those approved for the treatment of type 2 diabetes. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
Pediatrics: Safety and effectiveness have not been established. (8.4)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates reported in one clinical trial may not be easily compared to those rates reported in another clinical trial and may not reflect the rates actually observed in clinical practice.
The CYCLOSET safety trial was a 52-week, placebo-controlled study. A total of 3,070 patients were randomized to CYCLOSET (titrated to 1.6 to 4.8 mg daily, as tolerated) or placebo. The study population had a mean baseline age of 60 years (range 27-80) and 33% were 65 years of age or older. Approximately 43% of the patients were female, 68% were Caucasian, 17% were Black, 13% were Hispanic, and 1% were Asian. The mean baseline body mass index was 32 kg/m2. The mean duration of diabetes at baseline was 8 years, and the mean baseline HbA1c was 7.0% with a mean baseline fasting plasma glucose of 142 mg/dL. At baseline, 12% of patients were treated with diet only, 40% were treated with one oral antidiabetic agent, 33% were treated with two oral antidiabetic agents, and 16% were treated with insulin alone or insulin in combination with an oral antidiabetic agent. At baseline, 76% of patients reported a history of hypercholesterolemia, 75% reported a history of hypertension, 11% reported a history of revascularization surgery, 10% reported a history of myocardial infarction, 10% reported a history of angina, and 5% reported a history of stroke. Forty-seven percent of the CYCLOSET-treated patients and 32% of the placebo-treated patients prematurely discontinued treatment.
Table 1 summarizes the adverse reactions reported in ≥5% of patients treated with CYCLOSET in clinical trials regardless of investigator assessment of causality. The most commonly reported adverse reactions (nausea, fatigue, vomiting, headache, dizziness) lasted a median of 14 days and were more likely to occur during the initial titration of CYCLOSET. There were no differences in the pattern of common adverse reactions across race groups or age groups (<65 years old vs. >65 years old). In the 52-week CYCLOSET safety trial, 11.5% of CYCLOSET-treated women compared to 3.6% of placebo-treated women reported vomiting. In this same trial, 5.4% of CYCLOSET-treated men compared to 2.8% of placebo-treated men reported vomiting.
| Monotherapy | CYCLOSET 1.6 mg – 4.8 mg N (%) |
Placebo N (%) |
|---|---|---|
| N = 159 | N = 80 | N = 79 |
| Nausea | 26 (32.5) | 6 (7.6) |
| Rhinitis | 11 (13.8) | 3 (3.8) |
| Headache | 10 (12.5) | 7 (8.9) |
| Asthenia | 10 (12.5) | 5 (6.3) |
| Dizziness | 10 (12.5) | 6 (7.6) |
| Constipation | 9 (11.3) | 3 (3.8) |
| Sinusitis | 8 (10.0) | 2 (2.5) |
| Diarrhea | 7 (8.8) | 4 (5.1) |
| Amblyopia | 6 (7.5) | 1 (1.3) |
| Dyspepsia | 6 (7.5) | 2 (2.5) |
| Vomiting | 5 (6.3) | 1 (1.3) |
| Infection | 5 (6.3) | 4 (5.1) |
| Anorexia | 4 (5.0) | 1 (1.3) |
| Adjunct to Sulfonylurea (2 pooled 24-week studies) | ||
| N = 494 | N = 244 | N = 250 |
| Nausea | 62 (25.4) | 12 (4.8) |
| Asthenia | 46 (18.9) | 20 (8.0) |
| Headache | 41 (16.8) | 40 (16.0) |
| Flu syndrome | 23 (9.4) | 19 (7.6) |
| Constipation | 24 (9.8) | 11 (4.4) |
| Cold | 20 (8.2) | 20 (8.0) |
| Dizziness | 29 (11.9) | 14 (5.6) |
| Rhinitis | 26 (10.7) | 12 (4.8) |
| Sinusitis | 18 (7.4) | 16 (6.4) |
| Somnolence | 16 (6.6) | 5 (2.0) |
| Vomiting | 13 (5.3) | 8 (3.2) |
| Amblyopia | 13 (5.3) | 6 (2.4) |
|
52-Week Safety Trial The Safety Trial enrolled patients treated with diet or no more than 2 antidiabetic medications (metformin, insulin secretagogues such as a sulfonylurea, thiazolidinediones, alpha glucosidase inhibitors, and/or insulin).
|
||
| N = 3070 | N = 2054 | N = 1016 |
| Nausea | 661 (32.2) | 77 (7.6) |
| Dizziness | 303 (14.8) | 93 (9.2) |
| Fatigue | 285 (13.9) | 68 (6.7) |
| Headache | 235 (11.4) | 84 (8.3) |
| Vomiting | 167 (8.1) | 32 (3.1) |
| Diarrhea | 167 (8.1) | 81 (8.0) |
| Constipation | 119 (5.8) | 52 (5.1) |
2.3 Use With Concomitant Therapy
CYCLOSET dose should not exceed 1.6 mg once daily during concomitant use of a moderate CYP3A4 inhibitor (e.g., erythromycin). Avoid concomitant use of CYCLOSET and strong CYP3A4 inhibitors (e.g., azole antimycotics, HIV protease inhibitors) and ensure adequate washout of the strong CYP3A4 inhibitor drug before initiating CYCLOSET treatment [see Drug Interactions (7), Clinical Pharmacology (12.3)].
5.7 Risks in Postpartum Patients
CYCLOSET is contraindicated in postpartum patients. Serious and life-threatening adverse reactions including hypertension, myocardial infarction, seizures, stroke and psychosis have been reported postmarketing in postpartum women who were administered bromocriptine for inhibition of lactation [see Adverse Reactions (6.2)]. These risks may be higher in postpartum patients with cardiovascular disease. The indication for use of bromocriptine for inhibition of postpartum lactation was withdrawn from bromocriptine-containing products and is not approved for CYCLOSET.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
CYCLOSET 0.8 mg tablets are WHITE and round with "C" on one side and "9" on the other.
The tablets are supplied as follows:
NDC 73515-123-30 unit-of-use bottles of 200
NDC 73515-123-21 unit-of-use bottles of 21 (samples only).
5.6 Other Dopamine Receptor Agonists
Other dopamine receptor agonists are indicated for the treatment of Parkinson's disease, hyperprolactinemia, restless leg syndrome, acromegaly, and other disorders. The effectiveness and safety of CYCLOSET in patients who are already taking one of these other dopamine receptor agonists is unknown. Concomitant use is not recommended.
5.3 Impulse Control/compulsive Behaviors
There have been reports of patients experiencing intense urges to gamble, increased sexual urges, intense urges to spend money uncontrollably, and/or other intense urges, and the inability to control these urges while taking one or more of the medications, including bromocriptine, that increase central dopaminergic tone. In some cases, although not all, these urges were reported to have stopped when the dose was reduced, or the medication was discontinued. Because patients may not recognize these behaviors as abnormal, it is important to specifically ask patients or their caregivers about the development of new or increased gambling urges, sexual urges, uncontrolled spending, or other urges while being treated with CYCLOSET. Consider dose reduction or discontinuation if a patient develops such urges.
14.3 Changes in Lipids and Blood Pressure
CYCLOSET does not have an unfavorable effect on fasting plasma lipids.
CYCLOSET has not demonstrated an unfavorable hypertensive effect on blood pressure. Hypotension has been reported with use of CYCLOSET in clinical trials [see Warnings and Precautions (5.1)].
5.5 Interaction With Dopamine Receptor Antagonists
Dopamine receptor antagonists, including neuroleptic agents that have dopamine D2 receptor antagonist properties (e.g., clozapine, olanzapine, ziprasidone), may reduce the effectiveness of CYCLOSET, and CYCLOSET may reduce the effectiveness of these agents. CYCLOSET has not been studied in patients taking neuroleptic drugs. The concomitant use of CYCLOSET and dopamine receptor antagonists, including neuroleptic drugs, is not recommended.
Principal Display Panel 0.8 Mg Tablet Bottle Label
NDC 73515-123-30
CYCLOSET®
bromocriptine mesylate tablets
0.8 mg
200 Tablets
Rx only
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
- CYCLOSET should not be used to treat type 1 diabetes or diabetic ketoacidosis.
- Limited efficacy data in combination with thiazolidinediones.
- Efficacy has not been confirmed in combination with insulin.
Section 42230-3 (42230-3)
| Patient Information CYCLOSET® (Sikloset) (bromocriptine mesylate tablets) for oral use |
||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 08/2020 | |||
| Read this Patient Information that comes with CYCLOSET before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. | ||||
| What is CYCLOSET? | ||||
|
||||
| Who should not take CYCLOSET? | ||||
| Do not take CYCLOSET if you: | ||||
|
||||
| Talk to your healthcare provider before taking CYCLOSET if you have any of these conditions. | ||||
| What should I tell my healthcare provider before taking CYCLOSET? | ||||
| Before taking CYCLOSET, tell your healthcare provider about all of your medical conditions, including if you: | ||||
|
||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | ||||
| Especially tell your healthcare provider if you take: | ||||
|
||||
| Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure. | ||||
| CYCLOSET may affect the way other medicines work, and other medicines may affect how CYCLOSET works. | ||||
| Know the medicines you take. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine. | ||||
| How should I take CYCLOSET? | ||||
|
||||
| What are the possible side effects of CYCLOSET? | ||||
| CYCLOSET may cause serious side effects, including: | ||||
|
||||
| The most common side effects of CYCLOSET include: | ||||
|
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. | ||||
| These are not all the possible side effects of CYCLOSET. For more information, ask your healthcare provider or pharmacist. | ||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
| How should I store CYCLOSET? | ||||
|
||||
| Keep CYCLOSET and all medicines out of the reach of children. | ||||
| General information about the safe and effective use of CYCLOSET. | ||||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use CYCLOSET for a condition for which it was not prescribed. Do not give CYCLOSET to other people, even if they have the same symptoms that you have. It may harm them. | ||||
| This leaflet summarizes the most important information about CYCLOSET. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about CYCLOSET that is written for health professionals. | ||||
| What are the ingredients in CYCLOSET? | ||||
| Active ingredient: bromocriptine mesylate | ||||
| Inactive ingredients: lactose, corn starch, magnesium stearate, colloidal silicon dioxide, and citric acid | ||||
|
Manufactured for:
VeroScience, LLC Tiverton, RI 02878 |
||||
|
Distributed by:
Avvisto Therapeutics, LLC Tiverton, RI 02878 USA |
||||
| For information for healthcare professionals, call 1-888-621-1215. | ||||
| CYCLOSET is a registered trademark of: VeroScience, LLC Tiverton, RI 02878 |
||||
| For more information, go to www.CYCLOSET.com or call 1-888-621-1215. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
Store and dispense: At 20-25°C (68-77°F) in a tight, light-resistant container. See USP Controlled Room Temperature.
10 Overdosage (10 OVERDOSAGE)
With another formulation of bromocriptine mesylate, the most commonly reported signs and symptoms associated with acute overdose were nausea, vomiting, constipation, diaphoresis, dizziness, pallor, severe hypotension, malaise, confusion, lethargy, drowsiness, delusions, hallucinations, and repetitive yawning. The lethal dose has not been established.
Treatment of overdose consists of removal of the drug by emesis (if conscious), gastric lavage, activated charcoal, or saline catharsis. Careful supervision and recording of fluid intake and output is essential. Hypotension should be treated by placing the patient in the Trendelenburg position and administering intravenous fluids. If satisfactory relief of hypotension cannot be achieved by using the above measures to their fullest extent, vasopressors should be considered.
2.2 Titration
CYCLOSET should be initiated at one tablet (0.8 mg) and increased by one tablet per week until a maximum daily dose of 6 tablets (4.8 mg) or until the maximal tolerated number of tablets between 2 and 6 per day is reached.
11 Description (11 DESCRIPTION)
CYCLOSET Tablets contain micronized bromocriptine mesylate, an ergot derivative. Bromocriptine mesylate is chemically designated [Ergotaman-3',6',18-trione, 2-bromo-12'-hydroxy-2'-(1-methylethyl)-5'-(2-methylpropyl)-, monomethanesulfonate (salt), (5'α)-]. CYCLOSET is a single enantiomer with absolute configuration 5R, 8R, 2'R, 5'S, 11'S, 12'S.
The structural formula of bromocriptine is shown below:
Bromocriptine mesylate in CYCLOSET is a white or slightly colored micronized crystalline powder with a molecular formula of C32H40BrN5O5∙CH4SO3 and a molecular weight of 750.72. CYCLOSET Tablets contain bromocriptine mesylate USP in an amount equivalent to 0.8 mg. of bromocriptine. Each tablet contains the following inactive ingredients: lactose, corn starch, magnesium stearate, colloidal silicon dioxide, and citric acid.
5.4 Somnolence
CYCLOSET may cause somnolence. In a 52-week, randomized clinical trial, 4.3% of CYCLOSET-treated patients and 1.3% of placebo-treated patients reported somnolence. None of these events were reported as serious, and the majority of patients reported resolution of somnolence over time. Inform patients of the risk of somnolence, particularly when initiating therapy with CYCLOSET. Patients experiencing somnolence should refrain from driving or operating heavy machinery.
5.1 Hypotension
Hypotension, including orthostatic hypotension, can occur, particularly upon initiation of CYCLOSET therapy and with dose escalation. In a 52-week, randomized clinical trial of 3070 patients, hypotension was reported in 2.2% of patients randomized to CYCLOSET compared to 0.8% of patients randomized to placebo. Among CYCLOSET-treated patients reporting symptomatic hypotension, 98% were on at least one blood pressure medication compared to 73% on such medication in the total study population. In this trial, six CYCLOSET-treated patients (0.3%) reported orthostatic hypotension compared to 2 (0.2%) placebo-treated patients. All six patients were taking antihypertensive medications. Hypotension can result in syncope. In this trial, syncope due to any cause was reported in 1.6% of CYCLOSET-treated patients and 0.7% of placebo-treated patients [see Adverse Reactions (6.1)]. As a precaution, assessment of orthostatic vital signs is recommended prior to initiation of CYCLOSET and periodically thereafter. Advise patients during early treatment with CYCLOSET to make slow postural changes and to avoid situations that could lead to serious injury if syncope was to occur. Use caution in patients taking antihypertensive medications.
14.1 Monotherapy
A total of 159 adults with type 2 diabetes, overweight (body mass index ≥26.0 kg/m2 for males and ≥28.0 kg/m2 for females), and inadequate glycemic control (HbA1c 7.5-11%) participated in a 24-week, placebo-controlled, monotherapy trial that evaluated the efficacy and safety of CYCLOSET as an adjunct to diet and exercise. Mean body weight at baseline was 93 kg in the CYCLOSET group and 96 kg in the placebo group. Mean HbA1c at baseline was 9.0% in the CYCLOSET group and 8.8% in the placebo group. Mean duration of diabetes at baseline was 5 years in the CYCLOSET group and 4 years in the placebo group. Of the 80 patients in the CYCLOSET group, 69% (N = 55) achieved the maximum daily dose of 4.8 mg. CYCLOSET improved HbA1c and fasting plasma glucose compared to placebo (Table 2). Mean change from baseline in body weight was +0.2 kg in the CYCLOSET group (N = 78) and +0.5 kg in the placebo group (N = 77).
| CYCLOSET N = 80 (1.6 - 4.8 mg) |
Placebo N = 79 |
|
|---|---|---|
| P-value calculated by ANOVA | ||
| HbA1c (%) | N = 74 | N = 74 |
| Baseline (mean) | 9.0 | 8.8 |
| Change from baseline (adj. mean) | -0.1 | 0.3 |
| Difference from placebo (adj. mean) | -0.4 p = 0.05
|
|
| Fasting Plasma Glucose (mg/dL) | N = 76 | N = 75 |
| Baseline (mean) | 215 | 205 |
| Change from baseline (adj. mean) | 0 | 23 |
| Difference from placebo (adj. mean) | -23 p = 0.005
|
8.4 Pediatric Use
The safety and effectiveness of CYCLOSET in pediatric patients have not been established.
8.5 Geriatric Use
In the two clinical trials of CYCLOSET add-on to sulfonylurea therapy and in the monotherapy trial, a total of 54 patients randomized to CYCLOSET were ≥65 years old. In the 52-week safety trial, 601 of the 2,054 CYCLOSET-treated patients (29%) were ≥65 years old. No overall differences in safety or effectiveness were observed between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out [See Clinical Studies (14)].
14 Clinical Studies (14 CLINICAL STUDIES)
A total of 3,723 patients with type 2 diabetes were randomized across 4 double-blind, placebo-controlled clinical trials conducted to evaluate the safety and glycemic efficacy of CYCLOSET. In the pooled 24-week monotherapy trial and the two 24-week add-on to sulfonylurea trials (N = 653), the mean age of the CYCLOSET-treated patients (N = 324) was 55 years, 71% were male and 73% Caucasian. In the 52-week safety trial (N = 3,070), the mean age for the entire study population was 60 years and 43% of patients were female, 68% were Caucasian, 17% were Black, 13% were Hispanic, and 1% were Asian.
In all 4 clinical trials, patients assigned to treatment with CYCLOSET received an initial dose of 0.8 mg, which was increased by 0.8 mg each week for 6 weeks (4.8 mg/day final dose) if no intolerance occurred or until the maximum tolerated dose ≥1.6 mg/day was reached. In patients with type 2 diabetes, treatment with CYCLOSET produced clinically significant improvements in HbA1c and postprandial glucose (PPG).
4 Contraindications (4 CONTRAINDICATIONS)
CYCLOSET is contraindicated in:
- Patients with known hypersensitivity to bromocriptine, ergot-related drugs, or any of the excipients in CYCLOSET.
- Patients with syncopal migraine. Bromocriptine increases the likelihood of a hypotensive episode among patients with syncopal migraine. Loss of consciousness during a migraine may reflect dopamine receptor hypersensitivity. CYCLOSET is a dopamine receptor agonist and may, therefore, potentiate the risk for syncope in these patients.
- Postpartum patients. Serious and life-threatening adverse reactions have been reported with bromocriptine use in this population [see Warnings and Precautions (5.7), Adverse Reactions (6.2)].
- Lactating patients. CYCLOSET contains bromocriptine which inhibits lactation [see Use in Specific Populations (8.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypotension [see Warnings and Precautions (5.1)]
- Psychotic Disorders [see Warnings and Precautions (5.2)]
- Somnolence [see Warnings and Precautions (5.4)]
- Risks in Postpartum Women [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- The active ingredient in CYCLOSET (bromocriptine mesylate) is highly bound to serum proteins. Therefore, CYCLOSET may increase the unbound fraction of other concomitantly used highly protein-bound therapies (e.g., salicylates, sulfonamides, chloramphenicol and probenecid), which may alter their effectiveness and risk for side effects.
- CYCLOSET is a dopamine receptor agonist. Concomitant use of dopamine receptor antagonists, such as neuroleptics (e.g., phenothiazines, butyrophenones, thioxanthenes), or metoclopramide may diminish the effectiveness of CYCLOSET, and CYCLOSET may diminish the effectiveness of these other therapies. The concurrent use of CYCLOSET with these agents has not been studied in clinical trials and is not recommended [see Warnings and Precautions (5.5)].
- CYCLOSET in combination with ergot-related drugs may cause an increase in the occurrence of ergot-related side effects, such as nausea, vomiting, and fatigue, and may also reduce the effectiveness of these ergot therapies when used to treat migraine. The concurrent use of these ergot agents within 6 hours of CYCLOSET dosing is not recommended.
- CYCLOSET is extensively metabolized by the liver via CYP3A4. Therefore, potent inhibitors or inducers of CYP3A4 may increase or reduce the circulating levels of CYCLOSET, respectively. Use caution when co-administering drugs that are inhibitors or inducers of CYP3A4. CYCLOSET dose should not exceed 1.6 mg once daily during concomitant use of a moderate CYP3A4 inhibitor (e.g., erythromycin). Concomitant use of strong CYP3A4 inhibitors (e.g., azole antimycotics, HIV protease inhibitors) with CYCLOSET should be avoided. Ensure adequate washout of the strong CYP3A4 inhibitor drug before initiating CYCLOSET treatment [see Clinical Pharmacology (12.3)].
- There are postmarketing reports of hypertension and tachycardia when bromocriptine was co-administered with sympathomimetic drugs (e.g., phenylpropanolamine and isometheptene) in postpartum women. There are limited clinical trial data supporting the safety of co-administering sympathomimetic drugs and CYCLOSET for more than 10 days. Therefore, concomitant use of these agents with CYCLOSET for more than 10 days duration is not recommended. Also, there are limited clinical trial data supporting the safety of selective 5-hydroxytryptamine1B (5-HT1B) agonists (e.g., sumatriptan) used concurrently with CYCLOSET, and the concomitant use of these agents with CYCLOSET should be avoided.
2.1 Recommended Dosing
The recommended dose of CYCLOSET is 1.6 mg to 4.8 mg administered once daily within two hours after waking in the morning. CYCLOSET should be taken with food to potentially reduce gastrointestinal side effects such as nausea. If the morning dose is missed, instruct patients to take their usual dose the following morning. Doses of CYCLOSET should not be doubled the following morning.
1 Indications and Usage (1 INDICATIONS AND USAGE)
CYCLOSET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
5.2 Psychotic Disorders
In patients with severe psychotic disorders, treatment with a dopamine receptor agonist such as CYCLOSET may exacerbate the disorder or may diminish the effectiveness of drugs used to treat the disorder. Therefore, the use of CYCLOSET in patients with severe psychotic disorders is not recommended.
12.1 Mechanism of Action
CYCLOSET contains bromocriptine mesylate, a sympatholytic, dopamine D2 receptor agonist. In patients with type 2 diabetes, timed morning administration of CYCLOSET is associated with increased insulin sensitivity and glucose disposal and reduced fasting and postprandial hyperglycemia throughout the meals of the day without raising plasma insulin levels.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypotension: Can cause orthostatic hypotension and syncope, particularly upon initiation or dose escalation. Use caution in patients taking antihypertensive medications. Assess orthostatic vital signs prior to initiation of CYCLOSET and periodically thereafter. Advise patients during early treatment to avoid situations that could lead to injury if syncope was to occur. (5.1, 6.1)
- Psychosis: May exacerbate psychotic disorders or reduce the effectiveness of drugs that treat psychosis. Use in patients with severe psychotic disorders is not recommended. (5.2)
- Impulse control/compulsive behaviors: Ask patients or their caregivers about new or increased gambling urges, sexual urges, uncontrolled spending, or other urges while being treated with CYCLOSET. Consider dose reduction or stopping CYCLOSET if a patient develops such urges. Use of CYCLOSET in patients with impulse control/compulsive behaviors is not recommended. (5.3, 6.2).
- Somnolence: May cause somnolence. Advise patients not to operate heavy machinery if symptoms of somnolence occur. (5.4)
- Interaction with dopamine antagonists: Concomitant use with dopamine antagonists such as neuroleptic agents may diminish the effectiveness of both drugs. Concomitant use is not recommended. (5.5, 7)
- Other dopamine receptor agonists: Effectiveness and safety are unknown in patients already taking dopamine receptor agonists for other indications. Concomitant use is not recommended. (5.6)
- Risks in Postpartum Patients: Serious and life-threatening adverse reactions have been reported. (5.7, 6.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Taken within two hours after waking in the morning with food (2.1)
- Initial dose is one tablet (0.8 mg) daily increased weekly by one tablet until maximal tolerated daily dose of 1.6 to 4.8 mg is achieved. (2.2)
- Limit dose to 1.6 mg daily during concomitant use of a moderate CYP3A4 inhibitor. Avoid concomitant use with strong CYP3A4 inhibitors. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
0.8 mg tablets are white and round, imprinted with "C" on one side and "9" on the other.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of other formulations of bromocriptine mesylate for indications for which CYCLOSET is not approved (e.g., hyperprolactinemia, acromegaly, and Parkinson's disease), generally at doses higher than those approved for the treatment of type 2 diabetes. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pediatrics: Safety and effectiveness have not been established. (8.4)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates reported in one clinical trial may not be easily compared to those rates reported in another clinical trial and may not reflect the rates actually observed in clinical practice.
The CYCLOSET safety trial was a 52-week, placebo-controlled study. A total of 3,070 patients were randomized to CYCLOSET (titrated to 1.6 to 4.8 mg daily, as tolerated) or placebo. The study population had a mean baseline age of 60 years (range 27-80) and 33% were 65 years of age or older. Approximately 43% of the patients were female, 68% were Caucasian, 17% were Black, 13% were Hispanic, and 1% were Asian. The mean baseline body mass index was 32 kg/m2. The mean duration of diabetes at baseline was 8 years, and the mean baseline HbA1c was 7.0% with a mean baseline fasting plasma glucose of 142 mg/dL. At baseline, 12% of patients were treated with diet only, 40% were treated with one oral antidiabetic agent, 33% were treated with two oral antidiabetic agents, and 16% were treated with insulin alone or insulin in combination with an oral antidiabetic agent. At baseline, 76% of patients reported a history of hypercholesterolemia, 75% reported a history of hypertension, 11% reported a history of revascularization surgery, 10% reported a history of myocardial infarction, 10% reported a history of angina, and 5% reported a history of stroke. Forty-seven percent of the CYCLOSET-treated patients and 32% of the placebo-treated patients prematurely discontinued treatment.
Table 1 summarizes the adverse reactions reported in ≥5% of patients treated with CYCLOSET in clinical trials regardless of investigator assessment of causality. The most commonly reported adverse reactions (nausea, fatigue, vomiting, headache, dizziness) lasted a median of 14 days and were more likely to occur during the initial titration of CYCLOSET. There were no differences in the pattern of common adverse reactions across race groups or age groups (<65 years old vs. >65 years old). In the 52-week CYCLOSET safety trial, 11.5% of CYCLOSET-treated women compared to 3.6% of placebo-treated women reported vomiting. In this same trial, 5.4% of CYCLOSET-treated men compared to 2.8% of placebo-treated men reported vomiting.
| Monotherapy | CYCLOSET 1.6 mg – 4.8 mg N (%) |
Placebo N (%) |
|---|---|---|
| N = 159 | N = 80 | N = 79 |
| Nausea | 26 (32.5) | 6 (7.6) |
| Rhinitis | 11 (13.8) | 3 (3.8) |
| Headache | 10 (12.5) | 7 (8.9) |
| Asthenia | 10 (12.5) | 5 (6.3) |
| Dizziness | 10 (12.5) | 6 (7.6) |
| Constipation | 9 (11.3) | 3 (3.8) |
| Sinusitis | 8 (10.0) | 2 (2.5) |
| Diarrhea | 7 (8.8) | 4 (5.1) |
| Amblyopia | 6 (7.5) | 1 (1.3) |
| Dyspepsia | 6 (7.5) | 2 (2.5) |
| Vomiting | 5 (6.3) | 1 (1.3) |
| Infection | 5 (6.3) | 4 (5.1) |
| Anorexia | 4 (5.0) | 1 (1.3) |
| Adjunct to Sulfonylurea (2 pooled 24-week studies) | ||
| N = 494 | N = 244 | N = 250 |
| Nausea | 62 (25.4) | 12 (4.8) |
| Asthenia | 46 (18.9) | 20 (8.0) |
| Headache | 41 (16.8) | 40 (16.0) |
| Flu syndrome | 23 (9.4) | 19 (7.6) |
| Constipation | 24 (9.8) | 11 (4.4) |
| Cold | 20 (8.2) | 20 (8.0) |
| Dizziness | 29 (11.9) | 14 (5.6) |
| Rhinitis | 26 (10.7) | 12 (4.8) |
| Sinusitis | 18 (7.4) | 16 (6.4) |
| Somnolence | 16 (6.6) | 5 (2.0) |
| Vomiting | 13 (5.3) | 8 (3.2) |
| Amblyopia | 13 (5.3) | 6 (2.4) |
|
52-Week Safety Trial The Safety Trial enrolled patients treated with diet or no more than 2 antidiabetic medications (metformin, insulin secretagogues such as a sulfonylurea, thiazolidinediones, alpha glucosidase inhibitors, and/or insulin).
|
||
| N = 3070 | N = 2054 | N = 1016 |
| Nausea | 661 (32.2) | 77 (7.6) |
| Dizziness | 303 (14.8) | 93 (9.2) |
| Fatigue | 285 (13.9) | 68 (6.7) |
| Headache | 235 (11.4) | 84 (8.3) |
| Vomiting | 167 (8.1) | 32 (3.1) |
| Diarrhea | 167 (8.1) | 81 (8.0) |
| Constipation | 119 (5.8) | 52 (5.1) |
2.3 Use With Concomitant Therapy (2.3 Use with Concomitant Therapy)
CYCLOSET dose should not exceed 1.6 mg once daily during concomitant use of a moderate CYP3A4 inhibitor (e.g., erythromycin). Avoid concomitant use of CYCLOSET and strong CYP3A4 inhibitors (e.g., azole antimycotics, HIV protease inhibitors) and ensure adequate washout of the strong CYP3A4 inhibitor drug before initiating CYCLOSET treatment [see Drug Interactions (7), Clinical Pharmacology (12.3)].
5.7 Risks in Postpartum Patients
CYCLOSET is contraindicated in postpartum patients. Serious and life-threatening adverse reactions including hypertension, myocardial infarction, seizures, stroke and psychosis have been reported postmarketing in postpartum women who were administered bromocriptine for inhibition of lactation [see Adverse Reactions (6.2)]. These risks may be higher in postpartum patients with cardiovascular disease. The indication for use of bromocriptine for inhibition of postpartum lactation was withdrawn from bromocriptine-containing products and is not approved for CYCLOSET.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
CYCLOSET 0.8 mg tablets are WHITE and round with "C" on one side and "9" on the other.
The tablets are supplied as follows:
NDC 73515-123-30 unit-of-use bottles of 200
NDC 73515-123-21 unit-of-use bottles of 21 (samples only).
5.6 Other Dopamine Receptor Agonists
Other dopamine receptor agonists are indicated for the treatment of Parkinson's disease, hyperprolactinemia, restless leg syndrome, acromegaly, and other disorders. The effectiveness and safety of CYCLOSET in patients who are already taking one of these other dopamine receptor agonists is unknown. Concomitant use is not recommended.
5.3 Impulse Control/compulsive Behaviors (5.3 Impulse Control/Compulsive Behaviors)
There have been reports of patients experiencing intense urges to gamble, increased sexual urges, intense urges to spend money uncontrollably, and/or other intense urges, and the inability to control these urges while taking one or more of the medications, including bromocriptine, that increase central dopaminergic tone. In some cases, although not all, these urges were reported to have stopped when the dose was reduced, or the medication was discontinued. Because patients may not recognize these behaviors as abnormal, it is important to specifically ask patients or their caregivers about the development of new or increased gambling urges, sexual urges, uncontrolled spending, or other urges while being treated with CYCLOSET. Consider dose reduction or discontinuation if a patient develops such urges.
14.3 Changes in Lipids and Blood Pressure
CYCLOSET does not have an unfavorable effect on fasting plasma lipids.
CYCLOSET has not demonstrated an unfavorable hypertensive effect on blood pressure. Hypotension has been reported with use of CYCLOSET in clinical trials [see Warnings and Precautions (5.1)].
5.5 Interaction With Dopamine Receptor Antagonists (5.5 Interaction with Dopamine Receptor Antagonists)
Dopamine receptor antagonists, including neuroleptic agents that have dopamine D2 receptor antagonist properties (e.g., clozapine, olanzapine, ziprasidone), may reduce the effectiveness of CYCLOSET, and CYCLOSET may reduce the effectiveness of these agents. CYCLOSET has not been studied in patients taking neuroleptic drugs. The concomitant use of CYCLOSET and dopamine receptor antagonists, including neuroleptic drugs, is not recommended.
Principal Display Panel 0.8 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 0.8 mg Tablet Bottle Label)
NDC 73515-123-30
CYCLOSET®
bromocriptine mesylate tablets
0.8 mg
200 Tablets
Rx only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:16.141124 · Updated: 2026-03-14T22:43:09.785908