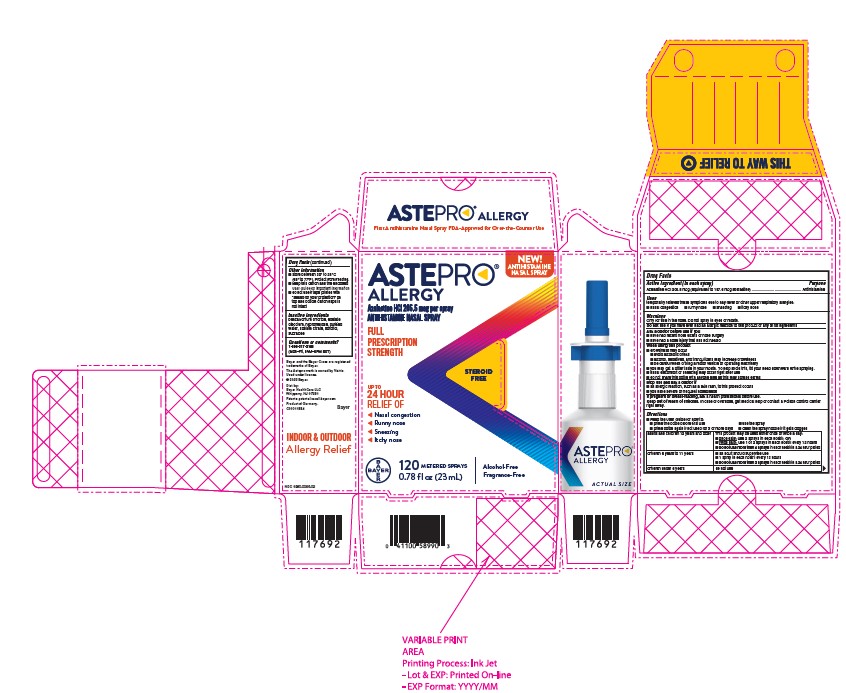
Astepro Allergy
e0640846-19a6-7f79-e053-2995a90a8176
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Warnings and Precautions
Warnings
Only for use in the nose. Do not spray in eyes or mouth.
Description
Drug Facts
Uses
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- nasal congestion
- runny nose
- sneezing
- itchy nose
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- an allergic reaction, such as a skin rash, to this product occurs
- you have severe or frequent nosebleeds
Section 50567-7
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- you may get a bitter taste in your mouth. To help avoid this, tilt your head downward while spraying.
- nasal discomfort or sneezing may occur right after use
- do not share this bottle with anyone else as this may spread germs
Section 50569-3
Ask a doctor before use if you
- have had recent nose ulcers or nose surgery
- have had a nose injury that has not healed
Section 50570-1
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Section 51727-6
Inactive ingredients
benzalkonium chloride, edetate disodium, hypromellose, purified water, sodium citrate, sorbitol, sucralose
Section 51945-4
Azelastine HCl 205.5 mcg per spray
ANTIHISTAMINE NASAL SPRAY
FULL PRESCRIPTION STRENGTH
STEROID FREE
UP TO 24 HOUR RELIEF OF
Nasal congestion
Runny nose
Sneezing
Itchy nose
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose
Antihistamine
Section 55106-9
Active ingredient
Azelastine HCl 205.5 mcg (equivalent to 187.6 mcg azelastine)
Directions
Read the User Guide for how to:
- prime the bottle before first use
- prime bottle again if not used for 3 or more days
- use the spray
- clean the spray nozzle if it gets clogged
| adults and children 12 years and older |
This product may be used either once or twice a day:
|
|
children 6 years to 11 years |
|
| children under 6 years | do not use |
Other Information
Other information
- store between 20° to 25° C (68° to 77° F). Protect from freezing.
- keep this carton and the enclosed User Guide for important information
- do not use if tape printed with “sealed for your protection” on top and bottom carton flaps is not intact
Questions Or Comments
Questions or comments?
1-800-317-2165 (Mon-Fri, 9AM-5PM EST)
Structured Label Content
Uses
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- nasal congestion
- runny nose
- sneezing
- itchy nose
Warnings and Precautions (34071-1)
Warnings
Only for use in the nose. Do not spray in eyes or mouth.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- an allergic reaction, such as a skin rash, to this product occurs
- you have severe or frequent nosebleeds
Section 50567-7 (50567-7)
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- you may get a bitter taste in your mouth. To help avoid this, tilt your head downward while spraying.
- nasal discomfort or sneezing may occur right after use
- do not share this bottle with anyone else as this may spread germs
Section 50569-3 (50569-3)
Ask a doctor before use if you
- have had recent nose ulcers or nose surgery
- have had a nose injury that has not healed
Section 50570-1 (50570-1)
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Section 51727-6 (51727-6)
Inactive ingredients
benzalkonium chloride, edetate disodium, hypromellose, purified water, sodium citrate, sorbitol, sucralose
Section 51945-4 (51945-4)
Azelastine HCl 205.5 mcg per spray
ANTIHISTAMINE NASAL SPRAY
FULL PRESCRIPTION STRENGTH
STEROID FREE
UP TO 24 HOUR RELIEF OF
Nasal congestion
Runny nose
Sneezing
Itchy nose
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Antihistamine
Section 55106-9 (55106-9)
Active ingredient
Azelastine HCl 205.5 mcg (equivalent to 187.6 mcg azelastine)
Directions
Read the User Guide for how to:
- prime the bottle before first use
- prime bottle again if not used for 3 or more days
- use the spray
- clean the spray nozzle if it gets clogged
| adults and children 12 years and older |
This product may be used either once or twice a day:
|
|
children 6 years to 11 years |
|
| children under 6 years | do not use |
Other Information (Other information)
Other information
- store between 20° to 25° C (68° to 77° F). Protect from freezing.
- keep this carton and the enclosed User Guide for important information
- do not use if tape printed with “sealed for your protection” on top and bottom carton flaps is not intact
Questions Or Comments (Questions or comments)
Questions or comments?
1-800-317-2165 (Mon-Fri, 9AM-5PM EST)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:29.056060 · Updated: 2026-03-14T23:11:00.469021