These Highlights Do Not Include All The Information Needed To Use Adefovir Dipivoxil Tablets Safely And Effectively. See Full Prescribing Information For Adefovir Dipivoxil Tablets.
e047f3b2-feae-4c5e-9d07-1fefb4c0ec25
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Severe acute exacerbations of hepatitis have been reported in patients who have discontinued anti-Hepatitis B therapy including Adefovir Dipivoxil Tablets. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-Hepatitis B therapy. If appropriate, resumption of anti-Hepatitis B therapy may be warranted [See Warnings and Precautions (5.1) ] . In patients at risk of or having underlying renal dysfunction, chronic administration of Adefovir Dipivoxil Tablets may result in nephrotoxicity. These patients should be monitored closely for renal function and may require dose adjustment [See Warnings and Precautions (5.2) and Dosage and Administration (2.2) ] . HIV resistance may emerge in chronic hepatitis B patients with unrecognized or untreated Human Immunodeficiency Virus (HIV) infection treated with antihepatitis B therapies, such as therapy with Adefovir Dipivoxil Tablets, that may have activity against HIV [See Warnings and Precautions (5.3) ] . Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with other antiretrovirals [See Warnings and Precautions (5 . 4 ) ] .
Indications and Usage
Adefovir Dipivoxil Tablets are indicated for the treatment of chronic hepatitis B in patients 12 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease. This indication is based on histological, virological, biochemical, and serological responses in adult patients with HBeAg+ and HBeAg- chronic hepatitis B with compensated liver function, and with clinical evidence of lamivudine-resistant hepatitis B virus with either compensated or decompensated liver function. For patients 12 to less than 18 years of age, the indication is based on virological and biochemical responses in patients with HBeAg+ chronic hepatitis B virus infection with compensated liver function.
Dosage and Administration
One tablet containing 10 mg adefovir dipivoxil once daily orally with or without food. ( 2.1 ) Dose adjustment in renal impairment for adults ( 2.2 ) Creatinine Clearance (mL/min) a Hemodialysis Patients Greater than or equal to 50 30 - 49 10 - 29 Recommended dose and dosing interval 10 mg every 24 hours 10 mg every 48 hours 10 mg every 72 hours 10 mg every 7 days following dialysis a Creatinine clearance calculated by Cockcroft-Gault method using lean or ideal body weight. No dose recommendations for ( 2.1 ): Non-hemodialysis patients with creatinine clearance less than 10 mL per minute. Adolescent patients with renal impairment.
Warnings and Precautions
Severe acute exacerbations of hepatitis: Monitor hepatic function closely at repeated intervals for at least several months in patients who discontinue Adefovir Dipivoxil Tablets. ( 5.1 ) Nephrotoxicity: Monitor renal function during therapy for all patients, particularly those with pre-existing or other risks for renal impairment. Dose adjustment may be required. ( 5.2 ) HIV Resistance: Offer HIV testing to all patients prior to initiating Adefovir Dipivoxil Tablets. Untreated HIV may result in HIV resistance. ( 5.3 ) Lactic acidosis and severe hepatomegaly with steatosis: If suspected, suspend treatment. ( 5.4 ) Coadministration with Other Products: Do not administer Adefovir Dipivoxil Tablets concurrently with VIREAD ® or other tenofovir-containing products. ( 5.5 ) Clinical Resistance: For patients with lamivudine-resistant HBV use adefovir dipivoxil in combination with lamivudine. For all patients, consider modifying treatment in case serum HBV DNA remains above 1000 copies/mL with continued treatment. ( 5.6 )
Contraindications
Adefovir Dipivoxil Tablets are contraindicated in patients with previously demonstrated hypersensitivity to any of the components of the product.
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Severe acute exacerbations of Hepatitis [See Boxed Warning , Warnings and Precautions (5.1) ] Nephrotoxicity [See Boxed Warning , Warnings and Precautions (5.2) ]
Drug Interactions
Since adefovir is eliminated by the kidney, coadministration of Adefovir Dipivoxil Tablets with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either adefovir and/or these coadministered drugs [See Clinical Pharmacology (12.3) ] . Patients should be monitored closely for adverse events when Adefovir Dipivoxil Tablets are coadministered with drugs that are excreted renally or with other drugs known to affect renal function [See Warnings and Precautions (5.2) ]. Adefovir Dipivoxil Tablets should not be administered in combination with VIREAD [See Warnings and Precautions (5.5) ].
Storage and Handling
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed ∑3 on one side and plain on the other side. They are packaged as follows: Bottles of 30 tablets (NDC 42794-003-08) containing polyester and desiccant and closed with a child-resistant closure.
How Supplied
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed ∑3 on one side and plain on the other side. They are packaged as follows: Bottles of 30 tablets (NDC 42794-003-08) containing polyester and desiccant and closed with a child-resistant closure.
Medication Information
Warnings and Precautions
Severe acute exacerbations of hepatitis: Monitor hepatic function closely at repeated intervals for at least several months in patients who discontinue Adefovir Dipivoxil Tablets. ( 5.1 ) Nephrotoxicity: Monitor renal function during therapy for all patients, particularly those with pre-existing or other risks for renal impairment. Dose adjustment may be required. ( 5.2 ) HIV Resistance: Offer HIV testing to all patients prior to initiating Adefovir Dipivoxil Tablets. Untreated HIV may result in HIV resistance. ( 5.3 ) Lactic acidosis and severe hepatomegaly with steatosis: If suspected, suspend treatment. ( 5.4 ) Coadministration with Other Products: Do not administer Adefovir Dipivoxil Tablets concurrently with VIREAD ® or other tenofovir-containing products. ( 5.5 ) Clinical Resistance: For patients with lamivudine-resistant HBV use adefovir dipivoxil in combination with lamivudine. For all patients, consider modifying treatment in case serum HBV DNA remains above 1000 copies/mL with continued treatment. ( 5.6 )
Indications and Usage
Adefovir Dipivoxil Tablets are indicated for the treatment of chronic hepatitis B in patients 12 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease. This indication is based on histological, virological, biochemical, and serological responses in adult patients with HBeAg+ and HBeAg- chronic hepatitis B with compensated liver function, and with clinical evidence of lamivudine-resistant hepatitis B virus with either compensated or decompensated liver function. For patients 12 to less than 18 years of age, the indication is based on virological and biochemical responses in patients with HBeAg+ chronic hepatitis B virus infection with compensated liver function.
Dosage and Administration
One tablet containing 10 mg adefovir dipivoxil once daily orally with or without food. ( 2.1 ) Dose adjustment in renal impairment for adults ( 2.2 ) Creatinine Clearance (mL/min) a Hemodialysis Patients Greater than or equal to 50 30 - 49 10 - 29 Recommended dose and dosing interval 10 mg every 24 hours 10 mg every 48 hours 10 mg every 72 hours 10 mg every 7 days following dialysis a Creatinine clearance calculated by Cockcroft-Gault method using lean or ideal body weight. No dose recommendations for ( 2.1 ): Non-hemodialysis patients with creatinine clearance less than 10 mL per minute. Adolescent patients with renal impairment.
Contraindications
Adefovir Dipivoxil Tablets are contraindicated in patients with previously demonstrated hypersensitivity to any of the components of the product.
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Severe acute exacerbations of Hepatitis [See Boxed Warning , Warnings and Precautions (5.1) ] Nephrotoxicity [See Boxed Warning , Warnings and Precautions (5.2) ]
Drug Interactions
Since adefovir is eliminated by the kidney, coadministration of Adefovir Dipivoxil Tablets with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either adefovir and/or these coadministered drugs [See Clinical Pharmacology (12.3) ] . Patients should be monitored closely for adverse events when Adefovir Dipivoxil Tablets are coadministered with drugs that are excreted renally or with other drugs known to affect renal function [See Warnings and Precautions (5.2) ]. Adefovir Dipivoxil Tablets should not be administered in combination with VIREAD [See Warnings and Precautions (5.5) ].
Storage and Handling
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed ∑3 on one side and plain on the other side. They are packaged as follows: Bottles of 30 tablets (NDC 42794-003-08) containing polyester and desiccant and closed with a child-resistant closure.
How Supplied
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed ∑3 on one side and plain on the other side. They are packaged as follows: Bottles of 30 tablets (NDC 42794-003-08) containing polyester and desiccant and closed with a child-resistant closure.
Description
Severe acute exacerbations of hepatitis have been reported in patients who have discontinued anti-Hepatitis B therapy including Adefovir Dipivoxil Tablets. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-Hepatitis B therapy. If appropriate, resumption of anti-Hepatitis B therapy may be warranted [See Warnings and Precautions (5.1) ] . In patients at risk of or having underlying renal dysfunction, chronic administration of Adefovir Dipivoxil Tablets may result in nephrotoxicity. These patients should be monitored closely for renal function and may require dose adjustment [See Warnings and Precautions (5.2) and Dosage and Administration (2.2) ] . HIV resistance may emerge in chronic hepatitis B patients with unrecognized or untreated Human Immunodeficiency Virus (HIV) infection treated with antihepatitis B therapies, such as therapy with Adefovir Dipivoxil Tablets, that may have activity against HIV [See Warnings and Precautions (5.3) ] . Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with other antiretrovirals [See Warnings and Precautions (5 . 4 ) ] .
Data
Animal Data
In an embryo-fetal development study, ADV was administered orally to pregnant rabbits (at 1, 5, or 20 mg/kg/day) during organogenesis (on gestation day 6 through 18). No adverse developmental effects were observed at up to the highest dose tested, at systemic exposure (C max) 40 times that in humans at the RHD of Adefovir Dipivoxil Tablets.
In a pre/post-natal development study, ADV was administered orally to pregnant rats (at 2.5, 10, or 40 mg/kg/day) from organogenesis, through late gestation, delivery, and lactation (gestation day 7 to lactation/postpartum day 20). Reduced body weight of the offspring due to maternal toxicity was observed at systemic exposure 23 times that in humans at the RHD of Adefovir Dipivoxil Tablets.
In an embryo-fetal development study, ADV was administered intravenously to pregnant rats (at 2.5, 10, and 20 mg/kg/day) during organogenesis (gestation day 6 through 15). Embryo-fetal toxicity including malformations (anasarca, depressed eye bulge, umbilical hernia and kinked tail) and skeletal variations (reduction of ossified metacarpal bones, increases in thoracic vertebrae and decreases in lumbar vertebrae) occurred at systemic exposure (C max) 38 times that in humans at the RHD of Adefovir Dipivoxil Tablets. No adverse developmental effects were observed at an exposure (C max) 12 times that in humans at the RHD of Adefovir Dipivoxil Tablets.
Section 42229-5
Pediatric Patients
The efficacy and safety of Adefovir Dipivoxil Tablets have not been studied in patients less than 18 years of age with different degrees of renal impairment and no data are available to make dosage recommendations in these patients [See Dosage and Administration (2.2)]. Caution should be exercised when prescribing Adefovir Dipivoxil Tablets to adolescents with underlying renal dysfunction, and renal function in these patients should be closely monitored.
Section 42230-3
FDA-Approved Patient Labeling
PATIENT INFORMATION
ADEFOVIR DIPIVOXIL (a-DEF-oh-vir dip-ih-VOX-il) TABLETS
Read this Patient Information before you start taking Adefovir Dipivoxil Tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
What is the most important information I should know about Adefovir Dipivoxil Tablets?
Adefovir Dipivoxil Tablets can cause serious side effects, including:
-
Worsening of hepatitis B infection.Your hepatitis B (HBV) infection may become worse (flare-up) if you take Adefovir Dipivoxil Tablets and then stop taking it. A “flare-up” is when your HBV infection suddenly returns in a worse way than before.
- Do not run out of Adefovir Dipivoxil Tablets. Refill your prescription or talk to your doctor before your Adefovir Dipivoxil Tablets are all gone.
- Do not stop taking Adefovir Dipivoxil Tablets without first talking with your doctor.
- If you stop taking Adefovir Dipivoxil Tablets, your doctor will need to check your health often and do blood tests regularly to check your HBV infection for at least several months.
-
Adefovir Dipivoxil Tablets
may cause severe kidney problems. Severe kidney problems can happen in anyone who takes Adefovir Dipivoxil Tablets, but certain people may have a higher risk of severe kidney problems with Adefovir Dipivoxil Tablets including:
- people who already have kidney problems or are at an increased risk for kidney problems, and
- people who take certain medicines that can cause kidney problems. Ask your doctor whether any of the medicines you currently take can cause kidney problems.
Your doctor should do blood tests to check your kidney function during treatment with Adefovir Dipivoxil Tablets.
If you have kidney problems before you start taking Adefovir Dipivoxil Tablets your doctor may change your dose of Adefovir Dipivoxil Tablets. If you develop kidney problems during treatment, your doctor may need to change your dose of Adefovir Dipivoxil Tablets or may stop your treatment.
- HIV resistance. Your doctor may test you for HIV-1 infection before you start Adefovir Dipivoxil Tablets. If you have both HBV and HIV-1 and you only take Adefovir Dipivoxil Tablets, the HIV-1 virus may develop resistance and become harder to treat.
- Build-up of acid in your blood (lactic acidosis). Lactic acidosis can happen in some people who take Adefovir Dipivoxil Tablets. Lactic acidosis is a serious medical emergency that can lead to death.
Call your doctor right away if you get any of the following symptoms which could be signs of lactic acidosis:
|
|
- Severe liver problems. In rare cases, severe liver problems can happen that lead to death. Call your doctor right away if you get any of the following signs or symptoms of liver problems.
|
|
You may be more likely to get lactic acidosis or serious liver problems if you are female, are very overweight (obese), or have been taking nucleoside analog medicines for a long time.
What are Adefovir Dipivoxil Tablets?
Adefovir Dipivoxil Tablets are a medicine used to treat people 12 years of age and older with chronic (long-lasting) infections with active hepatitis B virus.
Adefovir Dipivoxil Tablets are not for use in children under 12 years of age.
- Adefovir Dipivoxil Tablets may lower the amount of hepatitis B virus (HBV) in your body.
- Adefovir Dipivoxil Tablets may improve the condition of your liver.
Who should not take Adefovir Dipivoxil Tablets?
Do not take Adefovir Dipivoxil Tablets if you are allergic to any of the ingredients in Adefovir Dipivoxil Tablets. See the end of this leaflet for a complete list of the ingredients in Adefovir Dipivoxil Tablets.
What should I tell my healthcare provider before taking Adefovir Dipivoxil Tablets?
Before taking Adefovir Dipivoxil Tablets, tell your doctor about all of your medical
conditions, including if you:
- have or had kidney problems. Your dose and schedule of Adefovir Dipivoxil Tablets may need to be changed.
- are pregnant or plan to become pregnant. It is not known if Adefovir Dipivoxil Tablets will harm your unborn baby.
Pregnancy Registry: There is a pregnancy registry for women who take Adefovir Dipivoxil Tablets during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk with your doctor about how you can take part in this registry.
- are breastfeeding. It is not known if adefovir dipivoxil passes into your breast milk. Talk with your doctor about the best way to feed your baby during treatment with Adefovir Dipivoxil Tablets.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines may affect how Adefovir Dipivoxil Tablets work, especially medicines that affect how your kidneys work. Adefovir Dipivoxil Tablets can affect how your other medicines work. Your dose of Adefovir Dipivoxil Tablets and the other medicines may need to be changed. Do not start a new medicine without telling your doctor. Your doctor can tell you if it is safe to take Adefovir Dipivoxil Tablets with other medicines.
How should I take Adefovir Dipivoxil Tablets?
- Take Adefovir Dipivoxil Tablets exactly as your doctor tells you to take it.
- Your doctor will tell you how much Adefovir Dipivoxil Tablets to take, and when and how often to take it.
- Do not change your dose or stop Adefovir Dipivoxil Tablets without talking to your doctor. Your hepatitis may get worse if you change doses or stop.
- Take Adefovir Dipivoxil Tablets at the same time each day that your doctor tells you, to avoid missing doses.
- Take Adefovir Dipivoxil Tablets with or without food.
- Stay under the care of your doctor during treatment with Adefovir Dipivoxil Tablets.
- When your Adefovir Dipivoxil Tablets supply gets low, call your doctor or pharmacy for a refill. Do not run out of Adefovir Dipivoxil Tablets.
- If you take too much Adefovir Dipivoxil Tablets, call your doctor or go to the nearest hospital emergency room right away.
- It is not known how long you should take Adefovir Dipivoxil Tablets. You and your doctor will need to decide when it is best for you to stop taking Adefovir Dipivoxil Tablets. Some people get worsening of their hepatitis B infection when they stop taking Adefovir Dipivoxil Tablets. See “What is the most important information I should know about Adefovir Dipivoxil Tablets?".
What are the possible side effects of Adefovir Dipivoxil Tablets?
Adefovir Dipivoxil Tablets can cause serious side effects. See “What is the most important information I should know about Adefovir Dipivoxil Tablets?"
The most common side effects of Adefovir Dipivoxil Tablets are weakness, headache, stomach pain, and nausea.
These are not all the possible side effects of Adefovir Dipivoxil Tablets. For more information, ask your doctor or pharmacist.
Call your doctor for advice about side effects. You may report side effects to FDA at 1-800-FDA-1088
How should I store Adefovir Dipivoxil Tablets?
- Store Adefovir Dipivoxil Tablets at room temperature, between 68°F to 77°F (20°C to 25°C).
- Keep Adefovir Dipivoxil Tablets in its original container.
- Do not use Adefovir Dipivoxil Tablets if the seal over the bottle is broken or missing when you receive it.
Keep Adefovir Dipivoxil Tablets and all medicines out of the reach of children.
General information about the safe and effective use of Adefovir Dipivoxil Tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Adefovir Dipivoxil Tablets for a condition for which it was not prescribed. Do not give Adefovir Dipivoxil Tablets to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about
Adefovir Dipivoxil Tablets that is written for health professionals.
What are the Ingredients in Adefovir dipivoxil tablets?
Active Ingredient: adefovir dipivoxil
Inactive ingredients: copovidone, anhydrous lactose, microcrystalline cellulose, silicon dioxide, crospovidone and magnesium stearate
Manufactured by:
Sigmapharm Laboratories, LLC
Bensalem, PA 19020
HEPSERA, ATRIPLA, COMPLERA, EMTRIVA, STRIBILD, TRUVADA, and VIREAD are trademarks or registered trademarks of Gilead Sciences, Inc., or its related companies. Other brands listed are the trademarks of their respective owners.
OS003-09 REV.0821
This Patient Information has been approved by the U.S. Food and Drug Administration.
Section 44425-7
Store in original container at 25 °C (77 °F), excursions permitted to 15° - 30 °C (59° - 86 °F) (See USP Controlled Room Temperature).
Do not use if seal over bottle opening is broken or missing.
10 Overdosage
Doses of adefovir dipivoxil 500 mg daily for 2 weeks and 250 mg daily for 12 weeks have been associated with gastrointestinal side effects. If overdose occurs the patient must be monitored for evidence of toxicity, and standard supportive treatment applied as necessary.
Following a 10 mg single dose of Adefovir Dipivoxil Tablets, a four-hour hemodialysis session removed approximately 35% of the adefovir dose.
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to Adefovir Dipivoxil Tablets during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
8.2 Lactation
Risk Summary
It is not known whether adefovir is present in human breast milk, affects human milk production, or has effects on the breastfed infant.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Adefovir Dipivoxil Tablets and any potential adverse effects on the breastfed infant from Adefovir Dipivoxil Tablets or from the underlying maternal condition.
11 Description
Adefovir dipivoxil is a diester prodrug of adefovir. Adefovir is an acyclic nucleotide analog with activity against human hepatitis B virus (HBV).
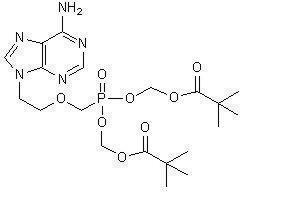
The chemical name of adefovir dipivoxil is 9-[2-[[bis[(pivaloyloxy)methoxy]-phosphinyl]-methoxy]ethyl]adenine. It has a molecular formula of C 20H 32N 5O 8P, a molecular weight of 501.48 and the following structural formula:
Adefovir dipivoxil is a white to off-white crystalline powder with an aqueous solubility of 19 mg/mL at pH 2.0 and 0.4 mg/mL at pH 7.2. It has an octanol/aqueous phosphate buffer (pH 7) partition coefficient (log p) of 1.91.
Adefovir Dipivoxil Tablets are for oral administration. Each tablet contains 10 mg of adefovir dipivoxil and the following inactive ingredients: copovidone, anhydrous lactose, microcrystalline cellulose, silicon dioxide, crospovidone and magnesium stearate.
12.4 Microbiology
8.4 Pediatric Use
8.5 Geriatric Use
Clinical studies of Adefovir Dipivoxil Tablets did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. In general, caution should be exercised when prescribing to elderly patients since they have greater frequency of decreased renal or cardiac function due to concomitant disease or other drug therapy.
5.2 Nephrotoxicity
Nephrotoxicity characterized by a delayed onset of gradual increases in serum creatinine and decreases in serum phosphorus was historically shown to be the treatment-limiting toxicity of adefovir dipivoxil therapy at substantially higher doses in HIV-infected patients (60 and 120 mg daily) and in chronic hepatitis B patients (30 mg daily). Chronic administration of Adefovir Dipivoxil Tablets (10 mg once daily) may result in delayed nephrotoxicity. The overall risk of nephrotoxicity in patients with adequate renal function is low. However, this is of special importance in patients at risk of or having underlying renal dysfunction and patients taking concomitant nephrotoxic agents such as cyclosporine, tacrolimus, aminoglycosides, vancomycin and non-steroidal anti-inflammatory drugs [See Adverse Reactions (6.2) and Clinical Pharmacology (12.3)] . It is recommended that creatinine clearance is calculated in all patients prior to initiating therapy with Adefovir Dipivoxil Tablets.
It is important to monitor renal function for all patients during treatment with Adefovir Dipivoxil Tablets, particularly for those with pre-existing or other risks for renal impairment. Patients with renal insufficiency at baseline or during treatment may require dose adjustment [See Dosage and Administration (2.2)] . The risks and benefits of Adefovir Dipivoxil Tablets treatment should be carefully evaluated prior to discontinuing Adefovir Dipivoxil Tablets in a patient with treatment-emergent nephrotoxicity.
5.3 Hiv Resistance
Prior to initiating adefovir dipivoxil therapy, HIV antibody testing should be offered to all patients. Treatment with anti-hepatitis B therapies, such as Adefovir Dipivoxil Tablets, that have activity against HIV in a chronic hepatitis B patient with unrecognized or untreated HIV infection may result in emergence of HIV resistance. Adefovir Dipivoxil Tablets has not been shown to suppress HIV RNA in patients; however, there are limited data on the use of Adefovir Dipivoxil Tablets to treat patients with chronic hepatitis B co-infected with HIV.
14 Clinical Studies
4 Contraindications
Adefovir Dipivoxil Tablets are contraindicated in patients with previously demonstrated hypersensitivity to any of the components of the product.
6 Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling:
- Severe acute exacerbations of Hepatitis [See Boxed Warning, Warnings and Precautions (5.1)]
- Nephrotoxicity [See Boxed Warning, Warnings and Precautions (5.2)]
7 Drug Interactions
Since adefovir is eliminated by the kidney, coadministration of Adefovir Dipivoxil Tablets with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either adefovir and/or these coadministered drugs [See Clinical Pharmacology (12.3)] .
Patients should be monitored closely for adverse events when Adefovir Dipivoxil Tablets are coadministered with drugs that are excreted renally or with other drugs known to affect renal function [See Warnings and Precautions (5.2)].
Adefovir Dipivoxil Tablets should not be administered in combination with VIREAD [See Warnings and Precautions (5.5)].
1 indications and Usage
Adefovir Dipivoxil Tablets are indicated for the treatment of chronic hepatitis B in patients 12 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
This indication is based on histological, virological, biochemical, and serological responses in adult patients with HBeAg+ and HBeAg- chronic hepatitis B with compensated liver function, and with clinical evidence of lamivudine-resistant hepatitis B virus with either compensated or decompensated liver function.
For patients 12 to less than 18 years of age, the indication is based on virological and biochemical responses in patients with HBeAg+ chronic hepatitis B virus infection with compensated liver function.
2.1 chronic Hepatitis B
The recommended dose of Adefovir Dipivoxil Tablets in chronic hepatitis B patients for patients 12 years of age and older with adequate renal function is 10 mg, once daily, taken orally, without regard to food. The optimal duration of treatment is unknown.
Adefovir Dipivoxil Tablets is not recommended for use in children less than 12 years of age.
6.3 Pediatric Patients
Assessment of adverse reactions is based on a placebo-controlled study (Study 518) in which 173 pediatric patients aged 2 to less than 18 years with chronic hepatitis B and compensated liver disease received double-blind treatment with Adefovir Dipivoxil Tablets (N=115), or placebo (N=58) for 48 weeks [See Clinical Studies (14.4) and Use In Specific Populations (8.4)] .
The safety profile of Adefovir Dipivoxil Tablets in patients 12 to less than 18 years of age (N=56) was similar to that observed in adults. No pediatric patients treated with Adefovir Dipivoxil Tablets developed a confirmed serum creatinine increase greater than or equal to 0.5 mg/dL from baseline or a confirmed phosphorus decrease to less than 2 mg/dL by Week 48.
5.6 Clinical Resistance
Resistance to adefovir dipivoxil can result in viral load rebound which may result in exacerbation of hepatitis B and, in the setting of diminished hepatic function, lead to liver decompensation and possible fatal outcome.
In order to reduce the risk of resistance in patients with lamivudine resistant HBV, adefovir dipivoxil should be used in combination with lamivudine and not as adefovir dipivoxil monotherapy.
In order to reduce the risk of resistance in all patients receiving adefovir dipivoxil monotherapy, a modification of treatment should be considered if serum HBV DNA remains above 1000 copies/mL with continued treatment.
Long-term (144 week) data from Study 438 (N=124) show that patients with HBV DNA levels greater than 1000 copies/mL at Week 48 of treatment with adefovir dipivoxil were at greater risk of developing resistance than patients with serum HBV DNA levels below 1000 copies/mL at Week 48 of therapy.
12.1 Mechanism of Action
Adefovir is an antiviral drug [See Microbiology (12.4)].
13 Nonclinical Toxicology
6.2 Special Risk Patients
Pre- and Post-Liver Transplantation Patients
Additional adverse reactions observed from an open-label study (Study 435) in pre- and post- liver transplantation patients with chronic hepatitis B and lamivudine-resistant hepatitis B administered Adefovir Dipivoxil Tablets once daily for up to 203 weeks include: abnormal renal function, renal failure, vomiting, rash, and pruritus.
Changes in renal function occurred in pre-and post-liver transplantation patients with risk factors for renal dysfunction, including concomitant use of cyclosporine and tacrolimus, renal insufficiency at baseline, hypertension, diabetes, and on-study transplantation. Therefore, the contributory role of Adefovir Dipivoxil Tablets to these changes in renal function is difficult to assess.
Increases in serum creatinine greater than or equal to 0.3 mg/dL from baseline were observed in 37% and 53% of pre-liver transplantation patients by Weeks 48 and 96, respectively, by Kaplan-Meier estimates. Increases in serum creatinine greater than or equal to 0.3 mg/dL from baseline were observed in 32% and 51% of post-liver transplantation patients by Weeks 48 and 96, respectively, by Kaplan-Meier estimates. Serum phosphorus values less than 2 mg/dL were observed in 3/226 (1.3%) of pre-liver transplantation patients and in 6/241 (2.5%) of post-liver transplantation patients by last study visit. Four percent (19 of 467) of patients discontinued treatment with Adefovir Dipivoxil Tablets due to renal adverse events.
5 Warnings and Precautions
2 Dosage and Administration
- One tablet containing 10 mg adefovir dipivoxil once daily orally with or without food. ( 2.1)
- Dose adjustment in renal impairment for adults ( 2.2)
| Creatinine Clearance (mL/min) a | Hemodialysis Patients | |||
| Greater than or equal to 50 | 30 - 49 | 10 - 29 | ||
| Recommended dose and dosing interval | 10 mg every 24 hours | 10 mg every 48 hours | 10 mg every 72 hours | 10 mg every 7 days following dialysis |
a Creatinine clearance calculated by Cockcroft-Gault method using lean or ideal body weight.
- No dose recommendations for (
2.1):
- Non-hemodialysis patients with creatinine clearance less than 10 mL per minute.
- Adolescent patients with renal impairment.
3 Dosage Forms and Strengths
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed Σ 3 on one side and plain on the other side.
8 Use in Specific Population
6.4 Post Marketing Experience
In addition to adverse reaction reports from clinical trials, the following possible adverse reactions have also been identified during post-approval use of adefovir dipivoxil. Because these events have been reported voluntarily from a population of unknown size, estimates of frequency cannot be made.
Metabolism and Nutrition Disorders: hypophosphatemia
Gastrointestinal Disorders: pancreatitis
Musculoskeletal System and Connective Tissue Disorders: myopathy, osteomalacia (manifested as bone pain and may contribute to fractures), both associated with proximal renal tubulopathy
Renal and Urinary Disorders: renal failure, Fanconi syndrome, proximal renal tubulopathy
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical and laboratory evidence of exacerbations of hepatitis have occurred after discontinuation of treatment with Adefovir Dipivoxil Tablets.
Adverse reactions to Adefovir Dipivoxil Tablets identified from placebo-controlled and open label studies include the following: asthenia, headache, abdominal pain, diarrhea, nausea, dyspepsia, flatulence, increased creatinine, and hypophosphatemia.
The incidence of these adverse reactions in studies 437 and 438, where 522 patients with chronic hepatitis B and compensated liver disease received double-blind treatment with Adefovir Dipivoxil Tablets (N=294) or placebo (N=228) for 48 weeks is presented in Table 2. Patients who received open-label Adefovir Dipivoxil Tablets for up to 240 weeks in Study 438 reported adverse reactions similar in nature and severity to those reported in the first 48 weeks.
| Adverse Reaction | Adefovir Dipivoxil Tablets 10 mg (N=294) | Placebo (N=228) |
| Asthenia | 13% | 14% |
| Headache | 9% | 10% |
| Abdominal Pain | 9% | 11% |
| Nausea | 5% | 8% |
| Flatulence | 4% | 4% |
| Diarrhea | 3% | 4% |
| Dyspepsia | 3% | 2% |
a In these studies, the overall incidence of adverse reactions with Adefovir Dipivoxil Tablets was similar to that reported with placebo. The incidence of adverse reactions is derived from treatment-related events as identified by the study investigators.
No patients treated with Adefovir Dipivoxil Tablets developed a confirmed serum creatinine increase greater than or equal to 0.5 mg/dL from baseline or confirmed phosphorus decrease to 2 mg/dL or less by Week 48. By Week 96, 2% of Adefovir Dipivoxil Tablets-treated patients, by Kaplan-Meier estimate, had increases in serum creatinine greater than or equal to 0.5 mg/dL from baseline (no placebo-controlled results were available for comparison beyond Week 48). For patients who chose to continue Adefovir Dipivoxil Tablets for up to 240 weeks in Study 438, 4 of 125 patients (3%) had a confirmed increase of 0.5 mg/dL from baseline. The creatinine elevation resolved in 1 patient who permanently discontinued treatment and remained stable in 3 patients who continued treatment. For 65 patients who chose to continue Adefovir Dipivoxil Tablets for up to 240 weeks in Study 437, 6 had a confirmed increase in serum creatinine of greater than or equal to 0.5 mg/dL from baseline with 2 patients discontinuing from the study due to the elevated serum creatinine concentration. See Adverse Reactions (6.2) for changes in serum creatinine in patients with underlying renal insufficiency at baseline.
14.4 Study 518 (pediatric Study)
Study 518 was a double-blind, placebo-controlled, study in which 173 pediatric patients (ages 2 to less than 18 years) with chronic hepatitis B (CHB) infection and elevated ALT were randomized 2:1 (115 receiving adefovir dipivoxil and 58 receiving placebo). Randomization was stratified by prior treatment and age 2 to less than 7 years old (cohort 1), 7 to less than 12 years old (cohort 2), and 12 to less than 18 years old (cohort 3). All patients in cohort 3 received 10 mg tablet formulation; all patients in cohorts 1 and 2 received an investigational suspension formulation (0.3 mg/kg/day cohort 1, 0.25 mg/kg/day cohort 2) once daily. The primary efficacy endpoint was HBV DNA less than 1000 copies/mL plus normalization of ALT at the end of Week 48.
In cohort 3 (N=83), significantly more patients treated with Adefovir Dipivoxil Tablets achieved the primary efficacy endpoint at the end of 48 weeks of blinded treatment (23%) when compared to placebo-treated patients (0%). The proportion of patients from cohorts 1 and 2 who responded to treatment with adefovir dipivoxil was not statistically significant when compared to the placebo arm, although the adefovir plasma concentrations in these patients were comparable to those observed in older patients. Overall, 22 of 115 (19%) of pediatric patients who received adefovir dipivoxil versus 1 of 58 (2%) of placebo treated patients responded to treatment by Week 48 [See Adverse Reactions (6.3), Use In Special Populations (8.4) and Clinical Pharmacology (12.3, 12.4)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Inform patients of the potential risks and benefits of Adefovir Dipivoxil Tablets and of alternative modes of therapy.
- Instruct patients to:
-Follow a regular dosing schedule to avoid missing doses.
-Immediately report any severe abdominal pain, muscle pain, yellowing of the eyes, dark urine, pale stools, and/or loss in appetite.
-Inform their doctor or pharmacist if they develop any unusual symptom(s), or if any known symptom persists or worsens.
- Advise patients that:
- The optimal duration of Adefovir Dipivoxil Tablets treatment and the relationship between treatment response and long-term outcomes such as hepatocellular carcinoma or decompensated cirrhosis are not known.
- Patients should not discontinue Adefovir Dipivoxil Tablets without first informing their physician [See Warnings and Precautions (5.1)].
- Routine laboratory monitoring and follow-up with a physician is important during Adefovir Dipivoxil Tablets therapy.
- Obtaining HIV antibody testing prior to starting Adefovir Dipivoxil Tablets is important [See Warnings and Precautions (5.3)].
- Adefovir Dipivoxil Tablets should not be administered concurrently with ATRIPLA or COMPLERA or STRIBILD or TRUVADA or VIREAD [See Warnings and Precautions (5.5)].
- Lamivudine-resistant patients should use Adefovir Dipivoxil Tablets in combination with lamivudine and not as Adefovir Dipivoxil Tablets monotherapy [See Warnings and Precautions(5.6)].
- Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant women exposed to Adefovir Dipivoxil [ see Use in Specific Populations (8.1)] .
Manufactured by:
Sigmapharm Laboratories, LLC
Bensalem, PA 19020
OS003-09 REV.0821
16 How Supplied / Storage and Handling
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed ∑3 on one side and plain on the other side. They are packaged as follows: Bottles of 30 tablets (NDC 42794-003-08) containing polyester and desiccant and closed with a child-resistant closure.
2.2 dose Adjustment in Renal Impairment
Significantly increased drug exposures were seen when Adefovir Dipivoxil Tablets was administered to adult patients with renal impairment [See Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. Therefore, the dosing interval of Adefovir Dipivoxil Tablets should be adjusted in adult patients with baseline creatinine clearance less than 50 mL per minute using the following suggested guidelines (See Table 1). The safety and effectiveness of these dosing interval adjustment guidelines have not been clinically evaluated.
Additionally, it is important to note that these guidelines were derived from data in patients with pre-existing renal impairment at baseline. They may not be appropriate for patients in whom renal insufficiency evolves during treatment with Adefovir Dipivoxil Tablets. Therefore, clinical response to treatment and renal function should be closely monitored in these patients.
Table 1 Dosing Interval Adjustment of Adefovir Dipivoxil Tablets in Adult Patients with Renal Impairment
| Creatinine Clearance (mL/min) a | Hemodialysis Patients | |||
| Greater than or equal to 50 | 30 - 49 | 10 - 29 | ||
| Recommended dose and dosing interval | 10 mg every 24 hours | 10 mg every 48 hours | 10 mg every 72 hours | 10 mg every 7 days following dialysis |
a Creatinine clearance calculated by Cockcroft-Gault method using lean or ideal body weight.
The pharmacokinetics of adefovir have not been evaluated in non-hemodialysis patients with creatinine clearance less than 10 mL per minute; therefore, no dosing recommendation is available for these patients.
No clinical data are available to make dosing recommendations in adolescent patients with renal insufficiency [See Warnings and Precautions (5.2)]
5.5 Coadministration With Other Products
Adefovir Dipivoxil Tablets should not be used concurrently with products containing tenofovir disoproxil fumarate or tenofovir alafenamide.
8.6 Patients With Impaired Renal Function
It is recommended that the dosing interval for Adefovir Dipivoxil Tablets be modified in adult patients with baseline creatinine clearance less than 50 mL per minute. The pharmacokinetics of adefovir have not been evaluated in non-hemodialysis patients with creatinine clearance less than 10 mL per minute or in adolescent patients with renal insufficiency; therefore, no dosing recommendations are available for these patients [See Dosage and Administration (2.2) and Warning and Precautions (5.2)].
13.2 Animal Toxicology And/or Pharmacology
Renal tubular nephropathy characterized by histological alterations and/or increases in BUN and serum creatinine was the primary dose-limiting toxicity associated with administration of adefovir dipivoxil in animals. Nephrotoxicity was observed in animals at systemic exposures approximately 3 - 10 times higher than those in humans at the recommended therapeutic dose of 10 mg/day.
14.1 Studies 437 and 438 (pivotal Studies)
HBeAg-Positive Chronic Hepatitis B
Study 437 was a randomized, double-blind, placebo-controlled, three-arm-study in patients with HBeAg-positive chronic hepatitis B that allowed for a comparison between placebo and Adefovir Dipivoxil Tablets. The median age of patients was 33 years. Seventy-four percent were male, 59% were Asian, 36% were Caucasian, and 24% had prior interferon-α treatment. At baseline, patients had a median total Knodell Histology Activity Index (HAI) score of 10, a median serum HBV DNA level as measured by the Roche Amplicor Monitor polymerase chain reaction (PCR) assay (LLOQ = 1000 copies/mL) of 8.36 log 10 copies/mL and a median ALT level of 2.3 times the upper limit of normal.
HBeAg-Negative (Anti-HBe Positive/HBV DNA Positive) Chronic Hepatitis B
Study 438 was a randomized, double-blind, placebo-controlled study in patients who were HBeAg-negative at screening, and anti-HBe positive. The median age of patients was 46 years. Eighty-three percent were male, 66% were Caucasian, 30% were Asian and 41% had prior interferon-α treatment. At baseline, the median total Knodell HAI score was 10, the median serum HBV DNA level as measured by the Roche Amplicor Monitor PCR assay (LLOQ = 1000 copies/mL) was 7.08 log 10 copies/mL, and the median ALT was 2.3 times the upper limit of normal.
The primary efficacy endpoint in both studies was histological improvement at Week 48; results of which are shown in Table 4.
| Study 437 | Study 438 | |||
|
Adefovir Dipivoxil Tablets
10 mg (N=168) |
Placebo
(N=161) |
Adefovir Dipivoxil Tablets 10 mg
(N=121) |
Placebo
(N=57) |
|
| Improvement b | 53% | 25% | 64% | 35% |
| No Improvement | 37% | 67% | 29% | 63% |
| Missing/Unassessable Data | 10% | 7% | 7% | 2% |
a Intent-to-Treat population (patients with ≥1 dose of study drug) with assessable baseline biopsies.
b Histological improvement defined as ≥2 point decrease in the Knodell necro-inflammatory score with no worsening of the Knodell fibrosis score.
Table 5 illustrates the changes in Ishak Fibrosis Score by treatment group.
Table 5 Changes in Ishak Fibrosis Score at Week 48
|
Number of Adequate
Biopsy Pairs |
Study 437 | Study 438 | ||
|
Adefovir Dipivoxil Tablets 10 mg
(N=152) |
Placebo
(N=149) |
Adefovir Dipivoxil Tablets 10 mg
(N=113) |
Placebo
(N=56) |
|
| Ishak Fibrosis Score
Improved a |
34% | 19% | 34% | 14% |
| Unchanged | 55% | 60% | 62% | 50% |
| Worsened a | 11% | 21% | 4% | 36% |
a Change of 1 point or more in Ishak Fibrosis Score.
At Week 48, improvement was seen with respect to mean change in serum HBV DNA (log 10 copies/mL), normalization of ALT, and HBeAg seroconversion as compared to placebo in patients receiving Adefovir Dipivoxil Tablets (Table 6).
Table 6 Change in Serum HBV DNA, ALT Normalization, and HBeAg Seroconversion at Week 48
| Study 437 | Study 438 | |||
|
Adefovir Dipivoxil Tablets 10 mg
(N=171) |
Placebo
(N=167) |
Adefovir Dipivoxil Tablets 10 mg
(N=123) |
Placebo
(N=61) |
|
| Mean change ± SD in serum HBV DNA form baseline (log 10 copies/mL) | -3.57 ± 1.64 | -0.98 ± 1.32 | -3.65 ± 1.14 | -1.32 ± 1.25 |
| ALT normalization | 48% | 16% | 72% | 29% |
| HBeAg seroconversion | 12% | 6% | NA a | NA a |
a Patients with HBeAg-negative disease cannot undergo HBeAg seroconversion.
Treatment Beyond 48 Weeks
In Study 437, continued treatment with Adefovir Dipivoxil Tablets to 72 weeks resulted in continued maintenance of mean reductions in serum HBV DNA observed at Week 48. An increase in the proportion of patients with ALT normalization was also observed in Study 437. The effect of continued treatment with Adefovir Dipivoxil Tablets on seroconversion is unknown.
In Study 438, patients who received Adefovir Dipivoxil Tablets during the first 48 weeks were re-randomized in a blinded manner to continue on Adefovir Dipivoxil Tablets or receive placebo for an additional 48 weeks. At Week 96, 50 of 70 (71%) of patients who continued treatment with Adefovir Dipivoxil Tablets had undetectable HBV DNA levels (less than 1000 copies/mL), and 47 of 64 (73%) of patients had ALT normalization. HBV DNA and ALT levels returned towards baseline in most patients who stopped treatment with Adefovir Dipivoxil Tablets.
From 141 eligible patients, there were 125 (89%) patients in Study 438 who chose to continue Adefovir Dipivoxil Tablets for up to 192 weeks or 240 weeks (4 years or 5 years). As these patients had already received Adefovir Dipivoxil Tablets for at least 48 weeks and appeared to be experiencing a benefit, they are not necessarily representative of patients initiating Adefovir Dipivoxil Tablets. Of these patients, 89/125 (71%) and 47/70 (67%) had an undetectable HBV DNA level (less than 1000 copies/mL) at Week 192 and Week 240, respectively. Of the patients who had an elevated ALT at baseline, 77/104 (74%) and 42/64 (66%) had a normal ALT at Week 192 and Week 240, respectively. Six (5%) patients experienced HBsAg loss.
5.4 Lactic Acidosis/severe Hepatomegaly With Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with antiretrovirals.
A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Particular caution should be exercised when administering nucleoside analogs to any patient with known risk factors for liver disease; however, cases have also been reported in patients with no known risk factors. Treatment with Adefovir Dipivoxil Tablets therapy should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
Principal Display Panel Adefovir Dipivoxil 10 Mg Carton Label
Sigmapharm Laboratories, LLC
NDC 42794- 003-08
Adefovir Dipivoxil Tablets
10 mg
PHARMACIST: DISPENSE THE PATIENT INFORMATION LEAFLET WITH DRUG PRODUCT
Rx Only
30 Tablets
Principal Display Panel Adefovir Dipivoxil 10 Mg bottle Label
Sigmapharm Laboratories, LLC
NDC 42794-003-08
Adefovir Dipivoxil Tablets
10 mg
PHARMACIST: DISPENSE THE PATIENT INFORMATION LEAFLET WITH DRUG PRODUCT
Rx Only
30 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term oral carcinogenicity studies of adefovir dipivoxil in mice and rats were carried out at exposures up to approximately 10 times (mice) and 4 times (rats) those observed in humans at the therapeutic dose for HBV infection. In both mouse and rat studies, adefovir dipivoxil was negative for carcinogenic findings. Adefovir dipivoxil was mutagenic in the in vitro mouse lymphoma cell assay (with or without metabolic activation). Adefovir induced chromosomal aberrations in the in vitro human peripheral blood lymphocyte assay without metabolic activation. Adefovir dipivoxil was not clastogenic in the in vivo mouse micronucleus assay and adefovir was not mutagenic in the Ames bacterial reverse mutation assay using S. typhimurium and E. coli strains in the presence or absence of metabolic activation. In reproductive toxicology studies, no evidence of impaired fertility was seen in male or female rats at systemic exposure approximately 19 times that achieved in humans at the therapeutic dose.
14.3 Study 461 (clinical Evidence of Lamivudine Resistance)
In Study 461, a double-blind, active controlled study in 59 chronic hepatitis B patients with clinical evidence of lamivudine-resistant hepatitis B virus, patients were randomized to receive either Adefovir Dipivoxil Tablets monotherapy or Adefovir Dipivoxil Tablets in combination with lamivudine 100 mg or lamivudine 100 mg alone. At Week 48, the mean ± SD decrease in serum HBV DNA as measured by the Roche Amplicor Monitor PCR assay (LLOQ = 1000 copies/mL) was 4.00 ± 1.41 log 10 copies/mL for patients treated with Adefovir Dipivoxil Tablets and 3.46 ± 1.10 log 10 copies/mL for patients treated with Adefovir Dipivoxil Tablets in combination with lamivudine. There was a mean decrease in serum HBV DNA of 0.31 ± 0.93 log 10 copies/mL in patients receiving lamivudine alone. ALT normalized in 47% of patients treated with Adefovir Dipivoxil Tablets, in 53% of patients treated with Adefovir Dipivoxil Tablets in combination with lamivudine, and 5% of patients treated with lamivudine alone. The significance of these findings as they relate to clinical outcomes is not known.
14.2 Study 435 (pre and Post Liver Transplantation Patients)
Adefovir Dipivoxil Tablets were also evaluated in an open-label, uncontrolled study of 467 chronic hepatitis B patients pre- (N=226) and post- (N=241) liver transplantation with clinical evidence of lamivudine-resistant hepatitis B virus (Study 435). At baseline, 60% of pre-liver transplantation patients were classified as Child-Pugh-Turcotte score of Class B or C. The median baseline HBV DNA as measured by the Roche Amplicor Monitor PCR assay (LLOQ = 1000 copies/mL) was 7.4 and 8.2 log 10 copies/mL, and the median baseline ALT was 1.8 and 2.0 times the upper limit of normal in pre- and post-liver transplantation patients, respectively. Results of this study are displayed in Table 7. Treatment with Adefovir Dipivoxil Tablets resulted in a similar reduction in serum HBV DNA regardless of the patterns of lamivudine-resistant HBV DNA polymerase mutations at baseline. The significance of the efficacy results listed in Table 7 as they relate to clinical outcomes is not known.
Table 7 Efficacy in Pre- and Post-Liver Transplantation Patients at Week 48
| Efficacy Parameter a |
Pre-Liver Transplantation
(N=226) |
Post-Liver Transplantation
(N=241) |
| Mean change ± SD in HBV DNA from baseline (log 10 copies/mL) | -3.7 ± 1.6
(N=117) |
-4.0 ± 1.6
(N=164) |
| Proportion with undetectable HBV DNA (less than 1000 copies/mL) b | 77/109 (71%) | 64/159 (40%) |
| Stable or improved Child-Pugh-Turcotte score | 86/90 (96%) | 107/115 (93%) |
| Normalization of: c ALT | 61/82 (74%) | 56/110 (51%) |
| Albumin | 43/54 (80%) | 21/26 (81%) |
| Bilirubin | 38/68 (58%) | 29/38 (76%) |
| Prothrombin time | 39/46 (85%) | 5/9 (56%) |
a Data are missing for 29% (HBV DNA) and 37% to 45% (CPT Score, Normalization of ALT, Albumin, Bilirubin, and PT) of total patients enrolled in the study.
b Denominator is the number of patients with serum HBV DNA ≥1000 copies/mL at baseline using the Roche Amplicor Monitor PCR Assay (LLOQ = 1000 copies/mL) and non-missing value at Week 48.
c Denominator is patients with abnormal values at baseline and non-missing value at Week 48.
5.1 Exacerbation of Hepatitis After Discontinuation of Treatment
Severe acute exacerbation of hepatitis has been reported in patients who have discontinued anti-hepatitis B therapy, including therapy with Adefovir Dipivoxil Tablets. Hepatic function should be monitored at repeated intervals with both clinical and laboratory follow-up for at least several months in patients who discontinue Adefovir Dipivoxil Tablets. If appropriate, resumption of anti-hepatitis B therapy may be warranted.
In clinical trials of Adefovir Dipivoxil Tablets, exacerbations of hepatitis (ALT elevations 10 times the upper limit of normal or greater) occurred in up to 25% of patients after discontinuation of Adefovir Dipivoxil Tablets. These events were identified in studies GS-98-437 and GS-98-438 (N=492). Most of these events occurred within 12 weeks of drug discontinuation. These exacerbations generally occurred in the absence of HBeAg seroconversion, and presented as serum ALT elevations in addition to re-emergence of viral replication. In the HBeAg-positive and HBeAg-negative studies in patients with compensated liver function, the exacerbations were not generally accompanied by hepatic decompensation. However, patients with advanced liver disease or cirrhosis may be at higher risk for hepatic decompensation. Although most events appear to have been self-limited or resolved with re-initiation of treatment, severe hepatitis exacerbations, including fatalities, have been reported. Therefore, patients should be closely monitored after stopping treatment.
Warning: Severe Acute Exacerbations of Hepatitis, Nephrotoxicity, Hiv Resistance, Lactic Acidosis and Severe Hepatomegaly With Steatosis
Severe acute exacerbations of hepatitis have been reported in patients who have discontinued anti-Hepatitis B therapy including Adefovir Dipivoxil Tablets. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-Hepatitis B therapy. If appropriate, resumption of anti-Hepatitis B therapy may be warranted [See Warnings and Precautions (5.1)] .
In patients at risk of or having underlying renal dysfunction, chronic administration of Adefovir Dipivoxil Tablets may result in nephrotoxicity. These patients should be monitored closely for renal function and may require dose adjustment [See Warnings and Precautions (5.2) and Dosage and Administration (2.2)] .
HIV resistance may emerge in chronic hepatitis B patients with unrecognized or untreated Human Immunodeficiency Virus (HIV) infection treated with antihepatitis B therapies, such as therapy with Adefovir Dipivoxil Tablets, that may have activity against HIV [See Warnings and Precautions (5.3)] .
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with other antiretrovirals [See Warnings and Precautions (5 . 4 )] .
Structured Label Content
Data
Animal Data
In an embryo-fetal development study, ADV was administered orally to pregnant rabbits (at 1, 5, or 20 mg/kg/day) during organogenesis (on gestation day 6 through 18). No adverse developmental effects were observed at up to the highest dose tested, at systemic exposure (C max) 40 times that in humans at the RHD of Adefovir Dipivoxil Tablets.
In a pre/post-natal development study, ADV was administered orally to pregnant rats (at 2.5, 10, or 40 mg/kg/day) from organogenesis, through late gestation, delivery, and lactation (gestation day 7 to lactation/postpartum day 20). Reduced body weight of the offspring due to maternal toxicity was observed at systemic exposure 23 times that in humans at the RHD of Adefovir Dipivoxil Tablets.
In an embryo-fetal development study, ADV was administered intravenously to pregnant rats (at 2.5, 10, and 20 mg/kg/day) during organogenesis (gestation day 6 through 15). Embryo-fetal toxicity including malformations (anasarca, depressed eye bulge, umbilical hernia and kinked tail) and skeletal variations (reduction of ossified metacarpal bones, increases in thoracic vertebrae and decreases in lumbar vertebrae) occurred at systemic exposure (C max) 38 times that in humans at the RHD of Adefovir Dipivoxil Tablets. No adverse developmental effects were observed at an exposure (C max) 12 times that in humans at the RHD of Adefovir Dipivoxil Tablets.
Section 42229-5 (42229-5)
Pediatric Patients
The efficacy and safety of Adefovir Dipivoxil Tablets have not been studied in patients less than 18 years of age with different degrees of renal impairment and no data are available to make dosage recommendations in these patients [See Dosage and Administration (2.2)]. Caution should be exercised when prescribing Adefovir Dipivoxil Tablets to adolescents with underlying renal dysfunction, and renal function in these patients should be closely monitored.
Section 42230-3 (42230-3)
FDA-Approved Patient Labeling
PATIENT INFORMATION
ADEFOVIR DIPIVOXIL (a-DEF-oh-vir dip-ih-VOX-il) TABLETS
Read this Patient Information before you start taking Adefovir Dipivoxil Tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
What is the most important information I should know about Adefovir Dipivoxil Tablets?
Adefovir Dipivoxil Tablets can cause serious side effects, including:
-
Worsening of hepatitis B infection.Your hepatitis B (HBV) infection may become worse (flare-up) if you take Adefovir Dipivoxil Tablets and then stop taking it. A “flare-up” is when your HBV infection suddenly returns in a worse way than before.
- Do not run out of Adefovir Dipivoxil Tablets. Refill your prescription or talk to your doctor before your Adefovir Dipivoxil Tablets are all gone.
- Do not stop taking Adefovir Dipivoxil Tablets without first talking with your doctor.
- If you stop taking Adefovir Dipivoxil Tablets, your doctor will need to check your health often and do blood tests regularly to check your HBV infection for at least several months.
-
Adefovir Dipivoxil Tablets
may cause severe kidney problems. Severe kidney problems can happen in anyone who takes Adefovir Dipivoxil Tablets, but certain people may have a higher risk of severe kidney problems with Adefovir Dipivoxil Tablets including:
- people who already have kidney problems or are at an increased risk for kidney problems, and
- people who take certain medicines that can cause kidney problems. Ask your doctor whether any of the medicines you currently take can cause kidney problems.
Your doctor should do blood tests to check your kidney function during treatment with Adefovir Dipivoxil Tablets.
If you have kidney problems before you start taking Adefovir Dipivoxil Tablets your doctor may change your dose of Adefovir Dipivoxil Tablets. If you develop kidney problems during treatment, your doctor may need to change your dose of Adefovir Dipivoxil Tablets or may stop your treatment.
- HIV resistance. Your doctor may test you for HIV-1 infection before you start Adefovir Dipivoxil Tablets. If you have both HBV and HIV-1 and you only take Adefovir Dipivoxil Tablets, the HIV-1 virus may develop resistance and become harder to treat.
- Build-up of acid in your blood (lactic acidosis). Lactic acidosis can happen in some people who take Adefovir Dipivoxil Tablets. Lactic acidosis is a serious medical emergency that can lead to death.
Call your doctor right away if you get any of the following symptoms which could be signs of lactic acidosis:
|
|
- Severe liver problems. In rare cases, severe liver problems can happen that lead to death. Call your doctor right away if you get any of the following signs or symptoms of liver problems.
|
|
You may be more likely to get lactic acidosis or serious liver problems if you are female, are very overweight (obese), or have been taking nucleoside analog medicines for a long time.
What are Adefovir Dipivoxil Tablets?
Adefovir Dipivoxil Tablets are a medicine used to treat people 12 years of age and older with chronic (long-lasting) infections with active hepatitis B virus.
Adefovir Dipivoxil Tablets are not for use in children under 12 years of age.
- Adefovir Dipivoxil Tablets may lower the amount of hepatitis B virus (HBV) in your body.
- Adefovir Dipivoxil Tablets may improve the condition of your liver.
Who should not take Adefovir Dipivoxil Tablets?
Do not take Adefovir Dipivoxil Tablets if you are allergic to any of the ingredients in Adefovir Dipivoxil Tablets. See the end of this leaflet for a complete list of the ingredients in Adefovir Dipivoxil Tablets.
What should I tell my healthcare provider before taking Adefovir Dipivoxil Tablets?
Before taking Adefovir Dipivoxil Tablets, tell your doctor about all of your medical
conditions, including if you:
- have or had kidney problems. Your dose and schedule of Adefovir Dipivoxil Tablets may need to be changed.
- are pregnant or plan to become pregnant. It is not known if Adefovir Dipivoxil Tablets will harm your unborn baby.
Pregnancy Registry: There is a pregnancy registry for women who take Adefovir Dipivoxil Tablets during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk with your doctor about how you can take part in this registry.
- are breastfeeding. It is not known if adefovir dipivoxil passes into your breast milk. Talk with your doctor about the best way to feed your baby during treatment with Adefovir Dipivoxil Tablets.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines may affect how Adefovir Dipivoxil Tablets work, especially medicines that affect how your kidneys work. Adefovir Dipivoxil Tablets can affect how your other medicines work. Your dose of Adefovir Dipivoxil Tablets and the other medicines may need to be changed. Do not start a new medicine without telling your doctor. Your doctor can tell you if it is safe to take Adefovir Dipivoxil Tablets with other medicines.
How should I take Adefovir Dipivoxil Tablets?
- Take Adefovir Dipivoxil Tablets exactly as your doctor tells you to take it.
- Your doctor will tell you how much Adefovir Dipivoxil Tablets to take, and when and how often to take it.
- Do not change your dose or stop Adefovir Dipivoxil Tablets without talking to your doctor. Your hepatitis may get worse if you change doses or stop.
- Take Adefovir Dipivoxil Tablets at the same time each day that your doctor tells you, to avoid missing doses.
- Take Adefovir Dipivoxil Tablets with or without food.
- Stay under the care of your doctor during treatment with Adefovir Dipivoxil Tablets.
- When your Adefovir Dipivoxil Tablets supply gets low, call your doctor or pharmacy for a refill. Do not run out of Adefovir Dipivoxil Tablets.
- If you take too much Adefovir Dipivoxil Tablets, call your doctor or go to the nearest hospital emergency room right away.
- It is not known how long you should take Adefovir Dipivoxil Tablets. You and your doctor will need to decide when it is best for you to stop taking Adefovir Dipivoxil Tablets. Some people get worsening of their hepatitis B infection when they stop taking Adefovir Dipivoxil Tablets. See “What is the most important information I should know about Adefovir Dipivoxil Tablets?".
What are the possible side effects of Adefovir Dipivoxil Tablets?
Adefovir Dipivoxil Tablets can cause serious side effects. See “What is the most important information I should know about Adefovir Dipivoxil Tablets?"
The most common side effects of Adefovir Dipivoxil Tablets are weakness, headache, stomach pain, and nausea.
These are not all the possible side effects of Adefovir Dipivoxil Tablets. For more information, ask your doctor or pharmacist.
Call your doctor for advice about side effects. You may report side effects to FDA at 1-800-FDA-1088
How should I store Adefovir Dipivoxil Tablets?
- Store Adefovir Dipivoxil Tablets at room temperature, between 68°F to 77°F (20°C to 25°C).
- Keep Adefovir Dipivoxil Tablets in its original container.
- Do not use Adefovir Dipivoxil Tablets if the seal over the bottle is broken or missing when you receive it.
Keep Adefovir Dipivoxil Tablets and all medicines out of the reach of children.
General information about the safe and effective use of Adefovir Dipivoxil Tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Adefovir Dipivoxil Tablets for a condition for which it was not prescribed. Do not give Adefovir Dipivoxil Tablets to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about
Adefovir Dipivoxil Tablets that is written for health professionals.
What are the Ingredients in Adefovir dipivoxil tablets?
Active Ingredient: adefovir dipivoxil
Inactive ingredients: copovidone, anhydrous lactose, microcrystalline cellulose, silicon dioxide, crospovidone and magnesium stearate
Manufactured by:
Sigmapharm Laboratories, LLC
Bensalem, PA 19020
HEPSERA, ATRIPLA, COMPLERA, EMTRIVA, STRIBILD, TRUVADA, and VIREAD are trademarks or registered trademarks of Gilead Sciences, Inc., or its related companies. Other brands listed are the trademarks of their respective owners.
OS003-09 REV.0821
This Patient Information has been approved by the U.S. Food and Drug Administration.
Section 44425-7 (44425-7)
Store in original container at 25 °C (77 °F), excursions permitted to 15° - 30 °C (59° - 86 °F) (See USP Controlled Room Temperature).
Do not use if seal over bottle opening is broken or missing.
10 Overdosage (10 OVERDOSAGE)
Doses of adefovir dipivoxil 500 mg daily for 2 weeks and 250 mg daily for 12 weeks have been associated with gastrointestinal side effects. If overdose occurs the patient must be monitored for evidence of toxicity, and standard supportive treatment applied as necessary.
Following a 10 mg single dose of Adefovir Dipivoxil Tablets, a four-hour hemodialysis session removed approximately 35% of the adefovir dose.
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to Adefovir Dipivoxil Tablets during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
8.2 Lactation
Risk Summary
It is not known whether adefovir is present in human breast milk, affects human milk production, or has effects on the breastfed infant.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Adefovir Dipivoxil Tablets and any potential adverse effects on the breastfed infant from Adefovir Dipivoxil Tablets or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Adefovir dipivoxil is a diester prodrug of adefovir. Adefovir is an acyclic nucleotide analog with activity against human hepatitis B virus (HBV).
The chemical name of adefovir dipivoxil is 9-[2-[[bis[(pivaloyloxy)methoxy]-phosphinyl]-methoxy]ethyl]adenine. It has a molecular formula of C 20H 32N 5O 8P, a molecular weight of 501.48 and the following structural formula:
Adefovir dipivoxil is a white to off-white crystalline powder with an aqueous solubility of 19 mg/mL at pH 2.0 and 0.4 mg/mL at pH 7.2. It has an octanol/aqueous phosphate buffer (pH 7) partition coefficient (log p) of 1.91.
Adefovir Dipivoxil Tablets are for oral administration. Each tablet contains 10 mg of adefovir dipivoxil and the following inactive ingredients: copovidone, anhydrous lactose, microcrystalline cellulose, silicon dioxide, crospovidone and magnesium stearate.
12.4 Microbiology
8.4 Pediatric Use
8.5 Geriatric Use
Clinical studies of Adefovir Dipivoxil Tablets did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. In general, caution should be exercised when prescribing to elderly patients since they have greater frequency of decreased renal or cardiac function due to concomitant disease or other drug therapy.
5.2 Nephrotoxicity
Nephrotoxicity characterized by a delayed onset of gradual increases in serum creatinine and decreases in serum phosphorus was historically shown to be the treatment-limiting toxicity of adefovir dipivoxil therapy at substantially higher doses in HIV-infected patients (60 and 120 mg daily) and in chronic hepatitis B patients (30 mg daily). Chronic administration of Adefovir Dipivoxil Tablets (10 mg once daily) may result in delayed nephrotoxicity. The overall risk of nephrotoxicity in patients with adequate renal function is low. However, this is of special importance in patients at risk of or having underlying renal dysfunction and patients taking concomitant nephrotoxic agents such as cyclosporine, tacrolimus, aminoglycosides, vancomycin and non-steroidal anti-inflammatory drugs [See Adverse Reactions (6.2) and Clinical Pharmacology (12.3)] . It is recommended that creatinine clearance is calculated in all patients prior to initiating therapy with Adefovir Dipivoxil Tablets.
It is important to monitor renal function for all patients during treatment with Adefovir Dipivoxil Tablets, particularly for those with pre-existing or other risks for renal impairment. Patients with renal insufficiency at baseline or during treatment may require dose adjustment [See Dosage and Administration (2.2)] . The risks and benefits of Adefovir Dipivoxil Tablets treatment should be carefully evaluated prior to discontinuing Adefovir Dipivoxil Tablets in a patient with treatment-emergent nephrotoxicity.
5.3 Hiv Resistance (5.3 HIV Resistance)
Prior to initiating adefovir dipivoxil therapy, HIV antibody testing should be offered to all patients. Treatment with anti-hepatitis B therapies, such as Adefovir Dipivoxil Tablets, that have activity against HIV in a chronic hepatitis B patient with unrecognized or untreated HIV infection may result in emergence of HIV resistance. Adefovir Dipivoxil Tablets has not been shown to suppress HIV RNA in patients; however, there are limited data on the use of Adefovir Dipivoxil Tablets to treat patients with chronic hepatitis B co-infected with HIV.
14 Clinical Studies (14 CLINICAL STUDIES)
4 Contraindications (4 CONTRAINDICATIONS)
Adefovir Dipivoxil Tablets are contraindicated in patients with previously demonstrated hypersensitivity to any of the components of the product.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in other sections of the labeling:
- Severe acute exacerbations of Hepatitis [See Boxed Warning, Warnings and Precautions (5.1)]
- Nephrotoxicity [See Boxed Warning, Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Since adefovir is eliminated by the kidney, coadministration of Adefovir Dipivoxil Tablets with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either adefovir and/or these coadministered drugs [See Clinical Pharmacology (12.3)] .
Patients should be monitored closely for adverse events when Adefovir Dipivoxil Tablets are coadministered with drugs that are excreted renally or with other drugs known to affect renal function [See Warnings and Precautions (5.2)].
Adefovir Dipivoxil Tablets should not be administered in combination with VIREAD [See Warnings and Precautions (5.5)].
1 indications and Usage (1 INDICATIONS AND USAGE)
Adefovir Dipivoxil Tablets are indicated for the treatment of chronic hepatitis B in patients 12 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
This indication is based on histological, virological, biochemical, and serological responses in adult patients with HBeAg+ and HBeAg- chronic hepatitis B with compensated liver function, and with clinical evidence of lamivudine-resistant hepatitis B virus with either compensated or decompensated liver function.
For patients 12 to less than 18 years of age, the indication is based on virological and biochemical responses in patients with HBeAg+ chronic hepatitis B virus infection with compensated liver function.
2.1 chronic Hepatitis B (2.1 Chronic Hepatitis B)
The recommended dose of Adefovir Dipivoxil Tablets in chronic hepatitis B patients for patients 12 years of age and older with adequate renal function is 10 mg, once daily, taken orally, without regard to food. The optimal duration of treatment is unknown.
Adefovir Dipivoxil Tablets is not recommended for use in children less than 12 years of age.
6.3 Pediatric Patients
Assessment of adverse reactions is based on a placebo-controlled study (Study 518) in which 173 pediatric patients aged 2 to less than 18 years with chronic hepatitis B and compensated liver disease received double-blind treatment with Adefovir Dipivoxil Tablets (N=115), or placebo (N=58) for 48 weeks [See Clinical Studies (14.4) and Use In Specific Populations (8.4)] .
The safety profile of Adefovir Dipivoxil Tablets in patients 12 to less than 18 years of age (N=56) was similar to that observed in adults. No pediatric patients treated with Adefovir Dipivoxil Tablets developed a confirmed serum creatinine increase greater than or equal to 0.5 mg/dL from baseline or a confirmed phosphorus decrease to less than 2 mg/dL by Week 48.
5.6 Clinical Resistance
Resistance to adefovir dipivoxil can result in viral load rebound which may result in exacerbation of hepatitis B and, in the setting of diminished hepatic function, lead to liver decompensation and possible fatal outcome.
In order to reduce the risk of resistance in patients with lamivudine resistant HBV, adefovir dipivoxil should be used in combination with lamivudine and not as adefovir dipivoxil monotherapy.
In order to reduce the risk of resistance in all patients receiving adefovir dipivoxil monotherapy, a modification of treatment should be considered if serum HBV DNA remains above 1000 copies/mL with continued treatment.
Long-term (144 week) data from Study 438 (N=124) show that patients with HBV DNA levels greater than 1000 copies/mL at Week 48 of treatment with adefovir dipivoxil were at greater risk of developing resistance than patients with serum HBV DNA levels below 1000 copies/mL at Week 48 of therapy.
12.1 Mechanism of Action
Adefovir is an antiviral drug [See Microbiology (12.4)].
13 Nonclinical Toxicology (13 NONCLINICAL TOXICOLOGY)
6.2 Special Risk Patients
Pre- and Post-Liver Transplantation Patients
Additional adverse reactions observed from an open-label study (Study 435) in pre- and post- liver transplantation patients with chronic hepatitis B and lamivudine-resistant hepatitis B administered Adefovir Dipivoxil Tablets once daily for up to 203 weeks include: abnormal renal function, renal failure, vomiting, rash, and pruritus.
Changes in renal function occurred in pre-and post-liver transplantation patients with risk factors for renal dysfunction, including concomitant use of cyclosporine and tacrolimus, renal insufficiency at baseline, hypertension, diabetes, and on-study transplantation. Therefore, the contributory role of Adefovir Dipivoxil Tablets to these changes in renal function is difficult to assess.
Increases in serum creatinine greater than or equal to 0.3 mg/dL from baseline were observed in 37% and 53% of pre-liver transplantation patients by Weeks 48 and 96, respectively, by Kaplan-Meier estimates. Increases in serum creatinine greater than or equal to 0.3 mg/dL from baseline were observed in 32% and 51% of post-liver transplantation patients by Weeks 48 and 96, respectively, by Kaplan-Meier estimates. Serum phosphorus values less than 2 mg/dL were observed in 3/226 (1.3%) of pre-liver transplantation patients and in 6/241 (2.5%) of post-liver transplantation patients by last study visit. Four percent (19 of 467) of patients discontinued treatment with Adefovir Dipivoxil Tablets due to renal adverse events.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- One tablet containing 10 mg adefovir dipivoxil once daily orally with or without food. ( 2.1)
- Dose adjustment in renal impairment for adults ( 2.2)
| Creatinine Clearance (mL/min) a | Hemodialysis Patients | |||
| Greater than or equal to 50 | 30 - 49 | 10 - 29 | ||
| Recommended dose and dosing interval | 10 mg every 24 hours | 10 mg every 48 hours | 10 mg every 72 hours | 10 mg every 7 days following dialysis |
a Creatinine clearance calculated by Cockcroft-Gault method using lean or ideal body weight.
- No dose recommendations for (
2.1):
- Non-hemodialysis patients with creatinine clearance less than 10 mL per minute.
- Adolescent patients with renal impairment.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed Σ 3 on one side and plain on the other side.
8 Use in Specific Population (8 USE IN SPECIFIC POPULATION)
6.4 Post Marketing Experience (6.4 Post-Marketing Experience)
In addition to adverse reaction reports from clinical trials, the following possible adverse reactions have also been identified during post-approval use of adefovir dipivoxil. Because these events have been reported voluntarily from a population of unknown size, estimates of frequency cannot be made.
Metabolism and Nutrition Disorders: hypophosphatemia
Gastrointestinal Disorders: pancreatitis
Musculoskeletal System and Connective Tissue Disorders: myopathy, osteomalacia (manifested as bone pain and may contribute to fractures), both associated with proximal renal tubulopathy
Renal and Urinary Disorders: renal failure, Fanconi syndrome, proximal renal tubulopathy
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical and laboratory evidence of exacerbations of hepatitis have occurred after discontinuation of treatment with Adefovir Dipivoxil Tablets.
Adverse reactions to Adefovir Dipivoxil Tablets identified from placebo-controlled and open label studies include the following: asthenia, headache, abdominal pain, diarrhea, nausea, dyspepsia, flatulence, increased creatinine, and hypophosphatemia.
The incidence of these adverse reactions in studies 437 and 438, where 522 patients with chronic hepatitis B and compensated liver disease received double-blind treatment with Adefovir Dipivoxil Tablets (N=294) or placebo (N=228) for 48 weeks is presented in Table 2. Patients who received open-label Adefovir Dipivoxil Tablets for up to 240 weeks in Study 438 reported adverse reactions similar in nature and severity to those reported in the first 48 weeks.
| Adverse Reaction | Adefovir Dipivoxil Tablets 10 mg (N=294) | Placebo (N=228) |
| Asthenia | 13% | 14% |
| Headache | 9% | 10% |
| Abdominal Pain | 9% | 11% |
| Nausea | 5% | 8% |
| Flatulence | 4% | 4% |
| Diarrhea | 3% | 4% |
| Dyspepsia | 3% | 2% |
a In these studies, the overall incidence of adverse reactions with Adefovir Dipivoxil Tablets was similar to that reported with placebo. The incidence of adverse reactions is derived from treatment-related events as identified by the study investigators.
No patients treated with Adefovir Dipivoxil Tablets developed a confirmed serum creatinine increase greater than or equal to 0.5 mg/dL from baseline or confirmed phosphorus decrease to 2 mg/dL or less by Week 48. By Week 96, 2% of Adefovir Dipivoxil Tablets-treated patients, by Kaplan-Meier estimate, had increases in serum creatinine greater than or equal to 0.5 mg/dL from baseline (no placebo-controlled results were available for comparison beyond Week 48). For patients who chose to continue Adefovir Dipivoxil Tablets for up to 240 weeks in Study 438, 4 of 125 patients (3%) had a confirmed increase of 0.5 mg/dL from baseline. The creatinine elevation resolved in 1 patient who permanently discontinued treatment and remained stable in 3 patients who continued treatment. For 65 patients who chose to continue Adefovir Dipivoxil Tablets for up to 240 weeks in Study 437, 6 had a confirmed increase in serum creatinine of greater than or equal to 0.5 mg/dL from baseline with 2 patients discontinuing from the study due to the elevated serum creatinine concentration. See Adverse Reactions (6.2) for changes in serum creatinine in patients with underlying renal insufficiency at baseline.
14.4 Study 518 (pediatric Study) (14.4 Study 518 (Pediatric Study))
Study 518 was a double-blind, placebo-controlled, study in which 173 pediatric patients (ages 2 to less than 18 years) with chronic hepatitis B (CHB) infection and elevated ALT were randomized 2:1 (115 receiving adefovir dipivoxil and 58 receiving placebo). Randomization was stratified by prior treatment and age 2 to less than 7 years old (cohort 1), 7 to less than 12 years old (cohort 2), and 12 to less than 18 years old (cohort 3). All patients in cohort 3 received 10 mg tablet formulation; all patients in cohorts 1 and 2 received an investigational suspension formulation (0.3 mg/kg/day cohort 1, 0.25 mg/kg/day cohort 2) once daily. The primary efficacy endpoint was HBV DNA less than 1000 copies/mL plus normalization of ALT at the end of Week 48.
In cohort 3 (N=83), significantly more patients treated with Adefovir Dipivoxil Tablets achieved the primary efficacy endpoint at the end of 48 weeks of blinded treatment (23%) when compared to placebo-treated patients (0%). The proportion of patients from cohorts 1 and 2 who responded to treatment with adefovir dipivoxil was not statistically significant when compared to the placebo arm, although the adefovir plasma concentrations in these patients were comparable to those observed in older patients. Overall, 22 of 115 (19%) of pediatric patients who received adefovir dipivoxil versus 1 of 58 (2%) of placebo treated patients responded to treatment by Week 48 [See Adverse Reactions (6.3), Use In Special Populations (8.4) and Clinical Pharmacology (12.3, 12.4)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Inform patients of the potential risks and benefits of Adefovir Dipivoxil Tablets and of alternative modes of therapy.
- Instruct patients to:
-Follow a regular dosing schedule to avoid missing doses.
-Immediately report any severe abdominal pain, muscle pain, yellowing of the eyes, dark urine, pale stools, and/or loss in appetite.
-Inform their doctor or pharmacist if they develop any unusual symptom(s), or if any known symptom persists or worsens.
- Advise patients that:
- The optimal duration of Adefovir Dipivoxil Tablets treatment and the relationship between treatment response and long-term outcomes such as hepatocellular carcinoma or decompensated cirrhosis are not known.
- Patients should not discontinue Adefovir Dipivoxil Tablets without first informing their physician [See Warnings and Precautions (5.1)].
- Routine laboratory monitoring and follow-up with a physician is important during Adefovir Dipivoxil Tablets therapy.
- Obtaining HIV antibody testing prior to starting Adefovir Dipivoxil Tablets is important [See Warnings and Precautions (5.3)].
- Adefovir Dipivoxil Tablets should not be administered concurrently with ATRIPLA or COMPLERA or STRIBILD or TRUVADA or VIREAD [See Warnings and Precautions (5.5)].
- Lamivudine-resistant patients should use Adefovir Dipivoxil Tablets in combination with lamivudine and not as Adefovir Dipivoxil Tablets monotherapy [See Warnings and Precautions(5.6)].
- Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant women exposed to Adefovir Dipivoxil [ see Use in Specific Populations (8.1)] .
Manufactured by:
Sigmapharm Laboratories, LLC
Bensalem, PA 19020
OS003-09 REV.0821
16 How Supplied / Storage and Handling (16 HOW SUPPLIED / STORAGE AND HANDLING)
Adefovir Dipivoxil is available as tablets. Each tablet contains 10 mg of adefovir dipivoxil. The tablets are white, round, flat faced beveled edged tablets, debossed ∑3 on one side and plain on the other side. They are packaged as follows: Bottles of 30 tablets (NDC 42794-003-08) containing polyester and desiccant and closed with a child-resistant closure.
2.2 dose Adjustment in Renal Impairment (2.2 Dose Adjustment in Renal Impairment)
Significantly increased drug exposures were seen when Adefovir Dipivoxil Tablets was administered to adult patients with renal impairment [See Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. Therefore, the dosing interval of Adefovir Dipivoxil Tablets should be adjusted in adult patients with baseline creatinine clearance less than 50 mL per minute using the following suggested guidelines (See Table 1). The safety and effectiveness of these dosing interval adjustment guidelines have not been clinically evaluated.
Additionally, it is important to note that these guidelines were derived from data in patients with pre-existing renal impairment at baseline. They may not be appropriate for patients in whom renal insufficiency evolves during treatment with Adefovir Dipivoxil Tablets. Therefore, clinical response to treatment and renal function should be closely monitored in these patients.
Table 1 Dosing Interval Adjustment of Adefovir Dipivoxil Tablets in Adult Patients with Renal Impairment
| Creatinine Clearance (mL/min) a | Hemodialysis Patients | |||
| Greater than or equal to 50 | 30 - 49 | 10 - 29 | ||
| Recommended dose and dosing interval | 10 mg every 24 hours | 10 mg every 48 hours | 10 mg every 72 hours | 10 mg every 7 days following dialysis |
a Creatinine clearance calculated by Cockcroft-Gault method using lean or ideal body weight.
The pharmacokinetics of adefovir have not been evaluated in non-hemodialysis patients with creatinine clearance less than 10 mL per minute; therefore, no dosing recommendation is available for these patients.
No clinical data are available to make dosing recommendations in adolescent patients with renal insufficiency [See Warnings and Precautions (5.2)]
5.5 Coadministration With Other Products (5.5 Coadministration with Other Products)
Adefovir Dipivoxil Tablets should not be used concurrently with products containing tenofovir disoproxil fumarate or tenofovir alafenamide.
8.6 Patients With Impaired Renal Function (8.6 Patients with Impaired Renal Function)
It is recommended that the dosing interval for Adefovir Dipivoxil Tablets be modified in adult patients with baseline creatinine clearance less than 50 mL per minute. The pharmacokinetics of adefovir have not been evaluated in non-hemodialysis patients with creatinine clearance less than 10 mL per minute or in adolescent patients with renal insufficiency; therefore, no dosing recommendations are available for these patients [See Dosage and Administration (2.2) and Warning and Precautions (5.2)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Renal tubular nephropathy characterized by histological alterations and/or increases in BUN and serum creatinine was the primary dose-limiting toxicity associated with administration of adefovir dipivoxil in animals. Nephrotoxicity was observed in animals at systemic exposures approximately 3 - 10 times higher than those in humans at the recommended therapeutic dose of 10 mg/day.
14.1 Studies 437 and 438 (pivotal Studies) (14.1 Studies 437 and 438 (Pivotal Studies))
HBeAg-Positive Chronic Hepatitis B
Study 437 was a randomized, double-blind, placebo-controlled, three-arm-study in patients with HBeAg-positive chronic hepatitis B that allowed for a comparison between placebo and Adefovir Dipivoxil Tablets. The median age of patients was 33 years. Seventy-four percent were male, 59% were Asian, 36% were Caucasian, and 24% had prior interferon-α treatment. At baseline, patients had a median total Knodell Histology Activity Index (HAI) score of 10, a median serum HBV DNA level as measured by the Roche Amplicor Monitor polymerase chain reaction (PCR) assay (LLOQ = 1000 copies/mL) of 8.36 log 10 copies/mL and a median ALT level of 2.3 times the upper limit of normal.
HBeAg-Negative (Anti-HBe Positive/HBV DNA Positive) Chronic Hepatitis B
Study 438 was a randomized, double-blind, placebo-controlled study in patients who were HBeAg-negative at screening, and anti-HBe positive. The median age of patients was 46 years. Eighty-three percent were male, 66% were Caucasian, 30% were Asian and 41% had prior interferon-α treatment. At baseline, the median total Knodell HAI score was 10, the median serum HBV DNA level as measured by the Roche Amplicor Monitor PCR assay (LLOQ = 1000 copies/mL) was 7.08 log 10 copies/mL, and the median ALT was 2.3 times the upper limit of normal.
The primary efficacy endpoint in both studies was histological improvement at Week 48; results of which are shown in Table 4.
| Study 437 | Study 438 | |||
|
Adefovir Dipivoxil Tablets
10 mg (N=168) |
Placebo
(N=161) |
Adefovir Dipivoxil Tablets 10 mg
(N=121) |
Placebo
(N=57) |
|
| Improvement b | 53% | 25% | 64% | 35% |
| No Improvement | 37% | 67% | 29% | 63% |
| Missing/Unassessable Data | 10% | 7% | 7% | 2% |
a Intent-to-Treat population (patients with ≥1 dose of study drug) with assessable baseline biopsies.
b Histological improvement defined as ≥2 point decrease in the Knodell necro-inflammatory score with no worsening of the Knodell fibrosis score.
Table 5 illustrates the changes in Ishak Fibrosis Score by treatment group.
Table 5 Changes in Ishak Fibrosis Score at Week 48
|
Number of Adequate
Biopsy Pairs |
Study 437 | Study 438 | ||
|
Adefovir Dipivoxil Tablets 10 mg
(N=152) |
Placebo
(N=149) |
Adefovir Dipivoxil Tablets 10 mg
(N=113) |
Placebo
(N=56) |
|
| Ishak Fibrosis Score
Improved a |
34% | 19% | 34% | 14% |
| Unchanged | 55% | 60% | 62% | 50% |
| Worsened a | 11% | 21% | 4% | 36% |
a Change of 1 point or more in Ishak Fibrosis Score.
At Week 48, improvement was seen with respect to mean change in serum HBV DNA (log 10 copies/mL), normalization of ALT, and HBeAg seroconversion as compared to placebo in patients receiving Adefovir Dipivoxil Tablets (Table 6).
Table 6 Change in Serum HBV DNA, ALT Normalization, and HBeAg Seroconversion at Week 48
| Study 437 | Study 438 | |||
|
Adefovir Dipivoxil Tablets 10 mg
(N=171) |
Placebo
(N=167) |
Adefovir Dipivoxil Tablets 10 mg
(N=123) |
Placebo
(N=61) |
|
| Mean change ± SD in serum HBV DNA form baseline (log 10 copies/mL) | -3.57 ± 1.64 | -0.98 ± 1.32 | -3.65 ± 1.14 | -1.32 ± 1.25 |
| ALT normalization | 48% | 16% | 72% | 29% |
| HBeAg seroconversion | 12% | 6% | NA a | NA a |
a Patients with HBeAg-negative disease cannot undergo HBeAg seroconversion.
Treatment Beyond 48 Weeks
In Study 437, continued treatment with Adefovir Dipivoxil Tablets to 72 weeks resulted in continued maintenance of mean reductions in serum HBV DNA observed at Week 48. An increase in the proportion of patients with ALT normalization was also observed in Study 437. The effect of continued treatment with Adefovir Dipivoxil Tablets on seroconversion is unknown.
In Study 438, patients who received Adefovir Dipivoxil Tablets during the first 48 weeks were re-randomized in a blinded manner to continue on Adefovir Dipivoxil Tablets or receive placebo for an additional 48 weeks. At Week 96, 50 of 70 (71%) of patients who continued treatment with Adefovir Dipivoxil Tablets had undetectable HBV DNA levels (less than 1000 copies/mL), and 47 of 64 (73%) of patients had ALT normalization. HBV DNA and ALT levels returned towards baseline in most patients who stopped treatment with Adefovir Dipivoxil Tablets.
From 141 eligible patients, there were 125 (89%) patients in Study 438 who chose to continue Adefovir Dipivoxil Tablets for up to 192 weeks or 240 weeks (4 years or 5 years). As these patients had already received Adefovir Dipivoxil Tablets for at least 48 weeks and appeared to be experiencing a benefit, they are not necessarily representative of patients initiating Adefovir Dipivoxil Tablets. Of these patients, 89/125 (71%) and 47/70 (67%) had an undetectable HBV DNA level (less than 1000 copies/mL) at Week 192 and Week 240, respectively. Of the patients who had an elevated ALT at baseline, 77/104 (74%) and 42/64 (66%) had a normal ALT at Week 192 and Week 240, respectively. Six (5%) patients experienced HBsAg loss.
5.4 Lactic Acidosis/severe Hepatomegaly With Steatosis (5.4 Lactic Acidosis/Severe Hepatomegaly with Steatosis)
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with antiretrovirals.
A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Particular caution should be exercised when administering nucleoside analogs to any patient with known risk factors for liver disease; however, cases have also been reported in patients with no known risk factors. Treatment with Adefovir Dipivoxil Tablets therapy should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
Principal Display Panel Adefovir Dipivoxil 10 Mg Carton Label (PRINCIPAL DISPLAY PANEL- Adefovir Dipivoxil 10 mg Carton Label)
Sigmapharm Laboratories, LLC
NDC 42794- 003-08
Adefovir Dipivoxil Tablets
10 mg
PHARMACIST: DISPENSE THE PATIENT INFORMATION LEAFLET WITH DRUG PRODUCT
Rx Only
30 Tablets
Principal Display Panel Adefovir Dipivoxil 10 Mg bottle Label (PRINCIPAL DISPLAY PANEL- Adefovir Dipivoxil 10 mg Bottle Label)
Sigmapharm Laboratories, LLC
NDC 42794-003-08
Adefovir Dipivoxil Tablets
10 mg
PHARMACIST: DISPENSE THE PATIENT INFORMATION LEAFLET WITH DRUG PRODUCT
Rx Only
30 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term oral carcinogenicity studies of adefovir dipivoxil in mice and rats were carried out at exposures up to approximately 10 times (mice) and 4 times (rats) those observed in humans at the therapeutic dose for HBV infection. In both mouse and rat studies, adefovir dipivoxil was negative for carcinogenic findings. Adefovir dipivoxil was mutagenic in the in vitro mouse lymphoma cell assay (with or without metabolic activation). Adefovir induced chromosomal aberrations in the in vitro human peripheral blood lymphocyte assay without metabolic activation. Adefovir dipivoxil was not clastogenic in the in vivo mouse micronucleus assay and adefovir was not mutagenic in the Ames bacterial reverse mutation assay using S. typhimurium and E. coli strains in the presence or absence of metabolic activation. In reproductive toxicology studies, no evidence of impaired fertility was seen in male or female rats at systemic exposure approximately 19 times that achieved in humans at the therapeutic dose.
14.3 Study 461 (clinical Evidence of Lamivudine Resistance) (14.3 Study 461 (Clinical Evidence of Lamivudine Resistance))
In Study 461, a double-blind, active controlled study in 59 chronic hepatitis B patients with clinical evidence of lamivudine-resistant hepatitis B virus, patients were randomized to receive either Adefovir Dipivoxil Tablets monotherapy or Adefovir Dipivoxil Tablets in combination with lamivudine 100 mg or lamivudine 100 mg alone. At Week 48, the mean ± SD decrease in serum HBV DNA as measured by the Roche Amplicor Monitor PCR assay (LLOQ = 1000 copies/mL) was 4.00 ± 1.41 log 10 copies/mL for patients treated with Adefovir Dipivoxil Tablets and 3.46 ± 1.10 log 10 copies/mL for patients treated with Adefovir Dipivoxil Tablets in combination with lamivudine. There was a mean decrease in serum HBV DNA of 0.31 ± 0.93 log 10 copies/mL in patients receiving lamivudine alone. ALT normalized in 47% of patients treated with Adefovir Dipivoxil Tablets, in 53% of patients treated with Adefovir Dipivoxil Tablets in combination with lamivudine, and 5% of patients treated with lamivudine alone. The significance of these findings as they relate to clinical outcomes is not known.
14.2 Study 435 (pre and Post Liver Transplantation Patients) (14.2 Study 435 (Pre- and Post-Liver Transplantation Patients))
Adefovir Dipivoxil Tablets were also evaluated in an open-label, uncontrolled study of 467 chronic hepatitis B patients pre- (N=226) and post- (N=241) liver transplantation with clinical evidence of lamivudine-resistant hepatitis B virus (Study 435). At baseline, 60% of pre-liver transplantation patients were classified as Child-Pugh-Turcotte score of Class B or C. The median baseline HBV DNA as measured by the Roche Amplicor Monitor PCR assay (LLOQ = 1000 copies/mL) was 7.4 and 8.2 log 10 copies/mL, and the median baseline ALT was 1.8 and 2.0 times the upper limit of normal in pre- and post-liver transplantation patients, respectively. Results of this study are displayed in Table 7. Treatment with Adefovir Dipivoxil Tablets resulted in a similar reduction in serum HBV DNA regardless of the patterns of lamivudine-resistant HBV DNA polymerase mutations at baseline. The significance of the efficacy results listed in Table 7 as they relate to clinical outcomes is not known.
Table 7 Efficacy in Pre- and Post-Liver Transplantation Patients at Week 48
| Efficacy Parameter a |
Pre-Liver Transplantation
(N=226) |
Post-Liver Transplantation
(N=241) |
| Mean change ± SD in HBV DNA from baseline (log 10 copies/mL) | -3.7 ± 1.6
(N=117) |
-4.0 ± 1.6
(N=164) |
| Proportion with undetectable HBV DNA (less than 1000 copies/mL) b | 77/109 (71%) | 64/159 (40%) |
| Stable or improved Child-Pugh-Turcotte score | 86/90 (96%) | 107/115 (93%) |
| Normalization of: c ALT | 61/82 (74%) | 56/110 (51%) |
| Albumin | 43/54 (80%) | 21/26 (81%) |
| Bilirubin | 38/68 (58%) | 29/38 (76%) |
| Prothrombin time | 39/46 (85%) | 5/9 (56%) |
a Data are missing for 29% (HBV DNA) and 37% to 45% (CPT Score, Normalization of ALT, Albumin, Bilirubin, and PT) of total patients enrolled in the study.
b Denominator is the number of patients with serum HBV DNA ≥1000 copies/mL at baseline using the Roche Amplicor Monitor PCR Assay (LLOQ = 1000 copies/mL) and non-missing value at Week 48.
c Denominator is patients with abnormal values at baseline and non-missing value at Week 48.
5.1 Exacerbation of Hepatitis After Discontinuation of Treatment (5.1 Exacerbation of Hepatitis after Discontinuation of Treatment)
Severe acute exacerbation of hepatitis has been reported in patients who have discontinued anti-hepatitis B therapy, including therapy with Adefovir Dipivoxil Tablets. Hepatic function should be monitored at repeated intervals with both clinical and laboratory follow-up for at least several months in patients who discontinue Adefovir Dipivoxil Tablets. If appropriate, resumption of anti-hepatitis B therapy may be warranted.
In clinical trials of Adefovir Dipivoxil Tablets, exacerbations of hepatitis (ALT elevations 10 times the upper limit of normal or greater) occurred in up to 25% of patients after discontinuation of Adefovir Dipivoxil Tablets. These events were identified in studies GS-98-437 and GS-98-438 (N=492). Most of these events occurred within 12 weeks of drug discontinuation. These exacerbations generally occurred in the absence of HBeAg seroconversion, and presented as serum ALT elevations in addition to re-emergence of viral replication. In the HBeAg-positive and HBeAg-negative studies in patients with compensated liver function, the exacerbations were not generally accompanied by hepatic decompensation. However, patients with advanced liver disease or cirrhosis may be at higher risk for hepatic decompensation. Although most events appear to have been self-limited or resolved with re-initiation of treatment, severe hepatitis exacerbations, including fatalities, have been reported. Therefore, patients should be closely monitored after stopping treatment.
Warning: Severe Acute Exacerbations of Hepatitis, Nephrotoxicity, Hiv Resistance, Lactic Acidosis and Severe Hepatomegaly With Steatosis (WARNING: SEVERE ACUTE EXACERBATIONS OF HEPATITIS, NEPHROTOXICITY, HIV RESISTANCE, LACTIC ACIDOSIS AND SEVERE HEPATOMEGALY WITH STEATOSIS)
Severe acute exacerbations of hepatitis have been reported in patients who have discontinued anti-Hepatitis B therapy including Adefovir Dipivoxil Tablets. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-Hepatitis B therapy. If appropriate, resumption of anti-Hepatitis B therapy may be warranted [See Warnings and Precautions (5.1)] .
In patients at risk of or having underlying renal dysfunction, chronic administration of Adefovir Dipivoxil Tablets may result in nephrotoxicity. These patients should be monitored closely for renal function and may require dose adjustment [See Warnings and Precautions (5.2) and Dosage and Administration (2.2)] .
HIV resistance may emerge in chronic hepatitis B patients with unrecognized or untreated Human Immunodeficiency Virus (HIV) infection treated with antihepatitis B therapies, such as therapy with Adefovir Dipivoxil Tablets, that may have activity against HIV [See Warnings and Precautions (5.3)] .
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with other antiretrovirals [See Warnings and Precautions (5 . 4 )] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:38:01.611537 · Updated: 2026-03-14T21:53:03.632313