These Highlights Do Not Include All The Information Needed To Use Vabomere Safely And Effectively. See Full Prescribing Information For Vabomere.
e237fd0f-3050-42a3-b43e-b6fb4824e93d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
VABOMERE (meropenem and vaborbactam) is a combination of meropenem, a penem antibacterial, and vaborbactam, a beta-lactamase inhibitor, indicated for the treatment of patients 18 years and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by designated susceptible bacteria. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
Indications and Usage
VABOMERE (meropenem and vaborbactam) is a combination of meropenem, a penem antibacterial, and vaborbactam, a beta-lactamase inhibitor, indicated for the treatment of patients 18 years and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by designated susceptible bacteria. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
Dosage and Administration
Administer VABOMERE 4 grams (meropenem 2 grams and vaborbactam 2 grams) every 8 hours by intravenous infusion over 3 hours for up to 14 days, in patients 18 years of age and older with an estimated glomerular filtration rate (eGFR) ≥50 mL/min/1.73m 2 . ( 2.1 ) Dosage adjustment is recommended in patients with renal impairment who have an eGFR less than 50 mL/min/ 1.73m 2 . ( 2.2 ) eGFR As calculated using the Modification of Diet in Renal Disease (MDRD) formula; (mL/min/ 1.73m 2 ) Recommended Dosage Regimen for VABOMERE (meropenem and vaborbactam) All doses of VABOMERE are administered intravenously over 3 hours; , Doses adjusted for renal impairment should be administered after a hemodialysis session; , The total duration of treatment is for up to 14 days. Dosing Interval 30 to 49 VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) Every 8 hours 15 to 29 VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) Every 12 hours Less than 15 VABOMERE 1 gram (meropenem 0.5 grams and vaborbactam 0.5 grams) Every 12 hours See Full Prescribing Information for instructions for constituting supplied dry powder and subsequent required dilution. ( 2.3 ) See Full Prescribing Information for drug compatibilities. ( 2.4 )
Warnings and Precautions
Hypersensitivity reactions were reported with the use of VABOMERE. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta-lactam antibacterial drugs. Discontinue infusion if signs of acute hypersensitivity occur. ( 5.1 ) Seizures and other adverse Central Nervous System experiences have been reported during treatment with meropenem, a component of VABOMERE. ( 5.2 ) Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue VABOMERE and initiate appropriate therapy. ( 5.3 ) Clostridioides difficile -associated diarrhea has been reported with nearly all systemic antibacterial agents, including VABOMERE. Evaluate patients if diarrhea occurs. ( 5.4 ) Co-administration of meropenem with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. ( 5.5 , 7.1 )
Contraindications
VABOMERE is contraindicated in patients with known hypersensitivity to any components of VABOMERE (meropenem and vaborbactam), or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactam antibacterial drugs [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section: Hypersensitivity Reactions [see Warnings and Precautions (5.1) ] Seizure Potential [see Warnings and Precautions (5.2) ] Rhabdomyolysis [see Warnings and Precautions (5.3) ] Clostridioides difficile -associated Diarrhea [see Warnings and Precautions (5.4) ] Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid [see Warnings and Precautions (5.5) ] Thrombocytopenia [see Warnings and Precautions (5.6) ] Potential for Neuromotor Impairment [see Warnings and Precautions (5.7) ] Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.8) ] Overgrowth of Non-susceptible Organisms [see Warnings and Precautions (5.9) ]
Drug Interactions
Hormonal Contraceptives: Effectiveness may be reduced; use an effective alternative non-hormonal form of contraception or additional contraceptive method. ( 7.4 , 12.3 )
Storage and Handling
VABOMERE 2 grams (meropenem and vaborbactam) for injection is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials (NDC 70842-120-01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum overseal. Each vial is supplied in cartons of 6 vials (NDC 70842-120-06). Each vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate.
How Supplied
VABOMERE 2 grams (meropenem and vaborbactam) for injection is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials (NDC 70842-120-01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum overseal. Each vial is supplied in cartons of 6 vials (NDC 70842-120-06). Each vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate.
Medication Information
Warnings and Precautions
Hypersensitivity reactions were reported with the use of VABOMERE. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta-lactam antibacterial drugs. Discontinue infusion if signs of acute hypersensitivity occur. ( 5.1 ) Seizures and other adverse Central Nervous System experiences have been reported during treatment with meropenem, a component of VABOMERE. ( 5.2 ) Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue VABOMERE and initiate appropriate therapy. ( 5.3 ) Clostridioides difficile -associated diarrhea has been reported with nearly all systemic antibacterial agents, including VABOMERE. Evaluate patients if diarrhea occurs. ( 5.4 ) Co-administration of meropenem with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. ( 5.5 , 7.1 )
Indications and Usage
VABOMERE (meropenem and vaborbactam) is a combination of meropenem, a penem antibacterial, and vaborbactam, a beta-lactamase inhibitor, indicated for the treatment of patients 18 years and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by designated susceptible bacteria. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
Dosage and Administration
Administer VABOMERE 4 grams (meropenem 2 grams and vaborbactam 2 grams) every 8 hours by intravenous infusion over 3 hours for up to 14 days, in patients 18 years of age and older with an estimated glomerular filtration rate (eGFR) ≥50 mL/min/1.73m 2 . ( 2.1 ) Dosage adjustment is recommended in patients with renal impairment who have an eGFR less than 50 mL/min/ 1.73m 2 . ( 2.2 ) eGFR As calculated using the Modification of Diet in Renal Disease (MDRD) formula; (mL/min/ 1.73m 2 ) Recommended Dosage Regimen for VABOMERE (meropenem and vaborbactam) All doses of VABOMERE are administered intravenously over 3 hours; , Doses adjusted for renal impairment should be administered after a hemodialysis session; , The total duration of treatment is for up to 14 days. Dosing Interval 30 to 49 VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) Every 8 hours 15 to 29 VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) Every 12 hours Less than 15 VABOMERE 1 gram (meropenem 0.5 grams and vaborbactam 0.5 grams) Every 12 hours See Full Prescribing Information for instructions for constituting supplied dry powder and subsequent required dilution. ( 2.3 ) See Full Prescribing Information for drug compatibilities. ( 2.4 )
Contraindications
VABOMERE is contraindicated in patients with known hypersensitivity to any components of VABOMERE (meropenem and vaborbactam), or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactam antibacterial drugs [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section: Hypersensitivity Reactions [see Warnings and Precautions (5.1) ] Seizure Potential [see Warnings and Precautions (5.2) ] Rhabdomyolysis [see Warnings and Precautions (5.3) ] Clostridioides difficile -associated Diarrhea [see Warnings and Precautions (5.4) ] Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid [see Warnings and Precautions (5.5) ] Thrombocytopenia [see Warnings and Precautions (5.6) ] Potential for Neuromotor Impairment [see Warnings and Precautions (5.7) ] Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.8) ] Overgrowth of Non-susceptible Organisms [see Warnings and Precautions (5.9) ]
Drug Interactions
Hormonal Contraceptives: Effectiveness may be reduced; use an effective alternative non-hormonal form of contraception or additional contraceptive method. ( 7.4 , 12.3 )
Storage and Handling
VABOMERE 2 grams (meropenem and vaborbactam) for injection is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials (NDC 70842-120-01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum overseal. Each vial is supplied in cartons of 6 vials (NDC 70842-120-06). Each vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate.
How Supplied
VABOMERE 2 grams (meropenem and vaborbactam) for injection is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials (NDC 70842-120-01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum overseal. Each vial is supplied in cartons of 6 vials (NDC 70842-120-06). Each vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate.
Description
VABOMERE (meropenem and vaborbactam) is a combination of meropenem, a penem antibacterial, and vaborbactam, a beta-lactamase inhibitor, indicated for the treatment of patients 18 years and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by designated susceptible bacteria. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.2 )
Section 42229-5
Preparation
VABOMERE is supplied as a dry powder in a single-dose vial that must be constituted and further diluted prior to intravenous infusion as outlined below. VABOMERE does not contain preservatives. Aseptic technique must be used for constitution and dilution.
1. To prepare the required dose for intravenous infusion, constitute the appropriate number of vials, as determined from Table 2 below. Withdraw 20 mL of 0.9% Sodium Chloride Injection, USP, from an infusion bag and constitute each vial of VABOMERE.
2. Mix gently to dissolve. The constituted VABOMERE solution will have an approximate meropenem concentration of 0.05 gram/mL and an approximate vaborbactam concentration of 0.05 gram/mL. The final volume is approximately 21.3 mL. The constituted solution is not for direct injection.
3. The constituted solution must be diluted further, immediately, in a 0.9% Sodium Chloride Injection, USP infusion bag before intravenous infusion. The intravenous infusion of the diluted solution must be completed within 4 hours if stored at room temperature or 22 hours if stored refrigerated at 2°C to 8°C (36°F to 46°F).
4. To dilute the constituted solution, withdraw the full or partial constituted vial contents from each vial and add it back into the infusion bag in accordance with Table 2 below.
| VABOMERE Dose (meropenem and vaborbactam) |
Number of Vials to Constitute for Further Dilution | Volume to Withdraw from Each Constituted Vial for Further Dilution | Volume of Infusion Bag | Final Infusion Concentration of VABOMERE |
|---|---|---|---|---|
| 4 grams (2 grams-2 grams) |
2 vials | Entire contents (approximately 21 mL) |
250 mL | 16 mg/mL |
| 500 mL | 8 mg/mL | |||
| 1,000 mL | 4 mg/mL | |||
| 2 grams (1 gram-1 gram) |
1 vial | Entire contents (approximately 21 mL) |
125 mL | 16 mg/mL |
| 250 mL | 8 mg/mL | |||
| 500 mL | 4 mg/mL | |||
| 1 gram (0.5 gram-0.5 gram) |
1 vial | 10.5 mL (discard unused portion) |
70 mL | 14.3 mg/mL |
| 125 mL | 8 mg/mL | |||
| 250 mL | 4 mg/mL |
5. Visually inspect the diluted VABOMERE solution for particulate matter and discoloration prior to administration (the color of the VABOMERE infusion solution for administration ranges from colorless to light yellow). Discard unused portion after use.
Section 44425-7
Store VABOMERE vials at 20°C to 25°C (68°F to 77°F); excursions are permitted to 15°C to 30°C (59°F to 86°F) [see USP, Controlled Room Temperature (CRT)].
1.2. Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
In the event of overdose, discontinue VABOMERE and institute general supportive treatment.
Meropenem and vaborbactam can be removed by hemodialysis. In subjects with end-stage renal disease (ESRD) administered meropenem 1 gram and vaborbactam 1 gram, the mean total recovery in dialysate following a hemodialysis session was 38% and 53% of the administered dose of meropenem and vaborbactam, respectively.
No clinical information is available on the use of hemodialysis to treat VABOMERE overdosage.
15 References
- Kawamura S, Russell AW, Freeman SJ, and Siddall, RA: Reproductive and Developmental Toxicity of Meropenem in Rats. Chemotherapy, 40:S238-250 (1992).
8.2 Lactation
Meropenem has been reported to be excreted in human milk. It is unknown whether vaborbactam is excreted in human milk. No information is available on the effects of meropenem and vaborbactam on the breast-fed child or on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for VABOMERE and any potential adverse effects on the breast-fed child from VABOMERE or from the underlying maternal condition.
11 Description
VABOMERE (meropenem and vaborbactam) for injection is a combination product that contains meropenem, a synthetic penem antibacterial drug and vaborbactam, a cyclic boronic acid beta-lactamase inhibitor, for intravenous administration.
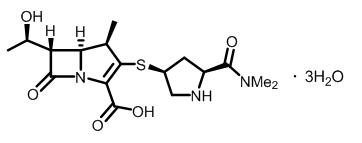
Meropenem, present as a trihydrate, is a white to light yellow crystalline powder, with a molecular weight of 437.52. The chemical name for meropenem trihydrate is (4R,5S,6S)-3-[[(3S,5S)-5-(dimethylcarbamoyl)-3-pyrrolidinyl]thio]-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid, trihydrate. The empirical formula of meropenem trihydrate is C17H25N3O5S∙3H2O and its chemical structure is:
Figure 1: Structure of Meropenem Trihydrate
Vaborbactam is a white to off-white powder, with a molecular weight of 297.14. The chemical name for vaborbactam is (3R,6S)-2-hydroxy-3-[[2-(2-thienyl)acetyl]amino]-1,2-oxaborinane-6-acetic acid. Its empirical formula is C12H16BNO5S and its chemical structure is:
Figure 2: Structure of Vaborbactam
VABOMERE is supplied as a white to light yellow sterile powder for constitution that contains meropenem trihydrate, vaborbactam, and sodium carbonate. Each 50 mL glass vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate. The total sodium content of the mixture is approximately 0.25 grams (10.9 mEq)/vial.
Each vial is constituted and further diluted with 0.9% Sodium Chloride Injection, USP. Both the constituted solution and the diluted solution for intravenous infusion should be a colorless to light yellow solution [see Dosage and Administration (2.3)].
7.2 Probenecid
Probenecid competes with meropenem for active tubular secretion, resulting in increased plasma concentrations of meropenem. Co-administration of probenecid with VABOMERE is not recommended [see Clinical Pharmacology (12.3)].
7.1 Valproic Acid
Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid's glucuronide metabolite (VPA-g) back to valproic acid, thus decreasing the serum concentrations of valproic acid. If administration of VABOMERE is necessary, then supplemental anti-convulsant therapy should be considered [see Warnings and Precautions (5.5)].
8.4 Pediatric Use
The safety and effectiveness of VABOMERE in pediatric patients (younger than 18 years of age) has not been established. Studies of VABOMERE have not been conducted in patients younger than 18 years of age.
8.5 Geriatric Use
Of the 272 patients treated with VABOMERE in the Phase 3 cUTI trial, 48 (18%) patients were 65 years of age and older, while 39 (14%) patients were 75 years of age and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Meropenem, a component of VABOMERE, is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with renal impairment. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Population pharmacokinetic (PK) analysis found no clinically relevant change in pharmacokinetic parameters in elderly patients. No dosage adjustment based on age is required. Dosage adjustment for elderly patients should be based on renal function [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
5.3 Rhabdomyolysis
Rhabdomyolysis has been reported with the use of meropenem, a component of VABOMERE [see Adverse Reactions (6.2)]. If signs or symptoms of rhabdomyolysis such as muscle pain, tenderness or weakness, dark urine, or elevated creatine phosphokinase are observed, discontinue VABOMERE and initiate appropriate therapy.
4 Contraindications
VABOMERE is contraindicated in patients with known hypersensitivity to any components of VABOMERE (meropenem and vaborbactam), or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactam antibacterial drugs [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Seizure Potential [see Warnings and Precautions (5.2)]
- Rhabdomyolysis [see Warnings and Precautions (5.3)]
- Clostridioides difficile-associated Diarrhea [see Warnings and Precautions (5.4)]
- Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid [see Warnings and Precautions (5.5)]
- Thrombocytopenia [see Warnings and Precautions (5.6)]
- Potential for Neuromotor Impairment [see Warnings and Precautions (5.7)]
- Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.8)]
- Overgrowth of Non-susceptible Organisms [see Warnings and Precautions (5.9)]
7 Drug Interactions
5.6 Thrombocytopenia
In patients with renal impairment, thrombocytopenia has been observed in patients treated with meropenem, but no clinical bleeding has been reported [see Dosage and Administration (2.2), Adverse Reactions (6.1), Use in Specific Populations (8.5) and (8.6), and Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Pharmacokinetic studies conducted with meropenem and vaborbactam in subjects with renal impairment have shown that the plasma exposures of both meropenem and vaborbactam increased with decreasing renal function [see Clinical Pharmacology (12.3)]. Dosage adjustment for VABOMERE is recommended in patients with renal impairment (eGFR less than 50 mL/min/1.73m2) [see Dosage and Administration (2.2)].
For patients with changing renal function, monitor serum creatinine concentrations and eGFR at least daily and adjust the dosage of VABOMERE accordingly. Meropenem and vaborbactam are removed by hemodialysis. Following a single dose of VABOMERE, vaborbactam exposure was substantially greater when VABOMERE was administered after hemodialysis than before hemodialysis [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Similar to other beta-lactam antibacterial drugs, the percentage of time of a dosing interval that unbound plasma concentration of meropenem exceeds the meropenem-vaborbactam minimum inhibitory concentration (MIC) against the infecting organism has been shown to best correlate with efficacy in animal and in vitro models of infection. The ratio of the 24-hour unbound plasma vaborbactam AUC to meropenem-vaborbactam MIC is the index that best predicts efficacy of vaborbactam in combination with meropenem in animal and in vitro models of infection.
5.2 Seizure Potential
Seizures and other adverse Central Nervous System (CNS) experiences have been reported during treatment with meropenem, which is a component of VABOMERE. These experiences have occurred most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) or with bacterial meningitis and/or compromised renal function [see Adverse Reactions (6.1) and Drug Interactions (7.1)].
Close adherence to the recommended dosage regimens is urged, especially in patients with known factors that predispose to convulsive activity. Continue anti-convulsant therapy in patients with known seizure disorders. If focal tremors, myoclonus, or seizures occur, evaluate neurologically, place on anti-convulsant therapy if not already instituted, and reexamine the dosage of VABOMERE to determine whether it should be decreased or discontinued.
2.1 Recommended Dosage
The recommended dosage of VABOMERE is 4 grams (meropenem 2 grams and vaborbactam 2 grams) administered every 8 hours by intravenous (IV) infusion over 3 hours in patients 18 years of age and older with an estimated glomerular filtration rate (eGFR) greater than or equal to 50 mL/min/1.73m2. The duration of treatment is for up to 14 days.
2.4 Drug Compatibility
VABOMERE solution for administration by 3-hour infusion is only compatible with 0.9% Sodium Chloride Injection, USP.
Compatibility of VABOMERE solution for administration with other drugs has not been established.
1 Indications and Usage
VABOMERE (meropenem and vaborbactam) is a combination of meropenem, a penem antibacterial, and vaborbactam, a beta-lactamase inhibitor, indicated for the treatment of patients 18 years and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by designated susceptible bacteria. (1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.2)
12.1 Mechanism of Action
VABOMERE is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions
- Hypersensitivity reactions were reported with the use of VABOMERE. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta-lactam antibacterial drugs. Discontinue infusion if signs of acute hypersensitivity occur. (5.1)
- Seizures and other adverse Central Nervous System experiences have been reported during treatment with meropenem, a component of VABOMERE. (5.2)
- Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue VABOMERE and initiate appropriate therapy. (5.3)
- Clostridioides difficile-associated diarrhea has been reported with nearly all systemic antibacterial agents, including VABOMERE. Evaluate patients if diarrhea occurs. (5.4)
- Co-administration of meropenem with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. (5.5, 7.1)
2 Dosage and Administration
- Administer VABOMERE 4 grams (meropenem 2 grams and vaborbactam 2 grams) every 8 hours by intravenous infusion over 3 hours for up to 14 days, in patients 18 years of age and older with an estimated glomerular filtration rate (eGFR) ≥50 mL/min/1.73m2. (2.1)
- Dosage adjustment is recommended in patients with renal impairment who have an eGFR less than 50 mL/min/ 1.73m2. (2.2)
| eGFR As calculated using the Modification of Diet in Renal Disease (MDRD) formula; (mL/min/ 1.73m2) |
Recommended Dosage Regimen for VABOMERE (meropenem and vaborbactam) All doses of VABOMERE are administered intravenously over 3 hours;
,
Doses adjusted for renal impairment should be administered after a hemodialysis session;
,
The total duration of treatment is for up to 14 days.
|
Dosing Interval |
|---|---|---|
| 30 to 49 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 8 hours |
| 15 to 29 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 12 hours |
| Less than 15 | VABOMERE 1 gram (meropenem 0.5 grams and vaborbactam 0.5 grams) | Every 12 hours |
7.4 Hormonal Contraceptives
Hormonal contraceptives (e.g., combined oral contraceptives containing a progestin and an estrogen) are metabolized by CYP3A and other pregnane X receptor (PXR)-regulated enzymes. Therefore, the blood concentration and the effectiveness of hormonal contraceptives may be reduced when used with VABOMERE [see Clinical Pharmacology (12.3)]. Effective alternative non-hormonal forms of contraception or additional contraceptive methods are recommended for patients taking hormonal contraceptives when treated concomitantly with VABOMERE [see Use in Specific Populations (8.3) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths
VABOMERE 2 grams (meropenem and vaborbactam) for injection, is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials containing meropenem 1 gram (equivalent to 1.14 grams meropenem trihydrate) and vaborbactam 1 gram.
5.4 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including VABOMERE, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing isolates of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of meropenem, a component of VABOMERE, or with VABOMERE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Musculoskeletal Disorders: rhabdomyolysis
Hepatobiliary Disorders: drug-induced liver injury (DILI), including hepatitis and liver failure
5.1 Hypersensitivity Reactions
Hypersensitivity reactions were reported in patients treated with VABOMERE in the clinical trials [see Adverse Reactions (6.1)]. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions and serious skin reactions have been reported in patients receiving therapy with beta-lactam antibacterial drugs. These reactions are more likely to occur in individuals with a history of sensitivity to multiple allergens. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe hypersensitivity reactions when treated with another beta-lactam antibacterial drug. Before initiating therapy with VABOMERE, it is important to inquire about previous hypersensitivity reactions to penicillins, cephalosporins, other beta-lactam antibacterial drugs, and other allergens. If an allergic reaction to VABOMERE occurs, discontinue the drug immediately.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
VABOMERE was evaluated in a Phase 3 comparator-controlled clinical trial in cUTI, including pyelonephritis, which included 272 patients treated with VABOMERE and 273 patients treated with the comparator piperacillin/tazobactam 4.5 grams (piperacillin 4 g/tazobactam 0.5 g) every 8 hours. After a minimum of 15 doses of IV therapy, patients could be switched to oral levofloxacin (500 mg daily every 24 hours) to complete the treatment course. Mean duration of IV therapy was 8 days in both treatment groups. Mean duration of IV and oral therapy was 10 days; patients with baseline bacteremia could receive up to 14 days of treatment.
The mean age of patients treated with VABOMERE was 53 years (range 18 to 92 years), and 32% of patients were 65 years of age or older. Patients were predominantly female (66.5%) and White (93.4%). Most patients were enrolled in Europe (89.7%).
16 How Supplied/storage and Handling
VABOMERE 2 grams (meropenem and vaborbactam) for injection is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials (NDC 70842-120-01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum overseal. Each vial is supplied in cartons of 6 vials (NDC 70842-120-06).
Each vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate.
5.7 Potential for Neuromotor Impairment
Alert patients receiving VABOMERE on an outpatient basis regarding adverse reactions such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment. Until it is reasonably well established that VABOMERE is well tolerated, advise patients not to operate machinery or motorized vehicles [see Adverse Reactions (6.1) ].
Principal Display Panel 2 G Vial Carton
Rx only
NDC 70842-120-06
VABOMERE®
(meropenem and vaborbactam)
for injection
2 g per vial*
*Meropenem 1 gram (equivalent
to 1.14 g meropenem trihydrate)
and vaborbactam 1 g
For Intravenous Infusion Only
Single Dose Only
Discard Unused Portion After Use
MUST BE CONSTITUTED THEN DILUTED
See prescribing information for constitution
and dilution instructions and complete
directions for use.
Each vial contains meropenem 1 g,
vaborbactam 1 g, and sodium carbonate
0.575 g. The total sodium content of the
mixture is approximately 0.25 g (10.9 mEq).
Storage:
Store at 20°C to 25°C (68°F to 77°F);
excursions are permitted to 15°C to 30°C
(59°F to 86°F) [See USP Controlled
Room Temperature]
MEL042-R004
Contains 6 single dose 2 g vials
5.8 Development of Drug Resistant Bacteria
Prescribing VABOMERE in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of drug-resistant bacteria [see Indications and Usage (1.2)].
5.9 Overgrowth of Non Susceptible Organisms
As with other antibacterial drugs, prolonged use of VABOMERE may result in overgrowth of non-susceptible organisms. Repeated evaluation of the patient is essential. If superinfection does occur during therapy, appropriate measures should be taken.
8.3 Females and Males of Reproductive Potential
Use of VABOMERE may reduce the effectiveness of hormonal contraceptives. Advise patients taking hormonal contraceptives to use an effective alternative non-hormonal contraception or additional contraceptive method (e.g., barrier method of contraception) during treatment with VABOMERE [see Drug Interactions (7.4)].
7.3 Potential for Vabomere to Affect Other Drugs
When administering VABOMERE concomitantly with medicinal products that are predominantly metabolized by CYP1A2, CYP3A4, CYP2C, and/or are substrates of P-gp transporters, there is a potential risk of interaction which may result in decreased plasma concentrations and activity of the co-administered drug(s) [see Clinical Pharmacology (12.3)]. When VABOMERE is concomitantly administered with the substrates of CYP1A2, CYP3A4, CYP2C, and/or P-gp, refer to the prescribing information for these concomitant medications for guidance on need for dosage adjustments and/or need for frequent drug level monitoring when administered with a weak CYP inducer(s).
When administering VABOMERE concomitantly with medicinal products that are substrate of OAT3 transporters, there is a potential risk of interaction which may result in increased plasma concentrations and activity of the co-administered drug(s) [see Clinical Pharmacology (12.3)]. When VABOMERE is concomitantly administered with OAT3 substrate(s), refer to the prescribing information for these concomitant medication(s) for guidance on need for dosage adjustments and/or need for frequent drug level monitoring when administered with an OAT3 inhibitor(s).
2.2 Dosage Adjustments in Patients With Renal Impairment
Dosage adjustment is recommended in patients with renal impairment who have an eGFR less than 50 mL/min/1.73m2. The recommended dosage of VABOMERE in patients with varying degrees of renal function is presented in Table 1. For patients with changing renal function, monitor serum creatinine concentrations and eGFR at least daily and adjust the dosage of VABOMERE accordingly [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
Meropenem and vaborbactam are removed by hemodialysis [see Clinical Pharmacology (12.3)]. For patients maintained on hemodialysis, administer VABOMERE after a hemodialysis session.
| eGFR As calculated using the Modification of Diet in Renal Disease (MDRD) formula as follows: eGFR (mL/min/1.73m2) = 175 × (serum creatinine)-1.154 × (age)-0.203× (0.742 if female) × (1.212 if African American)
(mL/min/ 1.73m2) |
Recommended Dosage Regimen for VABOMERE (meropenem and vaborbactam) All doses of VABOMERE are administered intravenously over 3 hours.
,
Doses adjusted for renal impairment should be administered after a hemodialysis session.
,
The total duration of treatment is for up to 14 days.
|
Dosing Interval |
|---|---|---|
| 30 to 49 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 8 hours |
| 15 to 29 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 12 hours |
| Less than 15 | VABOMERE 1 gram (meropenem 0.5 grams and vaborbactam 0.5 grams) | Every 12 hours |
1.1. Complicated Urinary Tract Infections (cuti), Including Pyelonephritis
VABOMERE® is indicated for the treatment of patients 18 years of age and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by the following susceptible microorganisms: Escherichia coli, Klebsiella pneumoniae, and Enterobacter cloacae species complex.
14.1 Complicated Urinary Tract Infections (cuti), Including Pyelonephritis
A total of 545 adults with cUTI, including pyelonephritis were randomized into a double-blind, double dummy, multi-center trial comparing VABOMERE (meropenem 2 grams and vaborbactam 2 grams) to piperacillin/tazobactam (piperacillin 4 grams/tazobactam 0.5 grams) intravenously every 8 hours. Switch to an oral antibacterial drug, such as levofloxacin was allowed after a minimum of 15 doses of IV therapy.
The microbiologically modified intent to treat population (m-MITT) included all randomized patients who received any study drug and had at least 1 baseline uropathogen. Clinical and microbiological response at the end of IV treatment (EOIVT) required both a clinical outcome of cure or improvement and a microbiologic outcome of eradication (all baseline uropathogens at >105 CFU/mL are to be reduced to <104 CFU/mL). Clinical and microbiological response was also assessed at the Test of Cure (TOC) visit (approximately 7 days after completion of treatment) in the m-MITT population and required both a clinical outcome of cure and a microbiological outcome of eradication.
Patient demographic and baseline characteristics were balanced between treatment groups in the m-MITT population. Approximately 93% of patients were Caucasian and 66% were females in both treatment groups. The mean age was 54 years with 32% and 42% patients greater than 65 years of age in VABOMERE and piperacillin/tazobactam treatment groups, respectively. Mean body mass index was approximately 26.5 kg/m2 in both treatment groups. Concomitant bacteremia was identified in 12 (6%) and 15 (8%) patients at baseline in VABOMERE and piperacillin/tazobactam treatment groups respectively. The proportion of patients with diabetes mellitus at baseline was 17% and 19% in VABOMERE and piperacillin/tazobactam treatment groups, respectively. The majority of patients (approximately 90%) were enrolled from Europe, and approximately 2% of patients were enrolled from North America. Overall, in both treatment groups, 59% of patients had pyelonephritis and 40% had cUTI, with 21% and 19% of patients having a non-removable and removable source of infection, respectively.
Mean duration of IV treatment in both treatment groups was 8 days and mean total treatment duration (IV and oral) was 10 days; patients with baseline bacteremia could receive up to 14 days of therapy. Approximately 10% of patients in each treatment group in the m-MITT population had a levofloxacin-resistant pathogen at baseline and received levofloxacin as the oral switch therapy. This protocol violation may have impacted the assessment of the outcomes at the TOC visit. These patients were not excluded from the analysis presented in Table 6, as the decision to switch to oral levofloxacin was based on post-randomization factors.
VABOMERE demonstrated efficacy with regard to clinical and microbiological response at the EOIVT visit and TOC visits in the m-MITT population as shown in Table 6.
| VABOMERE n/N (%) |
Piperacillin/Tazobactam n/N (%) |
Difference (95% CI) |
|
|---|---|---|---|
| CI = confidence interval; EOIVT = End of Intravenous Treatment; TOC = Test of Cure | |||
|
Clinical cure or improvement AND microbiological eradication at the End of IV Treatment Visit End of IV Treatment visit includes patients with organisms resistant to piperacillin/tazobactam at baseline
|
183/186 (98.4) | 165/175 (94.3) | 4.1% (0.3%, 8.8%) |
|
Clinical cure AND microbiological eradication at the Test of Cure visit approximately 7 days after completion of treatment Test of Cure visit excludes patients with organisms resistant to piperacillin/tazobactam at baseline
|
124/162 (76.5) | 112/153 (73.2) | 3.3% (-6.2%, 13.0%) |
In the m-MITT population, the rate of clinical and microbiological response in VABOMERE- treated patients with concurrent bacteremia at baseline was 10/12 (83.3%).
In a subset of the E. coli and K. pneumoniae isolates, genotypic testing identified certain ESBL groups (e.g., TEM, CTX-M, SHV and OXA) in both treatment groups of the Phase 3 cUTI trial. The rates of clinical and microbiological response were similar in the ESBL-positive and ESBL-negative subset at EOIVT; at TOC, clinical and microbiological response was lower in the ESBL-positive as compared to ESBL-negative subset in both treatment groups.
5.5 Risk of Breakthrough Seizures Due to Drug Interaction With Valproic Acid
The concomitant use of VABOMERE and valproic acid or divalproex sodium is generally not recommended. Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. Consider administration of antibacterial drugs other than carbapenems to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of VABOMERE is necessary, consider supplemental anticonvulsant therapy [see Drug Interactions (7.1)].
Structured Label Content
Section 42229-5 (42229-5)
Preparation
VABOMERE is supplied as a dry powder in a single-dose vial that must be constituted and further diluted prior to intravenous infusion as outlined below. VABOMERE does not contain preservatives. Aseptic technique must be used for constitution and dilution.
1. To prepare the required dose for intravenous infusion, constitute the appropriate number of vials, as determined from Table 2 below. Withdraw 20 mL of 0.9% Sodium Chloride Injection, USP, from an infusion bag and constitute each vial of VABOMERE.
2. Mix gently to dissolve. The constituted VABOMERE solution will have an approximate meropenem concentration of 0.05 gram/mL and an approximate vaborbactam concentration of 0.05 gram/mL. The final volume is approximately 21.3 mL. The constituted solution is not for direct injection.
3. The constituted solution must be diluted further, immediately, in a 0.9% Sodium Chloride Injection, USP infusion bag before intravenous infusion. The intravenous infusion of the diluted solution must be completed within 4 hours if stored at room temperature or 22 hours if stored refrigerated at 2°C to 8°C (36°F to 46°F).
4. To dilute the constituted solution, withdraw the full or partial constituted vial contents from each vial and add it back into the infusion bag in accordance with Table 2 below.
| VABOMERE Dose (meropenem and vaborbactam) |
Number of Vials to Constitute for Further Dilution | Volume to Withdraw from Each Constituted Vial for Further Dilution | Volume of Infusion Bag | Final Infusion Concentration of VABOMERE |
|---|---|---|---|---|
| 4 grams (2 grams-2 grams) |
2 vials | Entire contents (approximately 21 mL) |
250 mL | 16 mg/mL |
| 500 mL | 8 mg/mL | |||
| 1,000 mL | 4 mg/mL | |||
| 2 grams (1 gram-1 gram) |
1 vial | Entire contents (approximately 21 mL) |
125 mL | 16 mg/mL |
| 250 mL | 8 mg/mL | |||
| 500 mL | 4 mg/mL | |||
| 1 gram (0.5 gram-0.5 gram) |
1 vial | 10.5 mL (discard unused portion) |
70 mL | 14.3 mg/mL |
| 125 mL | 8 mg/mL | |||
| 250 mL | 4 mg/mL |
5. Visually inspect the diluted VABOMERE solution for particulate matter and discoloration prior to administration (the color of the VABOMERE infusion solution for administration ranges from colorless to light yellow). Discard unused portion after use.
Section 44425-7 (44425-7)
Store VABOMERE vials at 20°C to 25°C (68°F to 77°F); excursions are permitted to 15°C to 30°C (59°F to 86°F) [see USP, Controlled Room Temperature (CRT)].
1.2. Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
In the event of overdose, discontinue VABOMERE and institute general supportive treatment.
Meropenem and vaborbactam can be removed by hemodialysis. In subjects with end-stage renal disease (ESRD) administered meropenem 1 gram and vaborbactam 1 gram, the mean total recovery in dialysate following a hemodialysis session was 38% and 53% of the administered dose of meropenem and vaborbactam, respectively.
No clinical information is available on the use of hemodialysis to treat VABOMERE overdosage.
15 References (15 REFERENCES)
- Kawamura S, Russell AW, Freeman SJ, and Siddall, RA: Reproductive and Developmental Toxicity of Meropenem in Rats. Chemotherapy, 40:S238-250 (1992).
8.2 Lactation
Meropenem has been reported to be excreted in human milk. It is unknown whether vaborbactam is excreted in human milk. No information is available on the effects of meropenem and vaborbactam on the breast-fed child or on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for VABOMERE and any potential adverse effects on the breast-fed child from VABOMERE or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
VABOMERE (meropenem and vaborbactam) for injection is a combination product that contains meropenem, a synthetic penem antibacterial drug and vaborbactam, a cyclic boronic acid beta-lactamase inhibitor, for intravenous administration.
Meropenem, present as a trihydrate, is a white to light yellow crystalline powder, with a molecular weight of 437.52. The chemical name for meropenem trihydrate is (4R,5S,6S)-3-[[(3S,5S)-5-(dimethylcarbamoyl)-3-pyrrolidinyl]thio]-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid, trihydrate. The empirical formula of meropenem trihydrate is C17H25N3O5S∙3H2O and its chemical structure is:
Figure 1: Structure of Meropenem Trihydrate
Vaborbactam is a white to off-white powder, with a molecular weight of 297.14. The chemical name for vaborbactam is (3R,6S)-2-hydroxy-3-[[2-(2-thienyl)acetyl]amino]-1,2-oxaborinane-6-acetic acid. Its empirical formula is C12H16BNO5S and its chemical structure is:
Figure 2: Structure of Vaborbactam
VABOMERE is supplied as a white to light yellow sterile powder for constitution that contains meropenem trihydrate, vaborbactam, and sodium carbonate. Each 50 mL glass vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate. The total sodium content of the mixture is approximately 0.25 grams (10.9 mEq)/vial.
Each vial is constituted and further diluted with 0.9% Sodium Chloride Injection, USP. Both the constituted solution and the diluted solution for intravenous infusion should be a colorless to light yellow solution [see Dosage and Administration (2.3)].
7.2 Probenecid
Probenecid competes with meropenem for active tubular secretion, resulting in increased plasma concentrations of meropenem. Co-administration of probenecid with VABOMERE is not recommended [see Clinical Pharmacology (12.3)].
7.1 Valproic Acid
Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid's glucuronide metabolite (VPA-g) back to valproic acid, thus decreasing the serum concentrations of valproic acid. If administration of VABOMERE is necessary, then supplemental anti-convulsant therapy should be considered [see Warnings and Precautions (5.5)].
8.4 Pediatric Use
The safety and effectiveness of VABOMERE in pediatric patients (younger than 18 years of age) has not been established. Studies of VABOMERE have not been conducted in patients younger than 18 years of age.
8.5 Geriatric Use
Of the 272 patients treated with VABOMERE in the Phase 3 cUTI trial, 48 (18%) patients were 65 years of age and older, while 39 (14%) patients were 75 years of age and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Meropenem, a component of VABOMERE, is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with renal impairment. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Population pharmacokinetic (PK) analysis found no clinically relevant change in pharmacokinetic parameters in elderly patients. No dosage adjustment based on age is required. Dosage adjustment for elderly patients should be based on renal function [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
5.3 Rhabdomyolysis
Rhabdomyolysis has been reported with the use of meropenem, a component of VABOMERE [see Adverse Reactions (6.2)]. If signs or symptoms of rhabdomyolysis such as muscle pain, tenderness or weakness, dark urine, or elevated creatine phosphokinase are observed, discontinue VABOMERE and initiate appropriate therapy.
4 Contraindications (4 CONTRAINDICATIONS)
VABOMERE is contraindicated in patients with known hypersensitivity to any components of VABOMERE (meropenem and vaborbactam), or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactam antibacterial drugs [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in the Warnings and Precautions section:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Seizure Potential [see Warnings and Precautions (5.2)]
- Rhabdomyolysis [see Warnings and Precautions (5.3)]
- Clostridioides difficile-associated Diarrhea [see Warnings and Precautions (5.4)]
- Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid [see Warnings and Precautions (5.5)]
- Thrombocytopenia [see Warnings and Precautions (5.6)]
- Potential for Neuromotor Impairment [see Warnings and Precautions (5.7)]
- Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.8)]
- Overgrowth of Non-susceptible Organisms [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.6 Thrombocytopenia
In patients with renal impairment, thrombocytopenia has been observed in patients treated with meropenem, but no clinical bleeding has been reported [see Dosage and Administration (2.2), Adverse Reactions (6.1), Use in Specific Populations (8.5) and (8.6), and Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Pharmacokinetic studies conducted with meropenem and vaborbactam in subjects with renal impairment have shown that the plasma exposures of both meropenem and vaborbactam increased with decreasing renal function [see Clinical Pharmacology (12.3)]. Dosage adjustment for VABOMERE is recommended in patients with renal impairment (eGFR less than 50 mL/min/1.73m2) [see Dosage and Administration (2.2)].
For patients with changing renal function, monitor serum creatinine concentrations and eGFR at least daily and adjust the dosage of VABOMERE accordingly. Meropenem and vaborbactam are removed by hemodialysis. Following a single dose of VABOMERE, vaborbactam exposure was substantially greater when VABOMERE was administered after hemodialysis than before hemodialysis [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Similar to other beta-lactam antibacterial drugs, the percentage of time of a dosing interval that unbound plasma concentration of meropenem exceeds the meropenem-vaborbactam minimum inhibitory concentration (MIC) against the infecting organism has been shown to best correlate with efficacy in animal and in vitro models of infection. The ratio of the 24-hour unbound plasma vaborbactam AUC to meropenem-vaborbactam MIC is the index that best predicts efficacy of vaborbactam in combination with meropenem in animal and in vitro models of infection.
5.2 Seizure Potential
Seizures and other adverse Central Nervous System (CNS) experiences have been reported during treatment with meropenem, which is a component of VABOMERE. These experiences have occurred most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) or with bacterial meningitis and/or compromised renal function [see Adverse Reactions (6.1) and Drug Interactions (7.1)].
Close adherence to the recommended dosage regimens is urged, especially in patients with known factors that predispose to convulsive activity. Continue anti-convulsant therapy in patients with known seizure disorders. If focal tremors, myoclonus, or seizures occur, evaluate neurologically, place on anti-convulsant therapy if not already instituted, and reexamine the dosage of VABOMERE to determine whether it should be decreased or discontinued.
2.1 Recommended Dosage
The recommended dosage of VABOMERE is 4 grams (meropenem 2 grams and vaborbactam 2 grams) administered every 8 hours by intravenous (IV) infusion over 3 hours in patients 18 years of age and older with an estimated glomerular filtration rate (eGFR) greater than or equal to 50 mL/min/1.73m2. The duration of treatment is for up to 14 days.
2.4 Drug Compatibility
VABOMERE solution for administration by 3-hour infusion is only compatible with 0.9% Sodium Chloride Injection, USP.
Compatibility of VABOMERE solution for administration with other drugs has not been established.
1 Indications and Usage (1 INDICATIONS AND USAGE)
VABOMERE (meropenem and vaborbactam) is a combination of meropenem, a penem antibacterial, and vaborbactam, a beta-lactamase inhibitor, indicated for the treatment of patients 18 years and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by designated susceptible bacteria. (1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE and other antibacterial drugs, VABOMERE should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.2)
12.1 Mechanism of Action
VABOMERE is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity reactions were reported with the use of VABOMERE. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta-lactam antibacterial drugs. Discontinue infusion if signs of acute hypersensitivity occur. (5.1)
- Seizures and other adverse Central Nervous System experiences have been reported during treatment with meropenem, a component of VABOMERE. (5.2)
- Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue VABOMERE and initiate appropriate therapy. (5.3)
- Clostridioides difficile-associated diarrhea has been reported with nearly all systemic antibacterial agents, including VABOMERE. Evaluate patients if diarrhea occurs. (5.4)
- Co-administration of meropenem with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. (5.5, 7.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer VABOMERE 4 grams (meropenem 2 grams and vaborbactam 2 grams) every 8 hours by intravenous infusion over 3 hours for up to 14 days, in patients 18 years of age and older with an estimated glomerular filtration rate (eGFR) ≥50 mL/min/1.73m2. (2.1)
- Dosage adjustment is recommended in patients with renal impairment who have an eGFR less than 50 mL/min/ 1.73m2. (2.2)
| eGFR As calculated using the Modification of Diet in Renal Disease (MDRD) formula; (mL/min/ 1.73m2) |
Recommended Dosage Regimen for VABOMERE (meropenem and vaborbactam) All doses of VABOMERE are administered intravenously over 3 hours;
,
Doses adjusted for renal impairment should be administered after a hemodialysis session;
,
The total duration of treatment is for up to 14 days.
|
Dosing Interval |
|---|---|---|
| 30 to 49 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 8 hours |
| 15 to 29 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 12 hours |
| Less than 15 | VABOMERE 1 gram (meropenem 0.5 grams and vaborbactam 0.5 grams) | Every 12 hours |
7.4 Hormonal Contraceptives
Hormonal contraceptives (e.g., combined oral contraceptives containing a progestin and an estrogen) are metabolized by CYP3A and other pregnane X receptor (PXR)-regulated enzymes. Therefore, the blood concentration and the effectiveness of hormonal contraceptives may be reduced when used with VABOMERE [see Clinical Pharmacology (12.3)]. Effective alternative non-hormonal forms of contraception or additional contraceptive methods are recommended for patients taking hormonal contraceptives when treated concomitantly with VABOMERE [see Use in Specific Populations (8.3) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
VABOMERE 2 grams (meropenem and vaborbactam) for injection, is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials containing meropenem 1 gram (equivalent to 1.14 grams meropenem trihydrate) and vaborbactam 1 gram.
5.4 Clostridioides Difficile (5.4 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including VABOMERE, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing isolates of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of meropenem, a component of VABOMERE, or with VABOMERE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Musculoskeletal Disorders: rhabdomyolysis
Hepatobiliary Disorders: drug-induced liver injury (DILI), including hepatitis and liver failure
5.1 Hypersensitivity Reactions
Hypersensitivity reactions were reported in patients treated with VABOMERE in the clinical trials [see Adverse Reactions (6.1)]. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions and serious skin reactions have been reported in patients receiving therapy with beta-lactam antibacterial drugs. These reactions are more likely to occur in individuals with a history of sensitivity to multiple allergens. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe hypersensitivity reactions when treated with another beta-lactam antibacterial drug. Before initiating therapy with VABOMERE, it is important to inquire about previous hypersensitivity reactions to penicillins, cephalosporins, other beta-lactam antibacterial drugs, and other allergens. If an allergic reaction to VABOMERE occurs, discontinue the drug immediately.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
VABOMERE was evaluated in a Phase 3 comparator-controlled clinical trial in cUTI, including pyelonephritis, which included 272 patients treated with VABOMERE and 273 patients treated with the comparator piperacillin/tazobactam 4.5 grams (piperacillin 4 g/tazobactam 0.5 g) every 8 hours. After a minimum of 15 doses of IV therapy, patients could be switched to oral levofloxacin (500 mg daily every 24 hours) to complete the treatment course. Mean duration of IV therapy was 8 days in both treatment groups. Mean duration of IV and oral therapy was 10 days; patients with baseline bacteremia could receive up to 14 days of treatment.
The mean age of patients treated with VABOMERE was 53 years (range 18 to 92 years), and 32% of patients were 65 years of age or older. Patients were predominantly female (66.5%) and White (93.4%). Most patients were enrolled in Europe (89.7%).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
VABOMERE 2 grams (meropenem and vaborbactam) for injection is supplied as a white to light yellow sterile powder for constitution in single-dose, clear glass vials (NDC 70842-120-01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum overseal. Each vial is supplied in cartons of 6 vials (NDC 70842-120-06).
Each vial contains 1 gram of meropenem (equivalent to 1.14 grams of meropenem trihydrate), 1 gram of vaborbactam, and 0.575 gram of sodium carbonate.
5.7 Potential for Neuromotor Impairment
Alert patients receiving VABOMERE on an outpatient basis regarding adverse reactions such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment. Until it is reasonably well established that VABOMERE is well tolerated, advise patients not to operate machinery or motorized vehicles [see Adverse Reactions (6.1) ].
Principal Display Panel 2 G Vial Carton (PRINCIPAL DISPLAY PANEL - 2 g Vial Carton)
Rx only
NDC 70842-120-06
VABOMERE®
(meropenem and vaborbactam)
for injection
2 g per vial*
*Meropenem 1 gram (equivalent
to 1.14 g meropenem trihydrate)
and vaborbactam 1 g
For Intravenous Infusion Only
Single Dose Only
Discard Unused Portion After Use
MUST BE CONSTITUTED THEN DILUTED
See prescribing information for constitution
and dilution instructions and complete
directions for use.
Each vial contains meropenem 1 g,
vaborbactam 1 g, and sodium carbonate
0.575 g. The total sodium content of the
mixture is approximately 0.25 g (10.9 mEq).
Storage:
Store at 20°C to 25°C (68°F to 77°F);
excursions are permitted to 15°C to 30°C
(59°F to 86°F) [See USP Controlled
Room Temperature]
MEL042-R004
Contains 6 single dose 2 g vials
5.8 Development of Drug Resistant Bacteria (5.8 Development of Drug-Resistant Bacteria)
Prescribing VABOMERE in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of drug-resistant bacteria [see Indications and Usage (1.2)].
5.9 Overgrowth of Non Susceptible Organisms (5.9 Overgrowth of Non-susceptible Organisms)
As with other antibacterial drugs, prolonged use of VABOMERE may result in overgrowth of non-susceptible organisms. Repeated evaluation of the patient is essential. If superinfection does occur during therapy, appropriate measures should be taken.
8.3 Females and Males of Reproductive Potential
Use of VABOMERE may reduce the effectiveness of hormonal contraceptives. Advise patients taking hormonal contraceptives to use an effective alternative non-hormonal contraception or additional contraceptive method (e.g., barrier method of contraception) during treatment with VABOMERE [see Drug Interactions (7.4)].
7.3 Potential for Vabomere to Affect Other Drugs (7.3 Potential for VABOMERE to Affect Other Drugs)
When administering VABOMERE concomitantly with medicinal products that are predominantly metabolized by CYP1A2, CYP3A4, CYP2C, and/or are substrates of P-gp transporters, there is a potential risk of interaction which may result in decreased plasma concentrations and activity of the co-administered drug(s) [see Clinical Pharmacology (12.3)]. When VABOMERE is concomitantly administered with the substrates of CYP1A2, CYP3A4, CYP2C, and/or P-gp, refer to the prescribing information for these concomitant medications for guidance on need for dosage adjustments and/or need for frequent drug level monitoring when administered with a weak CYP inducer(s).
When administering VABOMERE concomitantly with medicinal products that are substrate of OAT3 transporters, there is a potential risk of interaction which may result in increased plasma concentrations and activity of the co-administered drug(s) [see Clinical Pharmacology (12.3)]. When VABOMERE is concomitantly administered with OAT3 substrate(s), refer to the prescribing information for these concomitant medication(s) for guidance on need for dosage adjustments and/or need for frequent drug level monitoring when administered with an OAT3 inhibitor(s).
2.2 Dosage Adjustments in Patients With Renal Impairment (2.2 Dosage Adjustments in Patients with Renal Impairment)
Dosage adjustment is recommended in patients with renal impairment who have an eGFR less than 50 mL/min/1.73m2. The recommended dosage of VABOMERE in patients with varying degrees of renal function is presented in Table 1. For patients with changing renal function, monitor serum creatinine concentrations and eGFR at least daily and adjust the dosage of VABOMERE accordingly [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
Meropenem and vaborbactam are removed by hemodialysis [see Clinical Pharmacology (12.3)]. For patients maintained on hemodialysis, administer VABOMERE after a hemodialysis session.
| eGFR As calculated using the Modification of Diet in Renal Disease (MDRD) formula as follows: eGFR (mL/min/1.73m2) = 175 × (serum creatinine)-1.154 × (age)-0.203× (0.742 if female) × (1.212 if African American)
(mL/min/ 1.73m2) |
Recommended Dosage Regimen for VABOMERE (meropenem and vaborbactam) All doses of VABOMERE are administered intravenously over 3 hours.
,
Doses adjusted for renal impairment should be administered after a hemodialysis session.
,
The total duration of treatment is for up to 14 days.
|
Dosing Interval |
|---|---|---|
| 30 to 49 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 8 hours |
| 15 to 29 | VABOMERE 2 grams (meropenem 1 gram and vaborbactam 1 gram) | Every 12 hours |
| Less than 15 | VABOMERE 1 gram (meropenem 0.5 grams and vaborbactam 0.5 grams) | Every 12 hours |
1.1. Complicated Urinary Tract Infections (cuti), Including Pyelonephritis (1.1. Complicated Urinary Tract Infections (cUTI), including Pyelonephritis)
VABOMERE® is indicated for the treatment of patients 18 years of age and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by the following susceptible microorganisms: Escherichia coli, Klebsiella pneumoniae, and Enterobacter cloacae species complex.
14.1 Complicated Urinary Tract Infections (cuti), Including Pyelonephritis (14.1 Complicated Urinary Tract Infections (cUTI), including Pyelonephritis)
A total of 545 adults with cUTI, including pyelonephritis were randomized into a double-blind, double dummy, multi-center trial comparing VABOMERE (meropenem 2 grams and vaborbactam 2 grams) to piperacillin/tazobactam (piperacillin 4 grams/tazobactam 0.5 grams) intravenously every 8 hours. Switch to an oral antibacterial drug, such as levofloxacin was allowed after a minimum of 15 doses of IV therapy.
The microbiologically modified intent to treat population (m-MITT) included all randomized patients who received any study drug and had at least 1 baseline uropathogen. Clinical and microbiological response at the end of IV treatment (EOIVT) required both a clinical outcome of cure or improvement and a microbiologic outcome of eradication (all baseline uropathogens at >105 CFU/mL are to be reduced to <104 CFU/mL). Clinical and microbiological response was also assessed at the Test of Cure (TOC) visit (approximately 7 days after completion of treatment) in the m-MITT population and required both a clinical outcome of cure and a microbiological outcome of eradication.
Patient demographic and baseline characteristics were balanced between treatment groups in the m-MITT population. Approximately 93% of patients were Caucasian and 66% were females in both treatment groups. The mean age was 54 years with 32% and 42% patients greater than 65 years of age in VABOMERE and piperacillin/tazobactam treatment groups, respectively. Mean body mass index was approximately 26.5 kg/m2 in both treatment groups. Concomitant bacteremia was identified in 12 (6%) and 15 (8%) patients at baseline in VABOMERE and piperacillin/tazobactam treatment groups respectively. The proportion of patients with diabetes mellitus at baseline was 17% and 19% in VABOMERE and piperacillin/tazobactam treatment groups, respectively. The majority of patients (approximately 90%) were enrolled from Europe, and approximately 2% of patients were enrolled from North America. Overall, in both treatment groups, 59% of patients had pyelonephritis and 40% had cUTI, with 21% and 19% of patients having a non-removable and removable source of infection, respectively.
Mean duration of IV treatment in both treatment groups was 8 days and mean total treatment duration (IV and oral) was 10 days; patients with baseline bacteremia could receive up to 14 days of therapy. Approximately 10% of patients in each treatment group in the m-MITT population had a levofloxacin-resistant pathogen at baseline and received levofloxacin as the oral switch therapy. This protocol violation may have impacted the assessment of the outcomes at the TOC visit. These patients were not excluded from the analysis presented in Table 6, as the decision to switch to oral levofloxacin was based on post-randomization factors.
VABOMERE demonstrated efficacy with regard to clinical and microbiological response at the EOIVT visit and TOC visits in the m-MITT population as shown in Table 6.
| VABOMERE n/N (%) |
Piperacillin/Tazobactam n/N (%) |
Difference (95% CI) |
|
|---|---|---|---|
| CI = confidence interval; EOIVT = End of Intravenous Treatment; TOC = Test of Cure | |||
|
Clinical cure or improvement AND microbiological eradication at the End of IV Treatment Visit End of IV Treatment visit includes patients with organisms resistant to piperacillin/tazobactam at baseline
|
183/186 (98.4) | 165/175 (94.3) | 4.1% (0.3%, 8.8%) |
|
Clinical cure AND microbiological eradication at the Test of Cure visit approximately 7 days after completion of treatment Test of Cure visit excludes patients with organisms resistant to piperacillin/tazobactam at baseline
|
124/162 (76.5) | 112/153 (73.2) | 3.3% (-6.2%, 13.0%) |
In the m-MITT population, the rate of clinical and microbiological response in VABOMERE- treated patients with concurrent bacteremia at baseline was 10/12 (83.3%).
In a subset of the E. coli and K. pneumoniae isolates, genotypic testing identified certain ESBL groups (e.g., TEM, CTX-M, SHV and OXA) in both treatment groups of the Phase 3 cUTI trial. The rates of clinical and microbiological response were similar in the ESBL-positive and ESBL-negative subset at EOIVT; at TOC, clinical and microbiological response was lower in the ESBL-positive as compared to ESBL-negative subset in both treatment groups.
5.5 Risk of Breakthrough Seizures Due to Drug Interaction With Valproic Acid (5.5 Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid)
The concomitant use of VABOMERE and valproic acid or divalproex sodium is generally not recommended. Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. Consider administration of antibacterial drugs other than carbapenems to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of VABOMERE is necessary, consider supplemental anticonvulsant therapy [see Drug Interactions (7.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:42.643133 · Updated: 2026-03-14T22:51:48.679170