These Highlights Do Not Include All The Information Needed To Use Atropen®
e2d4307d-da8f-49f5-aac0-02355dd9ffb7
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ATROPEN is indicated for the treatment of poisoning by susceptible organophosphorus nerve agents having cholinesterase activity as well as organophosphorus or carbamate insecticides in adult and pediatric patients.
Indications and Usage
ATROPEN is indicated for the treatment of poisoning by susceptible organophosphorus nerve agents having cholinesterase activity as well as organophosphorus or carbamate insecticides in adult and pediatric patients.
Dosage and Administration
ATROPEN is a single-dose autoinjector intended as an initial treatment of the muscarinic symptoms of insecticide or nerve agent poisonings; definitive medical care should be sought immediately. ( 2.1 ) Dosage is dependent on weight. ( 2.2 ) Dosage for Mild Symptoms: If the patient experiences two or more mild symptoms, administer one injection intramuscularly into the mid-lateral thigh. If, at any time after the first dose, the patient develops any of the severe symptoms, administer two additional injections intramuscularly in rapid succession. ( 2.2 ) Dosage for Severe Symptoms: If the patient is either unconscious or has any of the severe symptoms, immediately administer three injections intramuscularly into the patient's mid-lateral thigh in rapid succession. ( 2.2 )
Warnings and Precautions
Cardiovascular (CV) Risks: Tachycardia, palpitations, premature ventricular contractions, flutter, fibrillation, etc. Use caution in patients with known CV disease or conduction problems. ( 5.1 ) Heat Injury: May inhibit sweating and lead to hyperthermia; avoid excessive exercising and heat exposure. ( 5.2 ) Acute Glaucoma: May precipitate in susceptible individuals. ( 5.3 ) Urinary Retention: May precipitate in patient with bladder outflow obstruction. ( 5.4 ) Pyloric Stenosis: May precipitate complete obstruction. ( 5.5 ) Exacerbation of Chronic Lung Disease: Atropine may cause inspissation of bronchial secretions and formation of dangerous viscid plugs in individuals with chronic lung disease; monitor respiratory status. ( 5.6 ) Hypersensitivity: Atropine may cause hypersensitivity reactions, including anaphylaxis. ( 5.7 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Cardiovascular Risks [see Warnings and Precautions ( 5.1 )] Heat Injury [see Warnings and Precautions ( 5.2 )] Acute Glaucoma [see Warnings and Precautions ( 5.3 )] Urinary Retention [see Warnings and Precautions ( 5.4 )] Pyloric Stenosis [see Warnings and Precautions ( 5.5 )] Exacerbation of Chronic Lung Disease [see Warnings and Precautions ( 5.6 )] Hypersensitivity [see Warnings and Precautions ( 5.7 )] The following adverse reactions associated with the use of atropine were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Drug Interactions
Pralidoxime: The signs of atropinization (flushing, mydriasis, tachycardia, dryness of the mouth and nose) may occur earlier than might be expected than when atropine is used alone. ( 7.1 ) Barbiturates: Atropine may potentiate the effect of barbiturates. ( 7.2 )
Storage and Handling
Store between 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC and 30ºC (between 59ºF and 86ºF) [See USP Controlled Room Temperature]. Not made with natural rubber latex. Keep from freezing. Protect from light. After the ATROPEN autoinjector has been activated, the empty container should be disposed of properly. It cannot be refilled, nor can the protruding needle be retracted.
How Supplied
ATROPEN is a prefilled single-dose autoinjector that contains a clear solution and is supplied in the following package configurations: Table 3: ATROPEN Package Configurations NDC Number Package Configuration Product Description Delivered Dose (atropine) NDC 11704-107-01 Carton of 1 ATROPEN 0.25 mg (yellow label) 0.21 mg/0.3 mL (equivalent to 0.25 mg/0.3 mL of atropine sulfate) NDC 11704-104-01 Carton of 1 ATROPEN 0.5 mg (blue label) 0.42 mg/0.7 mL (equivalent to 0.5 mg/0.7 mL of atropine sulfate) NDC 11704-105-01 Carton of 1 ATROPEN 1 mg (red label) 0.84 mg/0.7 mL (equivalent to 1 mg/0.7 mL of atropine sulfate) NDC 11704-106-01 Carton of 1 ATROPEN 2 mg (green label) 1.67 mg/0.7 mL (equivalent to 2 mg/0.7 mL of atropine sulfate) NDC 11704-101-01 (For military use only) 1 Autoinjector ATROPEN 2 mg 1.67 mg/0.7 mL (equivalent to 2 mg/0. 7 mL of atropine sulfate)
Medication Information
Warnings and Precautions
Cardiovascular (CV) Risks: Tachycardia, palpitations, premature ventricular contractions, flutter, fibrillation, etc. Use caution in patients with known CV disease or conduction problems. ( 5.1 ) Heat Injury: May inhibit sweating and lead to hyperthermia; avoid excessive exercising and heat exposure. ( 5.2 ) Acute Glaucoma: May precipitate in susceptible individuals. ( 5.3 ) Urinary Retention: May precipitate in patient with bladder outflow obstruction. ( 5.4 ) Pyloric Stenosis: May precipitate complete obstruction. ( 5.5 ) Exacerbation of Chronic Lung Disease: Atropine may cause inspissation of bronchial secretions and formation of dangerous viscid plugs in individuals with chronic lung disease; monitor respiratory status. ( 5.6 ) Hypersensitivity: Atropine may cause hypersensitivity reactions, including anaphylaxis. ( 5.7 )
Indications and Usage
ATROPEN is indicated for the treatment of poisoning by susceptible organophosphorus nerve agents having cholinesterase activity as well as organophosphorus or carbamate insecticides in adult and pediatric patients.
Dosage and Administration
ATROPEN is a single-dose autoinjector intended as an initial treatment of the muscarinic symptoms of insecticide or nerve agent poisonings; definitive medical care should be sought immediately. ( 2.1 ) Dosage is dependent on weight. ( 2.2 ) Dosage for Mild Symptoms: If the patient experiences two or more mild symptoms, administer one injection intramuscularly into the mid-lateral thigh. If, at any time after the first dose, the patient develops any of the severe symptoms, administer two additional injections intramuscularly in rapid succession. ( 2.2 ) Dosage for Severe Symptoms: If the patient is either unconscious or has any of the severe symptoms, immediately administer three injections intramuscularly into the patient's mid-lateral thigh in rapid succession. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Cardiovascular Risks [see Warnings and Precautions ( 5.1 )] Heat Injury [see Warnings and Precautions ( 5.2 )] Acute Glaucoma [see Warnings and Precautions ( 5.3 )] Urinary Retention [see Warnings and Precautions ( 5.4 )] Pyloric Stenosis [see Warnings and Precautions ( 5.5 )] Exacerbation of Chronic Lung Disease [see Warnings and Precautions ( 5.6 )] Hypersensitivity [see Warnings and Precautions ( 5.7 )] The following adverse reactions associated with the use of atropine were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Drug Interactions
Pralidoxime: The signs of atropinization (flushing, mydriasis, tachycardia, dryness of the mouth and nose) may occur earlier than might be expected than when atropine is used alone. ( 7.1 ) Barbiturates: Atropine may potentiate the effect of barbiturates. ( 7.2 )
Storage and Handling
Store between 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC and 30ºC (between 59ºF and 86ºF) [See USP Controlled Room Temperature]. Not made with natural rubber latex. Keep from freezing. Protect from light. After the ATROPEN autoinjector has been activated, the empty container should be disposed of properly. It cannot be refilled, nor can the protruding needle be retracted.
How Supplied
ATROPEN is a prefilled single-dose autoinjector that contains a clear solution and is supplied in the following package configurations: Table 3: ATROPEN Package Configurations NDC Number Package Configuration Product Description Delivered Dose (atropine) NDC 11704-107-01 Carton of 1 ATROPEN 0.25 mg (yellow label) 0.21 mg/0.3 mL (equivalent to 0.25 mg/0.3 mL of atropine sulfate) NDC 11704-104-01 Carton of 1 ATROPEN 0.5 mg (blue label) 0.42 mg/0.7 mL (equivalent to 0.5 mg/0.7 mL of atropine sulfate) NDC 11704-105-01 Carton of 1 ATROPEN 1 mg (red label) 0.84 mg/0.7 mL (equivalent to 1 mg/0.7 mL of atropine sulfate) NDC 11704-106-01 Carton of 1 ATROPEN 2 mg (green label) 1.67 mg/0.7 mL (equivalent to 2 mg/0.7 mL of atropine sulfate) NDC 11704-101-01 (For military use only) 1 Autoinjector ATROPEN 2 mg 1.67 mg/0.7 mL (equivalent to 2 mg/0. 7 mL of atropine sulfate)
Description
ATROPEN is indicated for the treatment of poisoning by susceptible organophosphorus nerve agents having cholinesterase activity as well as organophosphorus or carbamate insecticides in adult and pediatric patients.
Section 42229-5
Dosage for Mild Symptoms
Section 42230-3
Instructions for Use
ATROPEN (at-ROW-pen)
(atropine injection)
for intramuscular use
Self and Caregiver Instructions for Use of ATROPEN
Important: Follow these instructions only when ready to use ATROPEN. If possible, a healthcare provider or someone who has been trained to identify and treat the symptoms of nerve agent and insecticide poisoning should give the ATROPEN injection. If a healthcare provider is not available during an emergency, a patient or caregiver might need to give the ATROPEN injection.
Individuals should not rely only on ATROPEN for protection from nerve agent and insecticide poisoning. Exposed persons and caregivers need to wear clothing to protect their skin and goggles and masks to protect their face and eyes when available, to avoid exposure.
|
Step 1
Choose the Correct Dose |
|
| Adults and children weighing over 90 pounds (41 kg) (generally over 10 years of age 2 mg ATROPEN (green label) |
2 mg |
| Children weighing 40 pounds to 90 pounds (18 kg to 41 kg) (generally 4 to 10 years of age) 1 mg ATROPEN (red label) |
1 mg |
| Children weighing 15 pounds to 40 pounds (7 kg to 18 kg) (generally 6 months to 4 years of age) 0.5 mg ATROPEN (blue label) |
0.5 mg |
| Infants weighing less than 15 pounds (7 kg) (generally less than 6 months of age) 0.25 mg ATROPEN (yellow label) |
0.25 mg |
Caution: When the ATROPEN is activated, the needle will quickly extend from the green tip and black tip. Keep your fingers away from the green tip and black tip.
|
Step 2
Decide if Symptoms are Mild or Severe |
|
| If nerve agent or insecticide exposure is known or suspected, you may see the following mild and severe symptoms. You may not have all of the symptoms or see all symptoms in others exposed to a nerve agent or certain insecticide poisoning:
|
|
| Mild Symptoms | Severe Symptoms |
|
|
Symptoms in infants and young children:
The mild symptoms excessive, unexplained teary eyes, excessive, unexplained runny nose and increased saliva or sudden drooling, and the severe symptom problems controlling urine or stool (bowel movement) are sometimes seen in healthy infants and young children. In infants and young children, these symptoms are seen less often than the other mild and severe symptoms listed above. Symptoms must be looked at together when nerve agent or insecticide exposure is known or suspected.
The severe symptoms severe muscle twitching and general weakness can happen in infants. Infants may become drowsy or pass out, with muscle floppiness instead of muscle twitching, soon after exposure to nerve agents or insecticides.
|
Step 3 Treating Mild Symptoms |
|
First Dose: Give 1 dose of ATROPEN (1 injection) if you get 2 or more mild symptoms of nerve agent or insecticide exposure. Give the injection in the outer thigh. Ask a caregiver to check your symptoms to see if they continue or get worse. Get medical help right away. Additional Doses: If, any time after the first injection of ATROPEN, the exposed person gets any of the severe symptoms listed above, give 2 more doses of ATROPEN (2 injections) in the outer thigh quickly one right after the other. Use a new ATROPEN for each injection. If you have given yourself the first injection, someone else should give you the second and third ATROPEN injections, if possible. |
|
Treating Severe Symptoms |
|
If you get any of the severe symptoms of nerve agent or insecticide exposure listed above or you see an exposed person who has passed out (unconscious), give 3 doses of ATROPEN (3 injections) right away. Give the 3 injections in the outer thigh quickly, one right after the other, using a new ATROPEN for each injection. Get medical help right away. Warning: Giving more ATROPEN injections by mistake in people who do not have nerve agent or insecticide poisoning may cause an overdose of atropine. This can lead to being unable to see or walk well for several hours. People with heart problems may have serious side effects, that can lead to death. |
|
Step 4
Give the ATROPEN Injection |
|
|
Instructions for Use of 2 mg, 1 mg, and 0.5 mg ATROPEN
(see below for 0.25 mg dose instructions) |
|
|
|
(A) Snap the grooved end of the plastic sleeve down and over the yellow safety release. Remove the ATROPEN from the plastic sleeve. Caution: Do not place fingers on the green tip. |
|
|
(B) Firmly grasp the ATROPEN in 1 hand with the green tip pointed down. |
| (C) Pull off the yellow safety release with your other hand. | |
| (D) Aim and firmly jab the green tip straight down (at a 90 degree angle) against the outer thigh. The ATROPEN will activate and deliver the medicine. You can inject through clothing, but make sure pockets at the injection site are empty.
Note: People who do not have a lot of fat at the injection site and small children should also be injected in the outer thigh. Before giving the ATROPEN injection, pinch a fold of skin on the outer thigh to provide a thicker area for injection. |
|
|
|
(E) Hold the ATROPEN firmly in place for at least 10 seconds to allow the injection to finish. |
|
|
(F) After 10 seconds, remove the ATROPEN and massage the injection site for several seconds. Note: If you see the needle after removing the ATROPEN, the injection is complete. If you do not see the needle, check to make sure the yellow safety release has been removed. If the yellow safety release has been removed, repeat Step 4 (D) and Step 4 (E), but press harder to activate the ATROPEN. |
|
|
(G) After the injection, bend the needle back against the ATROPEN using a hard surface. Use the bent needle as a hook to pin the used ATROPEN to the clothing of the exposed person. You can also show the used ATROPEN to the first medical person you see. This will allow medical personnel to see the number of ATROPENs used and the dose. Move yourself and the exposed person away from the contaminated area right away. Get medical help right away. |
|
Instructions for Use of 0.25 mg ATROPEN
|
|
|
|
(A) Remove the plastic cap from the yellow tube and slide the ATROPEN out of the tube. Caution: Do not place fingers on the black tip. |
|
|
(B) Firmly grasp the ATROPEN in 1 hand with the black tip pointed down. |
|
|
(C) Pull off the gray safety release with your other hand. |
|
|
(D) Pinch a fold of skin on the outer thigh to provide a thicker area for injection. Aim and firmly jab the black tip straight down (at a 90 degree angle) against the outer thigh. The ATROPEN will activate and deliver the medicine. You can inject through clothing, but make sure pockets at the injection site are empty. |
|
|
(E) Hold the ATROPEN firmly in place for at least 10 seconds to allow the injection to finish. |
|
|
(F) After 10 seconds, remove the ATROPEN and massage the injection site for several seconds. Note: If you see the needle after removing the ATROPEN, the injection is complete. If you do not see the needle, check to make sure the gray safety release has been removed. If the gray safety release has been removed, repeat Step 4 (D) and Step 4 (E), but press harder to activate the ATROPEN. |
|
|
(G) After the injection, bend the needle back against the ATROPEN using a hard surface. Use the bent needle as a hook to pin the used ATROPEN to the clothing of the exposed person. You can also show the used ATROPEN to the first medical person you see. This will allow medical personnel to see the number of ATROPENs used and the dose. Move yourself and the exposed person away from the contaminated area right away. Get medical help right away. |
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
ATROPEN® is a registered trademark of
Meridian Medical Technologies®, LLC
St. Louis, MO 63146
1-833-739-0945
Revised: 09/2022
Section 51945-4
Principal Display Panel - 0.25 mg Carton Label
NDC 11704-107-01
MERIDIAN
MEDICAL TECHNOLOGIES™
ATROPEN
®
(atropine injection) 0.25 mg
Single-Dose Auto-Injector
for Intramuscular Use Only
Rx Only
For use in Nerve Agent and Insecticide Poisoning
Each auto-injector delivers an injection of 0.21 mg/0.3 mL atropine (equivalent
to 0.25 mg atropine sulfate), 1.5 mg citric acid monohydrate, 2.0 mg sodium
citrate dihydrate and 2.0 mg sodium chloride.
11 Description
Each prefilled ATROPEN single-dose autoinjector provides an intramuscular dose of atropine, a cholinergic muscarinic antagonist in a self-contained unit, designed for self- or caregiver-administration.
When activated, the ATROPEN 0.25 mg autoinjector delivers 0.21 mg atropine base (equivalent to 0.25 mg atropine sulfate) in 0.3 mL of sterile pyrogen-free solution containing citrate buffer, sodium chloride, and Water for Injection. The pH range is 4.0 to 5.0.
When activated, the ATROPEN 0.5 mg autoinjector delivers 0.42 mg atropine base (equivalent to 0.5 mg atropine sulfate), the ATROPEN 1 mg autoinjector delivers 0.84 mg atropine base (equivalent to 1 mg atropine sulfate), and the ATROPEN 2 mg autoinjector delivers 1.67 mg atropine base (equivalent to 2 mg atropine sulfate). Each 0.5 mg, 1 mg, and 2 mg ATROPEN autoinjector delivers atropine in 0.7 mL of sterile pyrogen-free solution containing 12.5 mg glycerin, 2.8 mg phenol, citrate buffer, and Water for Injection. The pH range is 4.0 to 5.0.
Atropine occurs as white crystals, usually needle-like, or as a white, crystalline powder. It is slightly soluble in water with a molecular weight of 289.38. Atropine, a naturally occurring belladonna alkaloid, is a racemic mixture of equal parts of d- and l-hyoscyamine, with activity due almost entirely to the levo isomer of the drug.
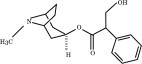
Chemically, atropine is designated as 1αH,5αH-Tropan-3α-ol (±) tropate (ester). Its empirical formula is C17H23NO3 and its structural formula is as follows:
5.2 Heat Injury
7.1 Pralidoxime
When atropine and pralidoxime are used together, the signs of atropinization (flushing, mydriasis, tachycardia, dryness of the mouth and nose) may occur earlier than might be expected when atropine is used alone because pralidoxime may potentiate the effect of atropine. Excitement and manic behavior immediately following recovery of consciousness have been reported in several cases. However, similar behavior has occurred in cases of organophosphate poisoning that were not treated with pralidoxime.
7.2 Barbiturates
Barbiturates are potentiated by the anticholinesterases; therefore, barbiturates should be used cautiously in the treatment of convulsions resulting from exposure to atropine.
16.1 How Supplied
ATROPEN is a prefilled single-dose autoinjector that contains a clear solution and is supplied in the following package configurations:
| NDC Number | Package Configuration | Product Description |
Delivered Dose
(atropine) |
| NDC 11704-107-01 | Carton of 1 | ATROPEN 0.25 mg (yellow label) | 0.21 mg/0.3 mL (equivalent to 0.25 mg/0.3 mL of atropine sulfate) |
| NDC 11704-104-01 | Carton of 1 | ATROPEN 0.5 mg (blue label) |
0.42 mg/0.7 mL (equivalent to 0.5 mg/0.7 mL of atropine sulfate) |
| NDC 11704-105-01 | Carton of 1 | ATROPEN 1 mg (red label) |
0.84 mg/0.7 mL (equivalent to 1 mg/0.7 mL of atropine sulfate) |
| NDC 11704-106-01 | Carton of 1 | ATROPEN 2 mg (green label) |
1.67 mg/0.7 mL (equivalent to 2 mg/0.7 mL of atropine sulfate) |
| NDC 11704-101-01 (For military use only) |
1 Autoinjector | ATROPEN 2 mg | 1.67 mg/0.7 mL (equivalent to 2 mg/0.7 mL of atropine sulfate) |
8.4 Pediatric Use
A review of published literature supports the safety and effectiveness of atropine in the setting of organophosphate insecticide poisoning in all pediatric age groups.
Adverse events seen in pediatric patients treated with atropine are similar to those that occur in adult patients, although central nervous system effects are often seen earlier and at lower doses [see Adverse Reactions (6)].
Overheating (atropine fever) caused by suppression of sweat gland activity may be more pronounced in infants and small children. Extreme hyperthermia in a newborn has been reported with as little as 0.065 mg orally.
8.5 Geriatric Use
Geriatric patients may be more susceptible to the effects of atropine. Because of the longer half-life of atropine in geriatric patients, they may require less frequent doses after the initial dose [see Clinical Pharmacology (12.3)].
5.3 Acute Glaucoma
ATROPEN may cause acute glaucoma and should be administered with caution in patients at risk for acute glaucoma or who have severe narrow angle glaucoma. Monitor for signs and symptoms of intraocular pressure, as appropriate.
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
- Cardiovascular Risks [see Warnings and Precautions (5.1)]
- Heat Injury [see Warnings and Precautions (5.2)]
- Acute Glaucoma [see Warnings and Precautions (5.3)]
- Urinary Retention [see Warnings and Precautions (5.4)]
- Pyloric Stenosis [see Warnings and Precautions (5.5)]
- Exacerbation of Chronic Lung Disease [see Warnings and Precautions (5.6)]
- Hypersensitivity [see Warnings and Precautions (5.7)]
The following adverse reactions associated with the use of atropine were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
7 Drug Interactions
5.5 Pyloric Stenosis
ATROPEN may cause complete pyloric obstruction in patients with partial pyloric stenosis. These patients should be monitored for gastrointestinal symptoms following administration of ATROPEN.
5.7 Hypersensitivity
Atropine can cause hypersensitivity reactions, including anaphylactic reactions [see Adverse Reactions (6)]. Medical supervision is necessary in patients who have had previous anaphylactic reactions to atropine and require treatment for organophosphorus or nerve agent poisoning.
12.2 Pharmacodynamics
Atropine reduces secretions in the mouth and respiratory passages, relieves airway constriction, and may reduce centrally-mediated respiratory paralysis. In severe organophosphorus poisoning, a fully atropinized patient may develop or continue to have respiratory failure and may require artificial respiration and suctioning of airway secretions. Atropine may cause thickening of secretions.
Atropine-induced parasympathetic inhibition may be preceded by a transient phase of stimulation, especially on the heart where small doses first slow the rate before characteristic tachycardia develops due to paralysis of vagal control. Atropine increases heart rate and reduces atrioventricular conduction time. Adequate atropine doses can prevent or abolish bradycardia or asystole produced by organophosphorus nerve agents.
Atropine may decrease the degree of partial heart block, which can occur after organophosphorus poisoning. In some patients with complete heart block, atropine may accelerate the idioventricular rate; in others, the rate is stabilized. In some patients with conduction defects, atropine may cause paradoxical atrioventricular block and nodal rhythm.
Atropine will not act on the neuromuscular junction and has no effect on muscle paralysis or weakness, fasciculations or tremors.
Atropine is not dependable in reversing the weakness or paralysis of the respiratory muscles. Severe difficulty in breathing requires artificial respiration in addition to the use of atropine.
Systemic doses of atropine slightly raise systolic and lower diastolic pressures and can produce significant postural hypotension. Such doses also slightly increase cardiac output and decrease central venous pressure. Atropine can dilate cutaneous blood vessels, particularly the “blush” area (atropine flush), may cause atropine “fever” due to suppression of sweat gland activity especially in infants and small children and may inhibit sweating, thereby causing hyperthermia, particularly in a warm environment or with exercise [see Warnings and Precautions (5.2)].
12.3 Pharmacokinetics
Atropine is well absorbed after intramuscular administration. Following 1.67 mg atropine given intramuscularly to adults by the 2 mg ATROPEN delivery system, the peak concentration (Cmax) is 9.6 ± 1.5 (mean ± SEM) ng/mL reached between 3-60 minutes (Tmax). The protein binding of atropine is 14 to 22% in plasma. Atropine is distributed throughout the various body tissues and fluids. Much of the drug is metabolized by enzymatic hydrolysis, particularly in the liver. Atropine has been reported to be excreted in human milk [see Use in Specific Populations (8.2)]. The unchanged drug excreted by urine is approximately 13 to 50%.
5.4 Urinary Retention
ATROPEN may cause urinary retention and should be administered with caution to patients with clinically significant bladder outflow obstruction.
2.2 Dosage Information
Different dose strengths of ATROPEN are available depending on the patient's age and weight (see Table 1).
| Age and Body Weight | Strength of each ATROPEN Injection |
|---|---|
| Adults and pediatric patients weighing over 41 kg (90 pounds) (generally over 10 years of age) |
ATROPEN 2 mg (green label) |
| Pediatric patients weighing 18 kg to 41 kg (40 pounds to 90 pounds) (generally 4 to 10 years of age) |
ATROPEN 1 mg (red label) |
| Pediatric patients weighing 7 kg to 18 kg (15 pounds to 40 pounds) (generally 6 months to 4 years of age) |
ATROPEN 0.5 mg (blue label) |
| Pediatric patients weighing less than 7 kg (15 pounds) (generally less than 6 months of age) |
ATROPEN 0.25 mg (yellow label) |
1 Indications and Usage
ATROPEN is indicated for the treatment of poisoning by susceptible organophosphorus nerve agents having cholinesterase activity as well as organophosphorus or carbamate insecticides in adult and pediatric patients.
12.1 Mechanism of Action
Atropine competitively blocks the effects of acetylcholine, including excess acetylcholine due to organophosphorus poisoning, at muscarinic cholinergic receptors on smooth muscle, cardiac muscle, secretory gland cells, and in peripheral autonomic ganglia and the central nervous system.
5.1 Cardiovascular Risks
Cardiovascular adverse reactions reported in the literature for atropine include, but are not limited to, sinus tachycardia, palpitations, premature ventricular contractions, atrial flutter, atrial fibrillation, ventricular flutter, ventricular fibrillation, cardiac syncope, asystole, and myocardial infarction [see Adverse Reactions (6)]. In patients with a recent myocardial infarction and/or severe coronary artery disease, there is a possibility that atropine-induced tachycardia may cause ischemia, extend or initiate myocardial infarcts, and stimulate ventricular ectopy and fibrillation. ATROPEN should be used with caution in patients with known cardiovascular disease or cardiac conduction problems.
16.2 Storage and Handling
Store between 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC and 30ºC (between 59ºF and 86ºF) [See USP Controlled Room Temperature]. Not made with natural rubber latex. Keep from freezing. Protect from light.
After the ATROPEN autoinjector has been activated, the empty container should be disposed of properly. It cannot be refilled, nor can the protruding needle be retracted.
5 Warnings and Precautions
- Cardiovascular (CV) Risks: Tachycardia, palpitations, premature ventricular contractions, flutter, fibrillation, etc. Use caution in patients with known CV disease or conduction problems. (5.1)
- Heat Injury: May inhibit sweating and lead to hyperthermia; avoid excessive exercising and heat exposure. (5.2)
- Acute Glaucoma: May precipitate in susceptible individuals. (5.3)
- Urinary Retention: May precipitate in patient with bladder outflow obstruction. (5.4)
- Pyloric Stenosis: May precipitate complete obstruction. (5.5)
- Exacerbation of Chronic Lung Disease: Atropine may cause inspissation of bronchial secretions and formation of dangerous viscid plugs in individuals with chronic lung disease; monitor respiratory status. (5.6)
- Hypersensitivity: Atropine may cause hypersensitivity reactions, including anaphylaxis. (5.7)
2 Dosage and Administration
- ATROPEN is a single-dose autoinjector intended as an initial treatment of the muscarinic symptoms of insecticide or nerve agent poisonings; definitive medical care should be sought immediately. (2.1)
- Dosage is dependent on weight. (2.2)
- Dosage for Mild Symptoms: If the patient experiences two or more mild symptoms, administer one injection intramuscularly into the mid-lateral thigh. If, at any time after the first dose, the patient develops any of the severe symptoms, administer two additional injections intramuscularly in rapid succession. (2.2)
- Dosage for Severe Symptoms: If the patient is either unconscious or has any of the severe symptoms, immediately administer three injections intramuscularly into the patient's mid-lateral thigh in rapid succession. (2.2)
3 Dosage Forms and Strengths
Injection: Each single-dose ATROPEN autoinjector contains a clear sterile solution of atropine. Four strengths of ATROPEN are available
- 0.25 mg/0.3 mL (yellow label): 0.21 mg atropine (equivalent to 0.25 mg atropine sulfate) in 0.3 mL
- 0.5 mg/0.7 mL (blue label): 0.42 mg atropine (equivalent to 0.5 mg atropine sulfate) in 0.7 mL
- 1 mg/0.7 mL (red label): 0.84 mg atropine (equivalent to 1 mg atropine sulfate) in 0.7 mL
- 2 mg/0.7 mL (green label): 1.67 mg atropine (equivalent to 2 mg atropine sulfate) in 0.7 mL
8 Use in Specific Populations
Geriatric patients may be more susceptible to the effects of atropine. (8.5)
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
2.1 Important Administration Information
- It is recommended that three ATROPEN autoinjectors be available for use in each patient at risk for organophosphorus or carbamate poisoning; one (1) for mild symptoms plus two (2) more for severe symptoms [see Dosage and Administration (2.2)]. Different dose strengths of ATROPEN are available depending on the patient's weight.
- ATROPEN should be used by persons who have had adequate training in the recognition and treatment of nerve agent or insecticide intoxication, but may be administered by a caregiver or self-administration if a trained provider is not available.
- Only administer ATROPEN to patients experiencing symptoms of organophosphorus or carbamate poisoning in a situation where exposure is known or suspected. ATROPEN is a single-dose autoinjector intended as an initial treatment of the muscarinic symptoms of insecticide or nerve agent poisonings (generally breathing difficulties due to increased secretions); definitive medical care should be sought immediately.
- ATROPEN should be administered as soon as symptoms of organophosphorus or carbamate poisoning appear.
- In severe poisonings, it may also be desirable to concurrently administer an anticonvulsant (preferably a benzodiazepine) if seizure is suspected in the unconscious individual since the classic tonic-clonic jerking may not be apparent due to the effects of the poison.
- A cholinesterase reactivator such as pralidoxime may serve as an important adjunct to atropine therapy.
- Close supervision of all treated patients is indicated for at least 48 to 72 hours.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit [see Dosage Forms and Strengths (3)].
5.6 Exacerbation of Chronic Lung Disease
Atropine may cause thickening of bronchial secretions and formation of dangerous viscid plugs in individuals with chronic lung disease. Respiratory status should be monitored in individuals with chronic lung disease following administration of ATROPEN.
Structured Label Content
Section 42229-5 (42229-5)
Dosage for Mild Symptoms
Section 42230-3 (42230-3)
Instructions for Use
ATROPEN (at-ROW-pen)
(atropine injection)
for intramuscular use
Self and Caregiver Instructions for Use of ATROPEN
Important: Follow these instructions only when ready to use ATROPEN. If possible, a healthcare provider or someone who has been trained to identify and treat the symptoms of nerve agent and insecticide poisoning should give the ATROPEN injection. If a healthcare provider is not available during an emergency, a patient or caregiver might need to give the ATROPEN injection.
Individuals should not rely only on ATROPEN for protection from nerve agent and insecticide poisoning. Exposed persons and caregivers need to wear clothing to protect their skin and goggles and masks to protect their face and eyes when available, to avoid exposure.
|
Step 1
Choose the Correct Dose |
|
| Adults and children weighing over 90 pounds (41 kg) (generally over 10 years of age 2 mg ATROPEN (green label) |
2 mg |
| Children weighing 40 pounds to 90 pounds (18 kg to 41 kg) (generally 4 to 10 years of age) 1 mg ATROPEN (red label) |
1 mg |
| Children weighing 15 pounds to 40 pounds (7 kg to 18 kg) (generally 6 months to 4 years of age) 0.5 mg ATROPEN (blue label) |
0.5 mg |
| Infants weighing less than 15 pounds (7 kg) (generally less than 6 months of age) 0.25 mg ATROPEN (yellow label) |
0.25 mg |
Caution: When the ATROPEN is activated, the needle will quickly extend from the green tip and black tip. Keep your fingers away from the green tip and black tip.
|
Step 2
Decide if Symptoms are Mild or Severe |
|
| If nerve agent or insecticide exposure is known or suspected, you may see the following mild and severe symptoms. You may not have all of the symptoms or see all symptoms in others exposed to a nerve agent or certain insecticide poisoning:
|
|
| Mild Symptoms | Severe Symptoms |
|
|
Symptoms in infants and young children:
The mild symptoms excessive, unexplained teary eyes, excessive, unexplained runny nose and increased saliva or sudden drooling, and the severe symptom problems controlling urine or stool (bowel movement) are sometimes seen in healthy infants and young children. In infants and young children, these symptoms are seen less often than the other mild and severe symptoms listed above. Symptoms must be looked at together when nerve agent or insecticide exposure is known or suspected.
The severe symptoms severe muscle twitching and general weakness can happen in infants. Infants may become drowsy or pass out, with muscle floppiness instead of muscle twitching, soon after exposure to nerve agents or insecticides.
|
Step 3 Treating Mild Symptoms |
|
First Dose: Give 1 dose of ATROPEN (1 injection) if you get 2 or more mild symptoms of nerve agent or insecticide exposure. Give the injection in the outer thigh. Ask a caregiver to check your symptoms to see if they continue or get worse. Get medical help right away. Additional Doses: If, any time after the first injection of ATROPEN, the exposed person gets any of the severe symptoms listed above, give 2 more doses of ATROPEN (2 injections) in the outer thigh quickly one right after the other. Use a new ATROPEN for each injection. If you have given yourself the first injection, someone else should give you the second and third ATROPEN injections, if possible. |
|
Treating Severe Symptoms |
|
If you get any of the severe symptoms of nerve agent or insecticide exposure listed above or you see an exposed person who has passed out (unconscious), give 3 doses of ATROPEN (3 injections) right away. Give the 3 injections in the outer thigh quickly, one right after the other, using a new ATROPEN for each injection. Get medical help right away. Warning: Giving more ATROPEN injections by mistake in people who do not have nerve agent or insecticide poisoning may cause an overdose of atropine. This can lead to being unable to see or walk well for several hours. People with heart problems may have serious side effects, that can lead to death. |
|
Step 4
Give the ATROPEN Injection |
|
|
Instructions for Use of 2 mg, 1 mg, and 0.5 mg ATROPEN
(see below for 0.25 mg dose instructions) |
|
|
|
(A) Snap the grooved end of the plastic sleeve down and over the yellow safety release. Remove the ATROPEN from the plastic sleeve. Caution: Do not place fingers on the green tip. |
|
|
(B) Firmly grasp the ATROPEN in 1 hand with the green tip pointed down. |
| (C) Pull off the yellow safety release with your other hand. | |
| (D) Aim and firmly jab the green tip straight down (at a 90 degree angle) against the outer thigh. The ATROPEN will activate and deliver the medicine. You can inject through clothing, but make sure pockets at the injection site are empty.
Note: People who do not have a lot of fat at the injection site and small children should also be injected in the outer thigh. Before giving the ATROPEN injection, pinch a fold of skin on the outer thigh to provide a thicker area for injection. |
|
|
|
(E) Hold the ATROPEN firmly in place for at least 10 seconds to allow the injection to finish. |
|
|
(F) After 10 seconds, remove the ATROPEN and massage the injection site for several seconds. Note: If you see the needle after removing the ATROPEN, the injection is complete. If you do not see the needle, check to make sure the yellow safety release has been removed. If the yellow safety release has been removed, repeat Step 4 (D) and Step 4 (E), but press harder to activate the ATROPEN. |
|
|
(G) After the injection, bend the needle back against the ATROPEN using a hard surface. Use the bent needle as a hook to pin the used ATROPEN to the clothing of the exposed person. You can also show the used ATROPEN to the first medical person you see. This will allow medical personnel to see the number of ATROPENs used and the dose. Move yourself and the exposed person away from the contaminated area right away. Get medical help right away. |
|
Instructions for Use of 0.25 mg ATROPEN
|
|
|
|
(A) Remove the plastic cap from the yellow tube and slide the ATROPEN out of the tube. Caution: Do not place fingers on the black tip. |
|
|
(B) Firmly grasp the ATROPEN in 1 hand with the black tip pointed down. |
|
|
(C) Pull off the gray safety release with your other hand. |
|
|
(D) Pinch a fold of skin on the outer thigh to provide a thicker area for injection. Aim and firmly jab the black tip straight down (at a 90 degree angle) against the outer thigh. The ATROPEN will activate and deliver the medicine. You can inject through clothing, but make sure pockets at the injection site are empty. |
|
|
(E) Hold the ATROPEN firmly in place for at least 10 seconds to allow the injection to finish. |
|
|
(F) After 10 seconds, remove the ATROPEN and massage the injection site for several seconds. Note: If you see the needle after removing the ATROPEN, the injection is complete. If you do not see the needle, check to make sure the gray safety release has been removed. If the gray safety release has been removed, repeat Step 4 (D) and Step 4 (E), but press harder to activate the ATROPEN. |
|
|
(G) After the injection, bend the needle back against the ATROPEN using a hard surface. Use the bent needle as a hook to pin the used ATROPEN to the clothing of the exposed person. You can also show the used ATROPEN to the first medical person you see. This will allow medical personnel to see the number of ATROPENs used and the dose. Move yourself and the exposed person away from the contaminated area right away. Get medical help right away. |
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
ATROPEN® is a registered trademark of
Meridian Medical Technologies®, LLC
St. Louis, MO 63146
1-833-739-0945
Revised: 09/2022
Section 51945-4 (51945-4)
Principal Display Panel - 0.25 mg Carton Label
NDC 11704-107-01
MERIDIAN
MEDICAL TECHNOLOGIES™
ATROPEN
®
(atropine injection) 0.25 mg
Single-Dose Auto-Injector
for Intramuscular Use Only
Rx Only
For use in Nerve Agent and Insecticide Poisoning
Each auto-injector delivers an injection of 0.21 mg/0.3 mL atropine (equivalent
to 0.25 mg atropine sulfate), 1.5 mg citric acid monohydrate, 2.0 mg sodium
citrate dihydrate and 2.0 mg sodium chloride.
11 Description (11 DESCRIPTION)
Each prefilled ATROPEN single-dose autoinjector provides an intramuscular dose of atropine, a cholinergic muscarinic antagonist in a self-contained unit, designed for self- or caregiver-administration.
When activated, the ATROPEN 0.25 mg autoinjector delivers 0.21 mg atropine base (equivalent to 0.25 mg atropine sulfate) in 0.3 mL of sterile pyrogen-free solution containing citrate buffer, sodium chloride, and Water for Injection. The pH range is 4.0 to 5.0.
When activated, the ATROPEN 0.5 mg autoinjector delivers 0.42 mg atropine base (equivalent to 0.5 mg atropine sulfate), the ATROPEN 1 mg autoinjector delivers 0.84 mg atropine base (equivalent to 1 mg atropine sulfate), and the ATROPEN 2 mg autoinjector delivers 1.67 mg atropine base (equivalent to 2 mg atropine sulfate). Each 0.5 mg, 1 mg, and 2 mg ATROPEN autoinjector delivers atropine in 0.7 mL of sterile pyrogen-free solution containing 12.5 mg glycerin, 2.8 mg phenol, citrate buffer, and Water for Injection. The pH range is 4.0 to 5.0.
Atropine occurs as white crystals, usually needle-like, or as a white, crystalline powder. It is slightly soluble in water with a molecular weight of 289.38. Atropine, a naturally occurring belladonna alkaloid, is a racemic mixture of equal parts of d- and l-hyoscyamine, with activity due almost entirely to the levo isomer of the drug.
Chemically, atropine is designated as 1αH,5αH-Tropan-3α-ol (±) tropate (ester). Its empirical formula is C17H23NO3 and its structural formula is as follows:
5.2 Heat Injury
7.1 Pralidoxime
When atropine and pralidoxime are used together, the signs of atropinization (flushing, mydriasis, tachycardia, dryness of the mouth and nose) may occur earlier than might be expected when atropine is used alone because pralidoxime may potentiate the effect of atropine. Excitement and manic behavior immediately following recovery of consciousness have been reported in several cases. However, similar behavior has occurred in cases of organophosphate poisoning that were not treated with pralidoxime.
7.2 Barbiturates
Barbiturates are potentiated by the anticholinesterases; therefore, barbiturates should be used cautiously in the treatment of convulsions resulting from exposure to atropine.
16.1 How Supplied
ATROPEN is a prefilled single-dose autoinjector that contains a clear solution and is supplied in the following package configurations:
| NDC Number | Package Configuration | Product Description |
Delivered Dose
(atropine) |
| NDC 11704-107-01 | Carton of 1 | ATROPEN 0.25 mg (yellow label) | 0.21 mg/0.3 mL (equivalent to 0.25 mg/0.3 mL of atropine sulfate) |
| NDC 11704-104-01 | Carton of 1 | ATROPEN 0.5 mg (blue label) |
0.42 mg/0.7 mL (equivalent to 0.5 mg/0.7 mL of atropine sulfate) |
| NDC 11704-105-01 | Carton of 1 | ATROPEN 1 mg (red label) |
0.84 mg/0.7 mL (equivalent to 1 mg/0.7 mL of atropine sulfate) |
| NDC 11704-106-01 | Carton of 1 | ATROPEN 2 mg (green label) |
1.67 mg/0.7 mL (equivalent to 2 mg/0.7 mL of atropine sulfate) |
| NDC 11704-101-01 (For military use only) |
1 Autoinjector | ATROPEN 2 mg | 1.67 mg/0.7 mL (equivalent to 2 mg/0.7 mL of atropine sulfate) |
8.4 Pediatric Use
A review of published literature supports the safety and effectiveness of atropine in the setting of organophosphate insecticide poisoning in all pediatric age groups.
Adverse events seen in pediatric patients treated with atropine are similar to those that occur in adult patients, although central nervous system effects are often seen earlier and at lower doses [see Adverse Reactions (6)].
Overheating (atropine fever) caused by suppression of sweat gland activity may be more pronounced in infants and small children. Extreme hyperthermia in a newborn has been reported with as little as 0.065 mg orally.
8.5 Geriatric Use
Geriatric patients may be more susceptible to the effects of atropine. Because of the longer half-life of atropine in geriatric patients, they may require less frequent doses after the initial dose [see Clinical Pharmacology (12.3)].
5.3 Acute Glaucoma
ATROPEN may cause acute glaucoma and should be administered with caution in patients at risk for acute glaucoma or who have severe narrow angle glaucoma. Monitor for signs and symptoms of intraocular pressure, as appropriate.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
- Cardiovascular Risks [see Warnings and Precautions (5.1)]
- Heat Injury [see Warnings and Precautions (5.2)]
- Acute Glaucoma [see Warnings and Precautions (5.3)]
- Urinary Retention [see Warnings and Precautions (5.4)]
- Pyloric Stenosis [see Warnings and Precautions (5.5)]
- Exacerbation of Chronic Lung Disease [see Warnings and Precautions (5.6)]
- Hypersensitivity [see Warnings and Precautions (5.7)]
The following adverse reactions associated with the use of atropine were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
7 Drug Interactions (7 DRUG INTERACTIONS)
5.5 Pyloric Stenosis
ATROPEN may cause complete pyloric obstruction in patients with partial pyloric stenosis. These patients should be monitored for gastrointestinal symptoms following administration of ATROPEN.
5.7 Hypersensitivity
Atropine can cause hypersensitivity reactions, including anaphylactic reactions [see Adverse Reactions (6)]. Medical supervision is necessary in patients who have had previous anaphylactic reactions to atropine and require treatment for organophosphorus or nerve agent poisoning.
12.2 Pharmacodynamics
Atropine reduces secretions in the mouth and respiratory passages, relieves airway constriction, and may reduce centrally-mediated respiratory paralysis. In severe organophosphorus poisoning, a fully atropinized patient may develop or continue to have respiratory failure and may require artificial respiration and suctioning of airway secretions. Atropine may cause thickening of secretions.
Atropine-induced parasympathetic inhibition may be preceded by a transient phase of stimulation, especially on the heart where small doses first slow the rate before characteristic tachycardia develops due to paralysis of vagal control. Atropine increases heart rate and reduces atrioventricular conduction time. Adequate atropine doses can prevent or abolish bradycardia or asystole produced by organophosphorus nerve agents.
Atropine may decrease the degree of partial heart block, which can occur after organophosphorus poisoning. In some patients with complete heart block, atropine may accelerate the idioventricular rate; in others, the rate is stabilized. In some patients with conduction defects, atropine may cause paradoxical atrioventricular block and nodal rhythm.
Atropine will not act on the neuromuscular junction and has no effect on muscle paralysis or weakness, fasciculations or tremors.
Atropine is not dependable in reversing the weakness or paralysis of the respiratory muscles. Severe difficulty in breathing requires artificial respiration in addition to the use of atropine.
Systemic doses of atropine slightly raise systolic and lower diastolic pressures and can produce significant postural hypotension. Such doses also slightly increase cardiac output and decrease central venous pressure. Atropine can dilate cutaneous blood vessels, particularly the “blush” area (atropine flush), may cause atropine “fever” due to suppression of sweat gland activity especially in infants and small children and may inhibit sweating, thereby causing hyperthermia, particularly in a warm environment or with exercise [see Warnings and Precautions (5.2)].
12.3 Pharmacokinetics
Atropine is well absorbed after intramuscular administration. Following 1.67 mg atropine given intramuscularly to adults by the 2 mg ATROPEN delivery system, the peak concentration (Cmax) is 9.6 ± 1.5 (mean ± SEM) ng/mL reached between 3-60 minutes (Tmax). The protein binding of atropine is 14 to 22% in plasma. Atropine is distributed throughout the various body tissues and fluids. Much of the drug is metabolized by enzymatic hydrolysis, particularly in the liver. Atropine has been reported to be excreted in human milk [see Use in Specific Populations (8.2)]. The unchanged drug excreted by urine is approximately 13 to 50%.
5.4 Urinary Retention
ATROPEN may cause urinary retention and should be administered with caution to patients with clinically significant bladder outflow obstruction.
2.2 Dosage Information
Different dose strengths of ATROPEN are available depending on the patient's age and weight (see Table 1).
| Age and Body Weight | Strength of each ATROPEN Injection |
|---|---|
| Adults and pediatric patients weighing over 41 kg (90 pounds) (generally over 10 years of age) |
ATROPEN 2 mg (green label) |
| Pediatric patients weighing 18 kg to 41 kg (40 pounds to 90 pounds) (generally 4 to 10 years of age) |
ATROPEN 1 mg (red label) |
| Pediatric patients weighing 7 kg to 18 kg (15 pounds to 40 pounds) (generally 6 months to 4 years of age) |
ATROPEN 0.5 mg (blue label) |
| Pediatric patients weighing less than 7 kg (15 pounds) (generally less than 6 months of age) |
ATROPEN 0.25 mg (yellow label) |
1 Indications and Usage (1 INDICATIONS AND USAGE)
ATROPEN is indicated for the treatment of poisoning by susceptible organophosphorus nerve agents having cholinesterase activity as well as organophosphorus or carbamate insecticides in adult and pediatric patients.
12.1 Mechanism of Action
Atropine competitively blocks the effects of acetylcholine, including excess acetylcholine due to organophosphorus poisoning, at muscarinic cholinergic receptors on smooth muscle, cardiac muscle, secretory gland cells, and in peripheral autonomic ganglia and the central nervous system.
5.1 Cardiovascular Risks
Cardiovascular adverse reactions reported in the literature for atropine include, but are not limited to, sinus tachycardia, palpitations, premature ventricular contractions, atrial flutter, atrial fibrillation, ventricular flutter, ventricular fibrillation, cardiac syncope, asystole, and myocardial infarction [see Adverse Reactions (6)]. In patients with a recent myocardial infarction and/or severe coronary artery disease, there is a possibility that atropine-induced tachycardia may cause ischemia, extend or initiate myocardial infarcts, and stimulate ventricular ectopy and fibrillation. ATROPEN should be used with caution in patients with known cardiovascular disease or cardiac conduction problems.
16.2 Storage and Handling
Store between 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC and 30ºC (between 59ºF and 86ºF) [See USP Controlled Room Temperature]. Not made with natural rubber latex. Keep from freezing. Protect from light.
After the ATROPEN autoinjector has been activated, the empty container should be disposed of properly. It cannot be refilled, nor can the protruding needle be retracted.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Cardiovascular (CV) Risks: Tachycardia, palpitations, premature ventricular contractions, flutter, fibrillation, etc. Use caution in patients with known CV disease or conduction problems. (5.1)
- Heat Injury: May inhibit sweating and lead to hyperthermia; avoid excessive exercising and heat exposure. (5.2)
- Acute Glaucoma: May precipitate in susceptible individuals. (5.3)
- Urinary Retention: May precipitate in patient with bladder outflow obstruction. (5.4)
- Pyloric Stenosis: May precipitate complete obstruction. (5.5)
- Exacerbation of Chronic Lung Disease: Atropine may cause inspissation of bronchial secretions and formation of dangerous viscid plugs in individuals with chronic lung disease; monitor respiratory status. (5.6)
- Hypersensitivity: Atropine may cause hypersensitivity reactions, including anaphylaxis. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- ATROPEN is a single-dose autoinjector intended as an initial treatment of the muscarinic symptoms of insecticide or nerve agent poisonings; definitive medical care should be sought immediately. (2.1)
- Dosage is dependent on weight. (2.2)
- Dosage for Mild Symptoms: If the patient experiences two or more mild symptoms, administer one injection intramuscularly into the mid-lateral thigh. If, at any time after the first dose, the patient develops any of the severe symptoms, administer two additional injections intramuscularly in rapid succession. (2.2)
- Dosage for Severe Symptoms: If the patient is either unconscious or has any of the severe symptoms, immediately administer three injections intramuscularly into the patient's mid-lateral thigh in rapid succession. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: Each single-dose ATROPEN autoinjector contains a clear sterile solution of atropine. Four strengths of ATROPEN are available
- 0.25 mg/0.3 mL (yellow label): 0.21 mg atropine (equivalent to 0.25 mg atropine sulfate) in 0.3 mL
- 0.5 mg/0.7 mL (blue label): 0.42 mg atropine (equivalent to 0.5 mg atropine sulfate) in 0.7 mL
- 1 mg/0.7 mL (red label): 0.84 mg atropine (equivalent to 1 mg atropine sulfate) in 0.7 mL
- 2 mg/0.7 mL (green label): 1.67 mg atropine (equivalent to 2 mg atropine sulfate) in 0.7 mL
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Geriatric patients may be more susceptible to the effects of atropine. (8.5)
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
2.1 Important Administration Information
- It is recommended that three ATROPEN autoinjectors be available for use in each patient at risk for organophosphorus or carbamate poisoning; one (1) for mild symptoms plus two (2) more for severe symptoms [see Dosage and Administration (2.2)]. Different dose strengths of ATROPEN are available depending on the patient's weight.
- ATROPEN should be used by persons who have had adequate training in the recognition and treatment of nerve agent or insecticide intoxication, but may be administered by a caregiver or self-administration if a trained provider is not available.
- Only administer ATROPEN to patients experiencing symptoms of organophosphorus or carbamate poisoning in a situation where exposure is known or suspected. ATROPEN is a single-dose autoinjector intended as an initial treatment of the muscarinic symptoms of insecticide or nerve agent poisonings (generally breathing difficulties due to increased secretions); definitive medical care should be sought immediately.
- ATROPEN should be administered as soon as symptoms of organophosphorus or carbamate poisoning appear.
- In severe poisonings, it may also be desirable to concurrently administer an anticonvulsant (preferably a benzodiazepine) if seizure is suspected in the unconscious individual since the classic tonic-clonic jerking may not be apparent due to the effects of the poison.
- A cholinesterase reactivator such as pralidoxime may serve as an important adjunct to atropine therapy.
- Close supervision of all treated patients is indicated for at least 48 to 72 hours.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit [see Dosage Forms and Strengths (3)].
5.6 Exacerbation of Chronic Lung Disease
Atropine may cause thickening of bronchial secretions and formation of dangerous viscid plugs in individuals with chronic lung disease. Respiratory status should be monitored in individuals with chronic lung disease following administration of ATROPEN.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:14.705985 · Updated: 2026-03-14T21:49:30.472732