001
e32f1a47-9a5b-73de-e053-2995a90a28a4
34390-5
HUMAN OTC DRUG LABEL
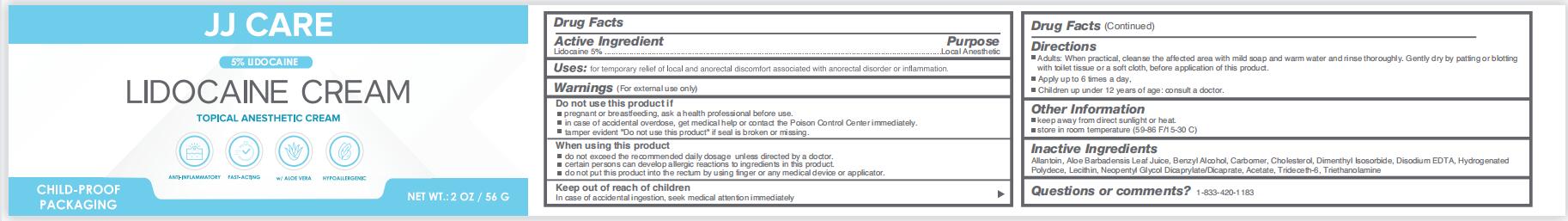
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 5%
Purpose
Local Anesthetic
Medication Information
Purpose
Local Anesthetic
Description
Lidocaine 5%
Uses
for temporary relief of local and anorectal discomfort associated with anorectal disorder or inflammation
Section 50565-1
Keep out of reach of children
Incase of accidental ingestion , seek medical attention immediately
Stop Use
n/a
Warnings
Warnings ( For extemal use only )
Directions
Adults : When practical , cleanse the affected area with mild soap and warm water and rinse thoroughly. Gently dry by patting or blottingwith toilet tissue or a soft cloth , before application of this product.
Apply up to 6 times a day,
Children up under 12 years of age: consult a doctor.
Do Not Use
Do not use this product if
pregnant or breastfeeding , ask a health professional before use.
in case of accidental overdose , get medical help or contact the Poison Control Center immediately.
tamper evident " Do not use this product " if seal is broken or missing.
Active Ingredient
Lidocaine 5%
Other Information
keep away from direct sunlight or heata.
store in room temperature ( 59-86 F/15-30 C )
Inactive Ingredients
Allantoin, Aloe Barbadensis Leaf Juice, Benzyl Alcohol, Carbomer, Cholesterol, Dimenthyl Isosorbide, Disodium EDTA , Hydrogenated Polydece, Lecithin, Neopentyl Glycol Dicaprylate/Dicaprate, Acetate, Trideceth-6, Triethanolamine
When Using This Product
do not exceed the recommended daily dosage unless directed by a doctor.
certain persons can develop allergic reactions to ingredients in this product.
do not put this product into the rectum by using finger or any medical device or applicator.
Package Label Principal Display Panel
Structured Label Content
Uses
for temporary relief of local and anorectal discomfort associated with anorectal disorder or inflammation
Section 50565-1 (50565-1)
Keep out of reach of children
Incase of accidental ingestion , seek medical attention immediately
Purpose
Local Anesthetic
Stop Use (Stop use)
n/a
Warnings
Warnings ( For extemal use only )
Directions
Adults : When practical , cleanse the affected area with mild soap and warm water and rinse thoroughly. Gently dry by patting or blottingwith toilet tissue or a soft cloth , before application of this product.
Apply up to 6 times a day,
Children up under 12 years of age: consult a doctor.
Do Not Use (Do not use)
Do not use this product if
pregnant or breastfeeding , ask a health professional before use.
in case of accidental overdose , get medical help or contact the Poison Control Center immediately.
tamper evident " Do not use this product " if seal is broken or missing.
Active Ingredient
Lidocaine 5%
Other Information
keep away from direct sunlight or heata.
store in room temperature ( 59-86 F/15-30 C )
Inactive Ingredients
Allantoin, Aloe Barbadensis Leaf Juice, Benzyl Alcohol, Carbomer, Cholesterol, Dimenthyl Isosorbide, Disodium EDTA , Hydrogenated Polydece, Lecithin, Neopentyl Glycol Dicaprylate/Dicaprate, Acetate, Trideceth-6, Triethanolamine
When Using This Product (When using this product)
do not exceed the recommended daily dosage unless directed by a doctor.
certain persons can develop allergic reactions to ingredients in this product.
do not put this product into the rectum by using finger or any medical device or applicator.
Package Label Principal Display Panel (Package Label - Principal Display Panel)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:16.888079 · Updated: 2026-03-14T23:00:39.941399