Clotrimazole Vaginal Cream, Usp
de3b6b64-dfa5-4abd-ba43-4ab59b4c2284
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Clotrimazole, USP 1% (50 mg in each applicatorful)
Purpose
Vaginal antifungal
Medication Information
Purpose
Vaginal antifungal
Description
Clotrimazole, USP 1% (50 mg in each applicatorful)
Uses
- treats vaginal yeast infections
- relieves external itching and irritation due to a vaginal yeast infection
Warnings
For vaginal use only
Directions
- before using this product, read the enclosed educational brochure for complete directions and information
- adults and children 12 years of age and over:
- vaginal cream: insert one applicatorful of cream into the vagina at bedtime for 7 days in a row. Wash applicator after each use.
- external cream: use the same tube of cream if you have itching and irritation on the skin outside the vagina. Squeeze a small amount of cream onto your fingertip. Apply to itchy, irritated skin outside the vagina. Use 2 times daily for up to 7 days as needed.
- children under 12 years of age: ask a doctor
Do Not Use
if you have never had a vaginal yeast infection diagnosed by a doctor
Questions?
Call 1-833-358-6431 Monday to Friday 9am to 7pm EST
Package Label
Other Information
- to open tube: unscrew cap, lift tab, and pull to remove foil seal prior to use
- do not use if foil seal on tube opening is broken or missing
- do not purchase if carton is open
- store at room temperature 20° to 25°C(68º to 77°F)
- before using any medication, read all label directions, Keep carton, it contains important information.
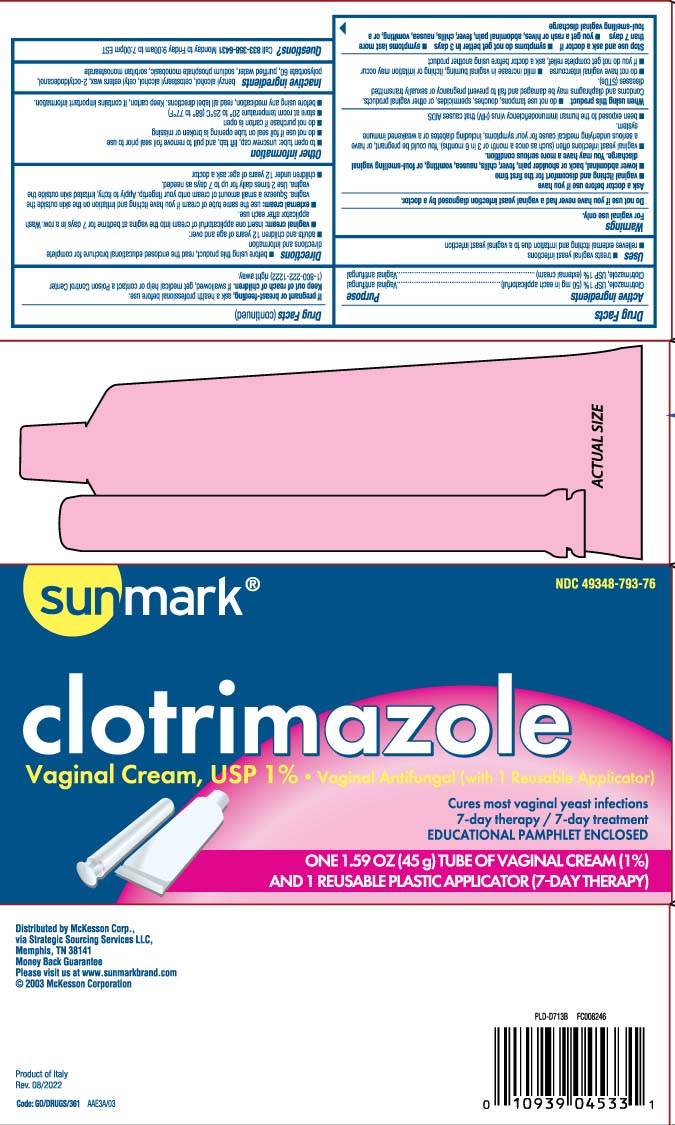
Active Ingredients
Clotrimazole, USP 1% (50 mg in each applicatorful)
Inactive Ingredients
benzyl alcohol, cetostearyl alcohol, cetyl esters wax, 2-octyldodecanol, polysorbate 60, purified water, sodium phosphate monobasic, sorbitan monostearate
Principal Display Panel
clotrimazole
Vaginal Cream, USP 1% • Vaginal Antifungal (with 1 Reusable Applicator)
Cures most vaginal yeast infections
7-day therapy / 7- day treatment
One 1.59 OZ (45g) TUBE OF VAGINAL CREAM (1%)
AND 1 REUSABLE PLASTIC APPLICATOR (7-DAY THERAPY
Distributed by McKesson Corp.,
via Strategic Sourcing Services LLC,
Memphis, TN 38141
When Using This Product
- do not use tampons, douches, spermicides, or other vaginal products. Condoms and diaphragms may be damaged and fail to prevent pregnancy or sexually transmitted diseases (STDs).
- do not have vaginal intercourse
- mild increase in vaginal burning, itching or irritation may occur
- if you do not get complete relief, ask a doctor before using another product
Stop Use and Ask A Doctor If
- symptoms do not get better in 3 days
- symptoms last more than 7 days.
- you get a rash or hives, abdominal pain, fever, chills, nausea, vomiting, or a foul-smelling vaginal discharge
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Ask A Doctor Before Use If You Have
- vaginal itching and discomfort for the first time
- lower abdominal, back or shoulder pain, fever, chills, nausea, vomiting, or foul-smelling vaginal discharge. You may have a more serious condition.
- vaginal yeast infections often (such as once a month or 3 in 6 months). You could be pregnant, or have a serious underlying medical cause for your symptoms, including diabetes or a weakened immune system.
- been exposed to the human immunodeficiency virus (HIV) that causes AIDS
Structured Label Content
Uses
- treats vaginal yeast infections
- relieves external itching and irritation due to a vaginal yeast infection
Purpose
Vaginal antifungal
Warnings
For vaginal use only
Directions
- before using this product, read the enclosed educational brochure for complete directions and information
- adults and children 12 years of age and over:
- vaginal cream: insert one applicatorful of cream into the vagina at bedtime for 7 days in a row. Wash applicator after each use.
- external cream: use the same tube of cream if you have itching and irritation on the skin outside the vagina. Squeeze a small amount of cream onto your fingertip. Apply to itchy, irritated skin outside the vagina. Use 2 times daily for up to 7 days as needed.
- children under 12 years of age: ask a doctor
Do Not Use (Do not use)
if you have never had a vaginal yeast infection diagnosed by a doctor
Questions?
Call 1-833-358-6431 Monday to Friday 9am to 7pm EST
Package Label (Package label)
Other Information (Other information)
- to open tube: unscrew cap, lift tab, and pull to remove foil seal prior to use
- do not use if foil seal on tube opening is broken or missing
- do not purchase if carton is open
- store at room temperature 20° to 25°C(68º to 77°F)
- before using any medication, read all label directions, Keep carton, it contains important information.
Active Ingredients (Active ingredients)
Clotrimazole, USP 1% (50 mg in each applicatorful)
Inactive Ingredients (Inactive ingredients)
benzyl alcohol, cetostearyl alcohol, cetyl esters wax, 2-octyldodecanol, polysorbate 60, purified water, sodium phosphate monobasic, sorbitan monostearate
Principal Display Panel (Principal display panel)
clotrimazole
Vaginal Cream, USP 1% • Vaginal Antifungal (with 1 Reusable Applicator)
Cures most vaginal yeast infections
7-day therapy / 7- day treatment
One 1.59 OZ (45g) TUBE OF VAGINAL CREAM (1%)
AND 1 REUSABLE PLASTIC APPLICATOR (7-DAY THERAPY
Distributed by McKesson Corp.,
via Strategic Sourcing Services LLC,
Memphis, TN 38141
When Using This Product (When using this product)
- do not use tampons, douches, spermicides, or other vaginal products. Condoms and diaphragms may be damaged and fail to prevent pregnancy or sexually transmitted diseases (STDs).
- do not have vaginal intercourse
- mild increase in vaginal burning, itching or irritation may occur
- if you do not get complete relief, ask a doctor before using another product
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- symptoms do not get better in 3 days
- symptoms last more than 7 days.
- you get a rash or hives, abdominal pain, fever, chills, nausea, vomiting, or a foul-smelling vaginal discharge
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- vaginal itching and discomfort for the first time
- lower abdominal, back or shoulder pain, fever, chills, nausea, vomiting, or foul-smelling vaginal discharge. You may have a more serious condition.
- vaginal yeast infections often (such as once a month or 3 in 6 months). You could be pregnant, or have a serious underlying medical cause for your symptoms, including diabetes or a weakened immune system.
- been exposed to the human immunodeficiency virus (HIV) that causes AIDS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:58.442032 · Updated: 2026-03-14T23:10:05.459246