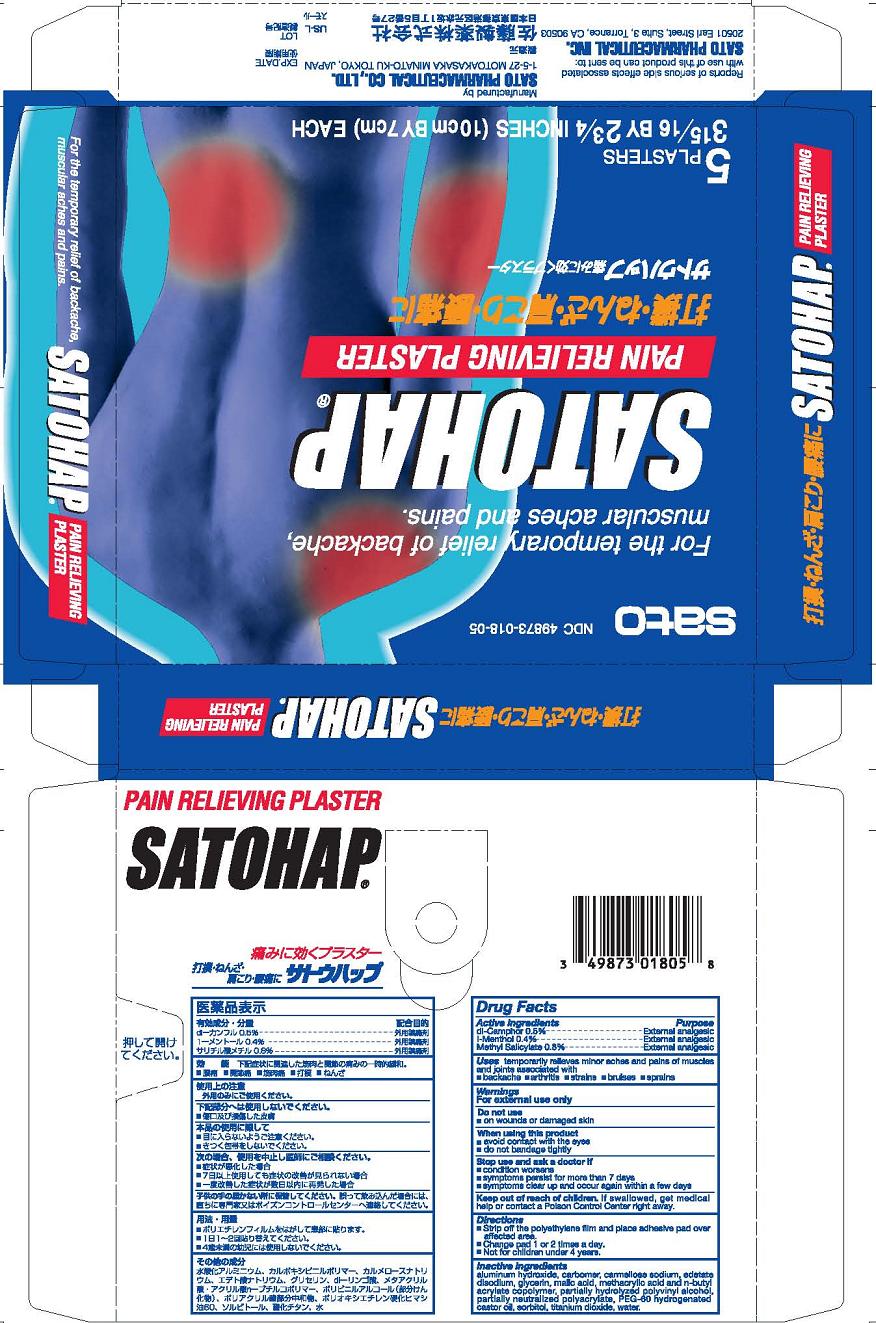
satohap plaster
df0e2b83-8f61-4ed8-bcc4-55e4ea782090
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients dl-Camphor 0.5% l-Menthol 0.4% Methyl Salicylate 0.8%
Medication Information
Warnings and Precautions
Warnings
For external use only
Active Ingredient
Active ingredients
dl-Camphor 0.5%
l-Menthol 0.4%
Methyl Salicylate 0.8%
Indications and Usage
Uses temporary relieves minor aches and pains of muscles and joints associated with
■ simple backache ■ arthritis ■ strains ■ bruises ■ sprains
Dosage and Administration
Directions
■ Strip off the polyethylene film and place adhesive pad over affected area.
■ Change pad 1 or 2 times a day
■ Not for children under 4 years.
Description
Active ingredients dl-Camphor 0.5% l-Menthol 0.4% Methyl Salicylate 0.8%
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
■ condition worsens
■ symptoms persist for more than 7 days
■ symptoms clear up and occur again withn a few days
Section 50567-7
When using this product
■ avoid contact with the eyes
■ do not bandage tightly
Section 50570-1
Do not use
■ on wounds or on damaged skin
Section 51727-6
Inactive ingredients
aluminum hydroxide gel, carbomer, carmellose sodium, edetate disodium, glycerin, malic acid, methacrylic acid and n-butyl acrylate copolymer, partially hydrolyzed polyvinyl alcohol, partially neutralized polyacrylate, PEG 60 hydrogenated castor oil, sorbitol, titanium dioxide, water.
Section 51945-4
Section 55105-1
Purpose
dl-Camphor External analgesic
l-Menthol External analgesic
Methyl Salicylate External analgesic
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only
Active Ingredient (55106-9)
Active ingredients
dl-Camphor 0.5%
l-Menthol 0.4%
Methyl Salicylate 0.8%
Indications and Usage (34067-9)
Uses temporary relieves minor aches and pains of muscles and joints associated with
■ simple backache ■ arthritis ■ strains ■ bruises ■ sprains
Dosage and Administration (34068-7)
Directions
■ Strip off the polyethylene film and place adhesive pad over affected area.
■ Change pad 1 or 2 times a day
■ Not for children under 4 years.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
■ condition worsens
■ symptoms persist for more than 7 days
■ symptoms clear up and occur again withn a few days
Section 50567-7 (50567-7)
When using this product
■ avoid contact with the eyes
■ do not bandage tightly
Section 50570-1 (50570-1)
Do not use
■ on wounds or on damaged skin
Section 51727-6 (51727-6)
Inactive ingredients
aluminum hydroxide gel, carbomer, carmellose sodium, edetate disodium, glycerin, malic acid, methacrylic acid and n-butyl acrylate copolymer, partially hydrolyzed polyvinyl alcohol, partially neutralized polyacrylate, PEG 60 hydrogenated castor oil, sorbitol, titanium dioxide, water.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose
dl-Camphor External analgesic
l-Menthol External analgesic
Methyl Salicylate External analgesic
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:20.692837 · Updated: 2026-03-14T23:01:49.492831