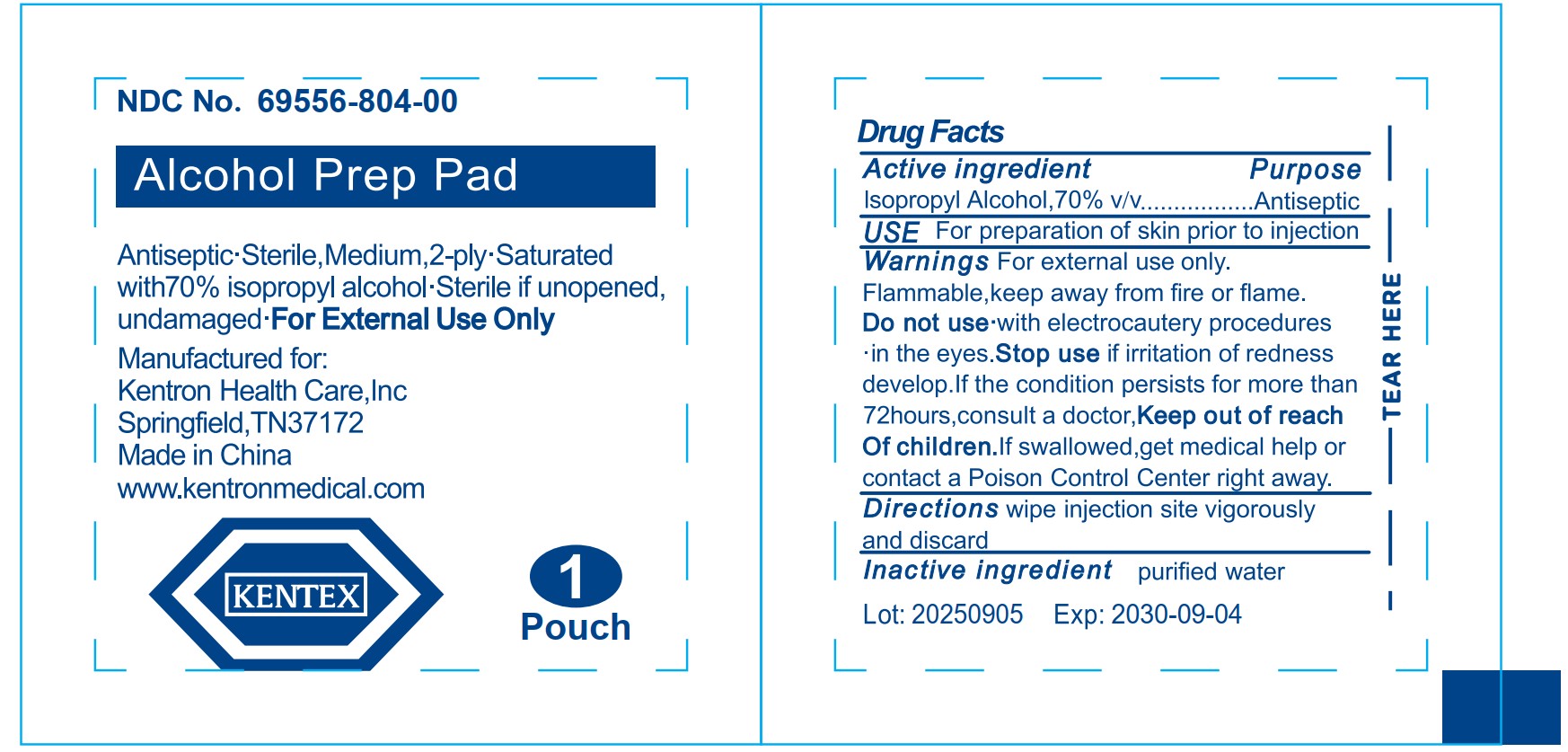
kentex alcohol prep pad saturated with 70% isopropyl alcohol
de7ffc63-bd14-42d8-b9fe-b7f8722b8882
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Isopropyl Alcohol,70% v/v
Purpose
Antiseptic
Medication Information
Warnings
For external use only.
Flammable,keep away from fire or flame.
Purpose
Antiseptic
Directions
wipe injection site vigorously and discard
Description
Isopropyl Alcohol,70% v/v
Use
For preparation of skin prior to injection
Do Not Use
• with electrocautery procedures
• in the eyes.
Drug Facts
Stop Use If
irritation of redness develop. If the condition persists for more than 72 hours, consuIt a doctor,
Active Ingredient
Isopropyl Alcohol,70% v/v
Package Labeling:
Inactive Ingredient
purified water
Keep Out of Reach of Children.
lf swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use (USE)
For preparation of skin prior to injection
Purpose
Antiseptic
Warnings
For external use only.
Flammable,keep away from fire or flame.
Directions
wipe injection site vigorously and discard
Do Not Use (Do not use)
• with electrocautery procedures
• in the eyes.
Drug Facts
Stop Use If (Stop use if)
irritation of redness develop. If the condition persists for more than 72 hours, consuIt a doctor,
Active Ingredient (Active ingredient)
Isopropyl Alcohol,70% v/v
Package Labeling:
Inactive Ingredient (Inactive ingredient)
purified water
Keep Out of Reach of Children. (Keep out of reach Of children.)
lf swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:41.245188 · Updated: 2026-03-14T23:13:00.870746