These Highlights Do Not Include All The Information Needed To Use Colchicine Safely And Effectively. See Full Prescribing Information For Colchicine Tablets, Usp.
df1dd89a-a1b3-48e8-b0e8-ad71b4cb0c96
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Colchicine Tablets, USP are an alkaloid indicated for: Prophylaxis and treatment of gout flares in adults ( 1.1 ). Familial Mediterranean fever (FMF) in adults and children 4 years or older ( 1.2 ).
Indications and Usage
Colchicine Tablets, USP are an alkaloid indicated for: Prophylaxis and treatment of gout flares in adults ( 1.1 ). Familial Mediterranean fever (FMF) in adults and children 4 years or older ( 1.2 ).
Dosage and Administration
The long-term use of colchicine is established for FMF and the prophylaxis of gout flares, but the safety and efficacy of repeat treatment for gout flares has not been evaluated. The dosing regimens for Colchicine Tablets, USP are different for each indication and must be individualized. The recommended dosage of Colchicine Tablets, USP depends on the patient's age, renal function, hepatic function and use of coadministered drugs [see Dosage and Administration (2.4 , 2.5 , 2.6) ] . Colchicine Tablets, USP are administered orally without regard to meals. Colchicine Tablets, USP are not an analgesic medication and should not be used to treat pain from other causes.
Warnings and Precautions
Fatal overdoses have been reported with colchicine in adults and children. Keep Colchicine Tablets, USP out of the reach of children ( 5.1 , 10 ). Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported ( 5.2 ). Monitor for toxicity and if present consider temporary interruption or discontinuation of colchicine ( 5.2 , 5.3 , 5.4 , 6 , 10 ). Drug interaction P-gp and/or CYP3A4 inhibitors: Coadministration of colchicine with P-gp and/or strong CYP3A4 inhibitors has resulted in life-threatening interactions and death ( 5.3 , 7 ). Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider temporary interruption or discontinuation of Colchicine Tablets, USP ( 5.4 , 7 ).
Contraindications
Patients with renal or hepatic impairment should not be given Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir). In these patients, life-threatening and fatal colchicine toxicity has been reported with colchicine taken in therapeutic doses.
Adverse Reactions
Prophylaxis of Gout Flares: The most commonly reported adverse reaction in clinical trials for the prophylaxis of gout was diarrhea. Treatment of Gout Flares: The most common adverse reactions reported in the clinical trial for gout were diarrhea (23%) and pharyngolaryngeal pain (3%). FMF: Most common adverse reactions (up to 20%) are abdominal pain, diarrhea, nausea and vomiting. These effects are usually mild, transient and reversible upon lowering the dose ( 6 ). To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals America, Inc. at 1-877-TAKEDA-7 (1-877-825-3327) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Colchicine is a P-gp and CYP3A4 substrate. Life-threatening and fatal drug interactions have been reported in patients treated with colchicine given with P-gp and strong CYP3A4 inhibitors. If treatment with a P-gp or strong CYP3A4 inhibitor is required in patients with normal renal and hepatic function, the patient's dose of colchicine may need to be reduced or interrupted [see Drug Interactions (7) ] . Use of Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir) is contraindicated in patients with renal or hepatic impairment [see Contraindications (4) ] .
Storage and Handling
Colchicine Tablets, USP 0.6 mg are purple, film-coated, capsule-shaped tablets debossed with "AR 374" on one side and scored on the other side. Bottles of 30 NDC 63629-2167-1 Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature]. Protect from light. DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
How Supplied
Colchicine Tablets, USP 0.6 mg are purple, film-coated, capsule-shaped tablets debossed with "AR 374" on one side and scored on the other side. Bottles of 30 NDC 63629-2167-1 Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature]. Protect from light. DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Medication Information
Warnings and Precautions
Fatal overdoses have been reported with colchicine in adults and children. Keep Colchicine Tablets, USP out of the reach of children ( 5.1 , 10 ). Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported ( 5.2 ). Monitor for toxicity and if present consider temporary interruption or discontinuation of colchicine ( 5.2 , 5.3 , 5.4 , 6 , 10 ). Drug interaction P-gp and/or CYP3A4 inhibitors: Coadministration of colchicine with P-gp and/or strong CYP3A4 inhibitors has resulted in life-threatening interactions and death ( 5.3 , 7 ). Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider temporary interruption or discontinuation of Colchicine Tablets, USP ( 5.4 , 7 ).
Indications and Usage
Colchicine Tablets, USP are an alkaloid indicated for: Prophylaxis and treatment of gout flares in adults ( 1.1 ). Familial Mediterranean fever (FMF) in adults and children 4 years or older ( 1.2 ).
Dosage and Administration
The long-term use of colchicine is established for FMF and the prophylaxis of gout flares, but the safety and efficacy of repeat treatment for gout flares has not been evaluated. The dosing regimens for Colchicine Tablets, USP are different for each indication and must be individualized. The recommended dosage of Colchicine Tablets, USP depends on the patient's age, renal function, hepatic function and use of coadministered drugs [see Dosage and Administration (2.4 , 2.5 , 2.6) ] . Colchicine Tablets, USP are administered orally without regard to meals. Colchicine Tablets, USP are not an analgesic medication and should not be used to treat pain from other causes.
Contraindications
Patients with renal or hepatic impairment should not be given Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir). In these patients, life-threatening and fatal colchicine toxicity has been reported with colchicine taken in therapeutic doses.
Adverse Reactions
Prophylaxis of Gout Flares: The most commonly reported adverse reaction in clinical trials for the prophylaxis of gout was diarrhea. Treatment of Gout Flares: The most common adverse reactions reported in the clinical trial for gout were diarrhea (23%) and pharyngolaryngeal pain (3%). FMF: Most common adverse reactions (up to 20%) are abdominal pain, diarrhea, nausea and vomiting. These effects are usually mild, transient and reversible upon lowering the dose ( 6 ). To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals America, Inc. at 1-877-TAKEDA-7 (1-877-825-3327) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Colchicine is a P-gp and CYP3A4 substrate. Life-threatening and fatal drug interactions have been reported in patients treated with colchicine given with P-gp and strong CYP3A4 inhibitors. If treatment with a P-gp or strong CYP3A4 inhibitor is required in patients with normal renal and hepatic function, the patient's dose of colchicine may need to be reduced or interrupted [see Drug Interactions (7) ] . Use of Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir) is contraindicated in patients with renal or hepatic impairment [see Contraindications (4) ] .
Storage and Handling
Colchicine Tablets, USP 0.6 mg are purple, film-coated, capsule-shaped tablets debossed with "AR 374" on one side and scored on the other side. Bottles of 30 NDC 63629-2167-1 Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature]. Protect from light. DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
How Supplied
Colchicine Tablets, USP 0.6 mg are purple, film-coated, capsule-shaped tablets debossed with "AR 374" on one side and scored on the other side. Bottles of 30 NDC 63629-2167-1 Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature]. Protect from light. DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Description
Colchicine Tablets, USP are an alkaloid indicated for: Prophylaxis and treatment of gout flares in adults ( 1.1 ). Familial Mediterranean fever (FMF) in adults and children 4 years or older ( 1.2 ).
2.2 Fmf
The recommended dosage of Colchicine Tablets, USP for FMF in adults is 1.2 to 2.4 mg daily.
Colchicine Tablets, USP should be increased as needed to control disease and as tolerated in increments of 0.3 mg/day to a maximum recommended daily dose. If intolerable side effects develop, the dose should be decreased in increments of 0.3 mg/day. The total daily Colchicine Tablets, USP dose may be administered in one to two divided doses.
Section 42229-5
Prophylaxis of Gout Flares
The recommended dosage of Colchicine Tablets, USP for prophylaxis of gout flares for adults and adolescents older than 16 years of age is 0.6 mg once or twice daily. The maximum recommended dose for prophylaxis of gout flares is 1.2 mg/day.
An increase in gout flares may occur after initiation of uric acid-lowering therapy, including pegloticase, febuxostat and allopurinol, due to changing serum uric acid levels resulting in mobilization of urate from tissue deposits. Colchicine Tablets, USP are recommended upon initiation of gout flare prophylaxis with uric acid-lowering therapy. Prophylactic therapy may be beneficial for at least the first six months of uric acid-lowering therapy.
Section 42231-1
| MEDICATION GUIDE Colchicine Tablets, USP for oral use |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | OS2008-01-75-02 COL345 R2 | Revised: July 2020 |
| Read the Medication Guide that comes with Colchicine Tablets, USP before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment. You and your healthcare provider should talk about Colchicine Tablets, USP when you start taking it and at regular checkups. | ||
|
What is the most important information that I should know about Colchicine Tablets, USP?
Colchicine Tablets, USP can cause serious side effects or death if levels of colchicine are too high in your body.
|
||
|
|
|
Ask your healthcare provider or pharmacist if you are not sure if you take any of the medicines listed above. This is not a complete list of all the medicines that can interact with Colchicine Tablets, USP.
|
||
|
What are Colchicine Tablets, USP?
Colchicine Tablets, USP are a prescription medicine used to:
|
||
|
Who should not take Colchicine Tablets, USP?
Do not take Colchicine Tablets, USP if you have liver or kidney problems and you take certain other medicines. Serious side effects, including death, have been reported in these patients even when taken as directed. See "What is the most important information that I should know about Colchicine Tablets, USP?". |
||
|
What should I tell my healthcare provider before starting Colchicine Tablets, USP?
See "What is the most important information that I should know about Colchicine Tablets, USP?". Before you take Colchicine Tablets, USP, tell your healthcare provider about all your medical conditions, including if you:
Using Colchicine Tablets, USP with certain other medicines, such as cholesterol-lowering medications and digoxin, can affect each other, causing serious side effects. Your healthcare provider may need to change your dose of Colchicine Tablets, USP. Talk to your healthcare provider about whether the medications you are taking might interact with Colchicine Tablets, USP and what side effects to look for. |
||
How should I take Colchicine Tablets, USP?
|
||
|
What should I avoid while taking Colchicine Tablets, USP?
Avoid eating grapefruit or drinking grapefruit juice while taking Colchicine Tablets, USP. It can increase your chances of getting serious side effects. |
||
|
What are the possible side effects of Colchicine Tablets, USP?
Colchicine Tablets, USP can cause serious side effects or even cause death. See "What is the most important information that I should know about Colchicine Tablets, USP?". Get medical help right away if you have:
FMF: The most common side effects of Colchicine Tablets, USP in people who have FMF are abdominal pain, diarrhea, nausea and vomiting. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Colchicine Tablets, USP. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store Colchicine Tablets, USP?
|
||
|
General Information about Colchicine Tablets, USP
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Colchicine Tablets, USP for a condition for which it was not prescribed. Do not give Colchicine Tablets, USP to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about Colchicine Tablets, USP. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Colchicine Tablets, USP that is written for healthcare professionals. |
||
|
What are the ingredients in Colchicine Tablets, USP?
Active Ingredient: colchicine. Inactive Ingredients: carnauba wax, FD&C blue #2, FD&C red #40, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, pregelatinized starch, sodium starch glycolate, titanium dioxide and triacetin. Distributed by: Par Pharmaceutical Chestnut Ridge, NY 10977 All trademarks are the property of their respective owners. For more information, call 1-877-TAKEDA-7 (1-877-825-3327). |
Section 51945-4
Colchicine 0.6 mg Tablet, #30
10 Overdosage
The exact dose of colchicine that produces significant toxicity is unknown. Fatalities have occurred after ingestion of a dose as low as 7 mg over a four day period, while other patients have survived after ingesting more than 60 mg. A review of 150 patients who overdosed on colchicine found that those who ingested less than 0.5 mg/kg survived and tended to have milder toxicities such as gastrointestinal symptoms, whereas those who took 0.5 to 0.8 mg/kg had more severe reactions such as myelosuppression. There was 100% mortality in those who ingested more than 0.8 mg/kg.
The first stage of acute colchicine toxicity typically begins within 24 hours of ingestion and includes gastrointestinal symptoms such as abdominal pain, nausea, vomiting, diarrhea and significant fluid loss, leading to volume depletion. Peripheral leukocytosis may also be seen. Life-threatening complications occur during the second stage, which occurs 24 to 72 hours after drug administration, attributed to multiorgan failure and its consequences. Death is usually a result of respiratory depression and cardiovascular collapse. If the patient survives, recovery of multiorgan injury may be accompanied by rebound leukocytosis and alopecia starting about one week after the initial ingestion.
Treatment of colchicine poisoning should begin with gastric lavage and measures to prevent shock. Otherwise, treatment is symptomatic and supportive. No specific antidote is known. Colchicine is not effectively removed by dialysis [see Clinical Pharmacology (12.3)].
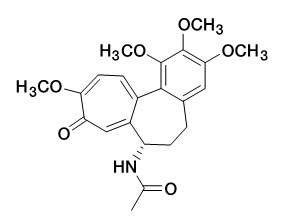
11 Description
Colchicine is an alkaloid chemically described as (S)N- (5,6,7,9-tetrahydro- 1,2,3, 10-tetramethoxy-9-oxobenzo [alpha] heptalen-7-yl) acetamide with a molecular formula of C22H25NO6 and a molecular weight of 399.4. The structural formula of colchicine is given below.
Colchicine occurs as a pale yellow powder that is soluble in water.
Colchicine Tablets, USP are supplied for oral administration as purple, film-coated, capsule-shaped tablets (0.1575" × 0.3030"), debossed with "AR 374" on one side and scored on the other, containing 0.6 mg of the active ingredient colchicine USP. Inactive ingredients: carnauba wax, FD&C blue #2, FD&C red #40, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, pregelatinized starch, sodium starch glycolate, titanium dioxide and triacetin.
1.1 Gout Flares
Colchicine Tablets, USP are indicated for prophylaxis and the treatment of acute gout flares.
-
Prophylaxis of Gout Flares
:
Colchicine Tablets, USP are indicated for prophylaxis of gout flares. -
Treatment of Gout Flares
:
Colchicine Tablets, USP are indicated for treatment of acute gout flares when taken at the first sign of a flare.
8.4 Pediatric Use
The safety and efficacy of colchicine in children of all ages with FMF has been evaluated in uncontrolled studies. There does not appear to be an adverse effect on growth in children with FMF treated long-term with colchicine.
Safety and effectiveness of colchicine in pediatric patients with gout has not been established.
8.5 Geriatric Use
Clinical studies with colchicine for prophylaxis and treatment of gout flares and for treatment of FMF did not include sufficient numbers of patients aged 65 years and older to determine whether they respond differently from younger patients. In general, dose selection for an elderly patient with gout should be cautious, reflecting the greater frequency of decreased renal function, concomitant disease or other drug therapy [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
5.1 Fatal Overdose
Fatal overdoses, both accidental and intentional, have been reported in adults and children who have ingested colchicine [see Overdosage (10)]. Colchicine Tablets, USP should be kept out of the reach of children.
14 Clinical Studies
The evidence for the efficacy of colchicine in patients with chronic gout is derived from the published literature. Two randomized clinical trials assessed the efficacy of colchicine 0.6 mg twice a day for the prophylaxis of gout flares in patients with gout initiating treatment with urate-lowering therapy. In both trials, treatment with colchicine decreased the frequency of gout flares.
The efficacy of a low-dosage regimen of oral colchicine (total dose 1.8 mg over one hour) for treatment of gout flares was assessed in a multicenter, randomized, double-blind, placebo-controlled, parallel group, one week, dose-comparison study. Patients meeting American College of Rheumatology criteria for gout were randomly assigned to three groups: high-dose colchicine (1.2 mg, then 0.6 mg hourly × 6 hours [4.8 mg total]); low-dose colchicine (1.2 mg, then 0.6 mg in one hour [1.8 mg total] followed by five placebo doses hourly); or placebo (two capsules, then one capsule hourly × six hours). Patients took the first dose within 12 hours of the onset of the flare and recorded pain intensity (11 point Likert scale) and adverse events over 72 hours. The efficacy of colchicine was measured based on response to treatment in the target joint, using patient self-assessment of pain at 24 hours following the time of first dose as recorded in the diary. A responder was one who achieved at least a 50% reduction in pain score at the 24 hour postdose assessment relative to the pretreatment score and did not use rescue medication prior to the actual time of 24 hour postdose assessment.
Rates of response were similar for the recommended low-dose treatment group (38%) and the nonrecommended high-dose group (33%) but were higher as compared to the placebo group (16%) as shown in Table 8.
| Colchicine Tablets, USP Dose Responders n (%) |
Placebo n (%) (n=58) |
% Differences in Proportion | ||
|---|---|---|---|---|
| Low-Dose (n=74) |
High-Dose (n=52) |
Low-Dose vs Placebo (95% CI) |
High-Dose vs Placebo (95% CI) |
|
| 28 (38%) | 17 (33%) | 9 (16%) | 22 (8, 37) | 17 (1, 33) |
Figure 1 shows the percentage of patients achieving varying degrees of improvement in pain from baseline at 24 hours.
The evidence for the efficacy of colchicine in patients with FMF is derived from the published literature. Three randomized, placebo-controlled studies were identified. The three placebo-controlled studies randomized a total of 48 adult patients diagnosed with FMF and reported similar efficacy endpoints as well as inclusion and exclusion criteria.
One of the studies randomized 15 patients with FMF to a six month crossover study during which five patients discontinued due to study noncompliance. The ten patients completing the study experienced five attacks over the course of 90 days while treated with colchicine compared to 59 attacks over the course of 90 days while treated with placebo. Similarly, the second study randomized 22 patients with FMF to a four month crossover study during which nine patients discontinued due to lack of efficacy while receiving placebo or study noncompliance. The 13 patients completing the study experienced 18 attacks over the course of 60 days while treated with colchicine compared to 68 attacks over the course of 60 days while treated with placebo. The third study was discontinued after an interim analysis of six of the 11 patients enrolled had completed the study; results could not be confirmed.
Open-label experience with colchicine in adults and children with FMF is consistent with the randomized, controlled trial experience and was utilized to support information on the safety profile of colchicine and for dosing recommendations.
4 Contraindications
Patients with renal or hepatic impairment should not be given Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir). In these patients, life-threatening and fatal colchicine toxicity has been reported with colchicine taken in therapeutic doses.
6 Adverse Reactions
- Prophylaxis of Gout Flares: The most commonly reported adverse reaction in clinical trials for the prophylaxis of gout was diarrhea.
- Treatment of Gout Flares: The most common adverse reactions reported in the clinical trial for gout were diarrhea (23%) and pharyngolaryngeal pain (3%).
- FMF: Most common adverse reactions (up to 20%) are abdominal pain, diarrhea, nausea and vomiting. These effects are usually mild, transient and reversible upon lowering the dose (6).
To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals America, Inc. at 1-877-TAKEDA-7 (1-877-825-3327) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
Colchicine is a substrate of the efflux transporter P-glycoprotein (P-gp). Of the cytochrome P450 enzymes tested, CYP3A4 was mainly involved in the metabolism of colchicine. If Colchicine Tablets, USP are administered with drugs that inhibit P-gp, most of which also inhibit CYP3A4, increased concentrations of colchicine are likely. Fatal drug interactions have been reported.
Physicians should ensure that patients are suitable candidates for treatment with Colchicine Tablets, USP and remain alert for signs and symptoms of toxicities related to increased colchicine exposure as a result of a drug interaction. Signs and symptoms of colchicine toxicity should be evaluated promptly and, if toxicity is suspected, Colchicine Tablets, USP should be discontinued immediately.
Table 4 provides recommendations as a result of other potentially significant drug interactions. Table 1 provides recommendations for strong and moderate CYP3A4 inhibitors and P-gp inhibitors.
| Concomitant Drug Class or Food | Noted or Anticipated Outcome | Clinical Comment |
|---|---|---|
|
HMG-Co A Reductase Inhibitors:
atorvastatin, fluvastatin, lovastatin, pravastatin, simvastatin |
Pharmacokinetic and/or pharmacodynamic interaction: the addition of one drug to a stable long-term regimen of the other has resulted in myopathy and rhabdomyolysis (including a fatality) | Weigh the potential benefits and risks and carefully monitor patients for any signs or symptoms of muscle pain, tenderness, or weakness, particularly during initial therapy; monitoring CPK (creatine phosphokinase) will not necessarily prevent the occurrence of severe myopathy. |
|
Other Lipid-Lowering Drugs:
fibrates, gemfibrozil |
||
|
Digitalis Glycosides:
digoxin |
P-gp substrate; rhabdomyolysis has been reported |
5.2 Blood Dyscrasias
Myelosuppression, leukopenia, granulocytopenia, thrombocytopenia, pancytopenia and aplastic anemia have been reported with colchicine used in therapeutic doses.
8.6 Renal Impairment
Colchicine is significantly excreted in urine in healthy subjects. Clearance of colchicine is decreased in patients with impaired renal function. Total body clearance of colchicine was reduced by 75% in patients with end-stage renal disease undergoing dialysis.
5.3 Drug Interactions
Colchicine is a P-gp and CYP3A4 substrate. Life-threatening and fatal drug interactions have been reported in patients treated with colchicine given with P-gp and strong CYP3A4 inhibitors. If treatment with a P-gp or strong CYP3A4 inhibitor is required in patients with normal renal and hepatic function, the patient's dose of colchicine may need to be reduced or interrupted [see Drug Interactions (7)]. Use of Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir) is contraindicated in patients with renal or hepatic impairment [see Contraindications (4)].
8.7 Hepatic Impairment
The clearance of colchicine may be significantly reduced and plasma half-life prolonged in patients with chronic hepatic impairment compared to healthy subjects [see Clinical Pharmacology (12.3)].
1 Indications and Usage
12.1 Mechanism of Action
The mechanism by which Colchicine Tablets, USP exert their beneficial effect in patients with FMF has not been fully elucidated; however, evidence suggests that colchicine may interfere with the intracellular assembly of the inflammasome complex present in neutrophils and monocytes that mediates activation of interleukin-1β. Additionally, colchicine disrupts cytoskeletal functions through inhibition of β-tubulin polymerization into microtubules, and consequently prevents the activation, degranulation and migration of neutrophils thought to mediate some gout symptoms.
5 Warnings and Precautions
- Fatal overdoses have been reported with colchicine in adults and children. Keep Colchicine Tablets, USP out of the reach of children (5.1, 10).
- Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported (5.2).
- Monitor for toxicity and if present consider temporary interruption or discontinuation of colchicine (5.2, 5.3, 5.4, 6, 10).
- Drug interaction P-gp and/or CYP3A4 inhibitors: Coadministration of colchicine with P-gp and/or strong CYP3A4 inhibitors has resulted in life-threatening interactions and death (5.3, 7).
- Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider temporary interruption or discontinuation of Colchicine Tablets, USP (5.4, 7).
5.4 Neuromuscular Toxicity
Colchicine-induced neuromuscular toxicity and rhabdomyolysis have been reported with chronic treatment in therapeutic doses. Patients with renal dysfunction and elderly patients, even those with normal renal and hepatic function, are at increased risk. Concomitant use of atorvastatin, simvastatin, pravastatin, fluvastatin, lovastatin, gemfibrozil, fenofibrate, fenofibric acid or benzafibrate (themselves associated with myotoxicity) or cyclosporine with Colchicine Tablets, USP may potentiate the development of myopathy [see Drug Interactions (7)]. Once colchicine is stopped, the symptoms generally resolve within one week to several months.
2 Dosage and Administration
The long-term use of colchicine is established for FMF and the prophylaxis of gout flares, but the safety and efficacy of repeat treatment for gout flares has not been evaluated. The dosing regimens for Colchicine Tablets, USP are different for each indication and must be individualized.
The recommended dosage of Colchicine Tablets, USP depends on the patient's age, renal function, hepatic function and use of coadministered drugs [see Dosage and Administration (2.4, 2.5, 2.6)].
Colchicine Tablets, USP are administered orally without regard to meals.
Colchicine Tablets, USP are not an analgesic medication and should not be used to treat pain from other causes.
9 Drug Abuse and Dependence
Tolerance, abuse or dependence with colchicine has not been reported.
3 Dosage Forms and Strengths
Tablets: 0.6 mg colchicine — purple capsule-shaped, film-coated with "AR 374" debossed on one side and scored on the other side.
6.2 Postmarketing Experience
Serious toxic manifestations associated with colchicine include myelosuppression, disseminated intravascular coagulation and injury to cells in the renal, hepatic, circulatory and central nervous systems.
These most often occur with excessive accumulation or overdosage [see Overdosage (10)].
The following adverse reactions have been identified with colchicine. These have been generally reversible upon temporarily interrupting treatment or lowering the dose of colchicine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Neurological: sensory motor neuropathy
Dermatological: alopecia, maculopapular rash, purpura, rash
Digestive: abdominal cramping, abdominal pain, diarrhea, lactose intolerance, nausea, vomiting
Hematological: leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, aplastic anemia
Hepatobiliary: elevated AST, elevated ALT
Musculoskeletal: myopathy, elevated CPK, myotonia, muscle weakness, muscle pain, rhabdomyolysis
Reproductive: azoospermia, oligospermia
8 Use in Specific Populations
- In the presence of mild to moderate renal or hepatic impairment, adjustment of dosing is not required for treatment of gout flare, prophylaxis of gout flare and FMF, but patients should be monitored closely (8.6).
- In patients with severe renal impairment for prophylaxis of gout flares, the starting dose should be 0.3 mg/day for gout flares, no dose adjustment is required, but a treatment course should be repeated no more than once every two weeks. In FMF patients, start with 0.3 mg/day, and any increase in dose should be done with close monitoring (8.6).
- In patients with severe hepatic impairment, a dose reduction may be needed in prophylaxis of gout flares and FMF patients; while a dose reduction may not be needed in gout flares, a treatment course should be repeated no more than once every two weeks (8.6, 8.7).
- For patients undergoing dialysis, the total recommended dose for prophylaxis of gout flares should be 0.3 mg given twice a week with close monitoring. For treatment of gout flares, the total recommended dose should be reduced to 0.6 mg (one tablet) × 1 dose and the treatment course should not be repeated more than once every two weeks. For FMF patients, the starting dose should be 0.3 mg/day and dosing can be increased with close monitoring (8.6).
- Females and Males of Reproductive Potential: Advise males that Colchicine Tablets, USP may transiently impair fertility (8.3).
- Geriatric Use: The recommended dose of colchicine should be based on renal function (8.5).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
Colchicine Tablets, USP 0.6 mg are purple, film-coated, capsule-shaped tablets debossed with "AR 374" on one side and scored on the other side.
Bottles of 30 NDC 63629-2167-1
Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature].
Protect from light.
DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
1.2 Familial Mediterranean Fever (fmf)
Colchicine Tablets, USP are indicated in adults and children four years or older for treatment of familial Mediterranean fever (FMF).
6.1 Clinical Trials Experience in Gout
Because clinical studies are conducted under widely varying and controlled conditions, adverse reaction rates observed in clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
In a randomized, double-blind, placebo-controlled trial in patients with a gout flare, gastrointestinal adverse reactions occurred in 26% of patients using the recommended dose (1.8 mg over one hour) of Colchicine Tablets, USP compared to 77% of patients taking a nonrecommended high-dose (4.8 mg over six hours) of colchicine and 20% of patients taking placebo. Diarrhea was the most commonly reported drug-related gastrointestinal adverse event. As shown in Table 3, diarrhea is associated with Colchicine Tablets, USP treatment. Diarrhea was more likely to occur in patients taking the high-dose regimen than the low-dose regimen. Severe diarrhea occurred in 19% and vomiting occurred in 17% of patients taking the nonrecommended high-dose colchicine regimen but did not occur in the recommended low-dose Colchicine Tablets, USP regimen.
| MedDRA System Organ Class MedDRA Preferred Term |
Colchicine Tablets, USP Dose | Placebo (N=59) n (%) |
|
|---|---|---|---|
| High (N=52) n (%) |
Low (N=74) n (%) |
||
| Number of Patients with at Least One Drug-Related TEAE | 40 (77) | 27 (37) | 16 (27) |
| Gastrointestinal Disorders | 40 (77) | 19 (26) | 12 (20) |
| Diarrhea | 40 (77) | 17 (23) | 8 (14) |
| Nausea | 9 (17) | 3 (4) | 3 (5) |
| Vomiting | 9 (17) | 0 | 0 |
| Abdominal Discomfort | 0 | 0 | 2 (3) |
| General Disorders and Administration Site Conditions | 4 (8) | 1 (1) | 1 (2) |
| Fatigue | 2 (4) | 1 (1) | 1 (2) |
| Metabolic and Nutrition Disorders | 0 | 3 (4) | 2 (3) |
| Gout | 0 | 3 (4) | 1 (2) |
| Nervous System Disorders | 1 (2) | 1 (1.4) | 2 (3) |
| Headache | 1 (2) | 1 (1) | 2 (3) |
| Respiratory Thoracic Mediastinal Disorders | 1 (2) | 2 (3) | 0 |
| Pharyngolaryngeal Pain | 1 (2) | 2 (3) | 0 |
2.5 Dose Modification in Renal Impairment
Colchicine dosing must be individualized according to the patient's renal function [see Use in Specific Populations (8.6)].
Clcr in mL/minute may be estimated from serum creatinine (mg/dL) determination using the following formula:
| [140-age (years) × weight (kg)] | |
| Clcr = | 72 × serum creatinine (mg/dL) × 0.85 for female patients |
Structured Label Content
2.2 Fmf (2.2 FMF)
The recommended dosage of Colchicine Tablets, USP for FMF in adults is 1.2 to 2.4 mg daily.
Colchicine Tablets, USP should be increased as needed to control disease and as tolerated in increments of 0.3 mg/day to a maximum recommended daily dose. If intolerable side effects develop, the dose should be decreased in increments of 0.3 mg/day. The total daily Colchicine Tablets, USP dose may be administered in one to two divided doses.
Section 42229-5 (42229-5)
Prophylaxis of Gout Flares
The recommended dosage of Colchicine Tablets, USP for prophylaxis of gout flares for adults and adolescents older than 16 years of age is 0.6 mg once or twice daily. The maximum recommended dose for prophylaxis of gout flares is 1.2 mg/day.
An increase in gout flares may occur after initiation of uric acid-lowering therapy, including pegloticase, febuxostat and allopurinol, due to changing serum uric acid levels resulting in mobilization of urate from tissue deposits. Colchicine Tablets, USP are recommended upon initiation of gout flare prophylaxis with uric acid-lowering therapy. Prophylactic therapy may be beneficial for at least the first six months of uric acid-lowering therapy.
Section 42231-1 (42231-1)
| MEDICATION GUIDE Colchicine Tablets, USP for oral use |
||
|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | OS2008-01-75-02 COL345 R2 | Revised: July 2020 |
| Read the Medication Guide that comes with Colchicine Tablets, USP before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment. You and your healthcare provider should talk about Colchicine Tablets, USP when you start taking it and at regular checkups. | ||
|
What is the most important information that I should know about Colchicine Tablets, USP?
Colchicine Tablets, USP can cause serious side effects or death if levels of colchicine are too high in your body.
|
||
|
|
|
Ask your healthcare provider or pharmacist if you are not sure if you take any of the medicines listed above. This is not a complete list of all the medicines that can interact with Colchicine Tablets, USP.
|
||
|
What are Colchicine Tablets, USP?
Colchicine Tablets, USP are a prescription medicine used to:
|
||
|
Who should not take Colchicine Tablets, USP?
Do not take Colchicine Tablets, USP if you have liver or kidney problems and you take certain other medicines. Serious side effects, including death, have been reported in these patients even when taken as directed. See "What is the most important information that I should know about Colchicine Tablets, USP?". |
||
|
What should I tell my healthcare provider before starting Colchicine Tablets, USP?
See "What is the most important information that I should know about Colchicine Tablets, USP?". Before you take Colchicine Tablets, USP, tell your healthcare provider about all your medical conditions, including if you:
Using Colchicine Tablets, USP with certain other medicines, such as cholesterol-lowering medications and digoxin, can affect each other, causing serious side effects. Your healthcare provider may need to change your dose of Colchicine Tablets, USP. Talk to your healthcare provider about whether the medications you are taking might interact with Colchicine Tablets, USP and what side effects to look for. |
||
How should I take Colchicine Tablets, USP?
|
||
|
What should I avoid while taking Colchicine Tablets, USP?
Avoid eating grapefruit or drinking grapefruit juice while taking Colchicine Tablets, USP. It can increase your chances of getting serious side effects. |
||
|
What are the possible side effects of Colchicine Tablets, USP?
Colchicine Tablets, USP can cause serious side effects or even cause death. See "What is the most important information that I should know about Colchicine Tablets, USP?". Get medical help right away if you have:
FMF: The most common side effects of Colchicine Tablets, USP in people who have FMF are abdominal pain, diarrhea, nausea and vomiting. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Colchicine Tablets, USP. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store Colchicine Tablets, USP?
|
||
|
General Information about Colchicine Tablets, USP
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Colchicine Tablets, USP for a condition for which it was not prescribed. Do not give Colchicine Tablets, USP to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about Colchicine Tablets, USP. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Colchicine Tablets, USP that is written for healthcare professionals. |
||
|
What are the ingredients in Colchicine Tablets, USP?
Active Ingredient: colchicine. Inactive Ingredients: carnauba wax, FD&C blue #2, FD&C red #40, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, pregelatinized starch, sodium starch glycolate, titanium dioxide and triacetin. Distributed by: Par Pharmaceutical Chestnut Ridge, NY 10977 All trademarks are the property of their respective owners. For more information, call 1-877-TAKEDA-7 (1-877-825-3327). |
Section 51945-4 (51945-4)
Colchicine 0.6 mg Tablet, #30
10 Overdosage (10 OVERDOSAGE)
The exact dose of colchicine that produces significant toxicity is unknown. Fatalities have occurred after ingestion of a dose as low as 7 mg over a four day period, while other patients have survived after ingesting more than 60 mg. A review of 150 patients who overdosed on colchicine found that those who ingested less than 0.5 mg/kg survived and tended to have milder toxicities such as gastrointestinal symptoms, whereas those who took 0.5 to 0.8 mg/kg had more severe reactions such as myelosuppression. There was 100% mortality in those who ingested more than 0.8 mg/kg.
The first stage of acute colchicine toxicity typically begins within 24 hours of ingestion and includes gastrointestinal symptoms such as abdominal pain, nausea, vomiting, diarrhea and significant fluid loss, leading to volume depletion. Peripheral leukocytosis may also be seen. Life-threatening complications occur during the second stage, which occurs 24 to 72 hours after drug administration, attributed to multiorgan failure and its consequences. Death is usually a result of respiratory depression and cardiovascular collapse. If the patient survives, recovery of multiorgan injury may be accompanied by rebound leukocytosis and alopecia starting about one week after the initial ingestion.
Treatment of colchicine poisoning should begin with gastric lavage and measures to prevent shock. Otherwise, treatment is symptomatic and supportive. No specific antidote is known. Colchicine is not effectively removed by dialysis [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Colchicine is an alkaloid chemically described as (S)N- (5,6,7,9-tetrahydro- 1,2,3, 10-tetramethoxy-9-oxobenzo [alpha] heptalen-7-yl) acetamide with a molecular formula of C22H25NO6 and a molecular weight of 399.4. The structural formula of colchicine is given below.
Colchicine occurs as a pale yellow powder that is soluble in water.
Colchicine Tablets, USP are supplied for oral administration as purple, film-coated, capsule-shaped tablets (0.1575" × 0.3030"), debossed with "AR 374" on one side and scored on the other, containing 0.6 mg of the active ingredient colchicine USP. Inactive ingredients: carnauba wax, FD&C blue #2, FD&C red #40, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, pregelatinized starch, sodium starch glycolate, titanium dioxide and triacetin.
1.1 Gout Flares
Colchicine Tablets, USP are indicated for prophylaxis and the treatment of acute gout flares.
-
Prophylaxis of Gout Flares
:
Colchicine Tablets, USP are indicated for prophylaxis of gout flares. -
Treatment of Gout Flares
:
Colchicine Tablets, USP are indicated for treatment of acute gout flares when taken at the first sign of a flare.
8.4 Pediatric Use
The safety and efficacy of colchicine in children of all ages with FMF has been evaluated in uncontrolled studies. There does not appear to be an adverse effect on growth in children with FMF treated long-term with colchicine.
Safety and effectiveness of colchicine in pediatric patients with gout has not been established.
8.5 Geriatric Use
Clinical studies with colchicine for prophylaxis and treatment of gout flares and for treatment of FMF did not include sufficient numbers of patients aged 65 years and older to determine whether they respond differently from younger patients. In general, dose selection for an elderly patient with gout should be cautious, reflecting the greater frequency of decreased renal function, concomitant disease or other drug therapy [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
5.1 Fatal Overdose
Fatal overdoses, both accidental and intentional, have been reported in adults and children who have ingested colchicine [see Overdosage (10)]. Colchicine Tablets, USP should be kept out of the reach of children.
14 Clinical Studies (14 CLINICAL STUDIES)
The evidence for the efficacy of colchicine in patients with chronic gout is derived from the published literature. Two randomized clinical trials assessed the efficacy of colchicine 0.6 mg twice a day for the prophylaxis of gout flares in patients with gout initiating treatment with urate-lowering therapy. In both trials, treatment with colchicine decreased the frequency of gout flares.
The efficacy of a low-dosage regimen of oral colchicine (total dose 1.8 mg over one hour) for treatment of gout flares was assessed in a multicenter, randomized, double-blind, placebo-controlled, parallel group, one week, dose-comparison study. Patients meeting American College of Rheumatology criteria for gout were randomly assigned to three groups: high-dose colchicine (1.2 mg, then 0.6 mg hourly × 6 hours [4.8 mg total]); low-dose colchicine (1.2 mg, then 0.6 mg in one hour [1.8 mg total] followed by five placebo doses hourly); or placebo (two capsules, then one capsule hourly × six hours). Patients took the first dose within 12 hours of the onset of the flare and recorded pain intensity (11 point Likert scale) and adverse events over 72 hours. The efficacy of colchicine was measured based on response to treatment in the target joint, using patient self-assessment of pain at 24 hours following the time of first dose as recorded in the diary. A responder was one who achieved at least a 50% reduction in pain score at the 24 hour postdose assessment relative to the pretreatment score and did not use rescue medication prior to the actual time of 24 hour postdose assessment.
Rates of response were similar for the recommended low-dose treatment group (38%) and the nonrecommended high-dose group (33%) but were higher as compared to the placebo group (16%) as shown in Table 8.
| Colchicine Tablets, USP Dose Responders n (%) |
Placebo n (%) (n=58) |
% Differences in Proportion | ||
|---|---|---|---|---|
| Low-Dose (n=74) |
High-Dose (n=52) |
Low-Dose vs Placebo (95% CI) |
High-Dose vs Placebo (95% CI) |
|
| 28 (38%) | 17 (33%) | 9 (16%) | 22 (8, 37) | 17 (1, 33) |
Figure 1 shows the percentage of patients achieving varying degrees of improvement in pain from baseline at 24 hours.
The evidence for the efficacy of colchicine in patients with FMF is derived from the published literature. Three randomized, placebo-controlled studies were identified. The three placebo-controlled studies randomized a total of 48 adult patients diagnosed with FMF and reported similar efficacy endpoints as well as inclusion and exclusion criteria.
One of the studies randomized 15 patients with FMF to a six month crossover study during which five patients discontinued due to study noncompliance. The ten patients completing the study experienced five attacks over the course of 90 days while treated with colchicine compared to 59 attacks over the course of 90 days while treated with placebo. Similarly, the second study randomized 22 patients with FMF to a four month crossover study during which nine patients discontinued due to lack of efficacy while receiving placebo or study noncompliance. The 13 patients completing the study experienced 18 attacks over the course of 60 days while treated with colchicine compared to 68 attacks over the course of 60 days while treated with placebo. The third study was discontinued after an interim analysis of six of the 11 patients enrolled had completed the study; results could not be confirmed.
Open-label experience with colchicine in adults and children with FMF is consistent with the randomized, controlled trial experience and was utilized to support information on the safety profile of colchicine and for dosing recommendations.
4 Contraindications (4 CONTRAINDICATIONS)
Patients with renal or hepatic impairment should not be given Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir). In these patients, life-threatening and fatal colchicine toxicity has been reported with colchicine taken in therapeutic doses.
6 Adverse Reactions (6 ADVERSE REACTIONS)
- Prophylaxis of Gout Flares: The most commonly reported adverse reaction in clinical trials for the prophylaxis of gout was diarrhea.
- Treatment of Gout Flares: The most common adverse reactions reported in the clinical trial for gout were diarrhea (23%) and pharyngolaryngeal pain (3%).
- FMF: Most common adverse reactions (up to 20%) are abdominal pain, diarrhea, nausea and vomiting. These effects are usually mild, transient and reversible upon lowering the dose (6).
To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals America, Inc. at 1-877-TAKEDA-7 (1-877-825-3327) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
Colchicine is a substrate of the efflux transporter P-glycoprotein (P-gp). Of the cytochrome P450 enzymes tested, CYP3A4 was mainly involved in the metabolism of colchicine. If Colchicine Tablets, USP are administered with drugs that inhibit P-gp, most of which also inhibit CYP3A4, increased concentrations of colchicine are likely. Fatal drug interactions have been reported.
Physicians should ensure that patients are suitable candidates for treatment with Colchicine Tablets, USP and remain alert for signs and symptoms of toxicities related to increased colchicine exposure as a result of a drug interaction. Signs and symptoms of colchicine toxicity should be evaluated promptly and, if toxicity is suspected, Colchicine Tablets, USP should be discontinued immediately.
Table 4 provides recommendations as a result of other potentially significant drug interactions. Table 1 provides recommendations for strong and moderate CYP3A4 inhibitors and P-gp inhibitors.
| Concomitant Drug Class or Food | Noted or Anticipated Outcome | Clinical Comment |
|---|---|---|
|
HMG-Co A Reductase Inhibitors:
atorvastatin, fluvastatin, lovastatin, pravastatin, simvastatin |
Pharmacokinetic and/or pharmacodynamic interaction: the addition of one drug to a stable long-term regimen of the other has resulted in myopathy and rhabdomyolysis (including a fatality) | Weigh the potential benefits and risks and carefully monitor patients for any signs or symptoms of muscle pain, tenderness, or weakness, particularly during initial therapy; monitoring CPK (creatine phosphokinase) will not necessarily prevent the occurrence of severe myopathy. |
|
Other Lipid-Lowering Drugs:
fibrates, gemfibrozil |
||
|
Digitalis Glycosides:
digoxin |
P-gp substrate; rhabdomyolysis has been reported |
5.2 Blood Dyscrasias
Myelosuppression, leukopenia, granulocytopenia, thrombocytopenia, pancytopenia and aplastic anemia have been reported with colchicine used in therapeutic doses.
8.6 Renal Impairment
Colchicine is significantly excreted in urine in healthy subjects. Clearance of colchicine is decreased in patients with impaired renal function. Total body clearance of colchicine was reduced by 75% in patients with end-stage renal disease undergoing dialysis.
5.3 Drug Interactions
Colchicine is a P-gp and CYP3A4 substrate. Life-threatening and fatal drug interactions have been reported in patients treated with colchicine given with P-gp and strong CYP3A4 inhibitors. If treatment with a P-gp or strong CYP3A4 inhibitor is required in patients with normal renal and hepatic function, the patient's dose of colchicine may need to be reduced or interrupted [see Drug Interactions (7)]. Use of Colchicine Tablets, USP in conjunction with P-gp or strong CYP3A4 inhibitors (this includes all protease inhibitors except fosamprenavir) is contraindicated in patients with renal or hepatic impairment [see Contraindications (4)].
8.7 Hepatic Impairment
The clearance of colchicine may be significantly reduced and plasma half-life prolonged in patients with chronic hepatic impairment compared to healthy subjects [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
12.1 Mechanism of Action
The mechanism by which Colchicine Tablets, USP exert their beneficial effect in patients with FMF has not been fully elucidated; however, evidence suggests that colchicine may interfere with the intracellular assembly of the inflammasome complex present in neutrophils and monocytes that mediates activation of interleukin-1β. Additionally, colchicine disrupts cytoskeletal functions through inhibition of β-tubulin polymerization into microtubules, and consequently prevents the activation, degranulation and migration of neutrophils thought to mediate some gout symptoms.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Fatal overdoses have been reported with colchicine in adults and children. Keep Colchicine Tablets, USP out of the reach of children (5.1, 10).
- Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported (5.2).
- Monitor for toxicity and if present consider temporary interruption or discontinuation of colchicine (5.2, 5.3, 5.4, 6, 10).
- Drug interaction P-gp and/or CYP3A4 inhibitors: Coadministration of colchicine with P-gp and/or strong CYP3A4 inhibitors has resulted in life-threatening interactions and death (5.3, 7).
- Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider temporary interruption or discontinuation of Colchicine Tablets, USP (5.4, 7).
5.4 Neuromuscular Toxicity
Colchicine-induced neuromuscular toxicity and rhabdomyolysis have been reported with chronic treatment in therapeutic doses. Patients with renal dysfunction and elderly patients, even those with normal renal and hepatic function, are at increased risk. Concomitant use of atorvastatin, simvastatin, pravastatin, fluvastatin, lovastatin, gemfibrozil, fenofibrate, fenofibric acid or benzafibrate (themselves associated with myotoxicity) or cyclosporine with Colchicine Tablets, USP may potentiate the development of myopathy [see Drug Interactions (7)]. Once colchicine is stopped, the symptoms generally resolve within one week to several months.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The long-term use of colchicine is established for FMF and the prophylaxis of gout flares, but the safety and efficacy of repeat treatment for gout flares has not been evaluated. The dosing regimens for Colchicine Tablets, USP are different for each indication and must be individualized.
The recommended dosage of Colchicine Tablets, USP depends on the patient's age, renal function, hepatic function and use of coadministered drugs [see Dosage and Administration (2.4, 2.5, 2.6)].
Colchicine Tablets, USP are administered orally without regard to meals.
Colchicine Tablets, USP are not an analgesic medication and should not be used to treat pain from other causes.
9 Drug Abuse and Dependence (9 DRUG ABUSE AND DEPENDENCE)
Tolerance, abuse or dependence with colchicine has not been reported.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 0.6 mg colchicine — purple capsule-shaped, film-coated with "AR 374" debossed on one side and scored on the other side.
6.2 Postmarketing Experience
Serious toxic manifestations associated with colchicine include myelosuppression, disseminated intravascular coagulation and injury to cells in the renal, hepatic, circulatory and central nervous systems.
These most often occur with excessive accumulation or overdosage [see Overdosage (10)].
The following adverse reactions have been identified with colchicine. These have been generally reversible upon temporarily interrupting treatment or lowering the dose of colchicine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Neurological: sensory motor neuropathy
Dermatological: alopecia, maculopapular rash, purpura, rash
Digestive: abdominal cramping, abdominal pain, diarrhea, lactose intolerance, nausea, vomiting
Hematological: leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, aplastic anemia
Hepatobiliary: elevated AST, elevated ALT
Musculoskeletal: myopathy, elevated CPK, myotonia, muscle weakness, muscle pain, rhabdomyolysis
Reproductive: azoospermia, oligospermia
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- In the presence of mild to moderate renal or hepatic impairment, adjustment of dosing is not required for treatment of gout flare, prophylaxis of gout flare and FMF, but patients should be monitored closely (8.6).
- In patients with severe renal impairment for prophylaxis of gout flares, the starting dose should be 0.3 mg/day for gout flares, no dose adjustment is required, but a treatment course should be repeated no more than once every two weeks. In FMF patients, start with 0.3 mg/day, and any increase in dose should be done with close monitoring (8.6).
- In patients with severe hepatic impairment, a dose reduction may be needed in prophylaxis of gout flares and FMF patients; while a dose reduction may not be needed in gout flares, a treatment course should be repeated no more than once every two weeks (8.6, 8.7).
- For patients undergoing dialysis, the total recommended dose for prophylaxis of gout flares should be 0.3 mg given twice a week with close monitoring. For treatment of gout flares, the total recommended dose should be reduced to 0.6 mg (one tablet) × 1 dose and the treatment course should not be repeated more than once every two weeks. For FMF patients, the starting dose should be 0.3 mg/day and dosing can be increased with close monitoring (8.6).
- Females and Males of Reproductive Potential: Advise males that Colchicine Tablets, USP may transiently impair fertility (8.3).
- Geriatric Use: The recommended dose of colchicine should be based on renal function (8.5).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Colchicine Tablets, USP 0.6 mg are purple, film-coated, capsule-shaped tablets debossed with "AR 374" on one side and scored on the other side.
Bottles of 30 NDC 63629-2167-1
Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature].
Protect from light.
DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
1.2 Familial Mediterranean Fever (fmf) (1.2 Familial Mediterranean Fever (FMF))
Colchicine Tablets, USP are indicated in adults and children four years or older for treatment of familial Mediterranean fever (FMF).
6.1 Clinical Trials Experience in Gout
Because clinical studies are conducted under widely varying and controlled conditions, adverse reaction rates observed in clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
In a randomized, double-blind, placebo-controlled trial in patients with a gout flare, gastrointestinal adverse reactions occurred in 26% of patients using the recommended dose (1.8 mg over one hour) of Colchicine Tablets, USP compared to 77% of patients taking a nonrecommended high-dose (4.8 mg over six hours) of colchicine and 20% of patients taking placebo. Diarrhea was the most commonly reported drug-related gastrointestinal adverse event. As shown in Table 3, diarrhea is associated with Colchicine Tablets, USP treatment. Diarrhea was more likely to occur in patients taking the high-dose regimen than the low-dose regimen. Severe diarrhea occurred in 19% and vomiting occurred in 17% of patients taking the nonrecommended high-dose colchicine regimen but did not occur in the recommended low-dose Colchicine Tablets, USP regimen.
| MedDRA System Organ Class MedDRA Preferred Term |
Colchicine Tablets, USP Dose | Placebo (N=59) n (%) |
|
|---|---|---|---|
| High (N=52) n (%) |
Low (N=74) n (%) |
||
| Number of Patients with at Least One Drug-Related TEAE | 40 (77) | 27 (37) | 16 (27) |
| Gastrointestinal Disorders | 40 (77) | 19 (26) | 12 (20) |
| Diarrhea | 40 (77) | 17 (23) | 8 (14) |
| Nausea | 9 (17) | 3 (4) | 3 (5) |
| Vomiting | 9 (17) | 0 | 0 |
| Abdominal Discomfort | 0 | 0 | 2 (3) |
| General Disorders and Administration Site Conditions | 4 (8) | 1 (1) | 1 (2) |
| Fatigue | 2 (4) | 1 (1) | 1 (2) |
| Metabolic and Nutrition Disorders | 0 | 3 (4) | 2 (3) |
| Gout | 0 | 3 (4) | 1 (2) |
| Nervous System Disorders | 1 (2) | 1 (1.4) | 2 (3) |
| Headache | 1 (2) | 1 (1) | 2 (3) |
| Respiratory Thoracic Mediastinal Disorders | 1 (2) | 2 (3) | 0 |
| Pharyngolaryngeal Pain | 1 (2) | 2 (3) | 0 |
2.5 Dose Modification in Renal Impairment
Colchicine dosing must be individualized according to the patient's renal function [see Use in Specific Populations (8.6)].
Clcr in mL/minute may be estimated from serum creatinine (mg/dL) determination using the following formula:
| [140-age (years) × weight (kg)] | |
| Clcr = | 72 × serum creatinine (mg/dL) × 0.85 for female patients |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:52.218699 · Updated: 2026-03-14T21:59:51.476921