These Highlights Do Not Include All The Information Needed To Use Tyenne Safely And Effectively. See Full Prescribing Information For Tyenne.
e400bbe6-43c5-4de6-82b9-cb44ce3cd906
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.6 ) 12/2024
Indications and Usage
TYENNE ® (tocilizumab-aazg) is an interleukin-6 (IL-6) receptor antagonist indicated for treatment of: Rheumatoid Arthritis (RA) ( 1.1 ) • Adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs). Giant Cell Arteritis (GCA) ( 1.2 ) • Adult patients with giant cell arteritis. Polyarticular Juvenile Idiopathic Arthritis (PJIA) ( 1.3 ) • Patients 2 years of age and older with active polyarticular juvenile idiopathic arthritis. Systemic Juvenile Idiopathic Arthritis (SJIA) ( 1.4 ) • Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
Dosage and Administration
For RA, pJIA and sJIA, TYENNE may be used alone or in combination with methotrexate; and in RA, other non-biologic DMARDs may be used. ( 2 ) General Administration and Dosing Information ( 2.1 ) • RA, GCA, PJIA and SJIA - It is recommended that TYENNE not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm 3 , platelet count below 100,000 per mm 3 , or ALT or AST above 1.5 times the upper limit of normal (ULN) ( 5.3 , 5.4 ). • In RA patients, TYENNE doses exceeding 800 mg per infusion are not recommended. ( 2.2 , 2.7 , 12.3 ) • In GCA patients, TYENNE doses exceeding 600 mg per infusion are not recommended. ( 2.3 , 12.3 ) Rheumatoid Arthritis ( 2.2 ) Recommended Adult Intravenous Dosage: When used in combination with non-biologic DMARDs or as monotherapy the recommended starting dose is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response. Recommended Adult Subcutaneous Dosage: Patients less than 100 kg weight 162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response Patients at or above 100 kg weight 162 mg administered subcutaneously every week Giant Cell Arteritis ( 2.3 ) Recommended Adult Intravenous Dosage: The recommended dose is 6 mg per kg every 4 weeks in combination with a tapering course of glucocorticoids. TYENNE can be used alone following discontinuation of glucocorticoids. Recommended Adult Subcutaneous Dosage: The recommended dose is 162 mg given once every week as a subcutaneous injection, in combination with a tapering course of glucocorticoids. A dose of 162 mg given once every other week as a subcutaneous injection, in combination with a tapering course of glucocorticoids, may be prescribed based on clinical considerations. TYENNE can be used alone following discontinuation of glucocorticoids. Polyarticular Juvenile Idiopathic Arthritis ( 2.4 ) Recommended Intravenous PJIA Dosage Every 4 Weeks Patients less than 30 kg weight 10 mg per kg Patients at or above 30 kg weight 8 mg per kg Recommended Subcutaneous PJIA Dosage Patients less than 30 kg weight 162 mg once every three weeks Patients at or above 30 kg weight 162 mg once every two weeks Systemic Juvenile Idiopathic Arthritis ( 2.5 ) Recommended Intravenous SJIA Dosage Every 2 Weeks Patients less than 30 kg weight 12 mg per kg Patients at or above 30 kg weight 8 mg per kg Recommended Subcutaneous SJIA Dosage Patients less than 30 kg weight 162 mg every two weeks Patients at or above 30 kg weight 162 mg every week Administration of Intravenous formulation ( 2.6 ) • For patients with RA, GCA, PJIA and SJIA patients at or above 30 kg, dilute to 100 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique. • For PJIA and SJIA patients less than 30 kg, dilute to 50 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique. • Administer as a single intravenous drip infusion over 1 hour; do not administer as bolus or push. Administration of Subcutaneous formulation ( 2.7 ) • Follow the Instructions for Use for prefilled syringe and prefilled autoinjector Dose Modifications ( 2.8 ) Recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia.
Warnings and Precautions
• Serious Infections - do not administer TYENNE during an active infection, including localized infections. If a serious infection develops, interrupt TYENNE until the infection is controlled. ( 5.1 ) • Gastrointestinal (GI) perforation-use with caution in patients who may be at increased risk. ( 5.2 ) • Hepatotoxicity - monitor patients for signs and symptoms of hepatic injury. Modify or discontinue TYENNE if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 2.8 , 5.3 ) • Laboratory monitoring recommended due to potential consequences of treatment-related changes in neutrophils, platelets, lipids, and liver function tests. ( 2.8 , 5.4 ) • Hypersensitivity reactions, including anaphylaxis and death and serious cutaneous reactions including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)- discontinue TYENNE, treat promptly, and monitor until reaction resolves ( 5.6 ) • Live vaccines - Avoid use with TYENNE ( 5.9 , 7.3 )
Contraindications
TYENNE is contraindicated in patients with known hypersensitivity to tocilizumab products [see Warnings and Precautions ( 5.6 )].
Adverse Reactions
The following serious adverse reactions are described elsewhere in labeling: • Serious Infections [see Warnings and Precautions ( 5.1 )] • Gastrointestinal Perforations [see Warnings and Precautions ( 5.2 )] • Laboratory Parameters [see Warnings and Precautions ( 5.4 )] • Immunosuppression [see Warnings and Precautions ( 5.5 )] • Hypersensitivity Reactions, Including Anaphylaxis [see Warnings and Precautions ( 5.6 )] • Demyelinating Disorders [see Warnings and Precautions ( 5.7 )] • Active Hepatic Disease and Hepatic Impairment [see Warnings and Precautions ( 5.8 )] Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
Storage and Handling
TYENNE (tocilizumab-aazg) injection is a preservative-free, sterile clear and colorless to pale yellow solution. The following packaging configurations are available:
How Supplied
TYENNE (tocilizumab-aazg) injection is a preservative-free, sterile clear and colorless to pale yellow solution. The following packaging configurations are available:
Medication Information
Warnings and Precautions
• Serious Infections - do not administer TYENNE during an active infection, including localized infections. If a serious infection develops, interrupt TYENNE until the infection is controlled. ( 5.1 ) • Gastrointestinal (GI) perforation-use with caution in patients who may be at increased risk. ( 5.2 ) • Hepatotoxicity - monitor patients for signs and symptoms of hepatic injury. Modify or discontinue TYENNE if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 2.8 , 5.3 ) • Laboratory monitoring recommended due to potential consequences of treatment-related changes in neutrophils, platelets, lipids, and liver function tests. ( 2.8 , 5.4 ) • Hypersensitivity reactions, including anaphylaxis and death and serious cutaneous reactions including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)- discontinue TYENNE, treat promptly, and monitor until reaction resolves ( 5.6 ) • Live vaccines - Avoid use with TYENNE ( 5.9 , 7.3 )
Indications and Usage
TYENNE ® (tocilizumab-aazg) is an interleukin-6 (IL-6) receptor antagonist indicated for treatment of: Rheumatoid Arthritis (RA) ( 1.1 ) • Adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs). Giant Cell Arteritis (GCA) ( 1.2 ) • Adult patients with giant cell arteritis. Polyarticular Juvenile Idiopathic Arthritis (PJIA) ( 1.3 ) • Patients 2 years of age and older with active polyarticular juvenile idiopathic arthritis. Systemic Juvenile Idiopathic Arthritis (SJIA) ( 1.4 ) • Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
Dosage and Administration
For RA, pJIA and sJIA, TYENNE may be used alone or in combination with methotrexate; and in RA, other non-biologic DMARDs may be used. ( 2 ) General Administration and Dosing Information ( 2.1 ) • RA, GCA, PJIA and SJIA - It is recommended that TYENNE not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm 3 , platelet count below 100,000 per mm 3 , or ALT or AST above 1.5 times the upper limit of normal (ULN) ( 5.3 , 5.4 ). • In RA patients, TYENNE doses exceeding 800 mg per infusion are not recommended. ( 2.2 , 2.7 , 12.3 ) • In GCA patients, TYENNE doses exceeding 600 mg per infusion are not recommended. ( 2.3 , 12.3 ) Rheumatoid Arthritis ( 2.2 ) Recommended Adult Intravenous Dosage: When used in combination with non-biologic DMARDs or as monotherapy the recommended starting dose is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response. Recommended Adult Subcutaneous Dosage: Patients less than 100 kg weight 162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response Patients at or above 100 kg weight 162 mg administered subcutaneously every week Giant Cell Arteritis ( 2.3 ) Recommended Adult Intravenous Dosage: The recommended dose is 6 mg per kg every 4 weeks in combination with a tapering course of glucocorticoids. TYENNE can be used alone following discontinuation of glucocorticoids. Recommended Adult Subcutaneous Dosage: The recommended dose is 162 mg given once every week as a subcutaneous injection, in combination with a tapering course of glucocorticoids. A dose of 162 mg given once every other week as a subcutaneous injection, in combination with a tapering course of glucocorticoids, may be prescribed based on clinical considerations. TYENNE can be used alone following discontinuation of glucocorticoids. Polyarticular Juvenile Idiopathic Arthritis ( 2.4 ) Recommended Intravenous PJIA Dosage Every 4 Weeks Patients less than 30 kg weight 10 mg per kg Patients at or above 30 kg weight 8 mg per kg Recommended Subcutaneous PJIA Dosage Patients less than 30 kg weight 162 mg once every three weeks Patients at or above 30 kg weight 162 mg once every two weeks Systemic Juvenile Idiopathic Arthritis ( 2.5 ) Recommended Intravenous SJIA Dosage Every 2 Weeks Patients less than 30 kg weight 12 mg per kg Patients at or above 30 kg weight 8 mg per kg Recommended Subcutaneous SJIA Dosage Patients less than 30 kg weight 162 mg every two weeks Patients at or above 30 kg weight 162 mg every week Administration of Intravenous formulation ( 2.6 ) • For patients with RA, GCA, PJIA and SJIA patients at or above 30 kg, dilute to 100 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique. • For PJIA and SJIA patients less than 30 kg, dilute to 50 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique. • Administer as a single intravenous drip infusion over 1 hour; do not administer as bolus or push. Administration of Subcutaneous formulation ( 2.7 ) • Follow the Instructions for Use for prefilled syringe and prefilled autoinjector Dose Modifications ( 2.8 ) Recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia.
Contraindications
TYENNE is contraindicated in patients with known hypersensitivity to tocilizumab products [see Warnings and Precautions ( 5.6 )].
Adverse Reactions
The following serious adverse reactions are described elsewhere in labeling: • Serious Infections [see Warnings and Precautions ( 5.1 )] • Gastrointestinal Perforations [see Warnings and Precautions ( 5.2 )] • Laboratory Parameters [see Warnings and Precautions ( 5.4 )] • Immunosuppression [see Warnings and Precautions ( 5.5 )] • Hypersensitivity Reactions, Including Anaphylaxis [see Warnings and Precautions ( 5.6 )] • Demyelinating Disorders [see Warnings and Precautions ( 5.7 )] • Active Hepatic Disease and Hepatic Impairment [see Warnings and Precautions ( 5.8 )] Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
Storage and Handling
TYENNE (tocilizumab-aazg) injection is a preservative-free, sterile clear and colorless to pale yellow solution. The following packaging configurations are available:
How Supplied
TYENNE (tocilizumab-aazg) injection is a preservative-free, sterile clear and colorless to pale yellow solution. The following packaging configurations are available:
Description
Warnings and Precautions ( 5.6 ) 12/2024
Data
Animal Data
An embryo-fetal developmental toxicity study was performed in which pregnant Cynomolgus monkeys were treated intravenously with tocilizumab at daily doses of 2, 10, or 50 mg/ kg during organogenesis from gestation day (GD) 20-50. Although there was no evidence for a teratogenic/dysmorphogenic effect at any dose, tocilizumab produced an increase in the incidence of abortion/embryo-fetal death at doses 1.25 times and higher the MRHD by the intravenous route at maternal intravenous doses of 10 and 50 mg/ kg. Testing of a murine analogue of tocilizumab in mice did not yield any evidence of harm to offspring during the pre- and postnatal development phase when dosed at 50 mg/kg intravenously with treatment every three days from implantation (GD 6) until post-partum day 21 (weaning). There was no evidence for any functional impairment of the development and behavior, learning ability, immune competence and fertility of the offspring.
Parturition is associated with significant increases of IL-6 in the cervix and myometrium. The literature suggests that inhibition of IL-6 signaling may interfere with cervical ripening and dilatation and myometrial contractile activity leading to potential delays of parturition. For mice deficient in IL-6 (ll6-/- null mice), parturition was delayed relative to wild-type (ll6+/+) mice.
Administration of recombinant IL-6 to ll6-/- null mice restored the normal timing of delivery.
Section 42229-5
Not Recommended for Concomitant Use with Biological DMARDs
Tocilizumab products have not been studied in combination with biological DMARDs such as TNF antagonists, IL-1R antagonists, anti-CD20 monoclonal antibodies and selective co-stimulation modulators because of the possibility of increased immunosuppression and increased risk of infection. Avoid using TYENNE with biological DMARDs.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 12/2024 | ||
|
Medication Guide
|
||
|
What is the most important information I should know about TYENNE?
TYENNE can cause serious side effects including:
|
||
|
Your healthcare provider should monitor you closely for signs and symptoms of TB during treatment with TYENNE.
Before starting TYENNE, tell your healthcare provider if you:
|
||
|
|
|
After starting TYENNE, call your healthcare provider right away if you have any symptoms of an infection. TYENNE can make you more likely to get infections or make worse any infection that you have.
|
||
|
|
|
|
||
|
Your healthcare provider will determine how often you will have follow-up blood tests. Make sure you get all your follow-up blood tests done as ordered by your healthcare provider.
|
||
|
See “What are the possible side effects with TYENNE?” for more information about side effects. |
||
|
What is TYENNE?
|
||
|
It is not known if TYENNE is safe and effective in children with PJIA or SJIA under 2 years of age or in children with conditions other than PJIA or SJIA. |
||
|
Do not take TYENNE: if you are allergic to tocilizumab products, or any of the ingredients in TYENNE. See the end of this Medication Guide for a complete list of ingredients in TYENNE. |
||
|
Before you receive TYENNE, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
Tell your healthcare provider about all of the medicines you take, including prescription, over-the-counter medicines, vitamins, and herbal supplements. TYENNE and other medicines may affect each other causing side effects.
|
||
|
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine. |
||
|
How will I receive TYENNE?
|
||
|
Under the skin (SC or subcutaneous injection) for Rheumatoid Arthritis, Giant Cell Arteritis, PJIA or SJIA:
|
||
|
What are the possible side effects with TYENNE? TYENNE can cause serious side effects, including:
|
||
|
|
|
|
||
|
The most common side effects of TYENNE include:
|
||
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
|
||
|
What are the ingredients in TYENNE?
|
Section 43683-2
Warnings and Precautions (5.6) 12/2024
Section 44425-7
Storage and Handling: Do not use beyond expiration date on the container, package, prefilled syringe, or autoinjector. TYENNE must be refrigerated at 36°F to 46°F (2ºC to 8ºC). Do not freeze. A single prefilled syringe (or autoinjector) may be stored at room temperature at or below 77°F (25°C) for a single period of up to 14 days. Protect the vials, syringes and autoinjectors from light by storage in the original package until time of use, and keep syringes and autoinjectors dry.
10 Overdosage
There are limited data available on overdoses with tocilizumab products. One case of accidental overdose was reported with intravenous tocilizumab in which a patient with multiple myeloma received a dose of 40 mg per kg. No adverse drug reactions were observed. No serious adverse drug reactions were observed in healthy volunteers who received single doses of up to 28 mg per kg, although all 5 patients at the highest dose of 28 mg per kg developed dose-limiting neutropenia.
In case of an overdose, it is recommended that the patient be monitored for signs and symptoms of adverse reactions. Patients who develop adverse reactions should receive appropriate symptomatic treatment.
11 Description
Tocilizumab-aazg is a recombinant humanized anti-human interleukin 6 (IL-6) receptor monoclonal antibody of the immunoglobulin IgG1κ (gamma 1, kappa) subclass with a typical H2L2 polypeptide structure. Each light chain and heavy chain consists of 214 and 448 amino acids (excluding the C-terminal lysine), respectively.
The four polypeptide chains are linked intra- and inter-molecularly by disulfide bonds. Tocilizumab-aazg has a molecular weight of approximately 148 kDa. The antibody is produced in mammalian (Chinese hamster ovary) cells.
Intravenous Infusion
TYENNE (tocilizumab-aazg) injection is a sterile, clear and colorless to pale yellow, histidine buffered preservative-free solution with a pH of approximately 6 for further dilution prior to intravenous infusion. Each single-dose vial is available at a concentration of 20 mg/mL containing 80 mg/4 mL, 200 mg/10 mL, or 400 mg/20 mL of TYENNE. Each mL of solution contains arginine (17.4 mg), histidine (3.1 mg), lactic acid (0.9 mg), polysorbate 80 (0.2 mg), sodium chloride (0.6 mg), and Water for Injection, USP. Hydrochloric acid and sodium hydroxide are added to adjust the pH.
Subcutaneous Injection
TYENNE (tocilizumab-aazg) injection is a sterile, clear, colorless to pale yellow, preservative-free, histidine buffered solution with a pH of approximately 6 for subcutaneous use. It is supplied in a ready-to-use, single-dose 0.9 mL prefilled syringe (PFS) with a needle safety device or in a ready-to-use, single-dose 0.9 mL autoinjector that delivers 162 mg tocilizumab-aazg, arginine (16.7 mg), histidine (2.0 mg), lactic acid (0.9 mg), polysorbate 80 (0.2 mg), sodium chloride (0.6 mg), and Water for Injection, USP. Hydrochloric acid and sodium hydroxide are added to adjust the pH.
5.9 Vaccinations
Avoid use of live vaccines concurrently with TYENNE as clinical safety has not been established. No data are available on the secondary transmission of infection from persons receiving live vaccines to patients receiving tocilizumab products.
No data are available on the effectiveness of vaccination in patients receiving tocilizumab products. Because IL-6 inhibition may interfere with the normal immune response to new antigens, it is recommended that all patients, particularly pediatric or elderly patients, if possible, be brought up to date with all immunizations in agreement with current immunization guidelines prior to initiating TYENNE therapy. The interval between live vaccinations and initiation of TYENNE therapy should be in accordance with current vaccination guidelines regarding immunosuppressive agents.
7.3 Live Vaccines
Avoid use of live vaccines concurrently with TYENNE [see Warnings and Precautions (5.9)].
8.4 Pediatric Use
TYENNE by intravenous use is indicated for the treatment of pediatric patients with:
-
•Active systemic juvenile idiopathic arthritis in patients 2 years of age and older
-
•Active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older
TYENNE by subcutaneous use is indicated for the treatment of pediatric patients with:
-
•Active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older
-
•Active systemic juvenile idiopathic arthritis in patients 2 years of age and older
The safety and effectiveness of TYENNE in pediatric patients with conditions other than PJIA or SJIA have not been established. The safety and effectiveness in pediatric patients below the age of 2 have not been established in PJIA or SJIA.
8.5 Geriatric Use
Of the 2644 patients who received tocilizumab in Studies I to V [see Clinical Studies (14)], a total of 435 rheumatoid arthritis patients were 65 years of age and older, including 50 patients 75 years and older. Of the 1069 patients who received tocilizumab-SC in studies SC-I and SC-II there were 295 patients 65 years of age and older, including 41 patients 75 years and older. The frequency of serious infection among tocilizumab treated subjects 65 years of age and older was higher than those under the age of 65. As there is a higher incidence of infections in the elderly population in general, caution should be used when treating the elderly.
5.3 Hepatotoxicity
Serious cases of hepatic injury have been observed in patients taking intravenous or subcutaneous tocilizumab products. Some of these cases have resulted in liver transplant or death. Time to onset for cases ranged from months to years after treatment initiation with tocilizumab products. While most cases presented with marked elevations of transaminases (> 5 times ULN), some cases presented with signs or symptoms of liver dysfunction and only mildly elevated transaminases.
During randomized controlled studies, treatment with tocilizumab was associated with a higher incidence of transaminase elevations [see Adverse Reactions (6.1, 6.2, 6.5, 6.7)]. Increased frequency and magnitude of these elevations was observed when potentially hepatotoxic drugs (e.g., MTX) were used in combination with tocilizumab.
For RA and GCA patients, obtain a liver test panel (serum alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase, and total bilirubin) before initiating TYENNE, every 4 to 8 weeks after start of therapy for the first 6 months of treatment and every 3 months thereafter. It is not recommended to initiate TYENNE treatment in RA or GCA patients with elevated transaminases ALT or AST greater than 1.5x ULN. In patients who develop elevated ALT or AST greater than 5x ULN, discontinue TYENNE. For recommended modifications based upon increase in transaminases [see Dosage and Administration (2.8)].
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, such as fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient is found to have abnormal liver tests (e.g., ALT greater than three times the upper limit of the reference range, serum total bilirubin greater than two times the upper limit of the reference range), TYENNE treatment should be interrupted and investigation done to establish the probable cause. TYENNE should only be restarted in patients with another explanation for the liver test abnormalities after normalization of the liver tests.
A similar pattern of liver enzyme elevation is noted with tocilizumab products treatment in the PJIA and SJIA populations. Monitor liver test panel at the time of the second administration and thereafter every 4 to 8 weeks for PJIA and every 2 to 4 weeks for SJIA.
4 Contraindications
TYENNE is contraindicated in patients with known hypersensitivity to tocilizumab products [see Warnings and Precautions (5.6)].
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in labeling:
-
•Serious Infections [see Warnings and Precautions (5.1)]
-
•Gastrointestinal Perforations [see Warnings and Precautions (5.2)]
-
•Laboratory Parameters [see Warnings and Precautions (5.4)]
-
•Immunosuppression [see Warnings and Precautions (5.5)]
-
•Hypersensitivity Reactions, Including Anaphylaxis [see Warnings and Precautions (5.6)]
-
•Demyelinating Disorders [see Warnings and Precautions (5.7)]
-
•Active Hepatic Disease and Hepatic Impairment [see Warnings and Precautions (5.8)]
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
8.7 Renal Impairment
No dose adjustment is required in patients with mild or moderate renal impairment. Tocilizumab products have not been studied in patients with severe renal impairment [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In clinical studies in RA patients with the 4 mg per kg and 8 mg per kg intravenous doses or the 162 mg weekly and every other weekly subcutaneous doses of tocilizumab, decreases in levels of C-reactive protein (CRP) to within normal ranges were seen as early as week 2. Changes in pharmacodynamic parameters were observed (i.e., decreases in rheumatoid factor, erythrocyte sedimentation rate (ESR), serum amyloid A, fibrinogen and increases in hemoglobin) with doses, however the greatest improvements were observed with 8 mg per kg tocilizumab. Pharmacodynamic changes were also observed to occur after tocilizumab administration in GCA, PJIA, and SJIA patients (decreases in CRP, ESR, and increases in hemoglobin). The relationship between these pharmacodynamic findings and clinical efficacy is not known.
In healthy subjects administered tocilizumab in doses from 2 to 28 mg per kg intravenously and 81 to 162 mg subcutaneously, absolute neutrophil counts decreased to the nadir 3 to 5 days following tocilizumab administration. Thereafter, neutrophils recovered towards baseline in a dose dependent manner. Rheumatoid arthritis and GCA patients demonstrated a similar pattern of absolute neutrophil counts following tocilizumab administration [see Warnings and Precautions (5.4)].
12.3 Pharmacokinetics
PK of tocilizumab is characterized by nonlinear elimination which is a combination of linear clearance and Michaelis-Menten elimination. The nonlinear part of tocilizumab elimination leads to an increase in exposure that is more than dose-proportional. The pharmacokinetic parameters of tocilizumab do not change with time. Due to the dependence of total clearance on tocilizumab serum concentrations, the half-life of tocilizumab is also concentration-dependent and varies depending on the serum concentration level. Population pharmacokinetic analyses in any patient population tested so far indicate no relationship between apparent clearance and the presence of anti-drug antibodies.
5.5 Immunosuppression
The impact of treatment with tocilizumab products on the development of malignancies is not known but malignancies were observed in clinical studies [see Adverse Reactions (6.1)]. TYENNE is an immunosuppressant, and treatment with immunosuppressants may result in an increased risk of malignancies.
5.1 Serious Infections
Serious and sometimes fatal infections due to bacterial, mycobacterial, invasive fungal, viral, protozoal, or other opportunistic pathogens have been reported in patients receiving immunosuppressive agents including tocilizumab products. The most common serious infections included pneumonia, urinary tract infection, cellulitis, herpes zoster, gastroenteritis, diverticulitis, sepsis and bacterial arthritis [see Adverse Reactions (6.1)]. Among opportunistic infections, tuberculosis, cryptococcus, aspergillosis, candidiasis, and pneumocystosis were reported with tocilizumab products. Other serious infections, not reported in clinical studies, may also occur (e.g., histoplasmosis, coccidioidomycosis, listeriosis). Patients have presented with disseminated rather than localized disease, and were often taking concomitant immunosuppressants such as methotrexate or corticosteroids which in addition to rheumatoid arthritis may predispose them to infections.
Do not administer TYENNE in patients with an active infection, including localized infections. The risks and benefits of treatment should be considered prior to initiating TYENNE in patients:
-
•with chronic or recurrent infection;
-
•who have been exposed to tuberculosis;
-
•with a history of serious or an opportunistic infection;
-
•who have resided or traveled in areas of endemic tuberculosis or endemic mycoses; or
-
•with underlying conditions that may predispose them to infection.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with TYENNE, as signs and symptoms of acute inflammation may be lessened due to suppression of the acute phase reactants [see Dosage and Administration (2.1), Adverse Reactions (6.1), and Patient Counseling Information (17)].
Hold TYENNE if a patient develops a serious infection, an opportunistic infection, or sepsis. A patient who develops a new infection during treatment with TYENNE should undergo a prompt and complete diagnostic workup appropriate for an immunocompromised patient, initiate appropriate antimicrobial therapy, and closely monitor the patient.
8.6 Hepatic Impairment
The safety and efficacy of tocilizumab products have not been studied in patients with hepatic impairment, including patients with positive HBV and HCV serology [see Warnings and Precautions (5.8)].
1 Indications and Usage
TYENNE® (tocilizumab-aazg) is an interleukin-6 (IL-6) receptor antagonist indicated for treatment of:
Rheumatoid Arthritis (RA) (1.1)
-
•Adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs).
Giant Cell Arteritis (GCA) (1.2)
-
•Adult patients with giant cell arteritis.
Polyarticular Juvenile Idiopathic Arthritis (PJIA) (1.3)
-
•Patients 2 years of age and older with active polyarticular juvenile idiopathic arthritis.
Systemic Juvenile Idiopathic Arthritis (SJIA) (1.4)
-
•Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
Clinical Considerations
Fetal/Neonatal adverse reactions
Monoclonal antibodies are increasingly transported across the placenta as pregnancy progresses, with the largest amount transferred during the third trimester. Risks and benefits should be considered prior to administering live or live-attenuated vaccines to infants exposed to TYENNE in utero [see Warnings and Precautions (5.9)].
Disease-associated Maternal Risk
Published data suggest that the risk of adverse pregnancy outcomes in women with rheumatoid arthritis is associated with increased disease activity. Adverse pregnancy outcomes include preterm delivery (before 37 weeks of gestation), low birth weight (less than 2500 g) infants, and small for gestational age at birth.
12.1 Mechanism of Action
Tocilizumab products bind to both soluble and membrane-bound IL-6 receptors (sIL-6R and mIL-6R), and have been shown to inhibit IL-6-mediated signaling through these receptors. IL-6 is a pleiotropic pro-inflammatory cytokine produced by a variety of cell types including T- and B-cells, lymphocytes, monocytes and fibroblasts. IL-6 has been shown to be involved in diverse physiological processes such as T-cell activation, induction of immunoglobulin secretion, initiation of hepatic acute phase protein synthesis, and stimulation of hematopoietic precursor cell proliferation and differentiation. IL-6 is also produced by synovial and endothelial cells leading to local production of IL-6 in joints affected by inflammatory processes such as rheumatoid arthritis.
5 Warnings and Precautions
-
•Serious Infections - do not administer TYENNE during an active infection, including localized infections. If a serious infection develops, interrupt TYENNE until the infection is controlled. (5.1)
-
•Gastrointestinal (GI) perforation-use with caution in patients who may be at increased risk. (5.2)
-
•Hepatotoxicity - monitor patients for signs and symptoms of hepatic injury. Modify or discontinue TYENNE if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. (2.8, 5.3)
-
•Laboratory monitoring recommended due to potential consequences of treatment-related changes in neutrophils, platelets, lipids, and liver function tests. (2.8, 5.4)
-
•Hypersensitivity reactions, including anaphylaxis and death and serious cutaneous reactions including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)- discontinue TYENNE, treat promptly, and monitor until reaction resolves (5.6)
-
•Live vaccines - Avoid use with TYENNE (5.9, 7.3)
2 Dosage and Administration
For RA, pJIA and sJIA, TYENNE may be used alone or in combination with methotrexate; and in RA, other non-biologic DMARDs may be used. (2)
General Administration and Dosing Information (2.1)
-
•RA, GCA, PJIA and SJIA - It is recommended that TYENNE not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm3, platelet count below 100,000 per mm3, or ALT or AST above 1.5 times the upper limit of normal (ULN) (5.3, 5.4).
-
•In RA patients, TYENNE doses exceeding 800 mg per infusion are not recommended. (2.2, 2.7, 12.3)
-
•In GCA patients, TYENNE doses exceeding 600 mg per infusion are not recommended. (2.3, 12.3)
Rheumatoid Arthritis (2.2)
Recommended Adult Intravenous Dosage:
When used in combination with non-biologic DMARDs or as monotherapy the recommended starting dose is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response.
Recommended Adult Subcutaneous Dosage:
|
Patients less than 100 kg weight |
162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response |
|
Patients at or above 100 kg weight |
162 mg administered subcutaneously every week |
Giant Cell Arteritis (2.3)
Recommended Adult Intravenous Dosage: The recommended dose is 6 mg per kg every 4 weeks in combination with a tapering course of glucocorticoids. TYENNE can be used alone following discontinuation of glucocorticoids.
Recommended Adult Subcutaneous Dosage: The recommended dose is 162 mg given once every week as a subcutaneous injection, in combination with a tapering course of glucocorticoids.
A dose of 162 mg given once every other week as a subcutaneous injection, in combination with a tapering course of glucocorticoids, may be prescribed based on clinical considerations.
TYENNE can be used alone following discontinuation of glucocorticoids.
Polyarticular Juvenile Idiopathic Arthritis (2.4)
|
Recommended Intravenous PJIA Dosage Every 4 Weeks |
|
|
Patients less than 30 kg weight |
10 mg per kg |
|
Patients at or above 30 kg weight |
8 mg per kg |
|
Recommended Subcutaneous PJIA Dosage |
|
|
Patients less than 30 kg weight |
162 mg once every three weeks |
|
Patients at or above 30 kg weight |
162 mg once every two weeks |
Systemic Juvenile Idiopathic Arthritis (2.5)
|
Recommended Intravenous SJIA Dosage Every 2 Weeks |
|
|
Patients less than 30 kg weight |
12 mg per kg |
|
Patients at or above 30 kg weight |
8 mg per kg |
|
Recommended Subcutaneous SJIA Dosage |
|
|
Patients less than 30 kg weight |
162 mg every two weeks |
|
Patients at or above 30 kg weight |
162 mg every week |
Administration of Intravenous formulation (2.6)
-
•For patients with RA, GCA, PJIA and SJIA patients at or above 30 kg, dilute to 100 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
-
•For PJIA and SJIA patients less than 30 kg, dilute to 50 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
-
•Administer as a single intravenous drip infusion over 1 hour; do not administer as bolus or push.
Administration of Subcutaneous formulation (2.7)
-
•Follow the Instructions for Use for prefilled syringe and prefilled autoinjector
Dose Modifications (2.8)
Recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia.
5.7 Demyelinating Disorders
The impact of treatment with tocilizumab products on demyelinating disorders is not known, but multiple sclerosis and chronic inflammatory demyelinating polyneuropathy were reported rarely in RA clinical studies. Monitor patients for signs and symptoms potentially indicative of demyelinating disorders. Prescribers should exercise caution in considering the use of TYENNE in patients with preexisting or recent onset demyelinating disorders.
9 Drug Abuse and Dependence
No studies on the potential for tocilizumab products to cause dependence have been performed. However, there is no evidence from the available data that tocilizumab products treatment results in dependence.
3 Dosage Forms and Strengths
Intravenous Infusion
Injection: 80 mg/4 mL (20 mg/mL), 200 mg/10 mL (20 mg/mL), 400 mg/20 mL (20 mg/mL) in single-dose vials for further dilution prior to intravenous infusion (3)
Subcutaneous Injection
Injection: 162 mg/0.9 mL in a single-dose prefilled syringe or single-dose prefilled autoinjector (3)
6.9 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of tocilizumab products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
1.1 Rheumatoid Arthritis (ra)
TYENNE® (tocilizumab-aazg) is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs).
8 Use in Specific Populations
1.2 Giant Cell Arteritis (gca)
TYENNE® (tocilizumab-aazg) is indicated for the treatment of giant cell arteritis (GCA) in adult patients.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
-
•Serious Infections
Inform patients that TYENNE may lower their resistance to infections [see Warnings and Precautions (5.1)]. Instruct the patient of the importance of contacting their doctor immediately when symptoms suggesting infection appear in order to assure rapid evaluation and appropriate treatment. -
•Gastrointestinal Perforation
Inform patients that some patients who have been treated with TYENNE have had serious side effects in the stomach and intestines [see Warnings and Precautions (5.2)]. Instruct the patient of the importance of contacting their doctor immediately when symptoms of fever, severe, persistent abdominal pain, and change in bowel habits appear to assure rapid evaluation and appropriate treatment. -
•Hypersensitivity and Serious Allergic Reactions
Inform patients that some patients who have been treated with TYENNE have developed serious allergic reactions, including anaphylaxis, as well as serious skin reactions [see Warnings and Precautions (5.6)]. Advise patients to stop taking TYENNE and seek immediate medical attention if they experience any symptom of serious allergic reactions (including rash, hives, and swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing).
5.2 Gastrointestinal Perforations
Events of gastrointestinal perforation have been reported in clinical trials, primarily as complications of diverticulitis in patients treated with tocilizumab. Use TYENNE with caution in patients who may be at increased risk for gastrointestinal perforation. Promptly evaluate patients presenting with fever, new onset abdominal symptoms, and a change in bowel habits for early identification of gastrointestinal perforation [see Adverse Reactions (6.1)].
Warning: Risk of Serious Infections
Patients treated with tocilizumab products including TYENNE are at increased risk for developing serious infections that may lead to hospitalization or death [see Warnings and Precautions (5.1), Adverse Reactions (6.1)]. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids.
If a serious infection develops, interrupt TYENNE until the infection is controlled.
Reported infections include:
-
•Active tuberculosis, which may present with pulmonary or extrapulmonary disease. Patients should be tested for latent tuberculosis before TYENNE use and during therapy. Treatment for latent infection should be initiated prior to TYENNE use.
-
•Invasive fungal infections, including candidiasis, aspergillosis, and pneumocystis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
-
•Bacterial, viral and other infections due to opportunistic pathogens.
The risks and benefits of treatment with TYENNE should be carefully considered prior to initiating therapy in patients with chronic or recurrent infection.
Patients should be closely monitored for the development of signs and symptoms of infection during and after treatment with TYENNE including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy [see Warnings and Precautions (5.1)] .
16 How Supplied/storage and Handling
TYENNE (tocilizumab-aazg) injection is a preservative-free, sterile clear and colorless to pale yellow solution. The following packaging configurations are available:
7.2 Interactions With Cyp450 Substrates
Cytochrome P450s in the liver are down-regulated by infection and inflammation stimuli including cytokines such as IL-6. Inhibition of IL-6 signaling in RA patients treated with tocilizumab products may restore CYP450 activities to higher levels than those in the absence of tocilizumab products leading to increased metabolism of drugs that are CYP450 substrates. In vitro studies showed that tocilizumab has the potential to affect expression of multiple CYP enzymes including CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP2D6 and CYP3A4. Its effect on CYP2C8 or transporters is unknown. In vivo studies with omeprazole, metabolized by CYP2C19 and CYP3A4, and simvastatin, metabolized by CYP3A4, showed up to a 28% and 57% decrease in exposure one week following a single dose of tocilizumab, respectively. The effect of tocilizumab products on CYP enzymes may be clinically relevant for CYP450 substrates with narrow therapeutic index, where the dose is individually adjusted.
Upon initiation or discontinuation of TYENNE, in patients being treated with these types of medicinal products, perform therapeutic monitoring of effect (e.g., warfarin) or drug concentration (e.g., cyclosporine or theophylline) and the individual dose of the medicinal product adjusted as needed. Exercise caution when coadministering TYENNE with CYP3A4 substrate drugs where decrease in effectiveness is undesirable, e.g., oral contraceptives, lovastatin, atorvastatin, etc. The effect of tocilizumab products on CYP450 enzyme activity may persist for several weeks after stopping therapy [see Clinical Pharmacology (12.3)].
Principal Display Panel – 4 Ml Vial Carton
NDC 65219-590-04 Rx only
Tyenne®
tocilizumab-aazg
Injection
80 mg / 4 mL
(20 mg/mL)
For intravenous infusion only
after dilution
Dispense a Medication
Guide to each patient
Carton contains:
1 Single-use Vial - Discard Unused Portion
1 Prescribing Information
1 Medication Guide
1 x 4 mL
2.2 Recommended Dosage for Rheumatoid Arthritis
TYENNE may be used as monotherapy or concomitantly with methotrexate or other non-biologic DMARDs as an intravenous infusion or as a subcutaneous injection.
Principal Display Panel – 10 Ml Vial Carton
NDC 65219-592-10 Rx only
Tyenne®
tocilizumab-aazg
Injection
200 mg / 10 mL
(20 mg/mL)
For intravenous infusion only
after dilution
Dispense a Medication
Guide to each patient
Carton contains:
1 Single-use Vial – Discard Unused Portion
1 Prescribing Information
1 Medication Guide
1 x 10 mL
Principal Display Panel – 20 Ml Vial Carton
NDC 65219-594-20 Rx only
Tyenne®
tocilizumab-aazg
Injection
400 mg / 20 mL
(20 mg/mL)
For intravenous infusion only
after dilution
Dispense a Medication
Guide to each patient
Carton contains:
1 Single-use Vial - Discard Unused Portion
1 Prescribing Information
1 Medication Guide
1 x 20 mL
1.4 Systemic Juvenile Idiopathic Arthritis (sjia)
TYENNE® (tocilizumab-aazg) is indicated for the treatment of active systemic juvenile idiopathic arthritis in patients 2 years of age and older.
5.8 Active Hepatic Disease and Hepatic Impairment
5.6 Hypersensitivity Reactions, Including Anaphylaxis
Hypersensitivity reactions, including anaphylaxis, have been reported in association with tocilizumab products and anaphylactic events with a fatal outcome have been reported with intravenous infusion of tocilizumab products. Anaphylaxis and other hypersensitivity reactions that required treatment discontinuation were reported in 0.1% (3 out of 2644) of patients in the 6-month controlled trials of intravenous tocilizumab, 0.2% (8 out of 4009) of patients in the intravenous all-exposure RA population, 0.7% (8 out of 1068) in the subcutaneous 6-month controlled RA trials, and in 0.7% (10 out of 1465) of patients in the subcutaneous all-exposure population. In the SJIA controlled trial with intravenous tocilizumab, 1 out of 112 patients (0.9%) experienced hypersensitivity reactions that required treatment discontinuation. In the PJIA controlled trial with intravenous tocilizumab, 0 out of 188 patients (0%) in the tocilizumab all-exposure population experienced hypersensitivity reactions that required treatment discontinuation. Reactions that required treatment discontinuation included generalized erythema, rash, and urticaria. Injection site reactions were categorized separately [see Adverse Reactions (6)].
In the postmarketing setting, events of hypersensitivity reactions, including anaphylaxis and death have occurred in patients treated with a range of doses of intravenous tocilizumab products, with or without concomitant therapies. Events have occurred in patients who received premedication. Hypersensitivity, including anaphylaxis events, have occurred both with and without previous hypersensitivity reactions and as early as the first infusion of tocilizumab products [see Adverse Reactions (6.9)]. In addition, serious cutaneous reactions, including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), have been reported in patients with autoinflammatory conditions treated with tocilizumab products.
TYENNE for intravenous use should only be infused by a healthcare professional with appropriate medical support to manage anaphylaxis. For TYENNE subcutaneous injection, advise patients to seek immediate medical attention if they experience any symptoms of a hypersensitivity reaction. If a hypersensitivity reaction occurs, stop immediately discontinue TYENNE treat promptly and monitor until signs and symptoms resolve.
Principal Display Panel – 4 Ml Vial Primary Label
NDC 65219-590-04 Rx only 4 mL
Tyenne®
(tocilizumab-aazg)
Injection
80 mg / 4 mL
(20 mg/mL)
For Intravenous Infusion
Only After Dilution.
Fresenius Kabi USA, LLC
US License No. 2146
1.3 Polyarticular Juvenile Idiopathic Arthritis (pjia)
TYENNE® (tocilizumab-aazg) is indicated for the treatment of active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older.
14.1 Rheumatoid Arthritis – Intravenous Administration
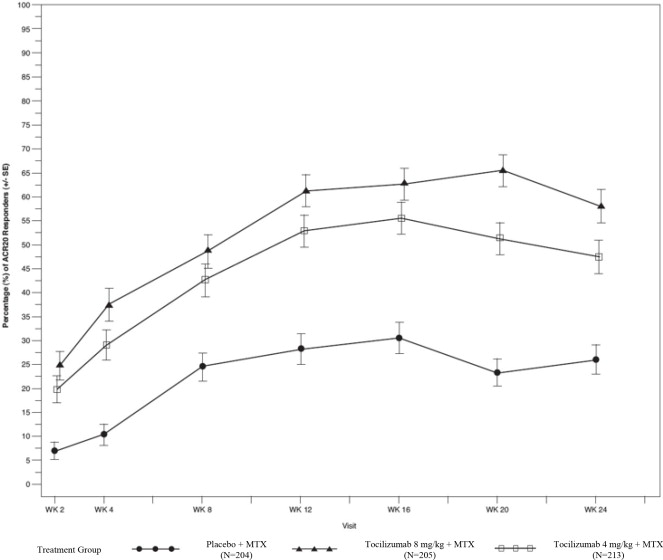
The efficacy and safety of intravenously administered tocilizumab was assessed in five randomized, double-blind, multicenter studies in patients greater than 18 years with active rheumatoid arthritis diagnosed according to American College of Rheumatology (ACR) criteria. Patients had at least 8 tender and 6 swollen joints at baseline. Tocilizumab was given intravenously every 4 weeks as monotherapy (Study I), in combination with methotrexate (MTX) (Studies II and III) or other disease-modifying anti-rheumatic drugs (DMARDs) (Study IV) in patients with an inadequate response to those drugs, or in combination with MTX in patients with an inadequate response to TNF antagonists (Study V).
Study I (NCT00109408) evaluated patients with moderate to severe active rheumatoid arthritis who had not been treated with MTX within 24 weeks prior to randomization, or who had not discontinued previous methotrexate treatment as a result of clinically important toxic effects or lack of response. In this study, 67% of patients were MTX-naïve, and over 40% of patients had rheumatoid arthritis less than 2 years. Patients received tocilizumab 8 mg per kg monotherapy or MTX alone (dose titrated over 8 weeks from 7.5 mg to a maximum of 20 mg weekly). The primary endpoint was the proportion of tocilizumab patients who achieved an ACR 20 response at Week 24.
Study II (NCT00106535) was a 104-week study with an optional 156-week extension phase that evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response to MTX. Patients received tocilizumab 8 mg per kg, tocilizumab 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). Upon completion of 52-weeks, patients received open-label treatment with tocilizumab 8 mg per kg through 104 weeks or they had the option to continue their double-blind treatment if they maintained a greater than 70% improvement in swollen/tender joint count. Two pre-specified interim analyses at week 24 and week 52 were conducted. The primary endpoint at week 24 was the proportion of patients who achieved an ACR 20 response. At weeks 52 and 104, the primary endpoints were change from baseline in modified total Sharp-Genant score and the area under the curve (AUC) of the change from baseline in HAQ-DI score.
Study III (NCT00106548) evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response to MTX. Patients received tocilizumab 8 mg per kg, tocilizumab 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
Study IV (NCT00106574) evaluated patients who had an inadequate response to their existing therapy, including one or more DMARDs. Patients received tocilizumab 8 mg per kg or placebo every four weeks, in combination with the stable DMARDs. The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
Study V (NCT00106522) evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response or were intolerant to one or more TNF antagonist therapies. The TNF antagonist therapy was discontinued prior to randomization. Patients received tocilizumab 8 mg per kg, tocilizumab 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
14.4 Giant Cell Arteritis – Intravenous Administration
Intravenously administered tocilizumab in patients with GCA was assessed in WP41152 (NCT03923738), an open-label PK-PD and safety study to determine the appropriate intravenous dose of tocilizumab that achieved comparable PK-PD profiles to the tocilizumab-SC regimen.
At enrollment, all patients (n=24) were in remission on tocilizumab-IV. In Period 1, all patients received open label tocilizumab-IV 7 mg/kg every 4 weeks for 20 weeks. Patients who completed Period 1 and remained in remission (n=22) were eligible to enter Period 2, and received open-label tocilizumab-IV 6 mg/kg every 4 weeks for 20 weeks.
The efficacy of intravenous tocilizumab 6 mg/kg in adult patients with GCA is based on pharmacokinetic exposure and extrapolation to the efficacy established for subcutaneous tocilizumab in patients with GCA [see Clinical Pharmacology (12.3) and Clinical Studies (14.3 )].
7.1 Concomitant Drugs for Treatment of Adult Indications
In RA patients, population pharmacokinetic analyses did not detect any effect of methotrexate (MTX), non-steroidal anti-inflammatory drugs or corticosteroids on tocilizumab clearance. Concomitant administration of a single intravenous dose of 10 mg/kg tocilizumab with 10-25 mg MTX once weekly had no clinically significant effect on MTX exposure. Tocilizumab products have not been studied in combination with biological DMARDs such as TNF antagonists [see Dosage and Administration (2.2)].
In GCA patients, no effect of concomitant corticosteroid on tocilizumab exposure was observed.
Principal Display Panel – 10 Ml Vial – Primary Label
NDC 65219-592-10 Rx only 10 mL
Tyenne®
(tocilizumab-aazg)
Injection
200 mg / 10 mL
(20 mg/mL)
For Intravenous Infusion
Only After Dilution.
Fresenius Kabi USA, LLC
US License No. 2146
Principal Display Panel – 20 Ml Vial – Primary Label
NDC 65219-594-20 Rx only 20 mL
Tyenne®
tocilizumab-aazg
Injection
400 mg / 20 mL
(20 mg/mL)
For Intravenous Infusion
Only After Dilution.
Fresenius Kabi USA, LLC
US License No. 2146
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to establish the carcinogenicity potential of tocilizumab products. Literature indicates that the IL-6 pathway can mediate anti-tumor responses by promoting increased immune cell surveillance of the tumor microenvironment. However, available published evidence also supports that IL-6 signaling through the IL-6 receptor may be involved in pathways that lead to tumorigenesis. The malignancy risk in humans from an antibody that disrupts signaling through the IL-6 receptor, such as tocilizumab, is presently unknown.
Fertility and reproductive performance were unaffected in male and female mice that received a murine analogue of tocilizumab administered by the intravenous route at a dose of 50 mg/kg every three days.
14.2 Rheumatoid Arthritis – Subcutaneous Administration
The efficacy and safety of subcutaneously administered tocilizumab was assessed in two double-blind, controlled, multicenter studies in patients with active RA. One study, SC-I (NCT01194414), was a non-inferiority study that compared the efficacy and safety of tocilizumab 162 mg administered every week subcutaneously to 8 mg per kg intravenously every four weeks. The second study, SC-II (NCT01232569), was a placebo-controlled superiority study that evaluated the safety and efficacy of tocilizumab 162 mg administered every other week subcutaneously to placebo. Both SC-I and SC-II required patients to be >18 years of age with moderate to severe active rheumatoid arthritis diagnosed according to ACR criteria who had at least 4 tender and 4 swollen joints at baseline (SC-I) or at least 8 tender and 6 swollen joints at baseline (SC-II), and an inadequate response to their existing DMARD therapy, where approximately 20% also had a history of inadequate response to at least one TNF inhibitor. All patients in both SC studies received background non-biologic DMARD(s).
In SC-I, 1262 patients were randomized 1:1 to receive tocilizumab-SC 162 mg every week or intravenous tocilizumab 8 mg/kg every four weeks in combination with DMARD(s). In SC-II, 656 patients were randomized 2:1 to tocilizumab-SC 162 mg every other week or placebo, in combination with DMARD(s). The primary endpoint in both studies was the proportion of patients who achieved an ACR20 response at Week 24.
The clinical response to 24 weeks of tocilizumab-SC therapy is shown in Table 7 . In SC-I, the primary outcome measure was ACR20 at Week 24. The pre-specified non-inferiority margin was a treatment difference of 12%. The study demonstrated non-inferiority of tocilizumab with respect to ACR20 at Week 24; ACR50, ACR70, and DAS28 responses are also shown in Table 7 . In SC-II, a greater portion of patients treated with tocilizumab 162 mg subcutaneously every other week achieved ACR20, ACR50, and ACR70 responses compared to placebo-treated patients (Table 7). Further, a greater proportion of patients treated with tocilizumab 162 mg subcutaneously every other week achieved a low level of disease activity as measured by a DAS28-ESR less than 2.6 at Week 24 compared to those treated with placebo (Table 7).
| TCZ = tocilizumab a Per Protocol Population b Intent To Treat Population |
||||
|
SC-Ia |
SC-IIb |
|||
|
TCZ SC 162 mg every week + DMARD |
TCZ IV 8mg/kg + DMARD |
TCZ SC 162 mg every other week + DMARD |
Placebo + DMARD |
|
|
N=558 |
N=537 |
N=437 |
N=219 |
|
|
ACR20 |
||||
|
Week 24 |
69% |
73.4% |
61% |
32% |
|
Weighted difference (95% CI) |
-4% (-9.2, 1.2) |
30% (22.0, 37.0) |
||
|
ACR50 |
||||
|
Week 24 |
47% |
49% |
40% |
12% |
|
Weighted difference (95% CI) |
-2% (-7.5, 4.0) |
28% (21.5, 34.4) |
||
|
ACR70 |
||||
|
Week 24 |
24% |
28% |
20% |
5% |
|
Weighted difference (95% CI) |
-4% (-9.0, 1.3) |
15% (9.8, 19.9) |
||
|
Change in DAS28 [Adjusted mean] |
||||
|
Week 24 |
-3.5 |
-3.5 |
-3.1 |
-1.7 |
|
Adjusted mean difference (95% CI) |
0 (-0.2, 0.1) |
-1.4 (-1.7; -1.1) |
||
|
DAS28 < 2.6 |
||||
|
Week 24 |
38.4% |
36.9% |
32.0% |
4.0% |
|
Weighted difference (95% CI) |
0.9 (-5.0, 6.8) |
28.6 (22.0, 35.2) |
The results of the components of the ACR response criteria and the percent of ACR20 responders by visit for tocilizumab-SC in Studies SC-I and SC-II were consistent with those observed for tocilizumab-IV.
14.3 Giant Cell Arteritis – Subcutaneous Administration
The efficacy and safety of subcutaneously administered tocilizumab was assessed in a single, randomized, double-blind, multicenter study in patients with active GCA. In Study WA28119 (NCT01791153), 251 screened patients with new-onset or relapsing GCA were randomized to one of four treatment arms. Two subcutaneous doses of tocilizumab (162 mg every week and 162 mg every other week) were compared to two different placebo control groups (pre-specified prednisone-taper regimen over 26 weeks and 52 weeks) randomized 2:1:1:1. The study consisted of a 52-week blinded period, followed by a 104-week open-label extension.
All patients received background glucocorticoid (prednisone) therapy. Each of the tocilizumab-treated groups and one of the placebo-treated groups followed a pre-specified prednisone-taper regimen with the aim to reach 0 mg by 26 weeks, while the second placebo-treated group followed a pre-specified prednisone-taper regimen with the aim to reach 0 mg by 52 weeks designed to be more in keeping with standard practice.
The primary efficacy endpoint was the proportion of patients achieving sustained remission from Week 12 through Week 52. Sustained remission was defined by a patient attaining a sustained (1) absence of GCA signs and symptoms from Week 12 through Week 52, (2) normalization of erythrocyte sedimentation rate (ESR) (to < 30 mm/hr without an elevation to ≥ 30 mm/hr attributable to GCA) from Week 12 through Week 52, (3) normalization of C-reactive protein (CRP) (to < 1 mg/dL, with an absence of successive elevations to ≥ 1mg/dL) from Week 12 through Week 52, and (4) successful adherence to the prednisone taper defined by not more than 100 mg of excess prednisone from Week 12 through Week 52. Tocilizumab 162 mg weekly and 162 mg every other week + 26 weeks prednisone taper both showed superiority in achieving sustained remission from Week 12 through Week 52 compared with placebo + 26 weeks prednisone taper ( Table 8 ). Both tocilizumab treatment arms also showed superiority compared to the placebo + 52 weeks prednisone taper ( Table 8 ).
|
a Sustained remission was achieved by a patient meeting all of the following components: absence of GCA signs and symptomsb, normalization of ESRc, normalization of CRPd and adherence to the prednisone taper regimene. b Patients who did not have any signs or symptoms of GCA recorded from Week 12 up to Week 52. c Patients who did not have an elevated ESR ≥30 mm/hr which was classified as attributed to GCA from Week 12 up to Week 52. d Patients who did not have two or more consecutive CRP records of ≥ 1mg/dL from Week 12 up to Week 52. e Patients who did not enter escape therapy and received ≤ 100mg of additional concomitant prednisone from Week 12 up to Week 52. |
||||
|
PBO + 26
|
PBO + 52
|
TCZ 162mg SC QW + 26
|
TCZ 162 mg SC Q2W + 26
|
|
|
Sustained remission a |
||||
|
Responders, n (%) |
7 (14.0%) |
9 (17.6%) |
56 (56.0%) |
26 (53.1%) |
|
Unadjusted difference in |
N/A |
N/A |
42.0% |
39.1% |
|
Unadjusted difference in |
N/A |
N/A |
38.4% |
35.4% |
|
Components of Sustained Remission |
||||
|
Sustained absence of GCA signs |
20 (40.0%) |
23 (45.1%) |
69 (69.0%) |
28 (57.1%) |
Patients not completing the study to week 52 were classified as non-responders in the primary and key secondary analysis: PBO+26: 6 (12.0%), PBO+52: 5 (9.8%), TCZ QW: 15 (15.0%), TCZ Q2W: 9 (18.4%).
CRP = C-reactive protein
ESR = erythrocyte sedimentation rate
PBO = placebo
Q2W = every other week dose
QW = every week dose
TCZ = tocilizumab
The estimated annual cumulative prednisone dose was lower in the two tocilizumab dose groups (medians of 1887 mg and 2207 mg on tocilizumab QW and Q2W, respectively) relative to the placebo arms (medians of 3804 mg and 3902 mg on placebo + 26 weeks prednisone and placebo + 52 weeks prednisone taper, respectively).
2.5 Recommended Dosage for Systemic Juvenile Idiopathic Arthritis
TYENNE may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change a dose based solely on a single visit body weight measurement, as weight may fluctuate.
2.4 Recommended Dosage for Polyarticular Juvenile Idiopathic Arthritis
TYENNE may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change dose based solely on a single visit body weight measurement, as weight may fluctuate.
14.7 Systemic Juvenile Idiopathic Arthritis Intravenous Administration
The efficacy of tocilizumab for the treatment of active SJIA was assessed in WA18221 (NCT00642460), a 12-week randomized, double blind, placebo-controlled, parallel group, 2-arm study. Patients treated with or without MTX, were randomized (tocilizumab:placebo = 2:1) to one of two treatment groups: 75 patients received tocilizumab infusions every two weeks at either 8 mg per kg for patients at or above 30 kg or 12 mg per kg for patients less than 30 kg and 37 were randomized to receive placebo infusions every two weeks. Corticosteroid tapering could occur from week six for patients who achieved a JIA ACR 70 response. After 12 weeks or at the time of escape, due to disease worsening, patients were treated with tocilizumab in the open-label extension phase at weight appropriate dosing.
The primary endpoint was the proportion of patients with at least 30% improvement in JIA ACR core set (JIA ACR 30 response) at Week 12 and absence of fever (no temperature at or above 37.5°C in the preceding 7 days). JIA ACR (American College of Rheumatology) responses are defined as the percentage improvement (e.g., 30%, 50%, 70%) in 3 of any 6 core outcome variables compared to baseline, with worsening in no more than 1 of the remaining variables by 30% or more.
Core outcome variables consist of physician global assessment, parent per patient global assessment, number of joints with active arthritis, number of joints with limitation of movement, erythrocyte sedimentation rate (ESR), and functional ability (childhood health assessment questionnaire-CHAQ).
Primary endpoint result and JIA ACR response rates at Week 12 are shown in Table 9 .
|
aThe weighted difference is the difference between the tocilizumab and Placebo response rates, adjusted for the stratification factors (weight, disease duration, background oral corticosteroid dose and background methotrexate use). b CI: confidence interval of the weighted difference. |
||
|
Tocilizumab
|
Placebo
|
|
|
Primary Endpoint: JIA ACR 30 response + absence of fever |
||
|
Responders |
85% |
24% |
|
Weighted difference (95% CI) |
62
|
- |
|
JIA ACR Response Rates at Week 12 |
||
|
JIA ACR 30 |
||
|
Responders Weighted difference a (95% CI) b |
91% |
24% |
|
JIA ACR 50 |
||
|
Responders Weighted difference a (95% CI) b |
85% |
11% |
|
JIA ACR 70 |
||
|
Responders Weighted difference a (95% CI) b |
71% |
8% |
The treatment effect of tocilizumab was consistent across all components of the JIA ACR response core variables. JIA ACR scores and absence of fever responses in the open label extension were consistent with the controlled portion of the study (data available through 44 weeks).
2.6 Preparation and Administration Instructions for Intravenous Infusion
TYENNE for intravenous infusion should be diluted by a healthcare professional using aseptic technique as follows:
-
•Use a sterile needle and syringe to prepare TYENNE.
-
•Patients less than 30 kg: use a 50 mL infusion bag or bottle of 0.9% or 0.45% Sodium Chloride Injection, USP, and then follow steps 1 and 2 below.
-
•Patients at or above 30 kg weight: use a 100 mL infusion bag or bottle, and then follow steps 1 and 2 below.
-
-Step 1. Withdraw a volume of 0.9% or 0.45% Sodium Chloride Injection, USP, equal to the volume of the TYENNE injection required for the patient's dose from the infusion bag or bottle [see Dosage and Administration (2.2, 2.4, 2.5)].
|
For Intravenous Use: Volume of TYENNE Injection per kg of Body Weight |
||
|
Dosage |
Indication |
Volume of TYENNE injection per kg of body weight |
|
4 mg/kg |
Adult RA |
0.2 mL/kg |
|
6 mg/kg |
Adult GCA |
0.3 mL/kg |
|
8 mg/kg |
Adult RA SJIA and PJIA (greater than or equal to 30 kg of body weight) |
0.4 mL/kg |
|
10 mg/kg |
PJIA (less than 30 kg of body weight) |
0.5 mL/kg |
|
12 mg/kg |
SJIA (less than 30 kg of body weight) |
0.6 mL/kg |
-
-Step 2. Withdraw the amount of TYENNE for intravenous infusion from the vial(s) and add slowly into the 0.9% or 0.45% Sodium Chloride Injection, USP infusion bag or bottle. To mix the solution, gently invert the bag to avoid foaming.
-
•The prepared solution for infusion should be used immediately. If not used immediately, the diluted tocilizumab solutions may be refrigerated at 36°F to 46°F (2°C to 8°C) up to 24 hours, or stored at room temperature at or below 77°F (25°C) for up to 4 hours and should be protected from light. Administration of diluted TYENNE solution must be completed within this period of time.
-
•TYENNE solutions do not contain preservatives; therefore, unused product remaining in the vials should not be used.
-
•Allow the fully diluted TYENNE solution to reach room temperature prior to infusion.
-
•The infusion should be administered over 60 minutes and must be administered with an infusion set. Do not administer as an intravenous push or bolus.
-
•TYENNE should not be infused concomitantly in the same intravenous line with other drugs. No physical or biochemical compatibility studies have been conducted to evaluate the co-administration of TYENNE with other drugs.
-
•Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If particulates and discolorations are noted, the product should not be used.
-
•Fully diluted TYENNE solutions are compatible with polypropylene, polyethylene and polyvinyl chloride infusion bags and bottles, and glass infusion bottles.
14.8 Systemic Juvenile Idiopathic Arthritis Subcutaneous Administration
Subcutaneously administered tocilizumab in pediatric patients with systemic juvenile idiopathic arthritis (SJIA) was assessed in WA28118 (NCT01904292), a 52-week, open-label, multicenter, PK-PD and safety study to determine the appropriate subcutaneous dose of tocilizumab that achieved comparable PK/PD profiles to the tocilizumab-IV regimen.
Eligible patients received tocilizumab subcutaneously dosed according to body weight, with patients weighing at or above 30 kg (n = 26) dosed with 162 mg of tocilizumab every week and patients weighing below 30 kg (n = 25) dosed with 162 mg of tocilizumab every 10 days (n=8) or every 2 weeks (n=17) for 52 weeks. Of these 51 patients, 26 (51%) were naïve to subcutaneous tocilizumab and 25 (49%) had been receiving tocilizumab intravenously and switched to subcutaneous tocilizumab at baseline.
The efficacy of subcutaneous tocilizumab in children 2 to 17 years of age is based on pharmacokinetic exposure and extrapolation of the established efficacy of intravenous tocilizumab in systemic JIA patients [see Clinical Pharmacology (12.3) and Clinical Studies (14.8 )].
2.7 Preparation and Administration Instructions for Subcutaneous Injection
-
•TYENNE for subcutaneous injection is not intended for intravenous drip infusion.
-
•Assess suitability of patient for subcutaneous home use and instruct patients to inform a healthcare professional before administering the next dose if they experience any symptoms of allergic reaction. Patients should seek immediate medical attention if they develop symptoms of serious allergic reactions. TYENNE subcutaneous injection is intended for use under the guidance of a healthcare practitioner. After proper training in subcutaneous injection technique, a patient may self-inject TYENNE or the patient's caregiver may administer TYENNE if a healthcare practitioner determines that it is appropriate. PJIA and SJIA patients may self-inject with the TYENNE prefilled syringe or autoinjector, or the patient's caregiver may administer TYENNE if both the healthcare practitioner and the parent/legal guardian determine it is appropriate [see Use in Specific Populations (8.4)].
Patients, or patient caregivers, should be instructed to follow the directions provided in the Instructions for Use (IFU) for additional details on medication administration. -
•Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use TYENNE prefilled syringes (PFS) or prefilled autoinjectors exhibiting particulate matter, cloudiness, or discoloration. TYENNE for subcutaneous administration should be clear and colorless to pale yellow. Do not use if any part of the PFS or autoinjector appears to be damaged.
-
•Patients using TYENNE for subcutaneous administration should be instructed to inject the full amount in the syringe (0.9 mL) or full amount in the autoinjector (0.9 mL), which provides 162 mg of TYENNE, according to the directions provided in the IFU.
-
•Injection sites should be rotated with each injection and should never be given into moles, scars, or areas where the skin is tender, bruised, red, hard, or not intact.
14.5 Polyarticular Juvenile Idiopathic Arthritis – Intravenous Administration
The efficacy of tocilizumab was assessed in a three-part study, WA19977 (NCT00988221), including an open- label extension in children 2 to 17 years of age with active polyarticular juvenile idiopathic arthritis (PJIA), who had an inadequate response to methotrexate or inability to tolerate methotrexate. Patients had at least 6 months of active disease (mean disease duration of 4.2 ± 3.7 years), with at least five joints with active arthritis (swollen or limitation of movement accompanied by pain and/or tenderness) and/or at least 3 active joints having limitation of motion (mean, 20 ± 14 active joints). The patients treated had subtypes of JIA that at disease onset included Rheumatoid Factor Positive or Negative Polyarticular JIA, or Extended Oligoarticular JIA. Treatment with a stable dose of methotrexate was permitted but was not required during the study. Concurrent use of disease modifying antirheumatic drugs (DMARDs), other than methotrexate, or other biologics (e.g., TNF antagonists or T cell costimulation modulator) were not permitted in the study.
Part I consisted of a 16-week active tocilizumab treatment lead-in period (n=188) followed by Part II, a 24-week randomized double-blind placebo-controlled withdrawal period, followed by Part III, a 64-week open-label period. Eligible patients weighing at or above 30 kg received tocilizumab at 8 mg/kg intravenously once every four weeks. Patients weighing less than 30 kg were randomized 1:1 to receive either tocilizumab 8 mg/kg or 10 mg/kg intravenously every four weeks. At the conclusion of the open-label Part I, 91% of patients taking background MTX in addition to tocilizumab and 83% of patients on tocilizumab monotherapy achieved an ACR 30 response at week 16 compared to baseline and entered the blinded withdrawal period (Part II) of the study.
The proportions of patients with JIA ACR 50/70 responses in Part I were 84.0%, and 64%, respectively for patients taking background MTX in addition to tocilizumab and 80% and 55% respectively for patients on tocilizumab monotherapy.
In Part II, patients (ITT, n=163) were randomized to tocilizumab (same dose received in Part I) or placebo in a 1:1 ratio that was stratified by concurrent methotrexate use and concurrent corticosteroid use. Each patient continued in Part II of the study until Week 40 or until the patient satisfied JIA ACR 30 flare criteria (relative to Week 16) and qualified for escape.
The primary endpoint was the proportion of patients with a JIA ACR 30 flare at week 40 relative to week 16. JIA ACR 30 flare was defined as 3 or more of the 6 core outcome variables worsening by at least 30% with no more than 1 of the remaining variables improving by more than 30% relative to Week 16.
Tocilizumab treated patients experienced significantly fewer disease flares compared to placebo-treated patients (26% [21/82] versus 48% [39/81]; adjusted difference in proportions -21%, 95% CI: -35%, -8%).
During the withdrawal phase (Part II), more patients treated with tocilizumab showed JIA ACR 30/50/70 responses at Week 40 compared to patients withdrawn to placebo.
14.6 Polyarticular Juvenile Idiopathic Arthritis – Subcutaneous Administration
Subcutaneously administered tocilizumab in pediatric patients with polyarticular juvenile idiopathic arthritis (PJIA) was assessed in WA28117 (NCT01904279), a 52-week, open-label, multicenter, PK-PD and safety study to determine the appropriate subcutaneous dose of tocilizumab that achieved comparable PK/PD profiles to the tocilizumab-IV regimen. PJIA patients aged 1 to 17 years with an inadequate response or inability to tolerate MTX, including patients with well-controlled disease on treatment with tocilizumab-IV and tocilizumab-naïve patients with active disease, were treated with subcutaneous tocilizumab based on body weight.
Patients weighing at or above 30 kg (n = 25) were treated with 162 mg of tocilizumab-SC every 2 weeks and patients weighing less than 30 kg (n = 27) received 162 mg of tocilizumab-SC every 3 weeks for 52 weeks. Of these 52 patients, 37 (71%) were naïve to tocilizumab and 15 (29%) had been receiving tocilizumab-IV and switched to tocilizumab-SC at baseline.
The efficacy of subcutaneous tocilizumab in children 2 to 17 years of age is based on pharmacokinetic exposure and extrapolation of the established efficacy of intravenous tocilizumab in polyarticular JIA patients and subcutaneous tocilizumab in patients with RA [see Clinical Pharmacology (12.3) and Clinical Studies (14.2 and 14.6)].
6.1 Clinical Trials Experience in Rheumatoid Arthritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv)
The tocilizumab-IV data in rheumatoid arthritis (RA) includes 5 double-blind, controlled, multicenter studies. In these studies, patients received doses of tocilizumab-IV 8 mg per kg monotherapy (288 patients), tocilizumab-IV 8 mg per kg in combination with DMARDs (including methotrexate) (1582 patients), or tocilizumab-IV 4 mg per kg in combination with methotrexate (774 patients).
The all exposure population includes all patients in registration studies who received at least one dose of tocilizumab-IV. Of the 4009 patients in this population, 3577 received treatment for at least 6 months, 3309 for at least one year; 2954 received treatment for at least 2 years and 2189 for 3 years.
All patients in these studies had moderately to severely active rheumatoid arthritis. The study population had a mean age of 52 years, 82% were female and 74% were Caucasian.
The most common serious adverse reactions were serious infections [see Warnings and Precautions (5.1)]. The most commonly reported adverse reactions in controlled studies up to 24 weeks (occurring in at least 5% of patients treated with tocilizumab-IV monotherapy or in combination with DMARDs) were upper respiratory tract infections, nasopharyngitis, headache, hypertension and increased ALT.
The proportion of patients who discontinued treatment due to any adverse reactions during the double-blind, placebo-controlled studies was 5% for patients taking tocilizumab-IV and 3% for placebo-treated patients. The most common adverse reactions that required discontinuation of tocilizumab-IV were increased hepatic transaminase values (per protocol requirement) and serious infections.
6.4 Clinical Trials Experience in Giant Cell Arteritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv)
The safety of tocilizumab-IV was studied in an open label PK-PD and safety study in 24 patients with GCA who were in remission on tocilizumab-IV at time of enrollment. Patients received tocilizumab 7 mg/kg every 4 weeks for 20 weeks, followed by 6 mg/kg every 4 weeks for 20 weeks. The total patient years exposure to treatment was 17.5 years. The overall safety profile observed for tocilizumab administered intravenously in GCA patients was consistent with the known safety profile of tocilizumab.
6.2 Clinical Trials Experience in Rheumatoid Arthritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc)
The tocilizumab-SC data in rheumatoid arthritis (RA) includes 2 double-blind, controlled, multicenter studies. Study SC-I was a non-inferiority study that compared the efficacy and safety of tocilizumab 162 mg administered every week subcutaneously and 8 mg/kg intravenously every four weeks in 1262 adult subjects with rheumatoid arthritis. Study SC-II was a placebo-controlled superiority study that evaluated the safety and efficacy of tocilizumab 162 mg administered every other week subcutaneously or placebo in 656 patients. All patients in both studies received background non-biologic DMARDs.
The safety observed for tocilizumab-SC administered subcutaneously was consistent with the known safety profile of intravenous tocilizumab, with the exception of injection site reactions (ISRs), which were more common with tocilizumab-SC compared with placebo SC injections (IV arm).
6.3 Clinical Trials Experience in Giant Cell Arteritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc)
The safety of subcutaneous tocilizumab has been studied in one Phase III study (WA28119) with 251 GCA patients. The total patient years duration in the tocilizumab-SC GCA all exposure population was 138.5 patient years during the 12-month double blind, placebo-controlled phase of the study. The overall safety profile observed in the tocilizumab-SC treatment groups was generally consistent with the known safety profile of tocilizumab. There was an overall higher incidence of infections in GCA patients relative to RA patients.
The rate of infection/serious infection events was 200.2/9.7 events per 100 patient years in the tocilizumab-SC weekly group and 160.2/4.4 events per 100 patient years in the tocilizumab-SC every other week group as compared to 156.0/4.2 events per 100 patient years in the placebo + 26 week prednisone taper and 210.2/12.5 events per 100 patient years in the placebo + 52 week taper groups.
6.7 Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv)
The data described below reflect exposure to tocilizumab-IV in one randomized, double-blind, placebo-controlled trial of 112 pediatric patients with SJIA 2 to 17 years of age who had an inadequate clinical response to nonsteroidal anti-inflammatory drugs (NSAIDs) or corticosteroids due to toxicity or lack of efficacy. At baseline, approximately half of the patients were taking 0.3 mg/kg/day corticosteroids or more, and almost 70% were taking methotrexate. The trial included a 12 week controlled phase followed by an open-label extension. In the 12 week double-blind, controlled portion of the clinical study 75 patients received treatment with tocilizumab-IV (8 or 12 mg per kg based upon body weight). After 12 weeks or at the time of escape, due to disease worsening, patients were treated with tocilizumab-IV in the open-label extension phase.
The most common adverse events (at least 5%) seen in tocilizumab-IV treated patients in the 12 week controlled portion of the study were: upper respiratory tract infection, headache, nasopharyngitis and diarrhea.
6.8 Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc)
The safety profile of tocilizumab-SC was studied in 51 pediatric patients 1 to 17 years of age with SJIA who had an inadequate clinical response to NSAIDs and corticosteroids. In general, the safety observed for tocilizumab administered subcutaneously was consistent with the known safety profile of intravenous tocilizumab, with the exception of ISRs where a higher frequency was observed in tocilizumab-SC treated SJIA patients compared to PJIA patients and adult RA or GCA patients [see Adverse Reactions (6.2, 6.3 and 6.6)].
6.5 Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv)
The safety of tocilizumab-IV was studied in 188 pediatric patients 2 to 17 years of age with PJIA who had an inadequate clinical response or were intolerant to methotrexate. The total patient exposure in the tocilizumab-IV all exposure population (defined as patients who received at least one dose of tocilizumab-IV) was 184.4 patient years. At baseline, approximately half of the patients were taking oral corticosteroids and almost 80% were taking methotrexate. In general, the types of adverse drug reactions in patients with PJIA were consistent with those seen in RA and SJIA patients [see Adverse Reactions (6.1 and 6.7)].
6.6 Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc)
The safety of tocilizumab-SC was studied in 52 pediatric patients 1 to 17 years of age with PJIA who had an inadequate clinical response or were intolerant to methotrexate. The total patient exposure in the PJIA tocilizumab-SC population (defined as patients who received at least one dose of tocilizumab-SC and accounting for treatment discontinuation) was 49.5 patient years.
In general, the safety observed for tocilizumab administered subcutaneously was consistent with the known safety profile of intravenous tocilizumab, with the exception of injection site reactions (ISRs), and neutropenia.
Structured Label Content
Data
Animal Data
An embryo-fetal developmental toxicity study was performed in which pregnant Cynomolgus monkeys were treated intravenously with tocilizumab at daily doses of 2, 10, or 50 mg/ kg during organogenesis from gestation day (GD) 20-50. Although there was no evidence for a teratogenic/dysmorphogenic effect at any dose, tocilizumab produced an increase in the incidence of abortion/embryo-fetal death at doses 1.25 times and higher the MRHD by the intravenous route at maternal intravenous doses of 10 and 50 mg/ kg. Testing of a murine analogue of tocilizumab in mice did not yield any evidence of harm to offspring during the pre- and postnatal development phase when dosed at 50 mg/kg intravenously with treatment every three days from implantation (GD 6) until post-partum day 21 (weaning). There was no evidence for any functional impairment of the development and behavior, learning ability, immune competence and fertility of the offspring.
Parturition is associated with significant increases of IL-6 in the cervix and myometrium. The literature suggests that inhibition of IL-6 signaling may interfere with cervical ripening and dilatation and myometrial contractile activity leading to potential delays of parturition. For mice deficient in IL-6 (ll6-/- null mice), parturition was delayed relative to wild-type (ll6+/+) mice.
Administration of recombinant IL-6 to ll6-/- null mice restored the normal timing of delivery.
Section 42229-5 (42229-5)
Not Recommended for Concomitant Use with Biological DMARDs
Tocilizumab products have not been studied in combination with biological DMARDs such as TNF antagonists, IL-1R antagonists, anti-CD20 monoclonal antibodies and selective co-stimulation modulators because of the possibility of increased immunosuppression and increased risk of infection. Avoid using TYENNE with biological DMARDs.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 12/2024 | ||
|
Medication Guide
|
||
|
What is the most important information I should know about TYENNE?
TYENNE can cause serious side effects including:
|
||
|
Your healthcare provider should monitor you closely for signs and symptoms of TB during treatment with TYENNE.
Before starting TYENNE, tell your healthcare provider if you:
|
||
|
|
|
After starting TYENNE, call your healthcare provider right away if you have any symptoms of an infection. TYENNE can make you more likely to get infections or make worse any infection that you have.
|
||
|
|
|
|
||
|
Your healthcare provider will determine how often you will have follow-up blood tests. Make sure you get all your follow-up blood tests done as ordered by your healthcare provider.
|
||
|
See “What are the possible side effects with TYENNE?” for more information about side effects. |
||
|
What is TYENNE?
|
||
|
It is not known if TYENNE is safe and effective in children with PJIA or SJIA under 2 years of age or in children with conditions other than PJIA or SJIA. |
||
|
Do not take TYENNE: if you are allergic to tocilizumab products, or any of the ingredients in TYENNE. See the end of this Medication Guide for a complete list of ingredients in TYENNE. |
||
|
Before you receive TYENNE, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
Tell your healthcare provider about all of the medicines you take, including prescription, over-the-counter medicines, vitamins, and herbal supplements. TYENNE and other medicines may affect each other causing side effects.
|
||
|
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine. |
||
|
How will I receive TYENNE?
|
||
|
Under the skin (SC or subcutaneous injection) for Rheumatoid Arthritis, Giant Cell Arteritis, PJIA or SJIA:
|
||
|
What are the possible side effects with TYENNE? TYENNE can cause serious side effects, including:
|
||
|
|
|
|
||
|
The most common side effects of TYENNE include:
|
||
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
|
||
|
What are the ingredients in TYENNE?
|
Section 43683-2 (43683-2)
Warnings and Precautions (5.6) 12/2024
Section 44425-7 (44425-7)
Storage and Handling: Do not use beyond expiration date on the container, package, prefilled syringe, or autoinjector. TYENNE must be refrigerated at 36°F to 46°F (2ºC to 8ºC). Do not freeze. A single prefilled syringe (or autoinjector) may be stored at room temperature at or below 77°F (25°C) for a single period of up to 14 days. Protect the vials, syringes and autoinjectors from light by storage in the original package until time of use, and keep syringes and autoinjectors dry.
10 Overdosage (10 OVERDOSAGE)
There are limited data available on overdoses with tocilizumab products. One case of accidental overdose was reported with intravenous tocilizumab in which a patient with multiple myeloma received a dose of 40 mg per kg. No adverse drug reactions were observed. No serious adverse drug reactions were observed in healthy volunteers who received single doses of up to 28 mg per kg, although all 5 patients at the highest dose of 28 mg per kg developed dose-limiting neutropenia.
In case of an overdose, it is recommended that the patient be monitored for signs and symptoms of adverse reactions. Patients who develop adverse reactions should receive appropriate symptomatic treatment.
11 Description (11 DESCRIPTION)
Tocilizumab-aazg is a recombinant humanized anti-human interleukin 6 (IL-6) receptor monoclonal antibody of the immunoglobulin IgG1κ (gamma 1, kappa) subclass with a typical H2L2 polypeptide structure. Each light chain and heavy chain consists of 214 and 448 amino acids (excluding the C-terminal lysine), respectively.
The four polypeptide chains are linked intra- and inter-molecularly by disulfide bonds. Tocilizumab-aazg has a molecular weight of approximately 148 kDa. The antibody is produced in mammalian (Chinese hamster ovary) cells.
Intravenous Infusion
TYENNE (tocilizumab-aazg) injection is a sterile, clear and colorless to pale yellow, histidine buffered preservative-free solution with a pH of approximately 6 for further dilution prior to intravenous infusion. Each single-dose vial is available at a concentration of 20 mg/mL containing 80 mg/4 mL, 200 mg/10 mL, or 400 mg/20 mL of TYENNE. Each mL of solution contains arginine (17.4 mg), histidine (3.1 mg), lactic acid (0.9 mg), polysorbate 80 (0.2 mg), sodium chloride (0.6 mg), and Water for Injection, USP. Hydrochloric acid and sodium hydroxide are added to adjust the pH.
Subcutaneous Injection
TYENNE (tocilizumab-aazg) injection is a sterile, clear, colorless to pale yellow, preservative-free, histidine buffered solution with a pH of approximately 6 for subcutaneous use. It is supplied in a ready-to-use, single-dose 0.9 mL prefilled syringe (PFS) with a needle safety device or in a ready-to-use, single-dose 0.9 mL autoinjector that delivers 162 mg tocilizumab-aazg, arginine (16.7 mg), histidine (2.0 mg), lactic acid (0.9 mg), polysorbate 80 (0.2 mg), sodium chloride (0.6 mg), and Water for Injection, USP. Hydrochloric acid and sodium hydroxide are added to adjust the pH.
5.9 Vaccinations
Avoid use of live vaccines concurrently with TYENNE as clinical safety has not been established. No data are available on the secondary transmission of infection from persons receiving live vaccines to patients receiving tocilizumab products.
No data are available on the effectiveness of vaccination in patients receiving tocilizumab products. Because IL-6 inhibition may interfere with the normal immune response to new antigens, it is recommended that all patients, particularly pediatric or elderly patients, if possible, be brought up to date with all immunizations in agreement with current immunization guidelines prior to initiating TYENNE therapy. The interval between live vaccinations and initiation of TYENNE therapy should be in accordance with current vaccination guidelines regarding immunosuppressive agents.
7.3 Live Vaccines
Avoid use of live vaccines concurrently with TYENNE [see Warnings and Precautions (5.9)].
8.4 Pediatric Use
TYENNE by intravenous use is indicated for the treatment of pediatric patients with:
-
•Active systemic juvenile idiopathic arthritis in patients 2 years of age and older
-
•Active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older
TYENNE by subcutaneous use is indicated for the treatment of pediatric patients with:
-
•Active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older
-
•Active systemic juvenile idiopathic arthritis in patients 2 years of age and older
The safety and effectiveness of TYENNE in pediatric patients with conditions other than PJIA or SJIA have not been established. The safety and effectiveness in pediatric patients below the age of 2 have not been established in PJIA or SJIA.
8.5 Geriatric Use
Of the 2644 patients who received tocilizumab in Studies I to V [see Clinical Studies (14)], a total of 435 rheumatoid arthritis patients were 65 years of age and older, including 50 patients 75 years and older. Of the 1069 patients who received tocilizumab-SC in studies SC-I and SC-II there were 295 patients 65 years of age and older, including 41 patients 75 years and older. The frequency of serious infection among tocilizumab treated subjects 65 years of age and older was higher than those under the age of 65. As there is a higher incidence of infections in the elderly population in general, caution should be used when treating the elderly.
5.3 Hepatotoxicity
Serious cases of hepatic injury have been observed in patients taking intravenous or subcutaneous tocilizumab products. Some of these cases have resulted in liver transplant or death. Time to onset for cases ranged from months to years after treatment initiation with tocilizumab products. While most cases presented with marked elevations of transaminases (> 5 times ULN), some cases presented with signs or symptoms of liver dysfunction and only mildly elevated transaminases.
During randomized controlled studies, treatment with tocilizumab was associated with a higher incidence of transaminase elevations [see Adverse Reactions (6.1, 6.2, 6.5, 6.7)]. Increased frequency and magnitude of these elevations was observed when potentially hepatotoxic drugs (e.g., MTX) were used in combination with tocilizumab.
For RA and GCA patients, obtain a liver test panel (serum alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase, and total bilirubin) before initiating TYENNE, every 4 to 8 weeks after start of therapy for the first 6 months of treatment and every 3 months thereafter. It is not recommended to initiate TYENNE treatment in RA or GCA patients with elevated transaminases ALT or AST greater than 1.5x ULN. In patients who develop elevated ALT or AST greater than 5x ULN, discontinue TYENNE. For recommended modifications based upon increase in transaminases [see Dosage and Administration (2.8)].
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, such as fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient is found to have abnormal liver tests (e.g., ALT greater than three times the upper limit of the reference range, serum total bilirubin greater than two times the upper limit of the reference range), TYENNE treatment should be interrupted and investigation done to establish the probable cause. TYENNE should only be restarted in patients with another explanation for the liver test abnormalities after normalization of the liver tests.
A similar pattern of liver enzyme elevation is noted with tocilizumab products treatment in the PJIA and SJIA populations. Monitor liver test panel at the time of the second administration and thereafter every 4 to 8 weeks for PJIA and every 2 to 4 weeks for SJIA.
4 Contraindications (4 CONTRAINDICATIONS)
TYENNE is contraindicated in patients with known hypersensitivity to tocilizumab products [see Warnings and Precautions (5.6)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in labeling:
-
•Serious Infections [see Warnings and Precautions (5.1)]
-
•Gastrointestinal Perforations [see Warnings and Precautions (5.2)]
-
•Laboratory Parameters [see Warnings and Precautions (5.4)]
-
•Immunosuppression [see Warnings and Precautions (5.5)]
-
•Hypersensitivity Reactions, Including Anaphylaxis [see Warnings and Precautions (5.6)]
-
•Demyelinating Disorders [see Warnings and Precautions (5.7)]
-
•Active Hepatic Disease and Hepatic Impairment [see Warnings and Precautions (5.8)]
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
8.7 Renal Impairment
No dose adjustment is required in patients with mild or moderate renal impairment. Tocilizumab products have not been studied in patients with severe renal impairment [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In clinical studies in RA patients with the 4 mg per kg and 8 mg per kg intravenous doses or the 162 mg weekly and every other weekly subcutaneous doses of tocilizumab, decreases in levels of C-reactive protein (CRP) to within normal ranges were seen as early as week 2. Changes in pharmacodynamic parameters were observed (i.e., decreases in rheumatoid factor, erythrocyte sedimentation rate (ESR), serum amyloid A, fibrinogen and increases in hemoglobin) with doses, however the greatest improvements were observed with 8 mg per kg tocilizumab. Pharmacodynamic changes were also observed to occur after tocilizumab administration in GCA, PJIA, and SJIA patients (decreases in CRP, ESR, and increases in hemoglobin). The relationship between these pharmacodynamic findings and clinical efficacy is not known.
In healthy subjects administered tocilizumab in doses from 2 to 28 mg per kg intravenously and 81 to 162 mg subcutaneously, absolute neutrophil counts decreased to the nadir 3 to 5 days following tocilizumab administration. Thereafter, neutrophils recovered towards baseline in a dose dependent manner. Rheumatoid arthritis and GCA patients demonstrated a similar pattern of absolute neutrophil counts following tocilizumab administration [see Warnings and Precautions (5.4)].
12.3 Pharmacokinetics
PK of tocilizumab is characterized by nonlinear elimination which is a combination of linear clearance and Michaelis-Menten elimination. The nonlinear part of tocilizumab elimination leads to an increase in exposure that is more than dose-proportional. The pharmacokinetic parameters of tocilizumab do not change with time. Due to the dependence of total clearance on tocilizumab serum concentrations, the half-life of tocilizumab is also concentration-dependent and varies depending on the serum concentration level. Population pharmacokinetic analyses in any patient population tested so far indicate no relationship between apparent clearance and the presence of anti-drug antibodies.
5.5 Immunosuppression
The impact of treatment with tocilizumab products on the development of malignancies is not known but malignancies were observed in clinical studies [see Adverse Reactions (6.1)]. TYENNE is an immunosuppressant, and treatment with immunosuppressants may result in an increased risk of malignancies.
5.1 Serious Infections
Serious and sometimes fatal infections due to bacterial, mycobacterial, invasive fungal, viral, protozoal, or other opportunistic pathogens have been reported in patients receiving immunosuppressive agents including tocilizumab products. The most common serious infections included pneumonia, urinary tract infection, cellulitis, herpes zoster, gastroenteritis, diverticulitis, sepsis and bacterial arthritis [see Adverse Reactions (6.1)]. Among opportunistic infections, tuberculosis, cryptococcus, aspergillosis, candidiasis, and pneumocystosis were reported with tocilizumab products. Other serious infections, not reported in clinical studies, may also occur (e.g., histoplasmosis, coccidioidomycosis, listeriosis). Patients have presented with disseminated rather than localized disease, and were often taking concomitant immunosuppressants such as methotrexate or corticosteroids which in addition to rheumatoid arthritis may predispose them to infections.
Do not administer TYENNE in patients with an active infection, including localized infections. The risks and benefits of treatment should be considered prior to initiating TYENNE in patients:
-
•with chronic or recurrent infection;
-
•who have been exposed to tuberculosis;
-
•with a history of serious or an opportunistic infection;
-
•who have resided or traveled in areas of endemic tuberculosis or endemic mycoses; or
-
•with underlying conditions that may predispose them to infection.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with TYENNE, as signs and symptoms of acute inflammation may be lessened due to suppression of the acute phase reactants [see Dosage and Administration (2.1), Adverse Reactions (6.1), and Patient Counseling Information (17)].
Hold TYENNE if a patient develops a serious infection, an opportunistic infection, or sepsis. A patient who develops a new infection during treatment with TYENNE should undergo a prompt and complete diagnostic workup appropriate for an immunocompromised patient, initiate appropriate antimicrobial therapy, and closely monitor the patient.
8.6 Hepatic Impairment
The safety and efficacy of tocilizumab products have not been studied in patients with hepatic impairment, including patients with positive HBV and HCV serology [see Warnings and Precautions (5.8)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
TYENNE® (tocilizumab-aazg) is an interleukin-6 (IL-6) receptor antagonist indicated for treatment of:
Rheumatoid Arthritis (RA) (1.1)
-
•Adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs).
Giant Cell Arteritis (GCA) (1.2)
-
•Adult patients with giant cell arteritis.
Polyarticular Juvenile Idiopathic Arthritis (PJIA) (1.3)
-
•Patients 2 years of age and older with active polyarticular juvenile idiopathic arthritis.
Systemic Juvenile Idiopathic Arthritis (SJIA) (1.4)
-
•Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
Clinical Considerations
Fetal/Neonatal adverse reactions
Monoclonal antibodies are increasingly transported across the placenta as pregnancy progresses, with the largest amount transferred during the third trimester. Risks and benefits should be considered prior to administering live or live-attenuated vaccines to infants exposed to TYENNE in utero [see Warnings and Precautions (5.9)].
Disease-associated Maternal Risk
Published data suggest that the risk of adverse pregnancy outcomes in women with rheumatoid arthritis is associated with increased disease activity. Adverse pregnancy outcomes include preterm delivery (before 37 weeks of gestation), low birth weight (less than 2500 g) infants, and small for gestational age at birth.
12.1 Mechanism of Action
Tocilizumab products bind to both soluble and membrane-bound IL-6 receptors (sIL-6R and mIL-6R), and have been shown to inhibit IL-6-mediated signaling through these receptors. IL-6 is a pleiotropic pro-inflammatory cytokine produced by a variety of cell types including T- and B-cells, lymphocytes, monocytes and fibroblasts. IL-6 has been shown to be involved in diverse physiological processes such as T-cell activation, induction of immunoglobulin secretion, initiation of hepatic acute phase protein synthesis, and stimulation of hematopoietic precursor cell proliferation and differentiation. IL-6 is also produced by synovial and endothelial cells leading to local production of IL-6 in joints affected by inflammatory processes such as rheumatoid arthritis.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Serious Infections - do not administer TYENNE during an active infection, including localized infections. If a serious infection develops, interrupt TYENNE until the infection is controlled. (5.1)
-
•Gastrointestinal (GI) perforation-use with caution in patients who may be at increased risk. (5.2)
-
•Hepatotoxicity - monitor patients for signs and symptoms of hepatic injury. Modify or discontinue TYENNE if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. (2.8, 5.3)
-
•Laboratory monitoring recommended due to potential consequences of treatment-related changes in neutrophils, platelets, lipids, and liver function tests. (2.8, 5.4)
-
•Hypersensitivity reactions, including anaphylaxis and death and serious cutaneous reactions including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)- discontinue TYENNE, treat promptly, and monitor until reaction resolves (5.6)
-
•Live vaccines - Avoid use with TYENNE (5.9, 7.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
For RA, pJIA and sJIA, TYENNE may be used alone or in combination with methotrexate; and in RA, other non-biologic DMARDs may be used. (2)
General Administration and Dosing Information (2.1)
-
•RA, GCA, PJIA and SJIA - It is recommended that TYENNE not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm3, platelet count below 100,000 per mm3, or ALT or AST above 1.5 times the upper limit of normal (ULN) (5.3, 5.4).
-
•In RA patients, TYENNE doses exceeding 800 mg per infusion are not recommended. (2.2, 2.7, 12.3)
-
•In GCA patients, TYENNE doses exceeding 600 mg per infusion are not recommended. (2.3, 12.3)
Rheumatoid Arthritis (2.2)
Recommended Adult Intravenous Dosage:
When used in combination with non-biologic DMARDs or as monotherapy the recommended starting dose is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response.
Recommended Adult Subcutaneous Dosage:
|
Patients less than 100 kg weight |
162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response |
|
Patients at or above 100 kg weight |
162 mg administered subcutaneously every week |
Giant Cell Arteritis (2.3)
Recommended Adult Intravenous Dosage: The recommended dose is 6 mg per kg every 4 weeks in combination with a tapering course of glucocorticoids. TYENNE can be used alone following discontinuation of glucocorticoids.
Recommended Adult Subcutaneous Dosage: The recommended dose is 162 mg given once every week as a subcutaneous injection, in combination with a tapering course of glucocorticoids.
A dose of 162 mg given once every other week as a subcutaneous injection, in combination with a tapering course of glucocorticoids, may be prescribed based on clinical considerations.
TYENNE can be used alone following discontinuation of glucocorticoids.
Polyarticular Juvenile Idiopathic Arthritis (2.4)
|
Recommended Intravenous PJIA Dosage Every 4 Weeks |
|
|
Patients less than 30 kg weight |
10 mg per kg |
|
Patients at or above 30 kg weight |
8 mg per kg |
|
Recommended Subcutaneous PJIA Dosage |
|
|
Patients less than 30 kg weight |
162 mg once every three weeks |
|
Patients at or above 30 kg weight |
162 mg once every two weeks |
Systemic Juvenile Idiopathic Arthritis (2.5)
|
Recommended Intravenous SJIA Dosage Every 2 Weeks |
|
|
Patients less than 30 kg weight |
12 mg per kg |
|
Patients at or above 30 kg weight |
8 mg per kg |
|
Recommended Subcutaneous SJIA Dosage |
|
|
Patients less than 30 kg weight |
162 mg every two weeks |
|
Patients at or above 30 kg weight |
162 mg every week |
Administration of Intravenous formulation (2.6)
-
•For patients with RA, GCA, PJIA and SJIA patients at or above 30 kg, dilute to 100 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
-
•For PJIA and SJIA patients less than 30 kg, dilute to 50 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
-
•Administer as a single intravenous drip infusion over 1 hour; do not administer as bolus or push.
Administration of Subcutaneous formulation (2.7)
-
•Follow the Instructions for Use for prefilled syringe and prefilled autoinjector
Dose Modifications (2.8)
Recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia.
5.7 Demyelinating Disorders
The impact of treatment with tocilizumab products on demyelinating disorders is not known, but multiple sclerosis and chronic inflammatory demyelinating polyneuropathy were reported rarely in RA clinical studies. Monitor patients for signs and symptoms potentially indicative of demyelinating disorders. Prescribers should exercise caution in considering the use of TYENNE in patients with preexisting or recent onset demyelinating disorders.
9 Drug Abuse and Dependence (9 DRUG ABUSE AND DEPENDENCE)
No studies on the potential for tocilizumab products to cause dependence have been performed. However, there is no evidence from the available data that tocilizumab products treatment results in dependence.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Intravenous Infusion
Injection: 80 mg/4 mL (20 mg/mL), 200 mg/10 mL (20 mg/mL), 400 mg/20 mL (20 mg/mL) in single-dose vials for further dilution prior to intravenous infusion (3)
Subcutaneous Injection
Injection: 162 mg/0.9 mL in a single-dose prefilled syringe or single-dose prefilled autoinjector (3)
6.9 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of tocilizumab products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
1.1 Rheumatoid Arthritis (ra) (1.1 Rheumatoid Arthritis (RA))
TYENNE® (tocilizumab-aazg) is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
1.2 Giant Cell Arteritis (gca) (1.2 Giant Cell Arteritis (GCA))
TYENNE® (tocilizumab-aazg) is indicated for the treatment of giant cell arteritis (GCA) in adult patients.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
-
•Serious Infections
Inform patients that TYENNE may lower their resistance to infections [see Warnings and Precautions (5.1)]. Instruct the patient of the importance of contacting their doctor immediately when symptoms suggesting infection appear in order to assure rapid evaluation and appropriate treatment. -
•Gastrointestinal Perforation
Inform patients that some patients who have been treated with TYENNE have had serious side effects in the stomach and intestines [see Warnings and Precautions (5.2)]. Instruct the patient of the importance of contacting their doctor immediately when symptoms of fever, severe, persistent abdominal pain, and change in bowel habits appear to assure rapid evaluation and appropriate treatment. -
•Hypersensitivity and Serious Allergic Reactions
Inform patients that some patients who have been treated with TYENNE have developed serious allergic reactions, including anaphylaxis, as well as serious skin reactions [see Warnings and Precautions (5.6)]. Advise patients to stop taking TYENNE and seek immediate medical attention if they experience any symptom of serious allergic reactions (including rash, hives, and swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing).
5.2 Gastrointestinal Perforations
Events of gastrointestinal perforation have been reported in clinical trials, primarily as complications of diverticulitis in patients treated with tocilizumab. Use TYENNE with caution in patients who may be at increased risk for gastrointestinal perforation. Promptly evaluate patients presenting with fever, new onset abdominal symptoms, and a change in bowel habits for early identification of gastrointestinal perforation [see Adverse Reactions (6.1)].
Warning: Risk of Serious Infections (WARNING: RISK OF SERIOUS INFECTIONS)
Patients treated with tocilizumab products including TYENNE are at increased risk for developing serious infections that may lead to hospitalization or death [see Warnings and Precautions (5.1), Adverse Reactions (6.1)]. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids.
If a serious infection develops, interrupt TYENNE until the infection is controlled.
Reported infections include:
-
•Active tuberculosis, which may present with pulmonary or extrapulmonary disease. Patients should be tested for latent tuberculosis before TYENNE use and during therapy. Treatment for latent infection should be initiated prior to TYENNE use.
-
•Invasive fungal infections, including candidiasis, aspergillosis, and pneumocystis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
-
•Bacterial, viral and other infections due to opportunistic pathogens.
The risks and benefits of treatment with TYENNE should be carefully considered prior to initiating therapy in patients with chronic or recurrent infection.
Patients should be closely monitored for the development of signs and symptoms of infection during and after treatment with TYENNE including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy [see Warnings and Precautions (5.1)] .
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
TYENNE (tocilizumab-aazg) injection is a preservative-free, sterile clear and colorless to pale yellow solution. The following packaging configurations are available:
7.2 Interactions With Cyp450 Substrates (7.2 Interactions with CYP450 Substrates)
Cytochrome P450s in the liver are down-regulated by infection and inflammation stimuli including cytokines such as IL-6. Inhibition of IL-6 signaling in RA patients treated with tocilizumab products may restore CYP450 activities to higher levels than those in the absence of tocilizumab products leading to increased metabolism of drugs that are CYP450 substrates. In vitro studies showed that tocilizumab has the potential to affect expression of multiple CYP enzymes including CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP2D6 and CYP3A4. Its effect on CYP2C8 or transporters is unknown. In vivo studies with omeprazole, metabolized by CYP2C19 and CYP3A4, and simvastatin, metabolized by CYP3A4, showed up to a 28% and 57% decrease in exposure one week following a single dose of tocilizumab, respectively. The effect of tocilizumab products on CYP enzymes may be clinically relevant for CYP450 substrates with narrow therapeutic index, where the dose is individually adjusted.
Upon initiation or discontinuation of TYENNE, in patients being treated with these types of medicinal products, perform therapeutic monitoring of effect (e.g., warfarin) or drug concentration (e.g., cyclosporine or theophylline) and the individual dose of the medicinal product adjusted as needed. Exercise caution when coadministering TYENNE with CYP3A4 substrate drugs where decrease in effectiveness is undesirable, e.g., oral contraceptives, lovastatin, atorvastatin, etc. The effect of tocilizumab products on CYP450 enzyme activity may persist for several weeks after stopping therapy [see Clinical Pharmacology (12.3)].
Principal Display Panel – 4 Ml Vial Carton (Principal Display Panel – 4 mL Vial - Carton)
NDC 65219-590-04 Rx only
Tyenne®
tocilizumab-aazg
Injection
80 mg / 4 mL
(20 mg/mL)
For intravenous infusion only
after dilution
Dispense a Medication
Guide to each patient
Carton contains:
1 Single-use Vial - Discard Unused Portion
1 Prescribing Information
1 Medication Guide
1 x 4 mL
2.2 Recommended Dosage for Rheumatoid Arthritis
TYENNE may be used as monotherapy or concomitantly with methotrexate or other non-biologic DMARDs as an intravenous infusion or as a subcutaneous injection.
Principal Display Panel – 10 Ml Vial Carton (Principal Display Panel – 10 mL Vial - Carton)
NDC 65219-592-10 Rx only
Tyenne®
tocilizumab-aazg
Injection
200 mg / 10 mL
(20 mg/mL)
For intravenous infusion only
after dilution
Dispense a Medication
Guide to each patient
Carton contains:
1 Single-use Vial – Discard Unused Portion
1 Prescribing Information
1 Medication Guide
1 x 10 mL
Principal Display Panel – 20 Ml Vial Carton (Principal Display Panel – 20 mL Vial - Carton)
NDC 65219-594-20 Rx only
Tyenne®
tocilizumab-aazg
Injection
400 mg / 20 mL
(20 mg/mL)
For intravenous infusion only
after dilution
Dispense a Medication
Guide to each patient
Carton contains:
1 Single-use Vial - Discard Unused Portion
1 Prescribing Information
1 Medication Guide
1 x 20 mL
1.4 Systemic Juvenile Idiopathic Arthritis (sjia) (1.4 Systemic Juvenile Idiopathic Arthritis (SJIA))
TYENNE® (tocilizumab-aazg) is indicated for the treatment of active systemic juvenile idiopathic arthritis in patients 2 years of age and older.
5.8 Active Hepatic Disease and Hepatic Impairment
5.6 Hypersensitivity Reactions, Including Anaphylaxis
Hypersensitivity reactions, including anaphylaxis, have been reported in association with tocilizumab products and anaphylactic events with a fatal outcome have been reported with intravenous infusion of tocilizumab products. Anaphylaxis and other hypersensitivity reactions that required treatment discontinuation were reported in 0.1% (3 out of 2644) of patients in the 6-month controlled trials of intravenous tocilizumab, 0.2% (8 out of 4009) of patients in the intravenous all-exposure RA population, 0.7% (8 out of 1068) in the subcutaneous 6-month controlled RA trials, and in 0.7% (10 out of 1465) of patients in the subcutaneous all-exposure population. In the SJIA controlled trial with intravenous tocilizumab, 1 out of 112 patients (0.9%) experienced hypersensitivity reactions that required treatment discontinuation. In the PJIA controlled trial with intravenous tocilizumab, 0 out of 188 patients (0%) in the tocilizumab all-exposure population experienced hypersensitivity reactions that required treatment discontinuation. Reactions that required treatment discontinuation included generalized erythema, rash, and urticaria. Injection site reactions were categorized separately [see Adverse Reactions (6)].
In the postmarketing setting, events of hypersensitivity reactions, including anaphylaxis and death have occurred in patients treated with a range of doses of intravenous tocilizumab products, with or without concomitant therapies. Events have occurred in patients who received premedication. Hypersensitivity, including anaphylaxis events, have occurred both with and without previous hypersensitivity reactions and as early as the first infusion of tocilizumab products [see Adverse Reactions (6.9)]. In addition, serious cutaneous reactions, including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), have been reported in patients with autoinflammatory conditions treated with tocilizumab products.
TYENNE for intravenous use should only be infused by a healthcare professional with appropriate medical support to manage anaphylaxis. For TYENNE subcutaneous injection, advise patients to seek immediate medical attention if they experience any symptoms of a hypersensitivity reaction. If a hypersensitivity reaction occurs, stop immediately discontinue TYENNE treat promptly and monitor until signs and symptoms resolve.
Principal Display Panel – 4 Ml Vial Primary Label (Principal Display Panel – 4 mL Vial - Primary Label)
NDC 65219-590-04 Rx only 4 mL
Tyenne®
(tocilizumab-aazg)
Injection
80 mg / 4 mL
(20 mg/mL)
For Intravenous Infusion
Only After Dilution.
Fresenius Kabi USA, LLC
US License No. 2146
1.3 Polyarticular Juvenile Idiopathic Arthritis (pjia) (1.3 Polyarticular Juvenile Idiopathic Arthritis (PJIA))
TYENNE® (tocilizumab-aazg) is indicated for the treatment of active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older.
14.1 Rheumatoid Arthritis – Intravenous Administration
The efficacy and safety of intravenously administered tocilizumab was assessed in five randomized, double-blind, multicenter studies in patients greater than 18 years with active rheumatoid arthritis diagnosed according to American College of Rheumatology (ACR) criteria. Patients had at least 8 tender and 6 swollen joints at baseline. Tocilizumab was given intravenously every 4 weeks as monotherapy (Study I), in combination with methotrexate (MTX) (Studies II and III) or other disease-modifying anti-rheumatic drugs (DMARDs) (Study IV) in patients with an inadequate response to those drugs, or in combination with MTX in patients with an inadequate response to TNF antagonists (Study V).
Study I (NCT00109408) evaluated patients with moderate to severe active rheumatoid arthritis who had not been treated with MTX within 24 weeks prior to randomization, or who had not discontinued previous methotrexate treatment as a result of clinically important toxic effects or lack of response. In this study, 67% of patients were MTX-naïve, and over 40% of patients had rheumatoid arthritis less than 2 years. Patients received tocilizumab 8 mg per kg monotherapy or MTX alone (dose titrated over 8 weeks from 7.5 mg to a maximum of 20 mg weekly). The primary endpoint was the proportion of tocilizumab patients who achieved an ACR 20 response at Week 24.
Study II (NCT00106535) was a 104-week study with an optional 156-week extension phase that evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response to MTX. Patients received tocilizumab 8 mg per kg, tocilizumab 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). Upon completion of 52-weeks, patients received open-label treatment with tocilizumab 8 mg per kg through 104 weeks or they had the option to continue their double-blind treatment if they maintained a greater than 70% improvement in swollen/tender joint count. Two pre-specified interim analyses at week 24 and week 52 were conducted. The primary endpoint at week 24 was the proportion of patients who achieved an ACR 20 response. At weeks 52 and 104, the primary endpoints were change from baseline in modified total Sharp-Genant score and the area under the curve (AUC) of the change from baseline in HAQ-DI score.
Study III (NCT00106548) evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response to MTX. Patients received tocilizumab 8 mg per kg, tocilizumab 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
Study IV (NCT00106574) evaluated patients who had an inadequate response to their existing therapy, including one or more DMARDs. Patients received tocilizumab 8 mg per kg or placebo every four weeks, in combination with the stable DMARDs. The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
Study V (NCT00106522) evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response or were intolerant to one or more TNF antagonist therapies. The TNF antagonist therapy was discontinued prior to randomization. Patients received tocilizumab 8 mg per kg, tocilizumab 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
14.4 Giant Cell Arteritis – Intravenous Administration
Intravenously administered tocilizumab in patients with GCA was assessed in WP41152 (NCT03923738), an open-label PK-PD and safety study to determine the appropriate intravenous dose of tocilizumab that achieved comparable PK-PD profiles to the tocilizumab-SC regimen.
At enrollment, all patients (n=24) were in remission on tocilizumab-IV. In Period 1, all patients received open label tocilizumab-IV 7 mg/kg every 4 weeks for 20 weeks. Patients who completed Period 1 and remained in remission (n=22) were eligible to enter Period 2, and received open-label tocilizumab-IV 6 mg/kg every 4 weeks for 20 weeks.
The efficacy of intravenous tocilizumab 6 mg/kg in adult patients with GCA is based on pharmacokinetic exposure and extrapolation to the efficacy established for subcutaneous tocilizumab in patients with GCA [see Clinical Pharmacology (12.3) and Clinical Studies (14.3 )].
7.1 Concomitant Drugs for Treatment of Adult Indications
In RA patients, population pharmacokinetic analyses did not detect any effect of methotrexate (MTX), non-steroidal anti-inflammatory drugs or corticosteroids on tocilizumab clearance. Concomitant administration of a single intravenous dose of 10 mg/kg tocilizumab with 10-25 mg MTX once weekly had no clinically significant effect on MTX exposure. Tocilizumab products have not been studied in combination with biological DMARDs such as TNF antagonists [see Dosage and Administration (2.2)].
In GCA patients, no effect of concomitant corticosteroid on tocilizumab exposure was observed.
Principal Display Panel – 10 Ml Vial – Primary Label (Principal Display Panel – 10 mL Vial – Primary Label)
NDC 65219-592-10 Rx only 10 mL
Tyenne®
(tocilizumab-aazg)
Injection
200 mg / 10 mL
(20 mg/mL)
For Intravenous Infusion
Only After Dilution.
Fresenius Kabi USA, LLC
US License No. 2146
Principal Display Panel – 20 Ml Vial – Primary Label (Principal Display Panel – 20 mL Vial – Primary Label)
NDC 65219-594-20 Rx only 20 mL
Tyenne®
tocilizumab-aazg
Injection
400 mg / 20 mL
(20 mg/mL)
For Intravenous Infusion
Only After Dilution.
Fresenius Kabi USA, LLC
US License No. 2146
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to establish the carcinogenicity potential of tocilizumab products. Literature indicates that the IL-6 pathway can mediate anti-tumor responses by promoting increased immune cell surveillance of the tumor microenvironment. However, available published evidence also supports that IL-6 signaling through the IL-6 receptor may be involved in pathways that lead to tumorigenesis. The malignancy risk in humans from an antibody that disrupts signaling through the IL-6 receptor, such as tocilizumab, is presently unknown.
Fertility and reproductive performance were unaffected in male and female mice that received a murine analogue of tocilizumab administered by the intravenous route at a dose of 50 mg/kg every three days.
14.2 Rheumatoid Arthritis – Subcutaneous Administration
The efficacy and safety of subcutaneously administered tocilizumab was assessed in two double-blind, controlled, multicenter studies in patients with active RA. One study, SC-I (NCT01194414), was a non-inferiority study that compared the efficacy and safety of tocilizumab 162 mg administered every week subcutaneously to 8 mg per kg intravenously every four weeks. The second study, SC-II (NCT01232569), was a placebo-controlled superiority study that evaluated the safety and efficacy of tocilizumab 162 mg administered every other week subcutaneously to placebo. Both SC-I and SC-II required patients to be >18 years of age with moderate to severe active rheumatoid arthritis diagnosed according to ACR criteria who had at least 4 tender and 4 swollen joints at baseline (SC-I) or at least 8 tender and 6 swollen joints at baseline (SC-II), and an inadequate response to their existing DMARD therapy, where approximately 20% also had a history of inadequate response to at least one TNF inhibitor. All patients in both SC studies received background non-biologic DMARD(s).
In SC-I, 1262 patients were randomized 1:1 to receive tocilizumab-SC 162 mg every week or intravenous tocilizumab 8 mg/kg every four weeks in combination with DMARD(s). In SC-II, 656 patients were randomized 2:1 to tocilizumab-SC 162 mg every other week or placebo, in combination with DMARD(s). The primary endpoint in both studies was the proportion of patients who achieved an ACR20 response at Week 24.
The clinical response to 24 weeks of tocilizumab-SC therapy is shown in Table 7 . In SC-I, the primary outcome measure was ACR20 at Week 24. The pre-specified non-inferiority margin was a treatment difference of 12%. The study demonstrated non-inferiority of tocilizumab with respect to ACR20 at Week 24; ACR50, ACR70, and DAS28 responses are also shown in Table 7 . In SC-II, a greater portion of patients treated with tocilizumab 162 mg subcutaneously every other week achieved ACR20, ACR50, and ACR70 responses compared to placebo-treated patients (Table 7). Further, a greater proportion of patients treated with tocilizumab 162 mg subcutaneously every other week achieved a low level of disease activity as measured by a DAS28-ESR less than 2.6 at Week 24 compared to those treated with placebo (Table 7).
| TCZ = tocilizumab a Per Protocol Population b Intent To Treat Population |
||||
|
SC-Ia |
SC-IIb |
|||
|
TCZ SC 162 mg every week + DMARD |
TCZ IV 8mg/kg + DMARD |
TCZ SC 162 mg every other week + DMARD |
Placebo + DMARD |
|
|
N=558 |
N=537 |
N=437 |
N=219 |
|
|
ACR20 |
||||
|
Week 24 |
69% |
73.4% |
61% |
32% |
|
Weighted difference (95% CI) |
-4% (-9.2, 1.2) |
30% (22.0, 37.0) |
||
|
ACR50 |
||||
|
Week 24 |
47% |
49% |
40% |
12% |
|
Weighted difference (95% CI) |
-2% (-7.5, 4.0) |
28% (21.5, 34.4) |
||
|
ACR70 |
||||
|
Week 24 |
24% |
28% |
20% |
5% |
|
Weighted difference (95% CI) |
-4% (-9.0, 1.3) |
15% (9.8, 19.9) |
||
|
Change in DAS28 [Adjusted mean] |
||||
|
Week 24 |
-3.5 |
-3.5 |
-3.1 |
-1.7 |
|
Adjusted mean difference (95% CI) |
0 (-0.2, 0.1) |
-1.4 (-1.7; -1.1) |
||
|
DAS28 < 2.6 |
||||
|
Week 24 |
38.4% |
36.9% |
32.0% |
4.0% |
|
Weighted difference (95% CI) |
0.9 (-5.0, 6.8) |
28.6 (22.0, 35.2) |
The results of the components of the ACR response criteria and the percent of ACR20 responders by visit for tocilizumab-SC in Studies SC-I and SC-II were consistent with those observed for tocilizumab-IV.
14.3 Giant Cell Arteritis – Subcutaneous Administration
The efficacy and safety of subcutaneously administered tocilizumab was assessed in a single, randomized, double-blind, multicenter study in patients with active GCA. In Study WA28119 (NCT01791153), 251 screened patients with new-onset or relapsing GCA were randomized to one of four treatment arms. Two subcutaneous doses of tocilizumab (162 mg every week and 162 mg every other week) were compared to two different placebo control groups (pre-specified prednisone-taper regimen over 26 weeks and 52 weeks) randomized 2:1:1:1. The study consisted of a 52-week blinded period, followed by a 104-week open-label extension.
All patients received background glucocorticoid (prednisone) therapy. Each of the tocilizumab-treated groups and one of the placebo-treated groups followed a pre-specified prednisone-taper regimen with the aim to reach 0 mg by 26 weeks, while the second placebo-treated group followed a pre-specified prednisone-taper regimen with the aim to reach 0 mg by 52 weeks designed to be more in keeping with standard practice.
The primary efficacy endpoint was the proportion of patients achieving sustained remission from Week 12 through Week 52. Sustained remission was defined by a patient attaining a sustained (1) absence of GCA signs and symptoms from Week 12 through Week 52, (2) normalization of erythrocyte sedimentation rate (ESR) (to < 30 mm/hr without an elevation to ≥ 30 mm/hr attributable to GCA) from Week 12 through Week 52, (3) normalization of C-reactive protein (CRP) (to < 1 mg/dL, with an absence of successive elevations to ≥ 1mg/dL) from Week 12 through Week 52, and (4) successful adherence to the prednisone taper defined by not more than 100 mg of excess prednisone from Week 12 through Week 52. Tocilizumab 162 mg weekly and 162 mg every other week + 26 weeks prednisone taper both showed superiority in achieving sustained remission from Week 12 through Week 52 compared with placebo + 26 weeks prednisone taper ( Table 8 ). Both tocilizumab treatment arms also showed superiority compared to the placebo + 52 weeks prednisone taper ( Table 8 ).
|
a Sustained remission was achieved by a patient meeting all of the following components: absence of GCA signs and symptomsb, normalization of ESRc, normalization of CRPd and adherence to the prednisone taper regimene. b Patients who did not have any signs or symptoms of GCA recorded from Week 12 up to Week 52. c Patients who did not have an elevated ESR ≥30 mm/hr which was classified as attributed to GCA from Week 12 up to Week 52. d Patients who did not have two or more consecutive CRP records of ≥ 1mg/dL from Week 12 up to Week 52. e Patients who did not enter escape therapy and received ≤ 100mg of additional concomitant prednisone from Week 12 up to Week 52. |
||||
|
PBO + 26
|
PBO + 52
|
TCZ 162mg SC QW + 26
|
TCZ 162 mg SC Q2W + 26
|
|
|
Sustained remission a |
||||
|
Responders, n (%) |
7 (14.0%) |
9 (17.6%) |
56 (56.0%) |
26 (53.1%) |
|
Unadjusted difference in |
N/A |
N/A |
42.0% |
39.1% |
|
Unadjusted difference in |
N/A |
N/A |
38.4% |
35.4% |
|
Components of Sustained Remission |
||||
|
Sustained absence of GCA signs |
20 (40.0%) |
23 (45.1%) |
69 (69.0%) |
28 (57.1%) |
Patients not completing the study to week 52 were classified as non-responders in the primary and key secondary analysis: PBO+26: 6 (12.0%), PBO+52: 5 (9.8%), TCZ QW: 15 (15.0%), TCZ Q2W: 9 (18.4%).
CRP = C-reactive protein
ESR = erythrocyte sedimentation rate
PBO = placebo
Q2W = every other week dose
QW = every week dose
TCZ = tocilizumab
The estimated annual cumulative prednisone dose was lower in the two tocilizumab dose groups (medians of 1887 mg and 2207 mg on tocilizumab QW and Q2W, respectively) relative to the placebo arms (medians of 3804 mg and 3902 mg on placebo + 26 weeks prednisone and placebo + 52 weeks prednisone taper, respectively).
2.5 Recommended Dosage for Systemic Juvenile Idiopathic Arthritis
TYENNE may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change a dose based solely on a single visit body weight measurement, as weight may fluctuate.
2.4 Recommended Dosage for Polyarticular Juvenile Idiopathic Arthritis
TYENNE may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change dose based solely on a single visit body weight measurement, as weight may fluctuate.
14.7 Systemic Juvenile Idiopathic Arthritis Intravenous Administration (14.7 Systemic Juvenile Idiopathic Arthritis - Intravenous Administration)
The efficacy of tocilizumab for the treatment of active SJIA was assessed in WA18221 (NCT00642460), a 12-week randomized, double blind, placebo-controlled, parallel group, 2-arm study. Patients treated with or without MTX, were randomized (tocilizumab:placebo = 2:1) to one of two treatment groups: 75 patients received tocilizumab infusions every two weeks at either 8 mg per kg for patients at or above 30 kg or 12 mg per kg for patients less than 30 kg and 37 were randomized to receive placebo infusions every two weeks. Corticosteroid tapering could occur from week six for patients who achieved a JIA ACR 70 response. After 12 weeks or at the time of escape, due to disease worsening, patients were treated with tocilizumab in the open-label extension phase at weight appropriate dosing.
The primary endpoint was the proportion of patients with at least 30% improvement in JIA ACR core set (JIA ACR 30 response) at Week 12 and absence of fever (no temperature at or above 37.5°C in the preceding 7 days). JIA ACR (American College of Rheumatology) responses are defined as the percentage improvement (e.g., 30%, 50%, 70%) in 3 of any 6 core outcome variables compared to baseline, with worsening in no more than 1 of the remaining variables by 30% or more.
Core outcome variables consist of physician global assessment, parent per patient global assessment, number of joints with active arthritis, number of joints with limitation of movement, erythrocyte sedimentation rate (ESR), and functional ability (childhood health assessment questionnaire-CHAQ).
Primary endpoint result and JIA ACR response rates at Week 12 are shown in Table 9 .
|
aThe weighted difference is the difference between the tocilizumab and Placebo response rates, adjusted for the stratification factors (weight, disease duration, background oral corticosteroid dose and background methotrexate use). b CI: confidence interval of the weighted difference. |
||
|
Tocilizumab
|
Placebo
|
|
|
Primary Endpoint: JIA ACR 30 response + absence of fever |
||
|
Responders |
85% |
24% |
|
Weighted difference (95% CI) |
62
|
- |
|
JIA ACR Response Rates at Week 12 |
||
|
JIA ACR 30 |
||
|
Responders Weighted difference a (95% CI) b |
91% |
24% |
|
JIA ACR 50 |
||
|
Responders Weighted difference a (95% CI) b |
85% |
11% |
|
JIA ACR 70 |
||
|
Responders Weighted difference a (95% CI) b |
71% |
8% |
The treatment effect of tocilizumab was consistent across all components of the JIA ACR response core variables. JIA ACR scores and absence of fever responses in the open label extension were consistent with the controlled portion of the study (data available through 44 weeks).
2.6 Preparation and Administration Instructions for Intravenous Infusion
TYENNE for intravenous infusion should be diluted by a healthcare professional using aseptic technique as follows:
-
•Use a sterile needle and syringe to prepare TYENNE.
-
•Patients less than 30 kg: use a 50 mL infusion bag or bottle of 0.9% or 0.45% Sodium Chloride Injection, USP, and then follow steps 1 and 2 below.
-
•Patients at or above 30 kg weight: use a 100 mL infusion bag or bottle, and then follow steps 1 and 2 below.
-
-Step 1. Withdraw a volume of 0.9% or 0.45% Sodium Chloride Injection, USP, equal to the volume of the TYENNE injection required for the patient's dose from the infusion bag or bottle [see Dosage and Administration (2.2, 2.4, 2.5)].
|
For Intravenous Use: Volume of TYENNE Injection per kg of Body Weight |
||
|
Dosage |
Indication |
Volume of TYENNE injection per kg of body weight |
|
4 mg/kg |
Adult RA |
0.2 mL/kg |
|
6 mg/kg |
Adult GCA |
0.3 mL/kg |
|
8 mg/kg |
Adult RA SJIA and PJIA (greater than or equal to 30 kg of body weight) |
0.4 mL/kg |
|
10 mg/kg |
PJIA (less than 30 kg of body weight) |
0.5 mL/kg |
|
12 mg/kg |
SJIA (less than 30 kg of body weight) |
0.6 mL/kg |
-
-Step 2. Withdraw the amount of TYENNE for intravenous infusion from the vial(s) and add slowly into the 0.9% or 0.45% Sodium Chloride Injection, USP infusion bag or bottle. To mix the solution, gently invert the bag to avoid foaming.
-
•The prepared solution for infusion should be used immediately. If not used immediately, the diluted tocilizumab solutions may be refrigerated at 36°F to 46°F (2°C to 8°C) up to 24 hours, or stored at room temperature at or below 77°F (25°C) for up to 4 hours and should be protected from light. Administration of diluted TYENNE solution must be completed within this period of time.
-
•TYENNE solutions do not contain preservatives; therefore, unused product remaining in the vials should not be used.
-
•Allow the fully diluted TYENNE solution to reach room temperature prior to infusion.
-
•The infusion should be administered over 60 minutes and must be administered with an infusion set. Do not administer as an intravenous push or bolus.
-
•TYENNE should not be infused concomitantly in the same intravenous line with other drugs. No physical or biochemical compatibility studies have been conducted to evaluate the co-administration of TYENNE with other drugs.
-
•Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If particulates and discolorations are noted, the product should not be used.
-
•Fully diluted TYENNE solutions are compatible with polypropylene, polyethylene and polyvinyl chloride infusion bags and bottles, and glass infusion bottles.
14.8 Systemic Juvenile Idiopathic Arthritis Subcutaneous Administration (14.8 Systemic Juvenile Idiopathic Arthritis - Subcutaneous Administration)
Subcutaneously administered tocilizumab in pediatric patients with systemic juvenile idiopathic arthritis (SJIA) was assessed in WA28118 (NCT01904292), a 52-week, open-label, multicenter, PK-PD and safety study to determine the appropriate subcutaneous dose of tocilizumab that achieved comparable PK/PD profiles to the tocilizumab-IV regimen.
Eligible patients received tocilizumab subcutaneously dosed according to body weight, with patients weighing at or above 30 kg (n = 26) dosed with 162 mg of tocilizumab every week and patients weighing below 30 kg (n = 25) dosed with 162 mg of tocilizumab every 10 days (n=8) or every 2 weeks (n=17) for 52 weeks. Of these 51 patients, 26 (51%) were naïve to subcutaneous tocilizumab and 25 (49%) had been receiving tocilizumab intravenously and switched to subcutaneous tocilizumab at baseline.
The efficacy of subcutaneous tocilizumab in children 2 to 17 years of age is based on pharmacokinetic exposure and extrapolation of the established efficacy of intravenous tocilizumab in systemic JIA patients [see Clinical Pharmacology (12.3) and Clinical Studies (14.8 )].
2.7 Preparation and Administration Instructions for Subcutaneous Injection
-
•TYENNE for subcutaneous injection is not intended for intravenous drip infusion.
-
•Assess suitability of patient for subcutaneous home use and instruct patients to inform a healthcare professional before administering the next dose if they experience any symptoms of allergic reaction. Patients should seek immediate medical attention if they develop symptoms of serious allergic reactions. TYENNE subcutaneous injection is intended for use under the guidance of a healthcare practitioner. After proper training in subcutaneous injection technique, a patient may self-inject TYENNE or the patient's caregiver may administer TYENNE if a healthcare practitioner determines that it is appropriate. PJIA and SJIA patients may self-inject with the TYENNE prefilled syringe or autoinjector, or the patient's caregiver may administer TYENNE if both the healthcare practitioner and the parent/legal guardian determine it is appropriate [see Use in Specific Populations (8.4)].
Patients, or patient caregivers, should be instructed to follow the directions provided in the Instructions for Use (IFU) for additional details on medication administration. -
•Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use TYENNE prefilled syringes (PFS) or prefilled autoinjectors exhibiting particulate matter, cloudiness, or discoloration. TYENNE for subcutaneous administration should be clear and colorless to pale yellow. Do not use if any part of the PFS or autoinjector appears to be damaged.
-
•Patients using TYENNE for subcutaneous administration should be instructed to inject the full amount in the syringe (0.9 mL) or full amount in the autoinjector (0.9 mL), which provides 162 mg of TYENNE, according to the directions provided in the IFU.
-
•Injection sites should be rotated with each injection and should never be given into moles, scars, or areas where the skin is tender, bruised, red, hard, or not intact.
14.5 Polyarticular Juvenile Idiopathic Arthritis – Intravenous Administration
The efficacy of tocilizumab was assessed in a three-part study, WA19977 (NCT00988221), including an open- label extension in children 2 to 17 years of age with active polyarticular juvenile idiopathic arthritis (PJIA), who had an inadequate response to methotrexate or inability to tolerate methotrexate. Patients had at least 6 months of active disease (mean disease duration of 4.2 ± 3.7 years), with at least five joints with active arthritis (swollen or limitation of movement accompanied by pain and/or tenderness) and/or at least 3 active joints having limitation of motion (mean, 20 ± 14 active joints). The patients treated had subtypes of JIA that at disease onset included Rheumatoid Factor Positive or Negative Polyarticular JIA, or Extended Oligoarticular JIA. Treatment with a stable dose of methotrexate was permitted but was not required during the study. Concurrent use of disease modifying antirheumatic drugs (DMARDs), other than methotrexate, or other biologics (e.g., TNF antagonists or T cell costimulation modulator) were not permitted in the study.
Part I consisted of a 16-week active tocilizumab treatment lead-in period (n=188) followed by Part II, a 24-week randomized double-blind placebo-controlled withdrawal period, followed by Part III, a 64-week open-label period. Eligible patients weighing at or above 30 kg received tocilizumab at 8 mg/kg intravenously once every four weeks. Patients weighing less than 30 kg were randomized 1:1 to receive either tocilizumab 8 mg/kg or 10 mg/kg intravenously every four weeks. At the conclusion of the open-label Part I, 91% of patients taking background MTX in addition to tocilizumab and 83% of patients on tocilizumab monotherapy achieved an ACR 30 response at week 16 compared to baseline and entered the blinded withdrawal period (Part II) of the study.
The proportions of patients with JIA ACR 50/70 responses in Part I were 84.0%, and 64%, respectively for patients taking background MTX in addition to tocilizumab and 80% and 55% respectively for patients on tocilizumab monotherapy.
In Part II, patients (ITT, n=163) were randomized to tocilizumab (same dose received in Part I) or placebo in a 1:1 ratio that was stratified by concurrent methotrexate use and concurrent corticosteroid use. Each patient continued in Part II of the study until Week 40 or until the patient satisfied JIA ACR 30 flare criteria (relative to Week 16) and qualified for escape.
The primary endpoint was the proportion of patients with a JIA ACR 30 flare at week 40 relative to week 16. JIA ACR 30 flare was defined as 3 or more of the 6 core outcome variables worsening by at least 30% with no more than 1 of the remaining variables improving by more than 30% relative to Week 16.
Tocilizumab treated patients experienced significantly fewer disease flares compared to placebo-treated patients (26% [21/82] versus 48% [39/81]; adjusted difference in proportions -21%, 95% CI: -35%, -8%).
During the withdrawal phase (Part II), more patients treated with tocilizumab showed JIA ACR 30/50/70 responses at Week 40 compared to patients withdrawn to placebo.
14.6 Polyarticular Juvenile Idiopathic Arthritis – Subcutaneous Administration
Subcutaneously administered tocilizumab in pediatric patients with polyarticular juvenile idiopathic arthritis (PJIA) was assessed in WA28117 (NCT01904279), a 52-week, open-label, multicenter, PK-PD and safety study to determine the appropriate subcutaneous dose of tocilizumab that achieved comparable PK/PD profiles to the tocilizumab-IV regimen. PJIA patients aged 1 to 17 years with an inadequate response or inability to tolerate MTX, including patients with well-controlled disease on treatment with tocilizumab-IV and tocilizumab-naïve patients with active disease, were treated with subcutaneous tocilizumab based on body weight.
Patients weighing at or above 30 kg (n = 25) were treated with 162 mg of tocilizumab-SC every 2 weeks and patients weighing less than 30 kg (n = 27) received 162 mg of tocilizumab-SC every 3 weeks for 52 weeks. Of these 52 patients, 37 (71%) were naïve to tocilizumab and 15 (29%) had been receiving tocilizumab-IV and switched to tocilizumab-SC at baseline.
The efficacy of subcutaneous tocilizumab in children 2 to 17 years of age is based on pharmacokinetic exposure and extrapolation of the established efficacy of intravenous tocilizumab in polyarticular JIA patients and subcutaneous tocilizumab in patients with RA [see Clinical Pharmacology (12.3) and Clinical Studies (14.2 and 14.6)].
6.1 Clinical Trials Experience in Rheumatoid Arthritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv) (6.1 Clinical Trials Experience in Rheumatoid Arthritis Patients Treated with Intravenous Tocilizumab (Tocilizumab-IV))
The tocilizumab-IV data in rheumatoid arthritis (RA) includes 5 double-blind, controlled, multicenter studies. In these studies, patients received doses of tocilizumab-IV 8 mg per kg monotherapy (288 patients), tocilizumab-IV 8 mg per kg in combination with DMARDs (including methotrexate) (1582 patients), or tocilizumab-IV 4 mg per kg in combination with methotrexate (774 patients).
The all exposure population includes all patients in registration studies who received at least one dose of tocilizumab-IV. Of the 4009 patients in this population, 3577 received treatment for at least 6 months, 3309 for at least one year; 2954 received treatment for at least 2 years and 2189 for 3 years.
All patients in these studies had moderately to severely active rheumatoid arthritis. The study population had a mean age of 52 years, 82% were female and 74% were Caucasian.
The most common serious adverse reactions were serious infections [see Warnings and Precautions (5.1)]. The most commonly reported adverse reactions in controlled studies up to 24 weeks (occurring in at least 5% of patients treated with tocilizumab-IV monotherapy or in combination with DMARDs) were upper respiratory tract infections, nasopharyngitis, headache, hypertension and increased ALT.
The proportion of patients who discontinued treatment due to any adverse reactions during the double-blind, placebo-controlled studies was 5% for patients taking tocilizumab-IV and 3% for placebo-treated patients. The most common adverse reactions that required discontinuation of tocilizumab-IV were increased hepatic transaminase values (per protocol requirement) and serious infections.
6.4 Clinical Trials Experience in Giant Cell Arteritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv) (6.4 Clinical Trials Experience in Giant Cell Arteritis Patients Treated With Intravenous Tocilizumab (Tocilizumab-IV))
The safety of tocilizumab-IV was studied in an open label PK-PD and safety study in 24 patients with GCA who were in remission on tocilizumab-IV at time of enrollment. Patients received tocilizumab 7 mg/kg every 4 weeks for 20 weeks, followed by 6 mg/kg every 4 weeks for 20 weeks. The total patient years exposure to treatment was 17.5 years. The overall safety profile observed for tocilizumab administered intravenously in GCA patients was consistent with the known safety profile of tocilizumab.
6.2 Clinical Trials Experience in Rheumatoid Arthritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc) (6.2 Clinical Trials Experience in Rheumatoid Arthritis Patients Treated with Subcutaneous Tocilizumab (Tocilizumab-SC))
The tocilizumab-SC data in rheumatoid arthritis (RA) includes 2 double-blind, controlled, multicenter studies. Study SC-I was a non-inferiority study that compared the efficacy and safety of tocilizumab 162 mg administered every week subcutaneously and 8 mg/kg intravenously every four weeks in 1262 adult subjects with rheumatoid arthritis. Study SC-II was a placebo-controlled superiority study that evaluated the safety and efficacy of tocilizumab 162 mg administered every other week subcutaneously or placebo in 656 patients. All patients in both studies received background non-biologic DMARDs.
The safety observed for tocilizumab-SC administered subcutaneously was consistent with the known safety profile of intravenous tocilizumab, with the exception of injection site reactions (ISRs), which were more common with tocilizumab-SC compared with placebo SC injections (IV arm).
6.3 Clinical Trials Experience in Giant Cell Arteritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc) (6.3 Clinical Trials Experience in Giant Cell Arteritis Patients Treated with Subcutaneous Tocilizumab (Tocilizumab-SC))
The safety of subcutaneous tocilizumab has been studied in one Phase III study (WA28119) with 251 GCA patients. The total patient years duration in the tocilizumab-SC GCA all exposure population was 138.5 patient years during the 12-month double blind, placebo-controlled phase of the study. The overall safety profile observed in the tocilizumab-SC treatment groups was generally consistent with the known safety profile of tocilizumab. There was an overall higher incidence of infections in GCA patients relative to RA patients.
The rate of infection/serious infection events was 200.2/9.7 events per 100 patient years in the tocilizumab-SC weekly group and 160.2/4.4 events per 100 patient years in the tocilizumab-SC every other week group as compared to 156.0/4.2 events per 100 patient years in the placebo + 26 week prednisone taper and 210.2/12.5 events per 100 patient years in the placebo + 52 week taper groups.
6.7 Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv) (6.7 Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated with Intravenous Tocilizumab (Tocilizumab-IV))
The data described below reflect exposure to tocilizumab-IV in one randomized, double-blind, placebo-controlled trial of 112 pediatric patients with SJIA 2 to 17 years of age who had an inadequate clinical response to nonsteroidal anti-inflammatory drugs (NSAIDs) or corticosteroids due to toxicity or lack of efficacy. At baseline, approximately half of the patients were taking 0.3 mg/kg/day corticosteroids or more, and almost 70% were taking methotrexate. The trial included a 12 week controlled phase followed by an open-label extension. In the 12 week double-blind, controlled portion of the clinical study 75 patients received treatment with tocilizumab-IV (8 or 12 mg per kg based upon body weight). After 12 weeks or at the time of escape, due to disease worsening, patients were treated with tocilizumab-IV in the open-label extension phase.
The most common adverse events (at least 5%) seen in tocilizumab-IV treated patients in the 12 week controlled portion of the study were: upper respiratory tract infection, headache, nasopharyngitis and diarrhea.
6.8 Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc) (6.8 Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated with Subcutaneous Tocilizumab (Tocilizumab-SC))
The safety profile of tocilizumab-SC was studied in 51 pediatric patients 1 to 17 years of age with SJIA who had an inadequate clinical response to NSAIDs and corticosteroids. In general, the safety observed for tocilizumab administered subcutaneously was consistent with the known safety profile of intravenous tocilizumab, with the exception of ISRs where a higher frequency was observed in tocilizumab-SC treated SJIA patients compared to PJIA patients and adult RA or GCA patients [see Adverse Reactions (6.2, 6.3 and 6.6)].
6.5 Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Intravenous Tocilizumab (tocilizumab Iv) (6.5 Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Intravenous Tocilizumab (Tocilizumab-IV))
The safety of tocilizumab-IV was studied in 188 pediatric patients 2 to 17 years of age with PJIA who had an inadequate clinical response or were intolerant to methotrexate. The total patient exposure in the tocilizumab-IV all exposure population (defined as patients who received at least one dose of tocilizumab-IV) was 184.4 patient years. At baseline, approximately half of the patients were taking oral corticosteroids and almost 80% were taking methotrexate. In general, the types of adverse drug reactions in patients with PJIA were consistent with those seen in RA and SJIA patients [see Adverse Reactions (6.1 and 6.7)].
6.6 Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Subcutaneous Tocilizumab (tocilizumab Sc) (6.6 Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Subcutaneous Tocilizumab (Tocilizumab-SC))
The safety of tocilizumab-SC was studied in 52 pediatric patients 1 to 17 years of age with PJIA who had an inadequate clinical response or were intolerant to methotrexate. The total patient exposure in the PJIA tocilizumab-SC population (defined as patients who received at least one dose of tocilizumab-SC and accounting for treatment discontinuation) was 49.5 patient years.
In general, the safety observed for tocilizumab administered subcutaneously was consistent with the known safety profile of intravenous tocilizumab, with the exception of injection site reactions (ISRs), and neutropenia.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:37.719096 · Updated: 2026-03-14T22:22:55.765381