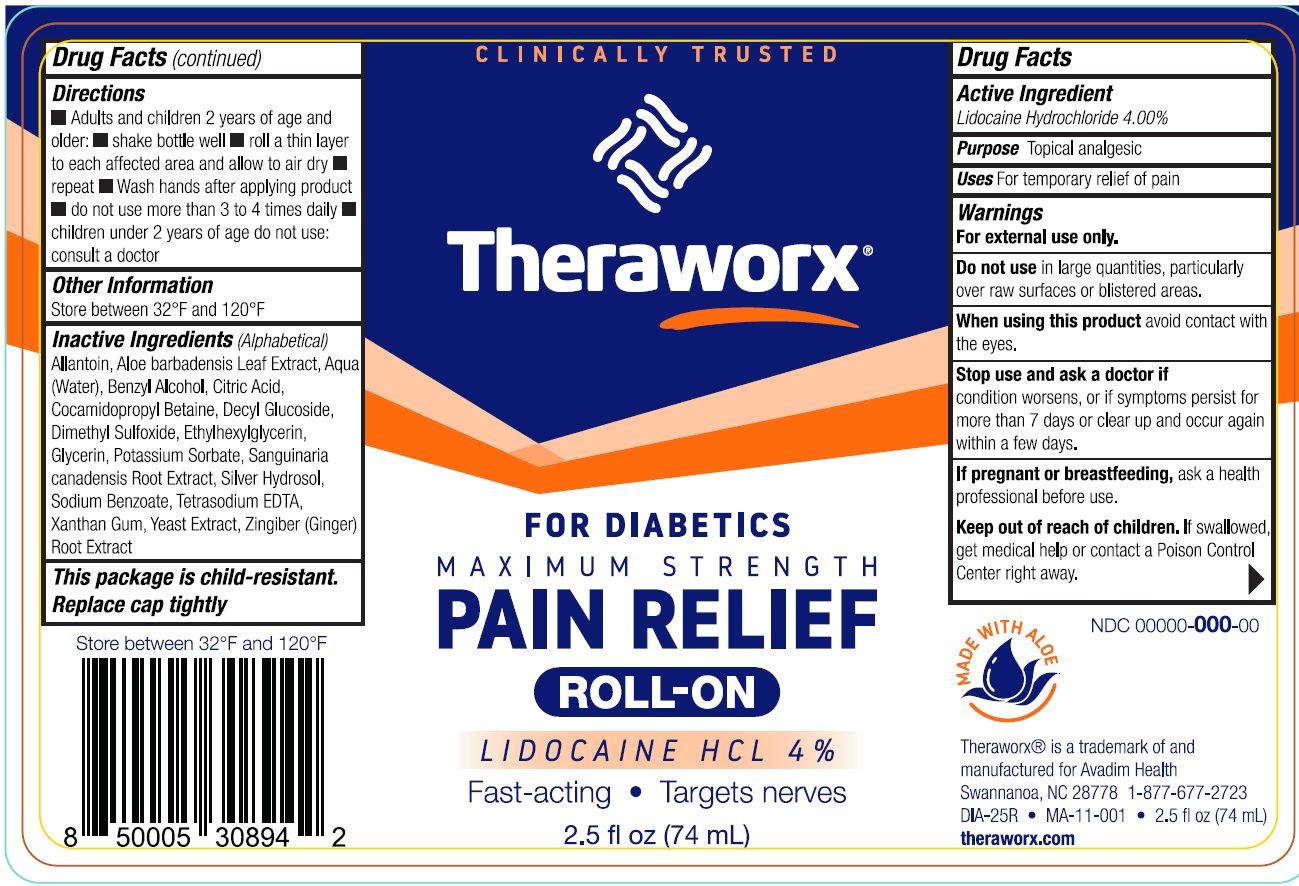
Theraworx Pain Relief Roll-on With Lidocaine For Diabetics
e07f5bf7-5a9a-468c-8c3f-2c6f0b17af29
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine Hydrochloride 4.00%
Purpose
Topical analgesic
Medication Information
Purpose
Topical analgesic
Description
Lidocaine Hydrochloride 4.00%
Uses
For temporary relief of pain
Warnings
For external use only.
Directions
- Adults and children 2 years of age and older:
- shake bottle well
- roll a thin layer to each affected area and allow to air dry
- repeat
- Wash hands after applying product
- do not use more than 3 to 4 times daily
- children under 2 years of age do not use: consult a doctor
Do Not Use
in large quantities, particularly over raw surfaces or blistered areas.
Drug Facts
Active Ingredient
Lidocaine Hydrochloride 4.00%
Other Information
Store between 32°F and 120°F
Package Labeling:
When Using This Product
avoid contact with the eyes.
Stop Use and Ask A Doctor If
condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days.
If Pregnant Or Breastfeeding,
ask a health professional before use.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Inactive Ingredients (alphabetical)
Allantoin, Aloe barbadensis Leaf Extract, Aqua (Water), Benzyl Alcohol, Citric Acid, Cocamidopropyl Betaine, Decyl Glucoside, Dimethyl Sulfoxide, Ethylhexylglycerin, Glycerin, Potassium Sorbate, Sanguinaria canadensis Root Extract, Silver Hydrosol, Sodium Benzoate, Tetrasodium EDTA, Xanthan Gum, Yeast Extract, Zingiber (Ginger) Root Extract
Structured Label Content
Uses
For temporary relief of pain
Purpose
Topical analgesic
Warnings
For external use only.
Directions
- Adults and children 2 years of age and older:
- shake bottle well
- roll a thin layer to each affected area and allow to air dry
- repeat
- Wash hands after applying product
- do not use more than 3 to 4 times daily
- children under 2 years of age do not use: consult a doctor
Do Not Use (Do not use)
in large quantities, particularly over raw surfaces or blistered areas.
Drug Facts
Active Ingredient
Lidocaine Hydrochloride 4.00%
Other Information
Store between 32°F and 120°F
Package Labeling:
When Using This Product (When using this product)
avoid contact with the eyes.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days.
If Pregnant Or Breastfeeding, (If pregnant or breastfeeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Inactive Ingredients (alphabetical) (Inactive Ingredients (Alphabetical))
Allantoin, Aloe barbadensis Leaf Extract, Aqua (Water), Benzyl Alcohol, Citric Acid, Cocamidopropyl Betaine, Decyl Glucoside, Dimethyl Sulfoxide, Ethylhexylglycerin, Glycerin, Potassium Sorbate, Sanguinaria canadensis Root Extract, Silver Hydrosol, Sodium Benzoate, Tetrasodium EDTA, Xanthan Gum, Yeast Extract, Zingiber (Ginger) Root Extract
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:08.578852 · Updated: 2026-03-14T23:03:01.087200