Drug Facts
e2e3c628-d241-42b6-8c1e-ef997ffa6e87
34390-5
Human OTC Drug Label
Drug Facts
Composition & Product
Identifiers & Packaging
Description
( in each extended-release tablet) Guaifenesin USP 1200 mg
Purpose
Expectorant
Medication Information
Purpose
Expectorant
Description
( in each extended-release tablet) Guaifenesin USP 1200 mg
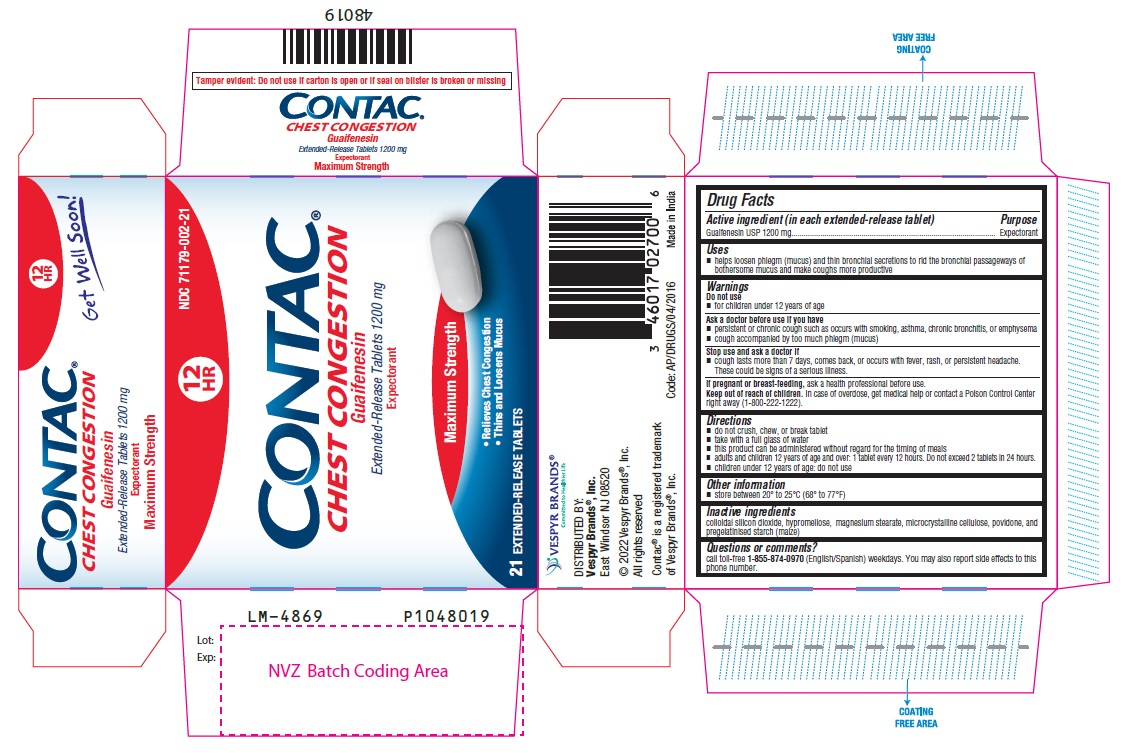
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Warnings
Do not use
- for children under 12 years of age
Directions
- do not crush, chew, or break tablet
- take with a full glass of water
- this product can be administered without regard for the timing of meals
- adults and children 12 years of age and over: 1 tablet every 12 hours. Do not exceed 2 tablets in 24 hours.
- children under 12 years of age: do not use
Active Ingredient
(
in each extended-release tablet)
Guaifenesin USP 1200 mg
Other Information
- store between 20° to 25°C (68° to 77°F)
Inactive Ingredients
colloidal silicon dioxide, hypromellose, magnesium stearate, microcrystalline cellulose, povidone, and pregelatinised starch (maize)
Questions Or Comments?
call toll-free 1-855-874-0970 (English/Spanish) weekdays. You may also report side effects to this phone number.
DISTRIBUTED BY:
Vespyr Brands®, Inc.
East Windsor, NJ 08520
Made in India
Code: AP/DRUGS/04/2016
Stop Use and Ask A Doctor If
- cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Before Use If You Have
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough accompanied by too much phlegm (mucus)
Package Label Principal Display Panel 1200 Mg Blister Carton (21 Tablets)
12 NDC 71179-002-21
HR
CONTAC®
CHEST CONGESTION
Guaifenesin
Extended-Release Tablets 1200 mg
Expectorant
Maximum Strength
- Relieves Chest Congestion
- Thins And Loosens Mucus
21
EXTENDED-RELEASE TABLETS
Structured Label Content
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Purpose
Expectorant
Warnings
Do not use
- for children under 12 years of age
Directions
- do not crush, chew, or break tablet
- take with a full glass of water
- this product can be administered without regard for the timing of meals
- adults and children 12 years of age and over: 1 tablet every 12 hours. Do not exceed 2 tablets in 24 hours.
- children under 12 years of age: do not use
Active Ingredient (Active ingredient)
(
in each extended-release tablet)
Guaifenesin USP 1200 mg
Other Information (Other information)
- store between 20° to 25°C (68° to 77°F)
Inactive Ingredients (Inactive ingredients)
colloidal silicon dioxide, hypromellose, magnesium stearate, microcrystalline cellulose, povidone, and pregelatinised starch (maize)
Questions Or Comments? (Questions or comments?)
call toll-free 1-855-874-0970 (English/Spanish) weekdays. You may also report side effects to this phone number.
DISTRIBUTED BY:
Vespyr Brands®, Inc.
East Windsor, NJ 08520
Made in India
Code: AP/DRUGS/04/2016
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough accompanied by too much phlegm (mucus)
Package Label Principal Display Panel 1200 Mg Blister Carton (21 Tablets) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1200 mg Blister Carton (21 Tablets))
12 NDC 71179-002-21
HR
CONTAC®
CHEST CONGESTION
Guaifenesin
Extended-Release Tablets 1200 mg
Expectorant
Maximum Strength
- Relieves Chest Congestion
- Thins And Loosens Mucus
21
EXTENDED-RELEASE TABLETS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:25.784375 · Updated: 2026-03-14T23:01:59.101977