Dulcolax Stimulant Laxative
e0dcae0f-6dca-4700-a415-de2274e6f779
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dulcolax Stimulant Laxative Tablets Drug Facts
Purpose
Stimulant laxative
Medication Information
Purpose
Stimulant laxative
Description
Dulcolax Stimulant Laxative Tablets Drug Facts
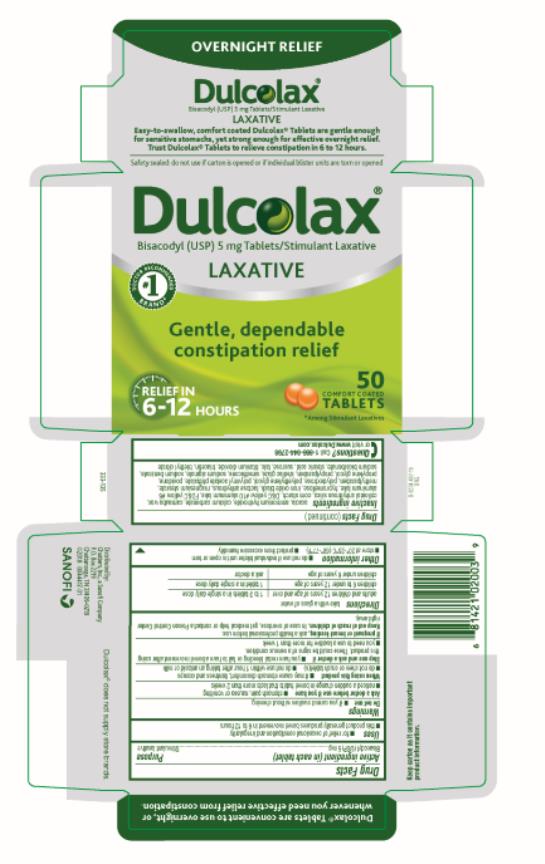
Use
● for relief of occasional constipation and irregularity
● this product generally produces bowel movement in 6 to 12 hours
Prin
Dulcolax
LAXATIVE
50 TABLETS
Section 42229-5
Dulcolax Stimulant Laxative Tablets
Drug Facts
Directions
| take with a glass of water | |
| adults and children 12 years of age and over | 1 to 3 tablets in a single daily dose |
| children 6 to under 12 years of age | 1 tablet in a single daily dose |
| children under 6 years of age | ask a doctor |
Do Not Use
● if you cannot swallow without chewing
Questions?
Call 1- 866 - 844 - 2798 or visit www.Dulcolax.com
Keep carton as it contains important product information.
Dulcolax Stimulant Laxative Tablets
Drug Facts
If Pregnant Or B
ask a health professional before use.
Active Ingredient
Bisacodyl (USP) 5 mg
Other Information
● contains FD&C Yellow No. 6
● do not use if individual blister unit is open or torn
● store at 20°-25°C (68°-77°F)
● protect from excessive humidity
Inactive Ingredients
acacia senegal gum, ammonium hydroxide, calcium carbonate, carnauba wax, colloidal anhydrous silica, corn starch, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, iron oxides, lactose, magnesium stearate, methylparaben, polydextrose, polyethylene glycol, polyvinyl acetate phthalate, povidone, propylene glycol, propylparaben, shellac, simethicone, sodium alginate, sodium benzoate, sodium bicarbonate, stearic acid, sucrose, talc, titanium dioxide, triacetin, triethyl citrate
Principal Display Panel
Dulcolax
LAXATIVE
200 TABLETS
When Using This Product
● it may cause stomach discomfort, faintness and cramps
● do not chew or crush tablet(s)
● do not use within 1 hour after taking an antacid or milk
Stop Use and Ask A Doctor If
● you have rectal bleeding or fail to have a bowel movement after using this product. These could be signs of a serious condition.
● you need to use a laxative for more than 1 week
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet)
Bisacodyl (USP) 5 mg
Ask A Doctor Before Use If You Have
● stomach pain, nausea or vomiting
● noticed a sudden change in bowel habits that lasts more than 2 weeks
Structured Label Content
Use
● for relief of occasional constipation and irregularity
● this product generally produces bowel movement in 6 to 12 hours
Prin (PRIN)
Dulcolax
LAXATIVE
50 TABLETS
Section 42229-5 (42229-5)
Dulcolax Stimulant Laxative Tablets
Drug Facts
Purpose
Stimulant laxative
Directions
| take with a glass of water | |
| adults and children 12 years of age and over | 1 to 3 tablets in a single daily dose |
| children 6 to under 12 years of age | 1 tablet in a single daily dose |
| children under 6 years of age | ask a doctor |
Do Not Use (Do not use)
● if you cannot swallow without chewing
Questions?
Call 1- 866 - 844 - 2798 or visit www.Dulcolax.com
Keep carton as it contains important product information.
Dulcolax Stimulant Laxative Tablets
Drug Facts
If Pregnant Or B (If pregnant or b)
ask a health professional before use.
Active Ingredient (Active ingredient)
Bisacodyl (USP) 5 mg
Other Information (Other information)
● contains FD&C Yellow No. 6
● do not use if individual blister unit is open or torn
● store at 20°-25°C (68°-77°F)
● protect from excessive humidity
Inactive Ingredients (Inactive ingredients)
acacia senegal gum, ammonium hydroxide, calcium carbonate, carnauba wax, colloidal anhydrous silica, corn starch, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, iron oxides, lactose, magnesium stearate, methylparaben, polydextrose, polyethylene glycol, polyvinyl acetate phthalate, povidone, propylene glycol, propylparaben, shellac, simethicone, sodium alginate, sodium benzoate, sodium bicarbonate, stearic acid, sucrose, talc, titanium dioxide, triacetin, triethyl citrate
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Dulcolax
LAXATIVE
200 TABLETS
When Using This Product (When using this product)
● it may cause stomach discomfort, faintness and cramps
● do not chew or crush tablet(s)
● do not use within 1 hour after taking an antacid or milk
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
● you have rectal bleeding or fail to have a bowel movement after using this product. These could be signs of a serious condition.
● you need to use a laxative for more than 1 week
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Bisacodyl (USP) 5 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
● stomach pain, nausea or vomiting
● noticed a sudden change in bowel habits that lasts more than 2 weeks
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:40.942504 · Updated: 2026-03-14T23:08:14.770901