Emerphed
e1323592-dc57-4c9d-b84d-442648a7b114
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
EMERPHED is indicated for the treatment of clinically important hypotension occurring in the setting of anesthesia.
Dosage and Administration
This is a pre-mixed formulation. Do not dilute before administration. ( 2 ) 5 mg to 10 mg administered by intravenous bolus. Additional boluses as needed, not to exceed a total dose of 50 mg. ( 2 )
Contraindications
None
Warnings and Precautions
Pressor Effects with Concomitant Use with Oxytocic Drugs: Pressor effect of sympathomimetic pressor amines is potentiated ( 5.1 ) Tachyphylaxis and Tolerance: Repeated administration of EMERPHED may cause tachyphylaxis ( 5.2 )
Adverse Reactions
The following adverse reactions associated with the use of ephedrine sulfate were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure. Gastrointestinal disorders: Nausea, vomiting Cardiac disorders: Tachycardia, palpitations (thumping heart), reactive hypertension, bradycardia, ventricular ectopics, R-R variability Nervous system disorders: Dizziness Psychiatric disorders: Restlessness
Drug Interactions
Interactions that Augment the Press or Effect Oxytocin and oxytocic drugs Clinical Impact: Serious postpartum hypertension has been described in patients who received both a vasopressor (i.e., methoxamine, phenylephrine, ephedrine) and an oxytocic (i.e., methylergonovine, ergonovine). Some of these patients experienced a stroke. Intervention: Carefully monitor the blood pressure of individuals who have received both EMERPHED and an oxytocic. Clonidine, propofol, monoamine oxidase inhibitors (MAOIs ), atropine Clinical Impact: These drugs augment the pressor effect of ephedrine. Intervention: Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. Interactions that Antagonize the Pressor Effect Clinical Impact: These drugs antagonize the pressor effect of ephedrine. Intervention: Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. Examples: α-adrenergic antagonists, β-adrenergic receptor antagonists, reserpine, quinidine, mephentermine Other Drug Interactions Guanethidine Clinical Impact: EMERPHED may inhibit the neuron blockage produced by guanethidine, resulting in loss of antihypertensive effectiveness. Intervention: Clinician should monitor patient for blood pressor response and adjust the dosage or choice of pressor accordingly. Rocuronium Clinical Impact: EMERPHED may reduce the onset time of neuromuscular blockade when used for intubation with rocuronium if administered simultaneously with anesthetic induction. Intervention: Be aware of this potential interaction. No treatment or other interventions are needed. Epidural anesthesia Clinical Impact: EMERPHED may decrease the efficacy of epidural blockade by hastening the regression of sensory analgesia. Intervention: Monitor and treat the patient according to clinical practice. Theophylline Clinical Impact: Concomitant use of EMERPHED may increase the frequency of nausea, nervousness, and insomnia. Intervention: Monitor patient for worsening symptoms and manage symptoms according to clinical practice. Cardiac glycosides Clinical Impact: Giving EMERPHED with a cardiac glycoside, such as digitalis, may increase the possibility of arrhythmias. Intervention: Carefully monitor patients on cardiac glycosides who are also administered ephedrine.
How Supplied
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base) and is supplied as follows: NDC Presentation 14789-250-07 10 mL clear glass, single- dose vial; strength 5 mg/mL 14789-250-10 10 mL vials packaged in a carton of 10 EMERPHED (ephedrine sulfate) injection 50 mg/10 mL (5 mg/mL) is not made with natural rubber latex. Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Store in carton until time of use. For single dose only. Discard unused portion. Patent, www.emerphed.com/patent Manufactured in Italy for: Nexus Pharmaceuticals, Inc. 400 Knightsbridge Parkway Lincolnshire, IL 60069 USA EPHPIR003
Storage and Handling
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base) and is supplied as follows: NDC Presentation 14789-250-07 10 mL clear glass, single- dose vial; strength 5 mg/mL 14789-250-10 10 mL vials packaged in a carton of 10 EMERPHED (ephedrine sulfate) injection 50 mg/10 mL (5 mg/mL) is not made with natural rubber latex. Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Store in carton until time of use. For single dose only. Discard unused portion. Patent, www.emerphed.com/patent Manufactured in Italy for: Nexus Pharmaceuticals, Inc. 400 Knightsbridge Parkway Lincolnshire, IL 60069 USA EPHPIR003
Description
EMERPHED is indicated for the treatment of clinically important hypotension occurring in the setting of anesthesia.
Medication Information
Warnings and Precautions
Pressor Effects with Concomitant Use with Oxytocic Drugs: Pressor effect of sympathomimetic pressor amines is potentiated ( 5.1 ) Tachyphylaxis and Tolerance: Repeated administration of EMERPHED may cause tachyphylaxis ( 5.2 )
Indications and Usage
EMERPHED is indicated for the treatment of clinically important hypotension occurring in the setting of anesthesia.
Dosage and Administration
This is a pre-mixed formulation. Do not dilute before administration. ( 2 ) 5 mg to 10 mg administered by intravenous bolus. Additional boluses as needed, not to exceed a total dose of 50 mg. ( 2 )
Contraindications
None
Adverse Reactions
The following adverse reactions associated with the use of ephedrine sulfate were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure. Gastrointestinal disorders: Nausea, vomiting Cardiac disorders: Tachycardia, palpitations (thumping heart), reactive hypertension, bradycardia, ventricular ectopics, R-R variability Nervous system disorders: Dizziness Psychiatric disorders: Restlessness
Drug Interactions
Interactions that Augment the Press or Effect Oxytocin and oxytocic drugs Clinical Impact: Serious postpartum hypertension has been described in patients who received both a vasopressor (i.e., methoxamine, phenylephrine, ephedrine) and an oxytocic (i.e., methylergonovine, ergonovine). Some of these patients experienced a stroke. Intervention: Carefully monitor the blood pressure of individuals who have received both EMERPHED and an oxytocic. Clonidine, propofol, monoamine oxidase inhibitors (MAOIs ), atropine Clinical Impact: These drugs augment the pressor effect of ephedrine. Intervention: Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. Interactions that Antagonize the Pressor Effect Clinical Impact: These drugs antagonize the pressor effect of ephedrine. Intervention: Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. Examples: α-adrenergic antagonists, β-adrenergic receptor antagonists, reserpine, quinidine, mephentermine Other Drug Interactions Guanethidine Clinical Impact: EMERPHED may inhibit the neuron blockage produced by guanethidine, resulting in loss of antihypertensive effectiveness. Intervention: Clinician should monitor patient for blood pressor response and adjust the dosage or choice of pressor accordingly. Rocuronium Clinical Impact: EMERPHED may reduce the onset time of neuromuscular blockade when used for intubation with rocuronium if administered simultaneously with anesthetic induction. Intervention: Be aware of this potential interaction. No treatment or other interventions are needed. Epidural anesthesia Clinical Impact: EMERPHED may decrease the efficacy of epidural blockade by hastening the regression of sensory analgesia. Intervention: Monitor and treat the patient according to clinical practice. Theophylline Clinical Impact: Concomitant use of EMERPHED may increase the frequency of nausea, nervousness, and insomnia. Intervention: Monitor patient for worsening symptoms and manage symptoms according to clinical practice. Cardiac glycosides Clinical Impact: Giving EMERPHED with a cardiac glycoside, such as digitalis, may increase the possibility of arrhythmias. Intervention: Carefully monitor patients on cardiac glycosides who are also administered ephedrine.
Storage and Handling
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base) and is supplied as follows: NDC Presentation 14789-250-07 10 mL clear glass, single- dose vial; strength 5 mg/mL 14789-250-10 10 mL vials packaged in a carton of 10 EMERPHED (ephedrine sulfate) injection 50 mg/10 mL (5 mg/mL) is not made with natural rubber latex. Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Store in carton until time of use. For single dose only. Discard unused portion. Patent, www.emerphed.com/patent Manufactured in Italy for: Nexus Pharmaceuticals, Inc. 400 Knightsbridge Parkway Lincolnshire, IL 60069 USA EPHPIR003
How Supplied
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base) and is supplied as follows: NDC Presentation 14789-250-07 10 mL clear glass, single- dose vial; strength 5 mg/mL 14789-250-10 10 mL vials packaged in a carton of 10 EMERPHED (ephedrine sulfate) injection 50 mg/10 mL (5 mg/mL) is not made with natural rubber latex. Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Store in carton until time of use. For single dose only. Discard unused portion. Patent, www.emerphed.com/patent Manufactured in Italy for: Nexus Pharmaceuticals, Inc. 400 Knightsbridge Parkway Lincolnshire, IL 60069 USA EPHPIR003
Description
EMERPHED is indicated for the treatment of clinically important hypotension occurring in the setting of anesthesia.
Section 42229-5
Risk Summary
Available data from randomized studies, case series, and reports of ephedrine sulfate use in pregnant women have not identified a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. However, there are clinical considerations [see Clinical Considerations ]. In animal reproduction studies, decreased fetal survival and fetal body weights were observed in the presence of maternal toxicity after normotensive pregnant rats were administered 60 mg/kg intravenous ephedrine sulfate (12 times the maximum recommended human dose (MRHD) of 50 mg/day). No malformations or embryofetal adverse effects were observed when pregnant rats or rabbits were treated with intravenous bolus doses of ephedrine sulfate during organogenesis at doses 1.9 and 7.7 times the MRHD, respectively [See data ].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 51945-4
Principal Display Panel – 5 mg/mL Carton Label
NDC 14789-250-10
EMERPHED
®
(ephedrine sulfate) Injection
50 mg/10 mL (5 mg/mL)
(equivalent to 38 mg/10 mL ephedrine base)
For Intravenous Use Only
Rx Only
Premixed formulation -
Do not dilute
10 x 10 mL Single-dose Vials
NEXUS
PHARMACEUTICALS
10. Overdosage
Overdose of EMERPHED can cause a rapid rise in blood pressure. In the case of an overdose, careful monitoring of blood pressure is recommended. If blood pressure continues to rise to an unacceptable level, parenteral antihypertensive agents can be administered at the discretion of the clinician.
11. Description
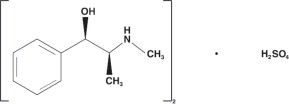
EMERPHED (ephedrine sulfate) injection is a clear, colorless, sterile solution for intravenous injection. The chemical name of ephedrine sulfate is benzenemethanol, α-[1-(methylamino)ethyl]-, [R-(R*,S*)]-, sulfate (2:1) (salt), and the molecular weight is 428.5 g/mol. Its structural formula is depicted below:
Ephedrine sulfate is freely soluble in water and ethanol, very slightly soluble in chloroform, and practically insoluble in ether. Each mL contains ephedrine sulfate, USP 5 mg (equivalent to 3.8 mg ephedrine base), 0.9% sodium chloride, USP in water for injection. The pH range is 4.5 to 7.0.
8.4 Pediatric Use
Safety and effectiveness of EMERPHED in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ephedrine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
14. Clinical Studies
The evidence for the efficacy of EMERPHED (ephedrine sulfate) injection is derived from the published literature. Increases in blood pressure following administration of ephedrine were observed in 14 studies, including 9 where ephedrine was used in pregnant women undergoing neuraxial anesthesia during Cesarean delivery, 1 study in non-obstetric surgery under neuraxial anesthesia, and 4 studies in patients undergoing surgery under general anesthesia. Ephedrine has been shown to raise systolic and mean blood pressure when administered as a bolus dose following the development of hypotension during anesthesia.
4. Contraindications
None
6. Adverse Reactions
The following adverse reactions associated with the use of ephedrine sulfate were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure.
Gastrointestinal disorders: Nausea, vomiting
Cardiac disorders: Tachycardia, palpitations (thumping heart), reactive hypertension, bradycardia, ventricular ectopics, R-R variability
Nervous system disorders: Dizziness
Psychiatric disorders: Restlessness
7. Drug Interactions
| Interactions that Augment the Press or Effect | |
| Oxytocin and oxytocic drugs | |
| Clinical Impact: | Serious postpartum hypertension has been described in patients who received both a vasopressor (i.e., methoxamine, phenylephrine, ephedrine) and an oxytocic (i.e., methylergonovine, ergonovine). Some of these patients experienced a stroke. |
| Intervention: | Carefully monitor the blood pressure of individuals who have received both EMERPHED and an oxytocic. |
| Clonidine, propofol, monoamine oxidase inhibitors (MAOIs ), atropine | |
| Clinical Impact: | These drugs augment the pressor effect of ephedrine. |
| Intervention: | Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. |
| Interactions that Antagonize the Pressor Effect | |
| Clinical Impact: | These drugs antagonize the pressor effect of ephedrine. |
| Intervention: | Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. |
| Examples: | α-adrenergic antagonists, β-adrenergic receptor antagonists, reserpine, quinidine, mephentermine |
| Other Drug Interactions | |
| Guanethidine | |
| Clinical Impact: | EMERPHED may inhibit the neuron blockage produced by guanethidine, resulting in loss of antihypertensive effectiveness. |
| Intervention: | Clinician should monitor patient for blood pressor response and adjust the dosage or choice of pressor accordingly. |
| Rocuronium | |
| Clinical Impact: | EMERPHED may reduce the onset time of neuromuscular blockade when used for intubation with rocuronium if administered simultaneously with anesthetic induction. |
| Intervention: | Be aware of this potential interaction. No treatment or other interventions are needed. |
| Epidural anesthesia | |
| Clinical Impact: | EMERPHED may decrease the efficacy of epidural blockade by hastening the regression of sensory analgesia. |
| Intervention: | Monitor and treat the patient according to clinical practice. |
| Theophylline | |
| Clinical Impact: | Concomitant use of EMERPHED may increase the frequency of nausea, nervousness, and insomnia. |
| Intervention: | Monitor patient for worsening symptoms and manage symptoms according to clinical practice. |
| Cardiac glycosides | |
| Clinical Impact: | Giving EMERPHED with a cardiac glycoside, such as digitalis, may increase the possibility of arrhythmias. |
| Intervention: | Carefully monitor patients on cardiac glycosides who are also administered ephedrine. |
8.6 Renal Impairment
Ephedrine and its metabolite are excreted in urine. In patients with renal impairment, excretion of ephedrine is likely to be affected with a corresponding increase in elimination half-life, which will lead to slow elimination of ephedrine and consequently prolonged pharmacological effect and potentially adverse reactions. Monitor patients with renal impairment carefully after the initial bolus dose for adverse events.
12.2 Pharmacodynamics
Ephedrine stimulates heart rate and cardiac output and variably increases peripheral resistance; as a result, ephedrine usually increases blood pressure. Stimulation of the α-adrenergic receptors of smooth muscle cells in the bladder base may increase the resistance to the outflow of urine. Activation of β- adrenergic receptors in the lungs promotes bronchodilation.
The overall cardiovascular effect from ephedrine is the result of a balance among α-1 adrenoceptor- mediated vasoconstriction, β-2 adrenoceptor- mediated vasoconstriction, and β-2 adrenoceptor-mediated vasodilatation. Stimulation of the β-1 adrenoceptors results in positive inotrope and chronotrope action.
Tachyphylaxis to the pressor effects of ephedrine may occur with repeated administration [see Warnings and Precautions 5.2 ].
12.3 Pharmacokinetics
Publications studying pharmacokinetics of oral administration of (-)-ephedrine support that (-)-ephedrine is metabolized into norephedrine. However, the metabolism pathway is unknown. Both the parent drug and the metabolite are excreted in urine. Limited data after IV administration of ephedrine support similar observations of urinary excretion of drug and metabolite. The plasma elimination half-life of ephedrine following oral administration was about 6 hours.
Ephedrine crosses the placental barrier [see Use in Specific Populations 8.1].
1. Indications and Usage
EMERPHED is indicated for the treatment of clinically important hypotension occurring in the setting of anesthesia.
12.1 Mechanism of Action
Ephedrine sulfate is a sympathomimetic amine that directly acts as an agonist at α- and β-adrenergic receptors and indirectly causes the release of norepinephrine from sympathetic neurons. Pressor effects by direct alpha- and beta-adrenergic receptor activation are mediated by increases in arterial pressures, cardiac output, and peripheral resistance. Indirect adrenergic stimulation is caused by norepinephrine release from sympathetic nerves.
5. Warnings and Precautions
2. Dosage and Administration
3. Dosage Forms and Strengths
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base).
5.2 Tolerance and Tachyphylaxis
Data indicate that repeated administration of ephedrine can result in tachyphylaxis. Be aware of this possibility when treating anesthesia-induced hypotension with EMERPHED and be prepared with an alternative pressor to mitigate unacceptable responsiveness.
16. How Supplied/storage and Handling
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base) and is supplied as follows:
| NDC | Presentation |
| 14789-250-07 | 10 mL clear glass, single- dose vial; strength 5 mg/mL |
| 14789-250-10 | 10 mL vials packaged in a carton of 10 |
EMERPHED (ephedrine sulfate) injection 50 mg/10 mL (5 mg/mL) is not made with natural rubber latex.
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Store in carton until time of use. For single dose only. Discard unused portion.
Patent, www.emerphed.com/patent
Manufactured in Italy for:
Nexus Pharmaceuticals, Inc.
400 Knightsbridge Parkway
Lincolnshire, IL 60069
USA
EPHPIR003
2.1 General Dosage and Administration Instructions
- This is a premixed formulation. Do not dilute prior to use.
- Discard any unused portion of EMERPHED.
- Inspect parenteral drug products visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
EMERPHED is a clear, colorless solution. Do not use if the solution is not clear or if particulate matter is present.
5.1 Pressor Effect With Concomitant Oxytocic Drugs
Serious postpartum hypertension has been described in patients who received both a vasopressor (i.e., methoxamine, phenylephrine, ephedrine) and an oxytocic (i.e., methylergonovine, ergonovine) [see Drug Interactions (7)]. Some of these patients experienced a stroke. Carefully monitor the blood pressure of individuals who have received both EMERPHED and an oxytocic.
5.3 Risk of Hypertension When Used Prophylactically
When used to prevent hypotension, ephedrine has been associated with an increased incidence of hypertension compared with when ephedrine is used to treat hypotension.
2.2 Dosing for the Treatment of Clinically Important Hypotension in the Setting of Anesthesia
The recommended dosages for the treatment of clinically important hypotension in the setting of anesthesia is an initial dose of 5 mg to 10 mg administered by intravenous bolus. Administer additional boluses as needed, not to exceed a total dosage of 50 mg.
- Adjust dosage according to the blood pressure goal (i.e., titrate to effect).
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
Available data from randomized studies, case series, and reports of ephedrine sulfate use in pregnant women have not identified a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. However, there are clinical considerations [see Clinical Considerations ]. In animal reproduction studies, decreased fetal survival and fetal body weights were observed in the presence of maternal toxicity after normotensive pregnant rats were administered 60 mg/kg intravenous ephedrine sulfate (12 times the maximum recommended human dose (MRHD) of 50 mg/day). No malformations or embryofetal adverse effects were observed when pregnant rats or rabbits were treated with intravenous bolus doses of ephedrine sulfate during organogenesis at doses 1.9 and 7.7 times the MRHD, respectively [See data ].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 51945-4 (51945-4)
Principal Display Panel – 5 mg/mL Carton Label
NDC 14789-250-10
EMERPHED
®
(ephedrine sulfate) Injection
50 mg/10 mL (5 mg/mL)
(equivalent to 38 mg/10 mL ephedrine base)
For Intravenous Use Only
Rx Only
Premixed formulation -
Do not dilute
10 x 10 mL Single-dose Vials
NEXUS
PHARMACEUTICALS
10. Overdosage (10. OVERDOSAGE)
Overdose of EMERPHED can cause a rapid rise in blood pressure. In the case of an overdose, careful monitoring of blood pressure is recommended. If blood pressure continues to rise to an unacceptable level, parenteral antihypertensive agents can be administered at the discretion of the clinician.
11. Description (11. DESCRIPTION)
EMERPHED (ephedrine sulfate) injection is a clear, colorless, sterile solution for intravenous injection. The chemical name of ephedrine sulfate is benzenemethanol, α-[1-(methylamino)ethyl]-, [R-(R*,S*)]-, sulfate (2:1) (salt), and the molecular weight is 428.5 g/mol. Its structural formula is depicted below:
Ephedrine sulfate is freely soluble in water and ethanol, very slightly soluble in chloroform, and practically insoluble in ether. Each mL contains ephedrine sulfate, USP 5 mg (equivalent to 3.8 mg ephedrine base), 0.9% sodium chloride, USP in water for injection. The pH range is 4.5 to 7.0.
8.4 Pediatric Use
Safety and effectiveness of EMERPHED in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ephedrine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
14. Clinical Studies (14. CLINICAL STUDIES)
The evidence for the efficacy of EMERPHED (ephedrine sulfate) injection is derived from the published literature. Increases in blood pressure following administration of ephedrine were observed in 14 studies, including 9 where ephedrine was used in pregnant women undergoing neuraxial anesthesia during Cesarean delivery, 1 study in non-obstetric surgery under neuraxial anesthesia, and 4 studies in patients undergoing surgery under general anesthesia. Ephedrine has been shown to raise systolic and mean blood pressure when administered as a bolus dose following the development of hypotension during anesthesia.
4. Contraindications (4. CONTRAINDICATIONS)
None
6. Adverse Reactions (6. ADVERSE REACTIONS)
The following adverse reactions associated with the use of ephedrine sulfate were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure.
Gastrointestinal disorders: Nausea, vomiting
Cardiac disorders: Tachycardia, palpitations (thumping heart), reactive hypertension, bradycardia, ventricular ectopics, R-R variability
Nervous system disorders: Dizziness
Psychiatric disorders: Restlessness
7. Drug Interactions (7. DRUG INTERACTIONS)
| Interactions that Augment the Press or Effect | |
| Oxytocin and oxytocic drugs | |
| Clinical Impact: | Serious postpartum hypertension has been described in patients who received both a vasopressor (i.e., methoxamine, phenylephrine, ephedrine) and an oxytocic (i.e., methylergonovine, ergonovine). Some of these patients experienced a stroke. |
| Intervention: | Carefully monitor the blood pressure of individuals who have received both EMERPHED and an oxytocic. |
| Clonidine, propofol, monoamine oxidase inhibitors (MAOIs ), atropine | |
| Clinical Impact: | These drugs augment the pressor effect of ephedrine. |
| Intervention: | Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. |
| Interactions that Antagonize the Pressor Effect | |
| Clinical Impact: | These drugs antagonize the pressor effect of ephedrine. |
| Intervention: | Carefully monitor the blood pressure of individuals who have received both EMERPHED and any of these drugs. |
| Examples: | α-adrenergic antagonists, β-adrenergic receptor antagonists, reserpine, quinidine, mephentermine |
| Other Drug Interactions | |
| Guanethidine | |
| Clinical Impact: | EMERPHED may inhibit the neuron blockage produced by guanethidine, resulting in loss of antihypertensive effectiveness. |
| Intervention: | Clinician should monitor patient for blood pressor response and adjust the dosage or choice of pressor accordingly. |
| Rocuronium | |
| Clinical Impact: | EMERPHED may reduce the onset time of neuromuscular blockade when used for intubation with rocuronium if administered simultaneously with anesthetic induction. |
| Intervention: | Be aware of this potential interaction. No treatment or other interventions are needed. |
| Epidural anesthesia | |
| Clinical Impact: | EMERPHED may decrease the efficacy of epidural blockade by hastening the regression of sensory analgesia. |
| Intervention: | Monitor and treat the patient according to clinical practice. |
| Theophylline | |
| Clinical Impact: | Concomitant use of EMERPHED may increase the frequency of nausea, nervousness, and insomnia. |
| Intervention: | Monitor patient for worsening symptoms and manage symptoms according to clinical practice. |
| Cardiac glycosides | |
| Clinical Impact: | Giving EMERPHED with a cardiac glycoside, such as digitalis, may increase the possibility of arrhythmias. |
| Intervention: | Carefully monitor patients on cardiac glycosides who are also administered ephedrine. |
8.6 Renal Impairment
Ephedrine and its metabolite are excreted in urine. In patients with renal impairment, excretion of ephedrine is likely to be affected with a corresponding increase in elimination half-life, which will lead to slow elimination of ephedrine and consequently prolonged pharmacological effect and potentially adverse reactions. Monitor patients with renal impairment carefully after the initial bolus dose for adverse events.
12.2 Pharmacodynamics
Ephedrine stimulates heart rate and cardiac output and variably increases peripheral resistance; as a result, ephedrine usually increases blood pressure. Stimulation of the α-adrenergic receptors of smooth muscle cells in the bladder base may increase the resistance to the outflow of urine. Activation of β- adrenergic receptors in the lungs promotes bronchodilation.
The overall cardiovascular effect from ephedrine is the result of a balance among α-1 adrenoceptor- mediated vasoconstriction, β-2 adrenoceptor- mediated vasoconstriction, and β-2 adrenoceptor-mediated vasodilatation. Stimulation of the β-1 adrenoceptors results in positive inotrope and chronotrope action.
Tachyphylaxis to the pressor effects of ephedrine may occur with repeated administration [see Warnings and Precautions 5.2 ].
12.3 Pharmacokinetics
Publications studying pharmacokinetics of oral administration of (-)-ephedrine support that (-)-ephedrine is metabolized into norephedrine. However, the metabolism pathway is unknown. Both the parent drug and the metabolite are excreted in urine. Limited data after IV administration of ephedrine support similar observations of urinary excretion of drug and metabolite. The plasma elimination half-life of ephedrine following oral administration was about 6 hours.
Ephedrine crosses the placental barrier [see Use in Specific Populations 8.1].
1. Indications and Usage (1. INDICATIONS AND USAGE)
EMERPHED is indicated for the treatment of clinically important hypotension occurring in the setting of anesthesia.
12.1 Mechanism of Action
Ephedrine sulfate is a sympathomimetic amine that directly acts as an agonist at α- and β-adrenergic receptors and indirectly causes the release of norepinephrine from sympathetic neurons. Pressor effects by direct alpha- and beta-adrenergic receptor activation are mediated by increases in arterial pressures, cardiac output, and peripheral resistance. Indirect adrenergic stimulation is caused by norepinephrine release from sympathetic nerves.
5. Warnings and Precautions (5. WARNINGS AND PRECAUTIONS)
2. Dosage and Administration (2. DOSAGE AND ADMINISTRATION)
3. Dosage Forms and Strengths (3. DOSAGE FORMS AND STRENGTHS)
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base).
5.2 Tolerance and Tachyphylaxis
Data indicate that repeated administration of ephedrine can result in tachyphylaxis. Be aware of this possibility when treating anesthesia-induced hypotension with EMERPHED and be prepared with an alternative pressor to mitigate unacceptable responsiveness.
16. How Supplied/storage and Handling (16. HOW SUPPLIED/STORAGE AND HANDLING)
EMERPHED (ephedrine sulfate) injection is a clear, colorless solution available as a single-dose vial that contains 50 mg/10 mL ephedrine sulfate, equivalent to 38 mg/10 mL ephedrine base (5 mg/mL ephedrine sulfate, equivalent to 3.8 mg/mL ephedrine base) and is supplied as follows:
| NDC | Presentation |
| 14789-250-07 | 10 mL clear glass, single- dose vial; strength 5 mg/mL |
| 14789-250-10 | 10 mL vials packaged in a carton of 10 |
EMERPHED (ephedrine sulfate) injection 50 mg/10 mL (5 mg/mL) is not made with natural rubber latex.
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Store in carton until time of use. For single dose only. Discard unused portion.
Patent, www.emerphed.com/patent
Manufactured in Italy for:
Nexus Pharmaceuticals, Inc.
400 Knightsbridge Parkway
Lincolnshire, IL 60069
USA
EPHPIR003
2.1 General Dosage and Administration Instructions
- This is a premixed formulation. Do not dilute prior to use.
- Discard any unused portion of EMERPHED.
- Inspect parenteral drug products visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
EMERPHED is a clear, colorless solution. Do not use if the solution is not clear or if particulate matter is present.
5.1 Pressor Effect With Concomitant Oxytocic Drugs (5.1 Pressor Effect with Concomitant Oxytocic Drugs)
Serious postpartum hypertension has been described in patients who received both a vasopressor (i.e., methoxamine, phenylephrine, ephedrine) and an oxytocic (i.e., methylergonovine, ergonovine) [see Drug Interactions (7)]. Some of these patients experienced a stroke. Carefully monitor the blood pressure of individuals who have received both EMERPHED and an oxytocic.
5.3 Risk of Hypertension When Used Prophylactically
When used to prevent hypotension, ephedrine has been associated with an increased incidence of hypertension compared with when ephedrine is used to treat hypotension.
2.2 Dosing for the Treatment of Clinically Important Hypotension in the Setting of Anesthesia
The recommended dosages for the treatment of clinically important hypotension in the setting of anesthesia is an initial dose of 5 mg to 10 mg administered by intravenous bolus. Administer additional boluses as needed, not to exceed a total dosage of 50 mg.
- Adjust dosage according to the blood pressure goal (i.e., titrate to effect).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:16.651096 · Updated: 2026-03-14T21:49:36.542485