Fexofenadine Hcl Tablets Usp, 60 Mg And 180 Mg
e0537b71-fdee-4122-9c09-868ce7352080
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Fexofenadine HCl USP, 60 mg (for 60 mg) Fexofenadine HCl USP, 180 mg (for 180 mg)
Purpose
Antihistamine
Medication Information
Purpose
Antihistamine
Description
Fexofenadine HCl USP, 60 mg (for 60 mg) Fexofenadine HCl USP, 180 mg (for 180 mg)
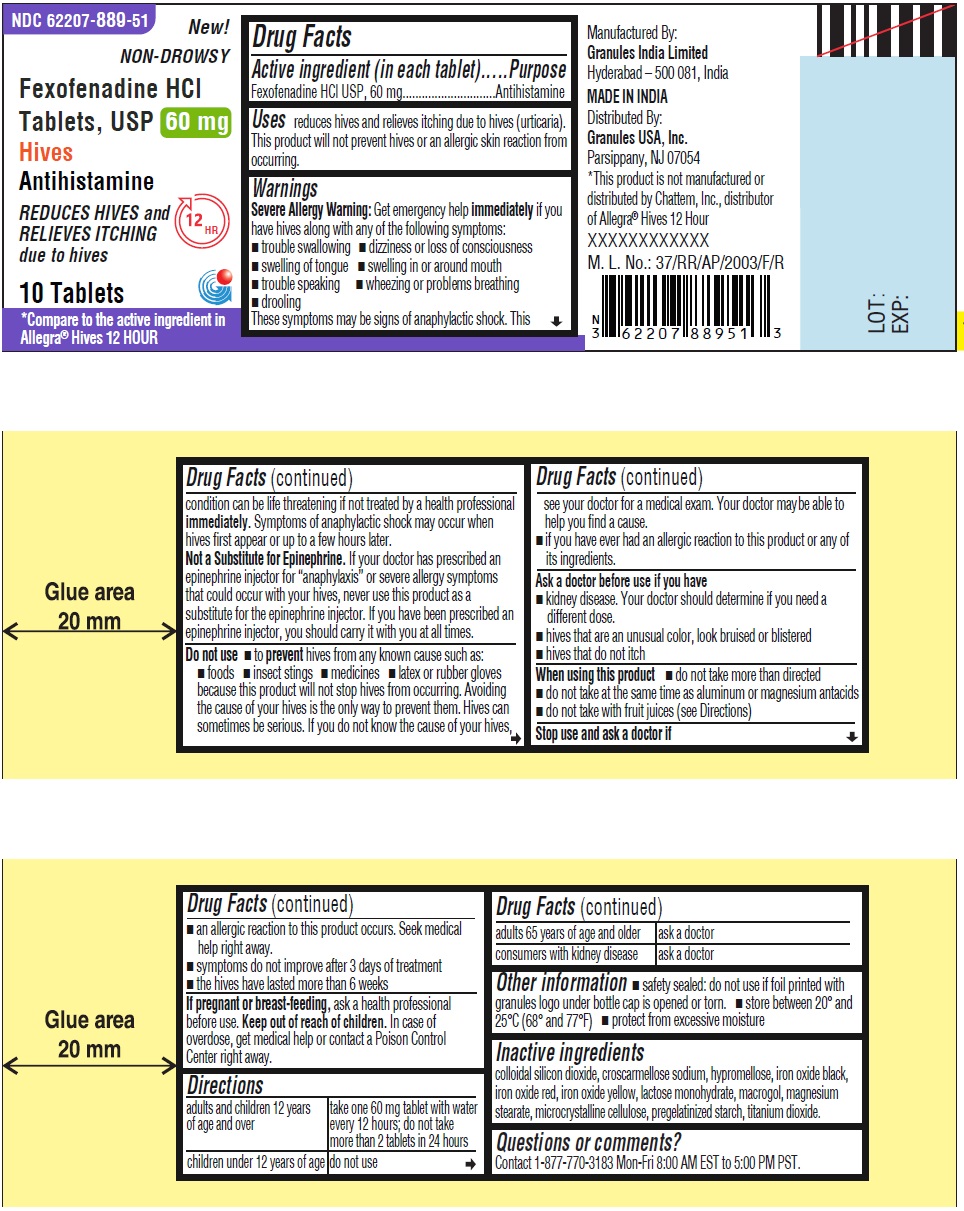
Uses
reduces hives and relieves itching due to hives (urticaria). This product will not prevent hives or an allergic skin reaction from occurring.
Warnings
Severe Allergy Warning:Get emergency help immediatelyif you have hives along with any of the following symptoms:
- trouble swallowing
- dizziness or loss of consciousness
- swelling of tongue
- swelling in or around mouth
- trouble speaking
- wheezing or problems breathing
- drooling
These symptoms may be signs of anaphylactic shock. This condition can be life threatening if not treated by a health professional
immediately. Symptoms of anaphylactic shock may occur when hives first appear or up to a few hours later.
Not a Substitute for Epinephrine.If your doctor has prescribed an epinephrine injector for “anaphylaxis” or severe allergy symptoms that could occur with your hives, never use this product as a substitute for the epinephrine injector. If you have been prescribed an epinephrine injector, you should carry it with you at all times.
Directions
| adults and children 12 years of age and over | take one 60 mg tablet with water every 12 hours; do not take more than 2 tablets in 24 hours (for 60 mg)
take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours (for 180 mg) |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Do Not Use
- to preventhives from any known cause such as:
- foods
- insect stings
- medicines
- latex or rubber gloves
because this product will not stop hives from occurring. Avoiding the cause of your hives is the only way to prevent them. Hives can sometimes be serious. If you do not know the cause of your hives, see your doctor for a medical exam. Your doctor may be able to help you find a cause.
- if you have ever had an allergic reaction to this product or any of its ingredients
Other Information
- safety sealed: do not use if foil printed with granules logo under bottle cap is opened or torn.
- store between 20º and 25ºC (68º and 77ºF)
- protect from excessive moisture
Inactive Ingredients
colloidal silicon dioxide, croscarmellose sodium, hypromellose, iron oxide black, iron oxide red, iron oxide yellow, lactose monohydrate, macrogol, magnesium stearate, microcrystalline cellulose, pregelatinized starch, titanium dioxide.
Questions Or Comments?
Contact 1-877-770-3183 Mon-Fri 8:00 AM EST to 5:00 PM PST
Manufactured By:
Granules India Limited
Hyderabad-500 081, India
MADE IN INDIA
Distributed By:
Granules USA, Inc.
Parsippany, NJ 07054
When Using This Product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Stop Use and Ask A Doctor If
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve after 3 days of treatment
- the hives have lasted more than 6 weeks
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet)
Fexofenadine HCl USP, 60 mg (for 60 mg)
Fexofenadine HCl USP, 180 mg (for 180 mg)
Ask A Doctor Before Use If You Have
- kidney disease. Your doctor should determine if you need a different dose.
- hives that are an unusual color, look bruised or blistered
- hives that do not itch
Principal Display Panel 60 Mg Blister Carton
NDC 62207-889-60
New!
NON-DROWSY
Fexofenadine HCl Tablets USP, 60 mg
Hives
Antihistamine
REDUCES HIVES and 12 HR
RELIEVES ITCHING
due to hives
72 (8 x 9) Tablets
*Compare to the active ingredient in Allegra ® Hives 12 HOUR
Principal Display Panel 180 Mg Blister Carton
NDC 62207-891-58 New!
NON-DROWSY
Fexofenadine HCl Tablets USP, 180 mg
Hives
Antihistamine
REDUCES HIVES and 24 HR
RELIEVES ITCHING
due to hives
70 (7 x 10) Tablets
*Compare to the active ingredient in Allegra ® Hives 24 HOUR
Principal Display Panel 60 Mg 10s Container Label
NDC 62207-889-51 New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP 60 mg
Hives
Antihistamine
REDUCES HIVES and 12 HR
RELIEVES ITCHING
due to hives
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 12 HOUR
Principal Display Panel 180 Mg 10s Container Label
NDC 62207-891-51 New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP 180 mg
Hives
Antihistamine
REDUCES HIVES and 24 HR
RELIEVES ITCHING
due to hives
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 24 HOUR
Principal Display Panel 60 Mg 10s Container Carton
NDC 62207-889-51
New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP
60 mg
Hives
Antihistamine
REDUCES HIVES and
RELIEVES ITCHING
due to hives
12 HR
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 12 HOUR
Principal Display Panel 180 Mg 10s Container Carton
NDC 62207-891-51
New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP
180 mg
Hives
Antihistamine
REDUCES HIVES and 24 HR
RELIEVES ITCHING
due to hives
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 24 HOUR
Structured Label Content
Uses
reduces hives and relieves itching due to hives (urticaria). This product will not prevent hives or an allergic skin reaction from occurring.
Purpose
Antihistamine
Warnings
Severe Allergy Warning:Get emergency help immediatelyif you have hives along with any of the following symptoms:
- trouble swallowing
- dizziness or loss of consciousness
- swelling of tongue
- swelling in or around mouth
- trouble speaking
- wheezing or problems breathing
- drooling
These symptoms may be signs of anaphylactic shock. This condition can be life threatening if not treated by a health professional
immediately. Symptoms of anaphylactic shock may occur when hives first appear or up to a few hours later.
Not a Substitute for Epinephrine.If your doctor has prescribed an epinephrine injector for “anaphylaxis” or severe allergy symptoms that could occur with your hives, never use this product as a substitute for the epinephrine injector. If you have been prescribed an epinephrine injector, you should carry it with you at all times.
Directions
| adults and children 12 years of age and over | take one 60 mg tablet with water every 12 hours; do not take more than 2 tablets in 24 hours (for 60 mg)
take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours (for 180 mg) |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Do Not Use (Do not use)
- to preventhives from any known cause such as:
- foods
- insect stings
- medicines
- latex or rubber gloves
because this product will not stop hives from occurring. Avoiding the cause of your hives is the only way to prevent them. Hives can sometimes be serious. If you do not know the cause of your hives, see your doctor for a medical exam. Your doctor may be able to help you find a cause.
- if you have ever had an allergic reaction to this product or any of its ingredients
Other Information (Other information)
- safety sealed: do not use if foil printed with granules logo under bottle cap is opened or torn.
- store between 20º and 25ºC (68º and 77ºF)
- protect from excessive moisture
Inactive Ingredients (Inactive ingredients)
colloidal silicon dioxide, croscarmellose sodium, hypromellose, iron oxide black, iron oxide red, iron oxide yellow, lactose monohydrate, macrogol, magnesium stearate, microcrystalline cellulose, pregelatinized starch, titanium dioxide.
Questions Or Comments? (Questions or comments?)
Contact 1-877-770-3183 Mon-Fri 8:00 AM EST to 5:00 PM PST
Manufactured By:
Granules India Limited
Hyderabad-500 081, India
MADE IN INDIA
Distributed By:
Granules USA, Inc.
Parsippany, NJ 07054
When Using This Product (When using this product)
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve after 3 days of treatment
- the hives have lasted more than 6 weeks
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Fexofenadine HCl USP, 60 mg (for 60 mg)
Fexofenadine HCl USP, 180 mg (for 180 mg)
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- kidney disease. Your doctor should determine if you need a different dose.
- hives that are an unusual color, look bruised or blistered
- hives that do not itch
Principal Display Panel 60 Mg Blister Carton (PRINCIPAL DISPLAY PANEL 60 mg - Blister Carton)
NDC 62207-889-60
New!
NON-DROWSY
Fexofenadine HCl Tablets USP, 60 mg
Hives
Antihistamine
REDUCES HIVES and 12 HR
RELIEVES ITCHING
due to hives
72 (8 x 9) Tablets
*Compare to the active ingredient in Allegra ® Hives 12 HOUR
Principal Display Panel 180 Mg Blister Carton (PRINCIPAL DISPLAY PANEL 180 mg - Blister Carton)
NDC 62207-891-58 New!
NON-DROWSY
Fexofenadine HCl Tablets USP, 180 mg
Hives
Antihistamine
REDUCES HIVES and 24 HR
RELIEVES ITCHING
due to hives
70 (7 x 10) Tablets
*Compare to the active ingredient in Allegra ® Hives 24 HOUR
Principal Display Panel 60 Mg 10s Container Label (PRINCIPAL DISPLAY PANEL 60 mg - 10s Container Label)
NDC 62207-889-51 New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP 60 mg
Hives
Antihistamine
REDUCES HIVES and 12 HR
RELIEVES ITCHING
due to hives
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 12 HOUR
Principal Display Panel 180 Mg 10s Container Label (PRINCIPAL DISPLAY PANEL 180 mg - 10s Container Label)
NDC 62207-891-51 New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP 180 mg
Hives
Antihistamine
REDUCES HIVES and 24 HR
RELIEVES ITCHING
due to hives
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 24 HOUR
Principal Display Panel 60 Mg 10s Container Carton (PRINCIPAL DISPLAY PANEL 60 mg - 10s Container Carton)
NDC 62207-889-51
New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP
60 mg
Hives
Antihistamine
REDUCES HIVES and
RELIEVES ITCHING
due to hives
12 HR
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 12 HOUR
Principal Display Panel 180 Mg 10s Container Carton (PRINCIPAL DISPLAY PANEL 180 mg - 10s Container Carton)
NDC 62207-891-51
New!
NON-DROWSY
Fexofenadine HCl
Tablets, USP
180 mg
Hives
Antihistamine
REDUCES HIVES and 24 HR
RELIEVES ITCHING
due to hives
10 Tablets
*Compare to the active ingredient in
Allegra ® Hives 24 HOUR
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:25.797467 · Updated: 2026-03-14T23:10:50.302579