Eplerenone Tablets
e2a51c12-bdc2-4b3b-8e9e-bf363c2817ed
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Eplerenone Tablets are an aldosterone antagonist indicated for: The treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions ( 1.2 ).
Dosage and Administration
Hypertension: 50 mg once daily, For inadequate response, increase to 50 mg twice daily. Higher dosages are not recommended. ( 2.2 ) For all patients: Measure serum potassium before starting Eplerenone Tablets and periodically thereafter. ( 2.3 )
Contraindications
For all patients: Serum potassium >5.5 mEq/L at initiation ( 4 ) Creatinine clearance ≤30 mL/min ( 4 ) Concomitant use with strong CYP3A inhibitors ( 4 , 7.1 ) For the treatment of hypertension: Type 2 diabetes with microalbuminuria ( 4 ) Serum creatinine >2.0 mg/dL in males, >1.8 mg/dL in females ( 4 ) Creatinine clearance <50 mL/min ( 4 ) Concomitant use of potassium supplements or potassium-sparing diuretics ( 4 )
Warnings and Precautions
Hyperkalemia: Patients with decreased renal function, diabetes, proteinuria or patients who are taking ARBs, NSAIDs or moderate CYP3A inhibitors are at increased risk. Monitor serum potassium levels and adjust dose as needed. ( 5.1 )
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Hyperkalemia [see Warnings and Precautions (5.1) ]
Drug Interactions
CYP3A Inhibitors: In patients with hypertension, initiate at 25 mg once daily. For inadequate blood pressure response, dosing may be increased to a maximum of 25 mg twice daily. ( 2.4 , 7.1 , 12.3 )
How Supplied
Eplerenone Tablets, 25 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side. NDC Number Size 51991-877-33 Bottle of 30 tablets 51991-877-90 Bottle of 90 tablets 51991-877-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box) Eplerenone Tablets, 50 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side. NDC Number Size 51991-878-33 Bottle of 30 tablets 51991-878-90 Bottle of 90 tablets 51991-878-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box)
Storage and Handling
Eplerenone Tablets, 25 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side. NDC Number Size 51991-877-33 Bottle of 30 tablets 51991-877-90 Bottle of 90 tablets 51991-877-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box) Eplerenone Tablets, 50 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side. NDC Number Size 51991-878-33 Bottle of 30 tablets 51991-878-90 Bottle of 90 tablets 51991-878-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box)
Description
Eplerenone Tablets are an aldosterone antagonist indicated for: The treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions ( 1.2 ).
Medication Information
Warnings and Precautions
Hyperkalemia: Patients with decreased renal function, diabetes, proteinuria or patients who are taking ARBs, NSAIDs or moderate CYP3A inhibitors are at increased risk. Monitor serum potassium levels and adjust dose as needed. ( 5.1 )
Indications and Usage
Eplerenone Tablets are an aldosterone antagonist indicated for: The treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions ( 1.2 ).
Dosage and Administration
Hypertension: 50 mg once daily, For inadequate response, increase to 50 mg twice daily. Higher dosages are not recommended. ( 2.2 ) For all patients: Measure serum potassium before starting Eplerenone Tablets and periodically thereafter. ( 2.3 )
Contraindications
For all patients: Serum potassium >5.5 mEq/L at initiation ( 4 ) Creatinine clearance ≤30 mL/min ( 4 ) Concomitant use with strong CYP3A inhibitors ( 4 , 7.1 ) For the treatment of hypertension: Type 2 diabetes with microalbuminuria ( 4 ) Serum creatinine >2.0 mg/dL in males, >1.8 mg/dL in females ( 4 ) Creatinine clearance <50 mL/min ( 4 ) Concomitant use of potassium supplements or potassium-sparing diuretics ( 4 )
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Hyperkalemia [see Warnings and Precautions (5.1) ]
Drug Interactions
CYP3A Inhibitors: In patients with hypertension, initiate at 25 mg once daily. For inadequate blood pressure response, dosing may be increased to a maximum of 25 mg twice daily. ( 2.4 , 7.1 , 12.3 )
Storage and Handling
Eplerenone Tablets, 25 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side. NDC Number Size 51991-877-33 Bottle of 30 tablets 51991-877-90 Bottle of 90 tablets 51991-877-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box) Eplerenone Tablets, 50 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side. NDC Number Size 51991-878-33 Bottle of 30 tablets 51991-878-90 Bottle of 90 tablets 51991-878-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box)
How Supplied
Eplerenone Tablets, 25 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side. NDC Number Size 51991-877-33 Bottle of 30 tablets 51991-877-90 Bottle of 90 tablets 51991-877-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box) Eplerenone Tablets, 50 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side. NDC Number Size 51991-878-33 Bottle of 30 tablets 51991-878-90 Bottle of 90 tablets 51991-878-11 Hospital Unit Dose (10 Blisters of 10 Tablets in a box)
Description
Eplerenone Tablets are an aldosterone antagonist indicated for: The treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions ( 1.2 ).
Section 42229-5
For All Patients
Eplerenone Tablets are contraindicated in all patients with:
- serum potassium >5.5 mEq/L at initiation,
- creatinine clearance ≤30 mL/min, or
- concomitant administration of strong CYP3A inhibitors (e.g., ketoconazole, itraconazole, nefazodone, troleandomycin, clarithromycin, ritonavir, and nelfinavir) [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [See USP Controlled Room Temperature].
7.3 Lithium
A drug interaction study of eplerenone with lithium has not been conducted. Lithium toxicity has been reported in patients receiving lithium concomitantly with diuretics. Serum lithium levels should be monitored frequently if Eplerenone Tablets are administered concomitantly with lithium.
10 Overdosage
No cases of human overdosage with eplerenone have been reported. Lethality was not observed in mice, rats, or dogs after single oral doses that provided Cmax exposures at least 25 times higher than in humans receiving eplerenone 100 mg/day. Dogs showed emesis, salivation, and tremors at a Cmax 41 times the human therapeutic Cmax, progressing to sedation and convulsions at higher exposures.
The most likely manifestation of human overdosage would be anticipated to be hypotension or hyperkalemia. Eplerenone cannot be removed by hemodialysis. Eplerenone has been shown to bind extensively to charcoal. If symptomatic hypotension should occur, supportive treatment should be instituted. If hyperkalemia develops, standard treatment should be initiated.
11 Description
Eplerenone Tablets contain eplerenone, a blocker of aldosterone binding at the mineralocorticoid receptor.
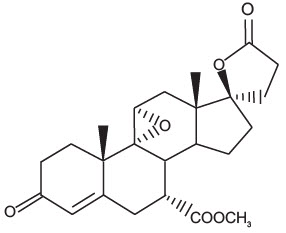
Eplerenone is chemically described as Pregn-4-ene-7,21-dicarboxylic acid, 9,11-epoxy-17-hydroxy-3-oxo-, γ-lactone, methyl ester, (7α,11α, 17α)-. Its empirical formula is C24H30O6 and it has a molecular weight of 414.50. The structural formula of eplerenone is represented below:
Eplerenone is an odorless, white to off-white crystalline powder. It is very slightly soluble in water, with its solubility essentially pH-independent. The octanol/water partition coefficient of eplerenone is approximately 7.1 at pH 7.0.
Eplerenone Tablets for oral administration contain 25 mg or 50 mg of eplerenone and the following inactive ingredients: croscarmellose sodium, D&C Yellow No. 10, FD&C Yellow No. 6, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, red iron oxide, sodium lauryl sulfate, talc, titanium dioxide, and yellow iron oxide.
1.2 Hypertension
Eplerenone Tablets are indicated for the treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular (CV) events, primarily strokes and MI. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes.
Control of high blood pressure should be part of comprehensive CV risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program's Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce CV morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent CV outcome benefit has been a reduction in the risk of stroke, but reductions in MI and CV mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased CV risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
2.2 Hypertension
The recommended starting dose of Eplerenone Tablets is 50 mg administered once daily. The full therapeutic effect of Eplerenone Tablets is apparent within 4 weeks. For patients with an inadequate blood pressure response to 50 mg once daily increase the dosage of eplerenone tablets to 50 mg twice daily. Higher dosages of Eplerenone Tablets are not recommended because they have no greater effect on blood pressure than 100 mg and are associated with an increased risk of hyperkalemia [see Clinical Studies (14.2)].
5.1 Hyperkalemia
The risk of hyperkalemia is higher in patients with impaired renal function, proteinuria, diabetes and those concomitantly treated with ARBs, NSAIDs and moderate CYP3A inhibitors. Minimize the risk of hyperkalemia with proper patient selection and monitoring [see Dosage and Administration (2.1), Contraindications (4), Adverse Reactions (6.2), and Drug Interactions (7)]. Monitor patients for the development of hyperkalemia until the effect of eplerenone is established. Patients who develop hyperkalemia (5.5-5.9 mEq/L) may continue eplerenone therapy with proper dose adjustment. Dose reduction decreases potassium levels. Patients on moderate CYP3A inhibitors that cannot be avoided should have their dose of eplerenone reduced [see Drug Interactions (7.2)].
14.2 Hypertension
The safety and efficacy of Eplerenone Tablets have been evaluated alone and in combination with other antihypertensive agents in clinical studies of 3091 hypertensive patients. The studies included 46% women, 14% Blacks, and 22% elderly (age ≥65). The studies excluded patients with elevated baseline serum potassium (>5.0 mEq/L) and elevated baseline serum creatinine (generally >1.5 mg/dL in males and >1.3 mg/dL in females).
Two fixed-dose, placebo-controlled, 8- to 12-week monotherapy studies in patients with baseline diastolic blood pressures of 95 to 114 mmHg were conducted to assess the antihypertensive effect of Eplerenone Tablets. In these two studies, 611 patients were randomized to Eplerenone Tablets and 140 patients to placebo. Patients received Eplerenone Tablets in doses of 25 mg to 400 mg daily as either a single daily dose or divided into two daily doses. The mean placebo-subtracted reductions in trough cuff blood pressure achieved by Eplerenone Tablets in these studies at doses up to 200 mg are shown in Figures 3 and 4.
Figure 3. Eplerenone Tablets Dose Response – Trough Cuff SBP Placebo-Subtracted Adjusted Mean Change from Baseline in Hypertension Studies
Figure 4. Eplerenone Tablets Dose Response – Trough Cuff DBP Placebo-Subtracted Adjusted Mean Change from Baseline in Hypertension Studies
Patients treated with Eplerenone Tablets 50 mg to 200 mg daily experienced significant decreases in sitting systolic and diastolic blood pressure at trough with differences from placebo of 6–13 mmHg (systolic) and 3–7 mmHg (diastolic). These effects were confirmed by assessments with 24-hour ambulatory blood pressure monitoring (ABPM). In these studies, assessments of 24-hour ABPM data demonstrated that Eplerenone Tablets, administered once or twice daily, maintained antihypertensive efficacy over the entire dosing interval. However, at a total daily dose of 100 mg, Eplerenone Tablets administered as 50 mg twice per day produced greater trough cuff (4/3 mmHg) and ABPM (2/1 mmHg) blood pressure reductions than 100 mg given once daily.
Blood pressure lowering was apparent within 2 weeks from the start of therapy with Eplerenone Tablets, with maximal antihypertensive effects achieved within 4 weeks. Stopping Eplerenone Tablets following treatment for 8 to 24 weeks in six studies did not lead to adverse event rates in the week following withdrawal of Eplerenone Tablets greater than following placebo or active control withdrawal. Blood pressures in patients not taking other antihypertensives rose 1 week after withdrawal of Eplerenone Tablets by about 6/3 mmHg, suggesting that the antihypertensive effect of eplerenone was maintained through 8 to 24 weeks.
Blood pressure reductions with Eplerenone Tablets in the two fixed-dose monotherapy studies and other studies using titrated doses, as well as concomitant treatments, were not significantly different when analyzed by age, gender, or race with one exception. In a study in patients with low renin hypertension, blood pressure reductions in Blacks were smaller than those in whites during the initial titration period with eplerenone.
Eplerenone has been studied concomitantly with treatment with ARB, calcium channel blockers, and beta-blockers. When administered concomitantly with one of these drugs, eplerenone tablets usually produced its expected antihypertensive effects.
8.4 Pediatric Use
The safety and effectiveness of eplerenone for treatment of hypertension have not been established in pediatric patients. In a 10-week study of 304 hypertensive pediatric patients ages 4 to 16 years treated with eplerenone up to 100 mg per day, doses that produced exposure similar to that in adults, eplerenone did not lower blood pressure effectively. In this study and in a 1-year pediatric safety study in 149 patients (age range 5 to 17 years), the incidence of reported adverse events was similar to that of adults. Eplerenone was not studied for treatment of hypertension in pediatric patients younger than 4 years of age because the study in older pediatric patients did not demonstrate effectiveness.
The safety and effectiveness of eplerenone has not been established in pediatric patients with heart failure.
4 Contraindications
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Hyperkalemia [see Warnings and Precautions (5.1)]
7 Drug Interactions
7.1 Cyp3a Inhibitors
Eplerenone metabolism is predominantly mediated via CYP3A. Do not use eplerenone with drugs that are strong inhibitors of CYP3A [see Contraindications (4) and Clinical Pharmacology (12.3)].
In patients with hypertension taking a moderate CYP3A inhibitor, initiate at 25 mg once daily. For inadequate blood pressure response, dosing may be increased to a maximum of 25 mg twice daily [see Dosage and Administration (2.3, 2.4) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
There was no significant change in average heart rate among patients treated with eplerenone in the combined clinical studies. No consistent effects of eplerenone on heart rate, QRS duration, or PR or QT interval were observed in 147 normal subjects evaluated for electrocardiographic changes during pharmacokinetic studies.
12.3 Pharmacokinetics
Eplerenone is cleared predominantly by cytochrome P450 (CYP) 3A4 metabolism, with an elimination half-life of 3 to 6 hours. Steady state is reached within 2 days. Absorption is not affected by food. Inhibitors of CYP3A (e.g., ketoconazole, saquinavir) increase blood levels of eplerenone.
1 Indications and Usage
Eplerenone Tablets are an aldosterone antagonist indicated for:
- The treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions (1.2).
12.1 Mechanism of Action
Eplerenone binds to the mineralocorticoid receptor and blocks the binding of aldosterone, a component of the renin-angiotensin-aldosterone-system (RAAS). Aldosterone synthesis, which occurs primarily in the adrenal gland, is modulated by multiple factors, including angiotensin II and non-RAAS mediators such as adrenocorticotropic hormone (ACTH) and potassium. Aldosterone binds to mineralocorticoid receptors in both epithelial (e.g., kidney) and nonepithelial (e.g., heart, blood vessels, and brain) tissues and increases blood pressure through induction of sodium reabsorption and possibly other mechanisms.
Eplerenone has been shown to produce sustained increases in plasma renin and serum aldosterone, consistent with inhibition of the negative regulatory feedback of aldosterone on renin secretion. The resulting increased plasma renin activity and aldosterone circulating levels do not overcome the effects of eplerenone.
Eplerenone selectively binds to human mineralocorticoid receptors relative to its binding to recombinant human glucocorticoid, progesterone, and androgen receptors.
2.3 Recommended Monitoring
Measure serum potassium before initiating eplerenone tablets therapy, within the first week, and at one month after the start of treatment or dose adjustment. Assess serum potassium periodically thereafter.
Check serum potassium and serum creatinine within 3-7 days of a patient initating a moderate CYP3A inhibitor, angiotensin-II blockers or non-steroidal-anti-inflammatories.
5 Warnings and Precautions
- Hyperkalemia: Patients with decreased renal function, diabetes, proteinuria or patients who are taking ARBs, NSAIDs or moderate CYP3A inhibitors are at increased risk. Monitor serum potassium levels and adjust dose as needed. (5.1)
2 Dosage and Administration
3 Dosage Forms and Strengths
- 25 mg tablets: yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side
- 50 mg tablets: yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of eplerenone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin: angioedema, rash
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise patients receiving eplerenone :
- Not to use potassium supplements or salt substitutes containing potassium without consulting the prescribing physician [see Warnings and Precautions (5.1)].
- To call their physician if they experience dizziness, diarrhea, vomiting, rapid or irregular heartbeat, lower extremity edema, or difficulty breathing [see Warnings and Precautions(5.1)].
16 How Supplied/storage and Handling
Eplerenone Tablets, 25 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side.
| NDC Number | Size |
|---|---|
| 51991-877-33 | Bottle of 30 tablets |
| 51991-877-90 | Bottle of 90 tablets |
| 51991-877-11 | Hospital Unit Dose (10 Blisters of 10 Tablets in a box) |
Eplerenone Tablets, 50 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side.
| NDC Number | Size |
|---|---|
| 51991-878-33 | Bottle of 30 tablets |
| 51991-878-90 | Bottle of 90 tablets |
| 51991-878-11 | Hospital Unit Dose (10 Blisters of 10 Tablets in a box) |
7.2 Angiotensin Ii Receptor Antagonists
The risk of hyperkalemia increases when eplerenone is used in combination with an ARB. A close monitoring of serum potassium and renal function is recommended, especially in patients at risk for impaired renal function, e.g., the elderly [see Warnings and Precautions (5.1)].
7.4 Nonsteroidal Anti Inflammatory Drugs (nsaids)
A drug interaction study of eplerenone with an NSAID has not been conducted. The administration of other potassium-sparing antihypertensives with NSAIDs has been shown to reduce the antihypertensive effect in some patients and result in severe hyperkalemia in patients with impaired renal function. Therefore, when eplerenone and NSAIDs are used concomitantly, monitor blood pressure and serum potassium levels.
Principal Display Panel 25 Mg Tablet Bottle Label
NDC 51991-877-90
Eplerenone Tablets
25 mg
breckenridge
A Towa
Company
Rx Only
90 Tablets
Principal Display Panel 50 Mg Tablet Bottle Label
NDC 51991-878-90
Eplerenone Tablets
50 mg
breckenridge
A Towa
Company
Rx Only
90 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Eplerenone was non-genotoxic in a battery of assays including in vitro bacterial mutagenesis (Ames test in Salmonella spp. and E. coli), in vitro mammalian cell mutagenesis (mouse lymphoma cells), in vitro chromosomal aberration (Chinese hamster ovary cells), in vivo rat bone marrow micronucleus formation, and in vivo/ex vivo unscheduled DNA synthesis in rat liver.
There was no drug-related tumor response in heterozygous P53 deficient mice when tested for 6 months at dosages up to 1000 mg/kg/day (systemic AUC exposures up to 9 times the exposure in humans receiving the 100 mg/day therapeutic dose). Statistically significant increases in benign thyroid tumors were observed after 2 years in both male and female rats when administered eplerenone 250 mg/kg/day (highest dose tested) and in male rats only at 75 mg/kg/day. These dosages provided systemic AUC exposures approximately 2 to 12 times higher than the average human therapeutic exposure at 100 mg/day. Repeat dose administration of eplerenone to rats increases the hepatic conjugation and clearance of thyroxin, which results in increased levels of TSH by a compensatory mechanism. Drugs that have produced thyroid tumors by this rodent-specific mechanism have not shown a similar effect in humans.
Male rats treated with eplerenone at 1000 mg/kg/day for 10 weeks (AUC 17 times that at the 100 mg/day human therapeutic dose) had decreased weights of seminal vesicles and epididymides and slightly decreased fertility. Dogs administered eplerenone at dosages of 15 mg/kg/day and higher (AUC 5 times that at the 100 mg/day human therapeutic dose) had dose-related prostate atrophy. The prostate atrophy was reversible after daily treatment for 1 year at 100 mg/kg/day. Dogs with prostate atrophy showed no decline in libido, sexual performance, or semen quality. Testicular weight and histology were not affected by eplerenone in any test animal species at any dosage.
2.4 Dose Modification for Use With Moderate Cyp3a Inhibitors
In patients with hypertension receiving a moderate CYP3A inhibitor, initiate at 25 mg once daily. For inadequate blood pressure response, dosing may be increased to a maximum of 25 mg twice daily [see Drug Interactions (7.1)].
Structured Label Content
Section 42229-5 (42229-5)
For All Patients
Eplerenone Tablets are contraindicated in all patients with:
- serum potassium >5.5 mEq/L at initiation,
- creatinine clearance ≤30 mL/min, or
- concomitant administration of strong CYP3A inhibitors (e.g., ketoconazole, itraconazole, nefazodone, troleandomycin, clarithromycin, ritonavir, and nelfinavir) [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [See USP Controlled Room Temperature].
7.3 Lithium
A drug interaction study of eplerenone with lithium has not been conducted. Lithium toxicity has been reported in patients receiving lithium concomitantly with diuretics. Serum lithium levels should be monitored frequently if Eplerenone Tablets are administered concomitantly with lithium.
10 Overdosage (10 OVERDOSAGE)
No cases of human overdosage with eplerenone have been reported. Lethality was not observed in mice, rats, or dogs after single oral doses that provided Cmax exposures at least 25 times higher than in humans receiving eplerenone 100 mg/day. Dogs showed emesis, salivation, and tremors at a Cmax 41 times the human therapeutic Cmax, progressing to sedation and convulsions at higher exposures.
The most likely manifestation of human overdosage would be anticipated to be hypotension or hyperkalemia. Eplerenone cannot be removed by hemodialysis. Eplerenone has been shown to bind extensively to charcoal. If symptomatic hypotension should occur, supportive treatment should be instituted. If hyperkalemia develops, standard treatment should be initiated.
11 Description (11 DESCRIPTION)
Eplerenone Tablets contain eplerenone, a blocker of aldosterone binding at the mineralocorticoid receptor.
Eplerenone is chemically described as Pregn-4-ene-7,21-dicarboxylic acid, 9,11-epoxy-17-hydroxy-3-oxo-, γ-lactone, methyl ester, (7α,11α, 17α)-. Its empirical formula is C24H30O6 and it has a molecular weight of 414.50. The structural formula of eplerenone is represented below:
Eplerenone is an odorless, white to off-white crystalline powder. It is very slightly soluble in water, with its solubility essentially pH-independent. The octanol/water partition coefficient of eplerenone is approximately 7.1 at pH 7.0.
Eplerenone Tablets for oral administration contain 25 mg or 50 mg of eplerenone and the following inactive ingredients: croscarmellose sodium, D&C Yellow No. 10, FD&C Yellow No. 6, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, red iron oxide, sodium lauryl sulfate, talc, titanium dioxide, and yellow iron oxide.
1.2 Hypertension
Eplerenone Tablets are indicated for the treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular (CV) events, primarily strokes and MI. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes.
Control of high blood pressure should be part of comprehensive CV risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program's Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce CV morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent CV outcome benefit has been a reduction in the risk of stroke, but reductions in MI and CV mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased CV risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
2.2 Hypertension
The recommended starting dose of Eplerenone Tablets is 50 mg administered once daily. The full therapeutic effect of Eplerenone Tablets is apparent within 4 weeks. For patients with an inadequate blood pressure response to 50 mg once daily increase the dosage of eplerenone tablets to 50 mg twice daily. Higher dosages of Eplerenone Tablets are not recommended because they have no greater effect on blood pressure than 100 mg and are associated with an increased risk of hyperkalemia [see Clinical Studies (14.2)].
5.1 Hyperkalemia
The risk of hyperkalemia is higher in patients with impaired renal function, proteinuria, diabetes and those concomitantly treated with ARBs, NSAIDs and moderate CYP3A inhibitors. Minimize the risk of hyperkalemia with proper patient selection and monitoring [see Dosage and Administration (2.1), Contraindications (4), Adverse Reactions (6.2), and Drug Interactions (7)]. Monitor patients for the development of hyperkalemia until the effect of eplerenone is established. Patients who develop hyperkalemia (5.5-5.9 mEq/L) may continue eplerenone therapy with proper dose adjustment. Dose reduction decreases potassium levels. Patients on moderate CYP3A inhibitors that cannot be avoided should have their dose of eplerenone reduced [see Drug Interactions (7.2)].
14.2 Hypertension
The safety and efficacy of Eplerenone Tablets have been evaluated alone and in combination with other antihypertensive agents in clinical studies of 3091 hypertensive patients. The studies included 46% women, 14% Blacks, and 22% elderly (age ≥65). The studies excluded patients with elevated baseline serum potassium (>5.0 mEq/L) and elevated baseline serum creatinine (generally >1.5 mg/dL in males and >1.3 mg/dL in females).
Two fixed-dose, placebo-controlled, 8- to 12-week monotherapy studies in patients with baseline diastolic blood pressures of 95 to 114 mmHg were conducted to assess the antihypertensive effect of Eplerenone Tablets. In these two studies, 611 patients were randomized to Eplerenone Tablets and 140 patients to placebo. Patients received Eplerenone Tablets in doses of 25 mg to 400 mg daily as either a single daily dose or divided into two daily doses. The mean placebo-subtracted reductions in trough cuff blood pressure achieved by Eplerenone Tablets in these studies at doses up to 200 mg are shown in Figures 3 and 4.
Figure 3. Eplerenone Tablets Dose Response – Trough Cuff SBP Placebo-Subtracted Adjusted Mean Change from Baseline in Hypertension Studies
Figure 4. Eplerenone Tablets Dose Response – Trough Cuff DBP Placebo-Subtracted Adjusted Mean Change from Baseline in Hypertension Studies
Patients treated with Eplerenone Tablets 50 mg to 200 mg daily experienced significant decreases in sitting systolic and diastolic blood pressure at trough with differences from placebo of 6–13 mmHg (systolic) and 3–7 mmHg (diastolic). These effects were confirmed by assessments with 24-hour ambulatory blood pressure monitoring (ABPM). In these studies, assessments of 24-hour ABPM data demonstrated that Eplerenone Tablets, administered once or twice daily, maintained antihypertensive efficacy over the entire dosing interval. However, at a total daily dose of 100 mg, Eplerenone Tablets administered as 50 mg twice per day produced greater trough cuff (4/3 mmHg) and ABPM (2/1 mmHg) blood pressure reductions than 100 mg given once daily.
Blood pressure lowering was apparent within 2 weeks from the start of therapy with Eplerenone Tablets, with maximal antihypertensive effects achieved within 4 weeks. Stopping Eplerenone Tablets following treatment for 8 to 24 weeks in six studies did not lead to adverse event rates in the week following withdrawal of Eplerenone Tablets greater than following placebo or active control withdrawal. Blood pressures in patients not taking other antihypertensives rose 1 week after withdrawal of Eplerenone Tablets by about 6/3 mmHg, suggesting that the antihypertensive effect of eplerenone was maintained through 8 to 24 weeks.
Blood pressure reductions with Eplerenone Tablets in the two fixed-dose monotherapy studies and other studies using titrated doses, as well as concomitant treatments, were not significantly different when analyzed by age, gender, or race with one exception. In a study in patients with low renin hypertension, blood pressure reductions in Blacks were smaller than those in whites during the initial titration period with eplerenone.
Eplerenone has been studied concomitantly with treatment with ARB, calcium channel blockers, and beta-blockers. When administered concomitantly with one of these drugs, eplerenone tablets usually produced its expected antihypertensive effects.
8.4 Pediatric Use
The safety and effectiveness of eplerenone for treatment of hypertension have not been established in pediatric patients. In a 10-week study of 304 hypertensive pediatric patients ages 4 to 16 years treated with eplerenone up to 100 mg per day, doses that produced exposure similar to that in adults, eplerenone did not lower blood pressure effectively. In this study and in a 1-year pediatric safety study in 149 patients (age range 5 to 17 years), the incidence of reported adverse events was similar to that of adults. Eplerenone was not studied for treatment of hypertension in pediatric patients younger than 4 years of age because the study in older pediatric patients did not demonstrate effectiveness.
The safety and effectiveness of eplerenone has not been established in pediatric patients with heart failure.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Hyperkalemia [see Warnings and Precautions (5.1)]
7 Drug Interactions (7 DRUG INTERACTIONS)
7.1 Cyp3a Inhibitors (7.1 CYP3A Inhibitors)
Eplerenone metabolism is predominantly mediated via CYP3A. Do not use eplerenone with drugs that are strong inhibitors of CYP3A [see Contraindications (4) and Clinical Pharmacology (12.3)].
In patients with hypertension taking a moderate CYP3A inhibitor, initiate at 25 mg once daily. For inadequate blood pressure response, dosing may be increased to a maximum of 25 mg twice daily [see Dosage and Administration (2.3, 2.4) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
There was no significant change in average heart rate among patients treated with eplerenone in the combined clinical studies. No consistent effects of eplerenone on heart rate, QRS duration, or PR or QT interval were observed in 147 normal subjects evaluated for electrocardiographic changes during pharmacokinetic studies.
12.3 Pharmacokinetics
Eplerenone is cleared predominantly by cytochrome P450 (CYP) 3A4 metabolism, with an elimination half-life of 3 to 6 hours. Steady state is reached within 2 days. Absorption is not affected by food. Inhibitors of CYP3A (e.g., ketoconazole, saquinavir) increase blood levels of eplerenone.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Eplerenone Tablets are an aldosterone antagonist indicated for:
- The treatment of hypertension in adults, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions (1.2).
12.1 Mechanism of Action
Eplerenone binds to the mineralocorticoid receptor and blocks the binding of aldosterone, a component of the renin-angiotensin-aldosterone-system (RAAS). Aldosterone synthesis, which occurs primarily in the adrenal gland, is modulated by multiple factors, including angiotensin II and non-RAAS mediators such as adrenocorticotropic hormone (ACTH) and potassium. Aldosterone binds to mineralocorticoid receptors in both epithelial (e.g., kidney) and nonepithelial (e.g., heart, blood vessels, and brain) tissues and increases blood pressure through induction of sodium reabsorption and possibly other mechanisms.
Eplerenone has been shown to produce sustained increases in plasma renin and serum aldosterone, consistent with inhibition of the negative regulatory feedback of aldosterone on renin secretion. The resulting increased plasma renin activity and aldosterone circulating levels do not overcome the effects of eplerenone.
Eplerenone selectively binds to human mineralocorticoid receptors relative to its binding to recombinant human glucocorticoid, progesterone, and androgen receptors.
2.3 Recommended Monitoring
Measure serum potassium before initiating eplerenone tablets therapy, within the first week, and at one month after the start of treatment or dose adjustment. Assess serum potassium periodically thereafter.
Check serum potassium and serum creatinine within 3-7 days of a patient initating a moderate CYP3A inhibitor, angiotensin-II blockers or non-steroidal-anti-inflammatories.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hyperkalemia: Patients with decreased renal function, diabetes, proteinuria or patients who are taking ARBs, NSAIDs or moderate CYP3A inhibitors are at increased risk. Monitor serum potassium levels and adjust dose as needed. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 25 mg tablets: yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side
- 50 mg tablets: yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of eplerenone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin: angioedema, rash
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients receiving eplerenone :
- Not to use potassium supplements or salt substitutes containing potassium without consulting the prescribing physician [see Warnings and Precautions (5.1)].
- To call their physician if they experience dizziness, diarrhea, vomiting, rapid or irregular heartbeat, lower extremity edema, or difficulty breathing [see Warnings and Precautions(5.1)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Eplerenone Tablets, 25 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "25" on other side.
| NDC Number | Size |
|---|---|
| 51991-877-33 | Bottle of 30 tablets |
| 51991-877-90 | Bottle of 90 tablets |
| 51991-877-11 | Hospital Unit Dose (10 Blisters of 10 Tablets in a box) |
Eplerenone Tablets, 50 mg, are yellowish red color diamond shaped biconvex film coated tablets, debossed with "E" on one side and "50" on other side.
| NDC Number | Size |
|---|---|
| 51991-878-33 | Bottle of 30 tablets |
| 51991-878-90 | Bottle of 90 tablets |
| 51991-878-11 | Hospital Unit Dose (10 Blisters of 10 Tablets in a box) |
7.2 Angiotensin Ii Receptor Antagonists (7.2 Angiotensin II Receptor Antagonists)
The risk of hyperkalemia increases when eplerenone is used in combination with an ARB. A close monitoring of serum potassium and renal function is recommended, especially in patients at risk for impaired renal function, e.g., the elderly [see Warnings and Precautions (5.1)].
7.4 Nonsteroidal Anti Inflammatory Drugs (nsaids) (7.4 Nonsteroidal Anti-Inflammatory Drugs (NSAIDs))
A drug interaction study of eplerenone with an NSAID has not been conducted. The administration of other potassium-sparing antihypertensives with NSAIDs has been shown to reduce the antihypertensive effect in some patients and result in severe hyperkalemia in patients with impaired renal function. Therefore, when eplerenone and NSAIDs are used concomitantly, monitor blood pressure and serum potassium levels.
Principal Display Panel 25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label)
NDC 51991-877-90
Eplerenone Tablets
25 mg
breckenridge
A Towa
Company
Rx Only
90 Tablets
Principal Display Panel 50 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 50 mg Tablet Bottle Label)
NDC 51991-878-90
Eplerenone Tablets
50 mg
breckenridge
A Towa
Company
Rx Only
90 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Eplerenone was non-genotoxic in a battery of assays including in vitro bacterial mutagenesis (Ames test in Salmonella spp. and E. coli), in vitro mammalian cell mutagenesis (mouse lymphoma cells), in vitro chromosomal aberration (Chinese hamster ovary cells), in vivo rat bone marrow micronucleus formation, and in vivo/ex vivo unscheduled DNA synthesis in rat liver.
There was no drug-related tumor response in heterozygous P53 deficient mice when tested for 6 months at dosages up to 1000 mg/kg/day (systemic AUC exposures up to 9 times the exposure in humans receiving the 100 mg/day therapeutic dose). Statistically significant increases in benign thyroid tumors were observed after 2 years in both male and female rats when administered eplerenone 250 mg/kg/day (highest dose tested) and in male rats only at 75 mg/kg/day. These dosages provided systemic AUC exposures approximately 2 to 12 times higher than the average human therapeutic exposure at 100 mg/day. Repeat dose administration of eplerenone to rats increases the hepatic conjugation and clearance of thyroxin, which results in increased levels of TSH by a compensatory mechanism. Drugs that have produced thyroid tumors by this rodent-specific mechanism have not shown a similar effect in humans.
Male rats treated with eplerenone at 1000 mg/kg/day for 10 weeks (AUC 17 times that at the 100 mg/day human therapeutic dose) had decreased weights of seminal vesicles and epididymides and slightly decreased fertility. Dogs administered eplerenone at dosages of 15 mg/kg/day and higher (AUC 5 times that at the 100 mg/day human therapeutic dose) had dose-related prostate atrophy. The prostate atrophy was reversible after daily treatment for 1 year at 100 mg/kg/day. Dogs with prostate atrophy showed no decline in libido, sexual performance, or semen quality. Testicular weight and histology were not affected by eplerenone in any test animal species at any dosage.
2.4 Dose Modification for Use With Moderate Cyp3a Inhibitors (2.4 Dose Modification for Use with Moderate CYP3A Inhibitors)
In patients with hypertension receiving a moderate CYP3A inhibitor, initiate at 25 mg once daily. For inadequate blood pressure response, dosing may be increased to a maximum of 25 mg twice daily [see Drug Interactions (7.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:06.531513 · Updated: 2026-03-14T22:31:57.634451